Abstract
Natural plant product Psyllium has anti-inflammatory activity that can modulate the function of cytokines. We determined the effect of Psyllium husk extract on interleukin (IL)-8 and NF-κB secretion by gastric epithelial cells in response to Helicobacter pylori. Human gastric adenocarcinoma cell line (AGS) cells were pretreated with Psyllium extract in different concentrations before H pylori infection. Cell culture supernatant was analyzed for IL-8 and NF-κB by ELISA. RNA from cells was used for real-time polymerase chain reaction for messenger RNA expression of IL-8. Psyllium extract 5 and 10 μg/mL markedly (P < .001) lowered basal IL-8 by 64.71% and 74.51%, respectively, and H pylori–stimulated IL-8 was also (P < .001) lowered by 41.67% and 66.67%, respectively. Psyllium 5 and 10 μg/mL also reduced (P < .0001) cagA-positive H pylori–induced IL-8 mRNA expression by 42.3% and 67.6%, respectively. Psyllium also reduced (P = .0001) NF-κB in response to H pylori strains confirming its role as an anti-inflammatory agent.
Plant-derived phenolic compounds and flavonoids exhibit anti-inflammatory activity by modulating the expression levels of various cytokines including interleukin-1 (IL-1), nuclear kappa B (NF-κB), tumor necrosis factor-α (TNF-α), cyclooxygenase (Cox-2), and so on. These plant-derived compounds have reported clinical benefits and pharmacologic effects. Plantago ovata (Psyllium, Ispaghul) is an annual herb of the Plantaginaceae family found widely in different countries. Decoction of its dried seeds is taken orally for diarrhea and as a demulcent.
1
Psyllium contains active principals, including 4-O-methylglucuronic acid, aucubin, campesterol, linoleic acid, oleic acid, palmitic acid,
Helicobacter pylori is a Gram-negative microaerobic organism that causes gastritis and is associated with peptic ulcers, gastric carcinomas, and mucosa-associated lymphomas. H pylori adheres to gastric epithelial cells and stimulates interleukin-8 (IL-8) production to attract components of the innate and adaptive immune systems to the site of infection. This results in neutrophil infiltration of the gastric epithelium that characterizes H pylori gastritis. H pylori infection activates NF-κB in gastric epithelial cells in vitro and in vivo. NF-κB is a transcriptional regulator of IL-8 production, and its activation after bacterial infection is an important defense response in gastrointestinal epithelial cells. Toll-like receptor 2 (TLR2) and TLR5 recognize H pylori and initiate signaling pathways that result in activation of NF-κB. H pylori induced a time-dependent expression of mRNA and protein for IL-8 via activation of NF-κB and increased the levels of IL-8 and IL-6, which were inhibited by NF-κB inhibitor, pyrrolidine dithiocarbamate. 9 NF-κB activity is inhibited by a variety of substances including glucocorticoids and nonsteroidal anti-inflammatory agents. 10,11 In gastric cancer-derived cell line, MKN45 was shown to produce an increased amount of IL-8 on coculture with live H pylori only, indicating that IL-8 production requires a direct contact between the cells and live bacteria. 12 In gastric carcinoma, the tumor cells were positive for IL-8. 13 H pylori directly induced IL-8 mRNA expression and protein secretion in established gastric epithelial cell lines and cagA positive and negative strains of H pylori varied in inducing epithelial IL-8 production. 14
The literature suggests that Psyllium contained chemical compounds and flavonoids that have the properties to counteract the immunological cytokine-mediated reactions involved in the immunopathogenesis of H pylori infection. Cell lines derived from normal human and patients are used to model and study drug effects, genetic and immune responses, and so on, as cells can be grown and treated with drugs under the same condition. The aim of this study was to determine the effect of Psyllium extract on the (a) production and expression of IL-8 by the AGS cells; (b) NF-κB by the AGS cells; and (c) determine whether the effect of Psyllium extract varied with the virulence marker of the H pylori–stimulated AGS.
