Abstract
Rosa canina fruits have been used traditionally for the treatment of diabetes mellitus and its complications. The aim of current study was to evaluate the in vitro mechanism of action of R canina in managing diabetes mellitus. Cell proliferation and cytotoxicity assay were performed on pancreatic β-cells, βTC6. The protective activity of the extract on streptozotocin-induced death in βTC6 cells was studied. The effect of R canina on the metabolism of glucose in HepG2, a hepatocellular carcinoma cell line, was evaluated. The effect of the extract on glucose diffusion across the dialysis membrane, which is a comfortable model for assessing cellular glucose absorption, was evaluated. The results obtained from current study confirmed that R canina extract can act as a growth factor for pancreatic β-cell line providing a novel mechanism for the observed antidiabetic effect of this natural agent. Further preclinical studies are necessary to evaluate the perfect mechanism of action of R canina in diabetes mellitus.
Diabetes mellitus, the most common endocrine disorder, is a chronic metabolic disease that has a remarkable influence on the health and quality of life of patients. 1,2 The number of the people who suffer from diabetes mellitus and its complications has increased dramatically in the recent past. It has been estimated that the number of people with diabetes has increased to 347 million worldwide in 2013 and that it cover 5.4% of the population by the year 2025. 3
Diabetes-associated hyperglycemia stimulates the generation of reactive oxygen species, which plays a pivotal role in the severity of complications of diabetes. 4 Therefore, ameliorating blood glucose level is a key approach for prevention and management of the diabetic complications and improving the quality of life in both type 1 and 2 diabetic patients. 5 Several antidiabetic drugs have been administrated in medical practices; however, insufficient prevention of secondary complications and occurrence of side effects are the main restriction of conventional therapeutic approaches. 6,7
Medicinal plants are considered as a giant source of biologically active substances that play a key role in past and modern therapeutic approaches. The ethnobotanical evidence on herbal medicines with the potential antidiabetic activities has been informed for nearly 800 plants. 8
Rosa canina (rose hip) is a well-known medicinal plant that is used traditionally for various metabolic and inflammatory diseases. Fruits of R canina are reported to have protective and healing activities in a wide range of diseases, including rheumatoid arthritis, osteoarthritis, rheumatism, gout, sciatica, hemorrhoids, influenza, common cold, biliary complaints, gallstones, kidney and the lower urinary tract disorders, and gastrointestinal disorders. This medicinal plant also is able to help digestion and prevent gastric ulcer and possesses laxative and diuretic properties. The fruit is considered as an effective medicinal plant against diabetes mellitus in Iranian and Turkish traditional medicine. 9,10
Experimental studies reported that rose hips act as a potent antioxidant, anti-inflammatory, and anti-mutagen agent. 11 It is recognized that R canina fruits encompass various phytochemical compounds including organic acids, amino acid, phospholipids, carotenoids, flavonoids, tannins, carbohydrates, unsaturated and polysaturated fatty acids, tocopherols, minerals, gallactolipid, pectin, ascorbic acid, and carotene. 12
Recent studies provided evidence that regeneration of pancreas β-cells mediated by its proliferation have a pivotal role in managing diabetes mellitus. 13,14 The aim of current study was to evaluate the mechanism of action of R canina extract in managing diabetes mellitus and its complications. We studied cell proliferation and cytotoxicity assay on rat pancreatic β-cells, βTC6, cell line. Its protective effect on streptozotocin (STZ)-induced death in βTC6 cells was studied. Due to the central role of the liver in the metabolism of glucose, glucose-lowering potential of R canina extract in HepG2, a human hepatocellular carcinoma cell line, was also evaluated. Finally, the effect of Rosa extract on glucose diffusion across the dialysis membrane, which is a comfortable model for assessing cellular glucose absorption, was evaluated in vitro. 15
Materials and Methods
Chemical and Reagents
Glucose assay kit (Reagents: Glucose Oxidase/Peroxidase G-3660, o-Dianisidine Reagent D-2679 Glucose Standard Solution G-3285), methylthiotetrazole (MTT), and streptozotocin (STZ) were purchased from Sigma Aldrich (St Loui, MO). Bovine serum albumin (BSA), cell culture medium (Dulbecco modified Eagle medium [DMEM]), trypsin, penicillin-streptomycin, and fetal bovine serum (FBS) were obtained from Gibco (Gaithersburg, MD).