Materials and Methods
Preparation of the Psyllium Extract and Its Fractions
The seed husk of Plantago ovata was purchased from a local market of Karachi, Pakistan. It had been cultivated in Jamshoro (Sindh, Pakistan). A specimen, voucher # Po-SH-09-06-81, was preserved in the herbarium of the Natural Product Research Division, Department of Biological and Biomedical Sciences, Aga Khan University, Karachi. The extract was prepared by using a method previously described, 1 with slight modification: psyllium husk was soaked in 70% methanol for 3 days and filtered through muslin cloth and Whatman (Maidstone, UK) No. 1 filter paper, simultaneously. This procedure was repeated 3 times, and all the filtrates were combined and evaporated on a rotary evaporator (model RE-111; Flawil, Switzerland) under reduced pressure (−760 mm Hg) to obtain, finally, a crude extract of Ispaghula. The yield of thick, dark brown, pasty-like mass was 1.75% (w/w). Activity-directed fractionation of the crude extract was carried out by following standard phytochemical procedures using different organic solvents. 15 Approximately 20 g Ispaghula extract was dissolved in 150 mL distilled water. An equal volume of petroleum spirit was added and the mixture was shaken vigorously in a separating funnel. After shaking the mixture separated into 2 layers and the petroleum spirit layer (upper) was removed. Extraction with petroleum spirit was repeated 2 more times. The petroleum spirit extracts were combined and evaporated to furnish the petroleum spirit fraction. The lower separated layer was placed in a separating funnel; chloroform was added, and extraction and separation were performed by following the previously mentioned procedure. The lower layer, in this case chloroform, was separated, and the procedure was repeated 2 times. The chloroform extracts were combined and evaporated to obtain the chloroform fraction. Ethyl acetate was added to the remaining, upper, layer and extraction and separation, followed by evaporation, were performed, resulting in the ethyl acetate fraction. The remaining lower layer was collected and concentrated to obtain the aqueous fraction.
Phytochemical Screening
Phytochemical analysis of the crude extract of Ispaghula was performed, qualitatively, for the presence of alkaloids, anthraquinones, coumarins, flavonoids, saponins, sterols, tannins, and terpenes according to standard methods. 16
Cell Culture
AGS cells (ATCC CRL 1739; human gastric adenocarcinoma epithelial cell line) were purchased from the American Type Cell Culture (Philadelphia, PA). Cells were grown in monolayer culture in RPMI 1640 (Sigma, Germany) medium supplemented with 100 μg/mL penicillin, 100 μg/mL streptomycin, 2.5 μg/mL amphotericin B, and 10% decomplement fetal bovine serum (Invitrogen, Paisley, UK). Cells were grown in 75 cm2 tissue culture flasks at 37°C in an atmosphere of 5% CO2 and 95% air and passaged every 3 to 5 days. In the H pylori in vitro experiment–induced IL-8 secretion, the cell culture medium was not supplemented with antibiotics.
Determination of DNA Fragmentation in AGS Cells
Cytotoxic effect of the Ispaghula extract was determined by quantitative DNA fragmentation assay as described previously. 17 Briefly, cells were either pretreated with or without extract for 4 hours and were lysed in a lysis buffer (10 mM Tris, 1 mM EDTA, 0.2% Triton X-100, pH 7.5) followed by centrifugation at 13 000 × g for 10 minutes. Each DNA sample in the supernatant and the resulting pellet were then precipitated in 12.5% trichloro acetic acid (TCA), at 4°C, and quantified using a diphenylamine reagent after hydrolysis in 5% TCA at 90°C for 20 minutes. The percentage of fragmented DNA for each sample was calculated as the amount of DNA in the supernatant divided by the total DNA for that sample (supernatant plus pellet). To examine cytotoxic effect of Ispaghula extract against AGS cells, DNA fragmentation assay was employed. DNA fragmentation is a hallmark of apoptosis, and was measured at the concentration of 5 μg/mL and 10 μg/mL at 4 hours of incubation with Ispaghula extract. The results revealed no significant induction of DNA fragmentation by the Ispaghula extract (2.5 ± 1.0%) when compared with untreated cells (2.4 ± 0.8%). These results suggested that the effect of these herbs in H pylori/cell coculture system will not be due to either alteration in H pylori viability or toxicity to the bacterial cells.
Bacterial Culture
Thirty-six clinical isolates of H pylori were used to infect AGS cells. H pylori were cultured using Columbia blood agar (Oxoid) supplemented with 10% defibrinated sheep blood and Dents supplement (containing vancomycin, trimethoprim, and polymyxin) and incubated at 37°C under microaerophilic conditions using anaerobic jars and Campygen strips (Oxoid, UK) for 3 to 5 days. Plates were then examined for bacterial growth and typical colonies were selected for identification. The identity of H pylori was confirmed by Gram stain and production of urease and catalase. H pylori isolates were defined as gram-negative spiral-shaped bacilli that were catalase positive and rapidly (less than 30 minutes) urease positive. H pylori ATCC 49503 (type strain) was used as a positive control for the culture conditions and identification tests. H pylori reference strain ATCC49503 was used as a control strain.
Extraction of Genomic DNA
The bacterial cells on chocolate agar plate was washed twice with phosphate buffer saline (PBS, pH 8.0) and then centrifuged at 3000 rpm for 20 minutes. H pylori DNA was extracted by a phenol/chloroform method similar to the method previously described. 18 Bacterial pellet was suspended in Tris–Cl buffer containing ethylenediaminetetraacetate (TE; pH 8.0) and lysozyme and was then incubated at 37°C for 30 minutes. The suspension was treated with sodium dodecyl sulfate, proteinase K, and RNase A. DNA was extracted with phenol/chloroform/isoamyl alcohol, precipitated by sodium acetate and ice-cold absolute alcohol, and washed with ice-cold alcohol (70%). The pellet of DNA was finally resuspended in TE buffer. DNA content and purity was determined by measuring the absorbance at 260 nm and 280 nm using a spectrophotometer (Beckman DU-600, USA).
Polymerase Chain Reaction
cagA and vacA Genotyping
Amplification of cagA was performed by polymerase chain reaction (PCR) using primers (Table 1) as previously described. 19 DNA from H pylori strains ATCC 43504 (vacAs1am1, cagA positive) and ATCC 51932 (vacA s2m2, cagA negative) was used to define the accuracy of the cagA. After PCR, the amplified PCR products were electrophoresed in 2% agarose gels containing 0.5% × Tris/acetate/ethylenediaminetetraacetic acid, stained with ethidium bromide, and visualized under a short wavelength ultraviolet light source.
Cytokines’ Primer Sequence.

Abbreviations: F, forward primer; R, reverse primer.
Real-Time Quantitative PCR With SBG
Total RNA was extracted from AGS cells as described previously. 20 Reverse transcription of the extracted RNA was performed using RNase H-deficient reverse transcriptase (Superscript II, Life Technologies) and oligo (dT) primers (Life Technologies). Aliquots (2 μL) of reverse transcription reaction mixture (20 μL) were used for quantitation of IL-8 and GAPDH gene expression by real-time PCR assays (Table 1). The SABG QRT PCR was used to quantify IL-8 and GAPDH gene expression (PE Applied Biosystems, Foster City, CA). The PCR reactions were performed using the SBG PCR kit (PE Applied Biosystems) as described previously. 21 After activation for 10 minutes at 95°C, 40 cycles of 15 seconds at 95°C, and 1 minute at 62°C was carried out in model icycler (Biorad). Real-time fluorescence measurements was recorded and the threshold cycle (Ct) value for each sample calculated by the above-mentioned sequence detector. 22 For GAPDH standard curves of Ct values was obtained from real-time PCR of PCRII GAPDH (Reference plasmids). Ct values for IL-8 and GAPDH transcripts from each experiment were plotted on the standard curves, and the amounts (in pg) of each transcript was calculated. The amounts of IL-8 transcripts (pg) were expressed relative to that of GAPDH (pg). Each experiment AGS cell sample was tested in duplicate, and the average of 2 Ct values was used in this study.