Plant Material and Sample Preparation
The ripe fruits of R canina were collected in July 2012 from Kermanshah province, west of Iran, and authenticated by Dr S. M Masoumi (Department of Plant Protection, Agricultural College, Razi University, Kermanshah, Iran). The ripe fruits of R canina were powdered after drying. A total of 100 g of plant powder was added to 1000 mL boiling water and mixed for 15 minutes. The whole content of the mixture was first filtered through an ordinary filter paper, and the filtrate was then passed through a No. 1 Whatman filter paper. The solution was transferred into the rotary evaporator for removing the surplus water and about 80% of water was removed. The final solution was kept in a water bath at 30°C.
Cell Culture
The βTC6 and HepG2 cell lines were obtained from Iran Genetic Resources Center and cultured in DMEM medium supplemented with 10% FBS, 100 U/mL penicillin, and 100 μg/mL streptomycin. βTC6 cells and HepG2 cells were cultured in a CO2 incubator (CB150 Binder) at 37°C in a humidified atmosphere enriched by 5% CO2 and subcultured every 3 to 4 days.
Viability and Proliferation Assay
The cytotoxic and proliferation effects of plant extract was determined against cell lines by a colorimetric assay using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) and compared with the untreated control. In the cytotoxic assay, cells were plated on to 96-well plates at a density of 5.0 × 104 cells/well and in a volume of 180 μL. One day after seeding, 20 μL of different concentrations of extract were added to each well. The cells were incubated for 24 hours and then 20 μL of MTT (5 mg/mL) was added in growth medium and then the plates transferred to a 37°C incubator for 3 to 4 hours. Supernatants were removed and the reduced MTT dye was solubilized with dimethyl sulfoxide (150 μL/well). Absorbance was determined on an ELISA plate reader (Biotek, H1 M) with a test wavelength of 540 nm and a reference wavelength of 630 nm to obtain the sample signal (OD570 to OD630). 16
Percentage of proliferation was calculated using the following formula:
Also, in vitro cell proliferation was assessed using MTT assay, and cells were seeded at a density of 5 × 104 cells/mL in a 96-well plate and incubated for 24, 48, 72, and 96 hours at 37°C in 5% CO2. Approximately 24 hours after seeding, when the cells were in the log growth phase, the cells were treated with plant extract in a concentration of 0.001 mg/mL dissolved in cell culture media, and continuously monitored for up to 96 hours. The cells that were treated with 20 μL sterile water served as the vehicle control cells. Cell attachment and proliferation were monitored every 24 hours using the MTT assay. 17,18
Morphological Studies
Morphological changes consistent with cell proliferation were observed using a normal light microscope. Cells were treated for 96 hours with Rosa extract (0.001 mg/mL). Untreated cells were used for a negative control. The morphological changes of the cells were observed 24, 48, 72, and 96 hours posttreatmet. 19
Death Induced by STZ Assay
βTC6 cells (5 × 104 cells/mL) were seeded in each well of a 96-well culture plate. After overnight incubation, 10 μL STZ (5 mM) dissolved in normal saline (pH 7.4) and 10 μL Rosa extract (1 mg/mL and 0.1 mg/mL) dissolved in sterile water were added to the cells at the same time. Treatment by 10 μL STZ (5 mM) with 10 μL sterile water was assigned as the STZ control. The cells were further incubated for 24 hours, and then the MTT assay was performed to measure cell viability. 20
Briefly medium was removed and 20 μL of MTT solution (5 mg/mL in PBS) was added to each well. After incubation for 3 hours at 37°C, MTT solution was removed and 150 μL of dimethyl sulfoxide was added to dissolve the crystals formed. Then absorbance at 540 nm was read using a microplate reader. The percentage of cell viability was calculated as (Absorbance(treatment)/Absorbance(negative control)) × 100%. 21
Effect of Rosa Extract on In Vitro Glucose Diffusion
A method described by Gallagher et al 22 was used to evaluate the effects of plant extract on glucose movement in vitro. This in vitro model consists of a dialysis tube (6 cm × 15 mm; MWCO: 2000) into which 6 mL of Rosa extract and 2 mL of NaCl (0.15 M) with D-glucose (1.65 mM) were added. The dialysis tube was sealed at each end and placed in a centrifuge tube containing 45 mL NaCl (0.15 M). The tubes were placed on an orbital shaker water bath and incubated at 37°C for 3 hours. The movement of glucose into the external solution was provided. Concentration of glucose within the dialysis tubing was measured and control test was conducted in the absence of Rosa extract. Glucose concentration was analyzed by enzymatic method using a glucose oxidase kit. 15
Glucose Consumption
The HepG2 cells were grown in DMEM (5.5 mmol/L glucose) containing 10% FBS. Two days before the experiments, the cells were plated into 96-well tissue culture plates with some wells left blank. After the cells reached confluence, the medium was replaced with DMEM supplemented with 0.2% BSA and glucose at various concentrations. After 12 hours, the medium was removed and the same BSA DMEM containing Rosa extract was added to wells as well as the blank wells. Finally, the medium was removed and its glucose concentration was assessed by the glucose oxidase method. The volume of glucose consumption was evaluated by the glucose concentration of blank wells subtracting the remaining glucose in the cell-plated wells. 23
Statistical Analyses
All tests were performed in triplicate and the results are presented as means ± standard deviation. Significant differences between the means of the experimental groups were identified with analysis of variance, followed by the Tukey–Kramer multiple comparisons test (GraphPad version 5.0; GraphPad Software Inc, San Diego, CA). Statistical significance was set at P < .05.