Effect of Psyllium Extract on Helicobacter Pylori Viability
Briefly, H pylori was either left untreated or treated with Psyllium extract at the concentration of 5 μg/mL and 10 μg/mL for 4 hours at 37°C. H pylori strain was then serially diluted and inoculated onto Columbia blood agar plates enriched with H pylori–selective supplements and incubated under microaerophilic conditions. After the incubation of 2 to 3 days, the bacterial colonies were counted and the colony forming units were calculated. Data are expressed as percentage of survival. The results are representative of at least 3 independent experiments. To evaluate the effect of psyllium extract in H pylori–infected cells, we first analyzed the viability of H pylori in the presence of this extract at 5 μg/mL and 10 μg/mL, which was lower than that of their MBC values reported earlier. 23
Cytokine Assays
Cell culture supernatants were harvested and analyzed for cytokines by ELISA techniques with a commercially available kit. Human IL-8 kit was obtained from BD OptEIA and NF-κB from Invitrogen (Camarillo, CA). All cytokine assays were calibrated against the World Health Organization international standards by the kit manufacturer. The lower limit of detection for the IL-8 assay was 0.8 pg/mL and for NF-κB it was less than 0.5 pg/mL.
IL-8 and NF-κB Protein Production by AGS Cells
We analyzed effect of Psyllium extract in 2 different concentrations on H pylori–induced IL-8 and NF-κB production by the AGS. Cells were pretreated with Psyllium extract in concentrations of 5 μg/mL and 10 μg/mL prior to H pylori infection. AGS cells were treated with Psyllium extract in cell culture medium before bacterial infection. The treated cells were then infected with H pylori isolates for 6 hours. The supernatants were collected and stored at −80°C before analysis. The level of IL-8 in supernatants from cell line culture was determined by using a sandwich enzyme-linked immunosorbent assay (ELISA) kit (IL-8 from BD systems and NF-κB from Invitrogen, Camarillo, CA), according to the manufacturer’s instruction.
Data Analysis and Statistics
All analysis of graphs, calculations, and statistical analysis were performed by using GraphPAD software (GraphPAD, San Diego, CA). Results are expressed as mean ± standard error of the mean (SEM; n = number of experiments) and the median effective concentrations (EC50 values) with 95% confidence intervals. P < .05 was considered significantly different. We used a nonparametric test to compare the effect of different concentrations of Psyllium extract on expression of IL-8 by the AGS cell lines.
Results
DNA Fragmentation Assay
The cytotoxic effect of Ispaghula extract was examined against the AGS cells. DNA fragmentation was measured at the concentration of 10 μg/mL at 4 hours of incubation with extract. The results revealed no significant induction of DNA fragmentation by the evaluated extract (Psyllium, 2.4 ± 1.0%) when compared with untreated cells (2.5 ± 0.6%). H pylori colonies were noted on the Columbia blood agar plates enriched with H pylori–selective supplements and incubated under microaerophilic conditions following treatment with Psyllium extract at the concentrations of 5 μg/mL and 10 μg/mL for 4 hours at 37°C after incubation for 3 days. These results suggest that the effect of the extract in H pylori/cell co-culture system will not be due to either alteration in H pylori viability or toxicity to the cells.
Effect of H pylori on AGS Cell Secretion of Interleukin-8
The mean basal AGS cell secretion of IL-8 was 51 ± 1 pg/mL, and the H pylori isolates stimulated mean IL-8 secretion was 773 ± 3 pg/mL (P < .001; Figure 1).
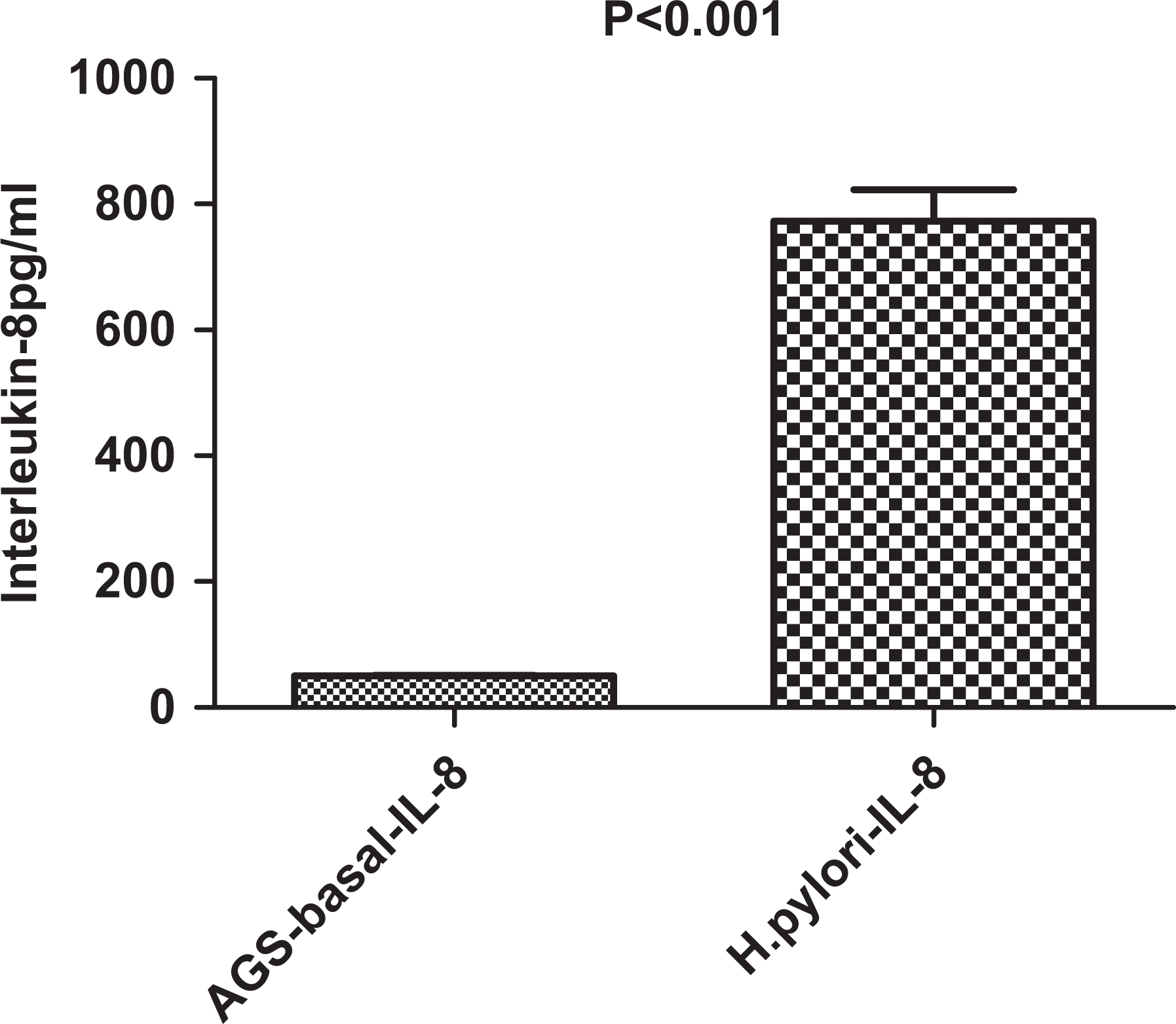
AGS cells without and with Helicobacter pylori (Wilcoxon rank test).