Result
Effect of Rosa Extract on βTC6 Cell Viability and Proliferation
The Rosa extract was evaluated for cytotoxic activity on βTC6 cells (Figure 1). The cells were treated for 24 hours with various concentrations of Rosa extract. Cell viability was determined by MTT assays. According to results obtained, Rosa extract at the concentrations of 0.00001 to 0.1 mg/mL was not toxic to βTC6 cells.
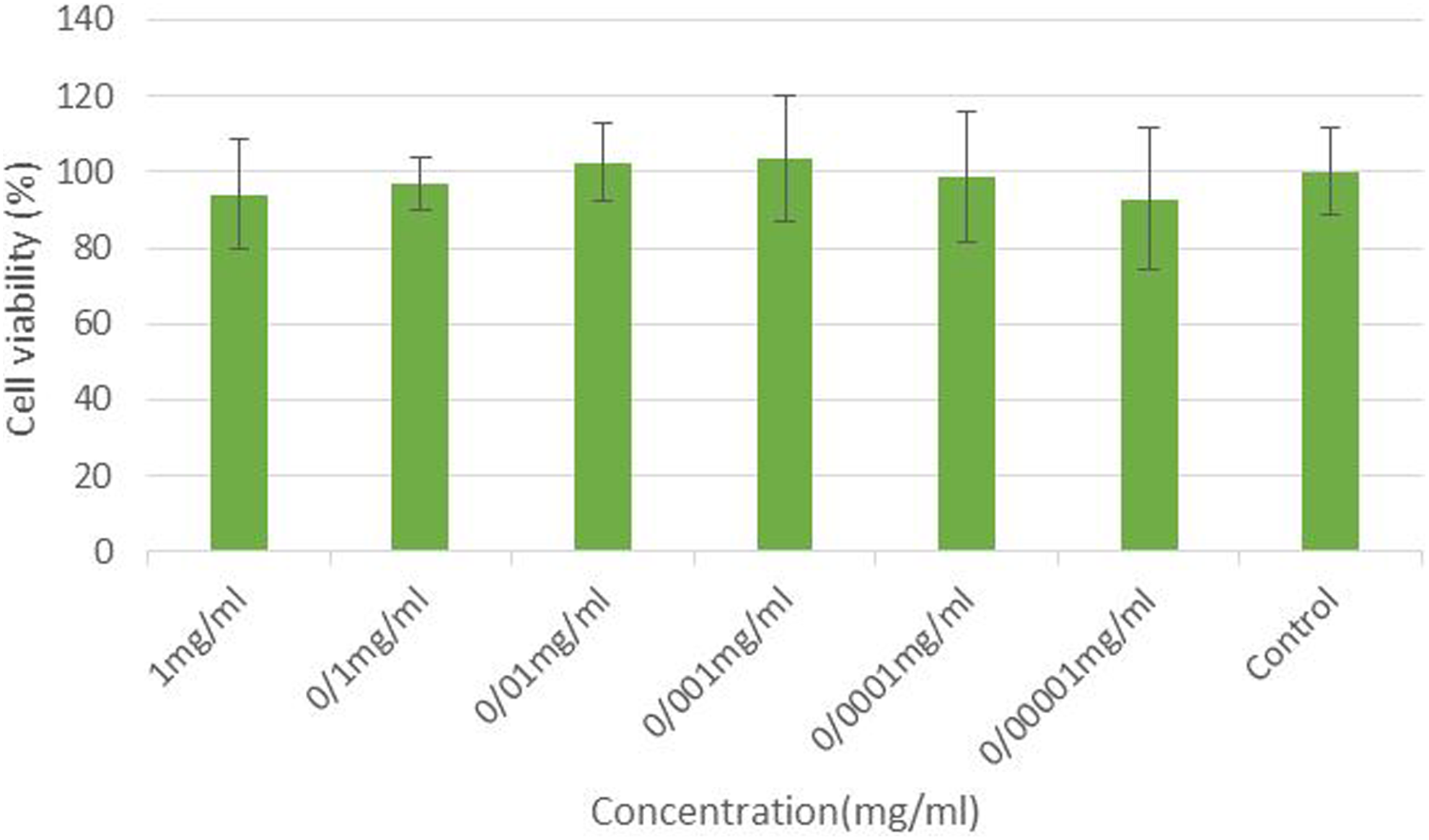
Cytotoxic activity of Rosa extract on βTC6 cells. βTC6 cells were cultured on a 96-well plate, at a concentration of 5 × 104 cells/mL in the absence or presence of different concentrations of Rosa extract (0.00001 to 1 mg/mL). Cell viability was measured using a cell proliferation kit (MTT), 24 hours after seeding. *Values are significantly different compared with control cells (P < .05).