Effect of Psyllium Extract on AGS Cell Secretion of Interleukin-8
The basal IL-8 AGS cell secretion was 51 ± 1 pg/mL. Psyllium extract 5 μg/mL lowered it to 64.71% (18 ± 3 pg/mL; P < .001) and 10 μg/mL to 74.51% (13 ± 3 pg/mL; P < .001; Figure 2A). The expression of IL-8 mRNA in AGS cells with Psyllium 5 μg/mL and 10 μg/mL was significantly (P < .001) reduced to 41.67% and 66.67%, respectively, when compared with (basal) control IL-8 mRNA level of AGS cells without Psyllium extract treatment (Figure 2B).

(A) AGS cells alone and with Psyllium 5 μg/mL (P < .001) and 10 μg/mL (P < .001) (Wilcoxon rank test). (B) AGS mRNA expression of AGS cells alone and with Psyllium 5 μg/mL (P < .001) and 10 μg/mL (P < .001) (Wilcoxon rank test).
Effect of Psyllium Extract on H pylori–Stimulated AGS Cell Secretion of Interleukin-8
The mean IL-8 concentration secreted by AGS cells when stimulated by H pylori isolates and acted on by Psyllium extract in a concentration of 5 μg/mL and 10 μg/mL was 343 ± 9 pg/mL and 198 ± 4 pg/mL, respectively (Figure 3A). The expression of IL-8 mRNA in H pylori–stimulated AGS cells with Psyllium 5 μg/mL and 10 μg/mL was also significantly (P < .001) reduced to 42.3% (P < .001) and 67.6%, respectively (Figure 3B).

(A) AGS cells with cagA-positive Helicobacter pylori alone and with Psyllium extract 5 μg/mL and 10 μg/mL (Wilcoxon-rank test). *Helicobacter pylori vs Psyllium 5 μg/mL (P < .001) and 10 μg/mL (P < .001). (B) AGS cell infected with cagA-positive Helicobacter pylori mRNA expression without Psyllium and with 5 μg/mL and 10 μg/mL (Wilcoxon rank test).
Effect of Psyllium Extract on H pylori cagA-Dependent IL-8 Secretion From AGS
The expression of IL-8 mRNA in H pylori cagA–positive stimulated AGS cells was found to be 100% (7.8 pg/mL) and following Psyllium extract 5 μg/mL and 10 μg/mL treatment significantly (P < .001) reduced the expression of IL-8 mRNA to 42.3% (4.5 ± 7 pg/mL) and 67.6% (2.5 pg/mL), respectively (Figure 3B).
Effect of Psyllium Extract on AGS Cell Secretion of NF-κB
The H pylori–stimulated AGS cell secretion of NF-κB varied from 0.9 to 3.2 pg/mL. The secretion of NF-κB AGS cells was greater in response to H pylori cagA–positive strains (P = .001; Figure 4). Following exposure to Psyllium extract 10 μg/mL treatment AGS secretion of NF-κB secretion was reduced in response to both H pylori cagA positive and negative strains (P = .0001; Figure 5). However, the lower limit of Psyllium extract was found ineffective.

Comparison of NF-κB level produced by AGS cells following exposure to Helicobacter pylori cagA positive and negative strains (Mann-Whitney U test).

Comparison of NF-κB level following exposure of Helicobacter pylori cagA positive and negative strains to Psyllium extract (Mann-Whitney U test).
Discussion
The gastrointestinal tract lining epithelium cells provide physical and chemical barriers that protect the host against the microorganisms. It plays an active role in host mucosal defenses. The Psyllium extract is not toxic to AGS cells or to H pylori as the results revealed lack of significant induction of DNA fragmentation and growth of H pylori colonies noted after the incubation of 3 days on the enriched Columbia blood agar plates incubated under microaerophilic conditions following treatment with Psyllium extract at the concentrations of 5 μg/mL and 10 μg/mL for 4 hours at 37°C.
This study demonstrated that Psyllium extract in vitro reduced IL-8 production from the AGS cells (Figure 1). This effect increased with an increase in the concentration of Psyllium extract. This IL-8 lowering effect of Psyllium was also associated with decreased expression of IL-8 mRNA expression in the gastric epithelial cells. H pylori increased IL-8 secretion from the cells (Figure 2A), and it was greater with H pylori isolates that were cagA positive (Figure 3A). Psyllium extract in different concentrations lowered H pylori–stimulated secretion of IL-8 from the epithelial cells (Figure 3A). This was also associated with reduced expression of IL-8 mRNA expression in the epithelial cells. It is known that IL-8 secretion by the H pylori is NF-κB mediated. Furthermore, the secretion of NF-κB from AGS cells was shown to be greater in response to H pylori cagA positive isolates (Figure 1; Mann-Whitney U test, P = .0011). Following exposure to Psyllium extract AGS secretion of NF-κB secretion was reduced in response to both H pylori cagA positive and negative strains.
These results are in keeping with previous studies that have shown that dietary fiber supplementation from Psyllium was associated with an increased production of short-chain fatty acids, for example, butyrate, that act synergistically in inhibiting the production of pro-inflammatory mediators. 8 TNF-α production by intestinal biopsies and lamina propria mononuclear cells was inhibited via NF-κB activation and IκBα degradation. 24 Psyllium extracts’ ability to reduce the cytokine secretion from the H pylori–stimulated gastric epithelial cells in vitro is explained on the basis of the active ingredient, that is, flavonoids such as apigenin, luteolin, baicalein, and so on. These are benzo-y-pyrone derivatives and are known to display inhibitory actions on enzyme systems critically involved in the initiation and maintenance of the inflammatory and immune response, including serine and threonine protein kinases, phospholipases, lipoxygenase, and cyclooxygenase. 21 Apigenin inhibit cytokine-induced gene expression in human endothelial cells, that is, it blocks the induction of intercellular adhesion molecule-1, vascular cell adhesion molecule-1, and E-selectin mRNA, suggesting that apigenin inhibits some step in the signal transduction pathway for cytokine activation of endothelial cells. 22 Apigenin also exhibited a dose- and time-dependent, reversible effect on adhesion protein expression as well as inhibiting adhesion protein upregulation at the transcriptional level. 22 This is in keeping with the result of this study, which demonstrated that increasing the extract concentration was associated with a greater decrease in the IL-8 production from AGS cells. Baicalein, previously demonstrated, significantly inhibited production of IL-6, IL-8, and MCP-1 in a dose-dependent manner in IL-1β-activated HMC-1. 22 It also inhibited the NF-κB activation via inhibition of IκBα phosphorylation and degradation. Luteolin present in the Psyllium also inhibits NF-κB signaling in immune cells. 25
The implication of this study is that the Psyllium can play a role in the treatment of H pylori–associated diseases that is infective and inflammatory in nature. Psyllium-containing compounds with gel forming properties will prevent H pylori contact with gastric epithelial cells. This contact plays an important role in the pathogenicity of the H pylori with essential contributions by outer membrane proteins, for example, HopQ, BabA, and its virulence marker, for example, cagA, vacA alleles. Treatment of AGS cells with Psyllium clearly decreases IL-8 expression in the presence and absence of H pylori coculture. It is beneficial as it prevents H pylori from eliciting a pro-inflammatory host cell response via the activation of NF-κB and mitogen-activated protein kinases. 26 Hence, Psyllium will keep immune cell function, including dendritic cells and T cells, from being modified by H pylori. The downregulation of pro-inflammatory responses mitigated by several mechanisms makes Psyllium useful for diverse indication. In conclusion, this study showed that Psyllium has an immune modifying effect on gastric epithelial cells. However, a randomized controlled study is required to establish the efficacy of Psyllium husk in the treatment of H pylori infection that can effect both basal and stimulated secretions of IL-8 by H pylori.
Footnotes
Acknowledgments
We are grateful to Ms Samra Javed for reviewing the text.
Author Contributions
JY conceived the idea; WJ, MHM, and ZA contributed to the development of the idea; JY, KT, and MHM did the work; JY wrote the manuscript; and MHM, WJ, ZA, and KT reviewed and approved the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Ethics review 2438-Med-12 committee and involved no human subjects.