The 0.001 mg/mL concentration of Rosa extract caused a significant (P < .05) increase in proliferation of βTC6 cells compared with control cells’ proliferation at the 24, 48, 72, and 96 hours after cell culture (Figure 2).

Rosa extract enhances proliferation of βTC6 cells. βTC6 cells were cultured on a 96-well plate, at a concentration of 2 × 104 cells/mL in the absence or presence of Rosa extract (0.001 mg/mL). Single dose proliferation was measured using a cell proliferation (MTT) assay 24, 48, 72, and 96 hours after seeding. Data are expressed as mean ± standard deviation of 2 determinations. *Values are significantly different compared with control cells (P < .05). Abs = absorbance.
Effect of Rosa Extract on Morphological Aspect of β-Cell
Control βTC6 cells (unexposed to Rosa extract) did not showed noticeable cell morphological changes (Figure 3). Treatment by Rosa extract with concentration of 0.001 mg/mL resulted in significant enhancement of cell number and morphological alterations in pancreatic βTC6 cells. As shown in Figure 3, β-cell treated with Rosa extract have spindle-shaped morphological properties, indicating that the cells are dividing.

Morphological and number changes in the βTC6 cells. βTC6 cells that were exposed to Rosa extract after 24 (A), 48 (B), 72 (C), and 96 (D) hours. Untreated control βTC6 cells after 24 (E), 48 (F), 72 (G), and 96 (H) hours. Cell number and morphological changes were examined using invert microscopy.
Inhibition of STZ-Induced βTC6 Cell Death
Exposure to STZ (5 mM) induced about 50% death of βTC6 cells. When concentrations of 1 and 0.1 mg/mL of Rosa extract was added, no significant protective effect against STZ-induced death of βTC6 cell line was observed by mentioned concentrations of plant preparation (Figure 4).

Effect of Rosa extract (Re) on STZ-induced cell death. About 50% inhibition of cell proliferation was induced by 5 mM STZ for 24 hours. Concentrations of 0.1 mg/mL and 1 mg/mL of Rosa extract were added together with 5 mM STZ and the percentage cell survival was determined by MTT assay. *Values are significantly different compared with STZ-control cells (P < .05).
Effect of Rosa Extract on Glucose Diffusion
In the present study, Rosa extract did not demonstrated significant inhibitory effects on glucose movement into external solution across dialysis membrane compared to control. In contrast, they exhibited an increasing effect on glucose movement from dialysis tube to external solution (Figure 5).
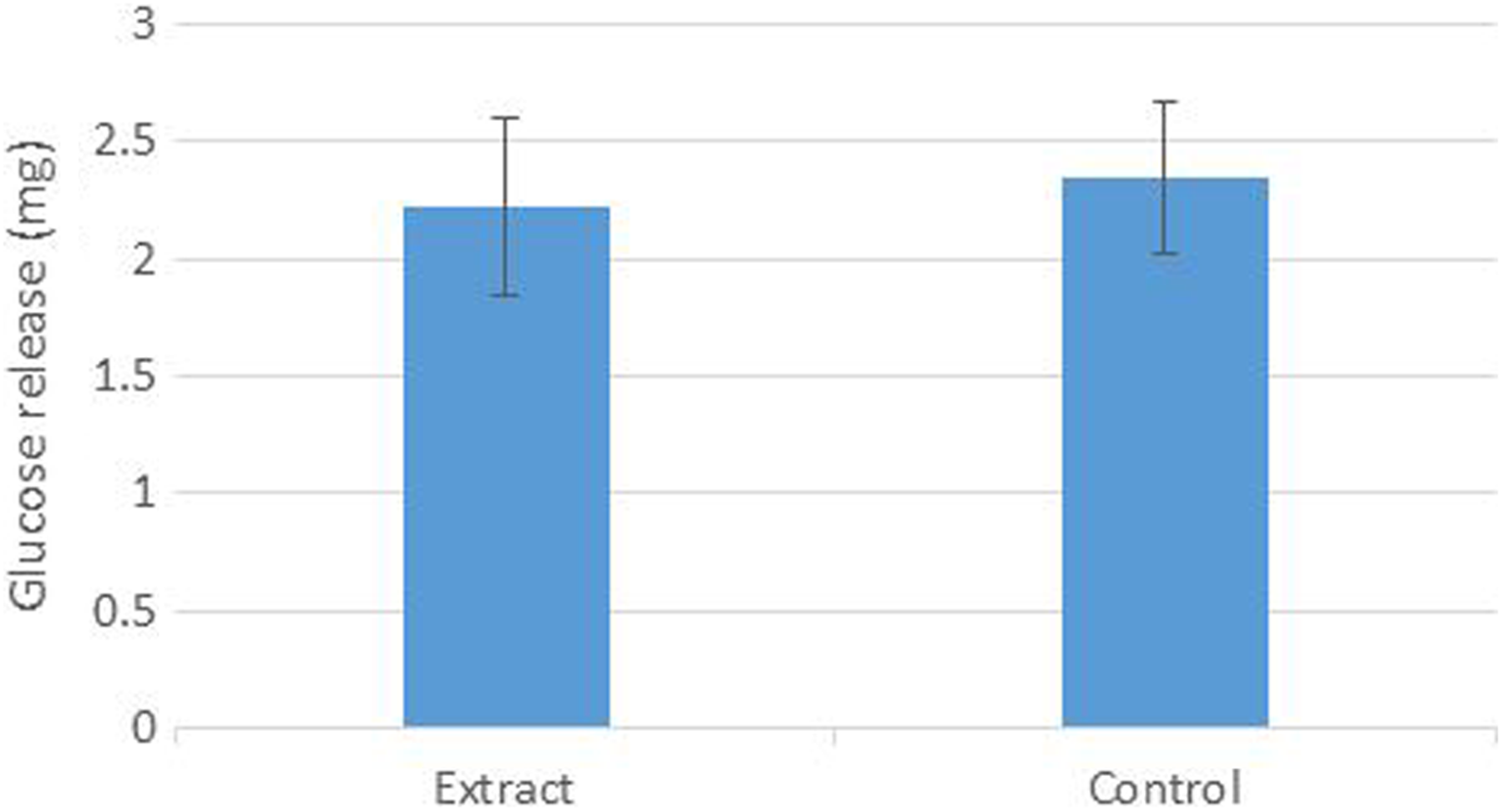
Effect of Rosa extract on the diffusion of glucose out of dialysis tube after 3 hours. Means are not significantly different compared with control as determined, P > .05.
Effect of Rosa Extract on HepG2 cells
Subsequent to 24-hour incubation of HepG2 cell line with 1 mg/mL concentration of Rosa extract under 11.1 mml/L glucose concentration, glucose consumption of hepatic cells was assessed (Figure 6). Treatment with Rosa extract did not cause significant glucose-lowering effect in HepG2 cell line exposed to immoderate hyperglycemia (11.1 mmol/L), in comparison with control group.

Glucose consumption was measured after 24-hour incubation of HepG2 cell line with 1 mg/mL concentration of Rosa extract under 11.1 mml/L glucose concentration. Means are not significantly different compared with control as determined, P > .05.
Discussion and Conclusion
The plant kingdom consist of an extensive source of bioactive compounds that can be considered as future drugs for various diseases. 24,25 Scientific evaluation of traditional medicinal approaches of nations of all over the world can result in exploration of novel therapeutic agents for management of complex diseases such as diabetes mellitus. 26
As diabetes mellitus is a major lifestyle disease nowadays, suitable management of the disease is necessary. Accumulating scientific evidence has confirmed the therapeutic benefits of medicinal plants with hypoglycemic effects in the management of diabetes mellitus. In Iranian folklore and traditional medicine, Rosa canina fruits have been used for the treatment of diabetes mellitus and its complications. In this study, we examined the efficiency of R canina fruits extract in in vitro models of antidiabetic mechanism of actions indicating that this plant actually can be considered as an adjunctive therapy for managing diabetes mellitus. Several bioassays were used and different cellular methods were carried out for clarifying the possible antidiabetic mechanism of the R canina fruits.
The extract with the ranges of 1 to 0.000001 mg/mL concentration applied in this study was not toxic in βTC6 cell line. The Rosa extract with the concentration of 0.001 mg/mL significantly increased proliferation of βTC6 cell line compared with control cells. Previous study on fruits of R canina demonstrated that this medicinal plant possesses hypoglycemic effect in animal model of diabetes mellitus. 27 One of the main causes of diabetes mellitus is defined as deficiency of pancreatic β-cell viability and performance. The finding of current study exhibited that enhancement of pancreatic β-cell proliferation plays a pivotal role in managing diabetes mellitus and its complications by this medicinal plant. 28,29
In progressive phases of type 2 diabetes mellitus, chronic hyperglycemia is toxic to the functions of β-cells, which can result in the necessity of insulin therapy. The glucotoxicity is a slow procedure defined by elevating β-cells dysfunction as a result of permanent β-cells loss by apoptosis. 21 In this study, the protective effect of Rosa extract against β-cells death was assessed. It was not confirmed that death of pancreatic βTC6 cell line induced by STZ is inhibited by pretreatment with Rosa extract, indicating that Rosa extract cannot protect β-cells against STZ-induced cell death, but this extract increased cell proliferation. It has been found that STZ cytotoxicity is due to its entrance to the β-cell through a glucose transporter (GLUT2), which can result in alkylation of DNA. Damage of DNA causes ADP-ribosylation activation and leads to STZ-induced cytotoxicity of β-cell. 30 Despite the potential of Rosa extract on improvement of cell proliferation, it cannot prevent pathological condition induced by STZ, which results in DNA damage of β-cell and cytotoxicity.
Liver possesses an essential character in glucose metabolism. In pathological condition of diabetes, hyperglycemia is associated with less glycogen production and more glycogenesis. 31 A large body of evidence confirmed that reduction of hepatic glucose output might be the leading step in the management of diabetes. Assessment of glucose consumption potential of Rosa extract in HepG2 cell line, which is phenotypically similar to human hepatocytes, did not resulted in significant alteration in glucose uptake in hepatic cells.
Finally, the potential of Rosa extracts to retard the diffusion and movement of glucose was evaluated in vitro. The results demonstrated that this medicinal plant did not inhibit glucose diffusion using an in vitro model of glucose absorption. However, the in vitro inhibitory activity on glucose diffusion cannot be always associated to in vivo activity; thus, the data must be proved in animal models.
The hypoglycemic potential of Rosa extract in type 1 diabetic rats has been demonstrated previously. Strengthening of antioxidant performance as well as scavenging free radicals play key roles in its antidiabetic activity. Trans-tiliroside, an active component of Rosa, is reported to improve glucose tolerance and insulin resistance–associated obesity in an animal model of diet-induced obesity. 32
Our data from cell studies confirmed that R canina extract can act as a growth factor for pancreatic β-cell line, providing a novel mechanism for the observed antidiabetic effect of this natural agent. In summary, although R canina extract could increase proliferation in βTC6 cell line, further preclinical and clinical studies are necessary to elucidate the perfect mechanism of action of this medicinal plant as an antidiabetic agent. In addition, further phytochemical investigation is recommended to isolate and identify the bioactive molecules of R canina fruits in inducing antidiabetic activity.
Footnotes
Authors’ Note
The study reported in this article was performed in partial fulfillment of the requirements for the PharmD degree of Fatemeh Niyazi, in the Faculty of Pharmacy, Kermanshah University of Medical Sciences, Kermanshah, Iran.
Author Contributions
FN contributed toward experimental procedures, data analyses, and prepared the final version of the article. MHF and AF contributed toward experiments, data analyses, and the preparation of the article. BS contributed toward all cellular procedures and data analyses. GB contributed toward data analysis and the preparation of the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by the Research Council of Kermanshah University of Medical Sciences (Grant Number: 94582).
Ethical Approval
This study did not require ethical approval as no human subjects were involved.

