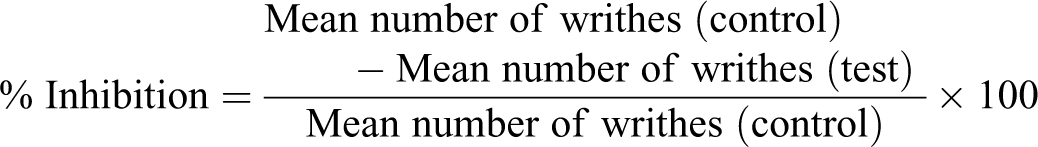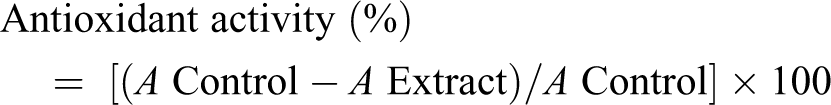Abstract
Asafetida is well known for its medicinal and therapeutic values in Iranian folk medicine. This study was conducted to investigate the antinociceptive and anti-inflammatory effects of asafetida. In hot plate test, asafetida exhibited a significant antinociceptive effect at all administered doses and the most effective dose was 10 mg/kg. The highest maximum potent effect was observed 15 minutes after asafetida administration. The antinociceptive effect of asafetida was not reversed by administration of any antagonist used in this study but asafetida showed a remarkable antioxidant and also inhibitory action against lipoxygenase activity. Paw weight was significantly reduced only in treated animals with 2.5 mg/kg asafetida. Results clearly indicate that the asafetida could be a potential source of anti-inflammatory and analgesic agent. These effects may be due to its effective constituents such as monoterpenes, flavonoids and phenolic components that have antioxidant properties and inhibit lipoxygenase activity.
Pain is a common situation that observed in different pathologic conditions such as ailments, wounds, and trauma. Treatment of pain and inflammation is one of the major challenges for these disorders. Since many patients suffering from chronic and subacute pain, management of pain is particularly challenging and need a multidisciplinary approach. The principal targets of effective pain control are to reduce pain threshold sensation and to improve quality of life. In recent years, people recognize and use the medicinal properties of many cultivated or wild plants to fight the disease. Plant drugs are frequently considered to be less toxic than synthetic ones. Medicinal plants are believed to be an important source of new chemical substances with potential therapeutic effects. 1 Plants of the genus Ferula belongs to the family of Apiaceae include about 130 species that distributed throughout central Asia and Mediterranean area. 2 Ferula assa-foetida L is one of these species that grows wildly in central area of Iran. The part used of this plant and several other species of Ferula is an oleo gum resin (asafetida) that obtained by incision of stem and root. 3 In Iranian folk medicine, asafetida is used as an antispasmodic, antihelminthic, carminative, and analgesic agent. 4 Recent pharmacological and biological studies have also shown several activities, such as antioxidant, 5 anticonvulsant, 6 cancer chemopreventive, 7 antiobesity, 8 antidiabetic, 9 antispasmodic, 10 atileishmania, 11 molluscicidal, 12 and antidementia 13 from this oleo gum resin. In some previous studies; possible antinociceptive effect of some Ferula species has been investigated in animal models. The antinociceptive and anti-inflammatory effects of Ferula gummosa was investigated by Mandegary et al. 14 They reported that the acetone extract of the root of Ferula gummosa could reduce pain in tail flick test but did not have any anti-inflammatory effects. In previous study, we reported the antinociceptive effect of asafetida in chronic and acute pain in mice. 15 There are also some reports regarding the antinociceptive, anti-inflammatory and antipyretic effects of some terpenoid compounds isolated from the plants of Ferula genus. 16,17 Phytochemistry of asafetida showed that gum fraction contains the glucuronic acid, galactose, arabinose, and rhamnose; its resin consists of umbelliferone, ferulic acid and its esters, coumarins, sesquiterpene coumarins and other terpenoids. 3 These evidences provided reliable reasons for investigation of the effects of asafetida on reduction of pain and inflammation. The purpose of this study is the investigating of antinociceptive and anti-inflammatory activity of Ferula assa-foetida oleo gum resin in mice. We also investigated possible mechanisms involved in antinociceptive effect of asafetida.
Materials and Methods
Animals
Male albino mice (25-30 g), 6 to 8 weeks old, bred in animal house of Medical School of Shahid Sadoghi University of medical sciences were selected. Animals were housed at controlled temperature (22°C ± 2°C) with a 12-hour light/dark cycle and with standard lab chow and tap water ad libitum. The experiments reported in this study were carried out in accordance with current ethical guidelines for the investigation of experimental pain in conscious animals. 18 The numbers of animals and intensities of noxious stimuli used were the minimum necessary to demonstrate the consistent effects of the drug treatments (n = 6).
Plant Oleo Gum Resin
Asafetida was collected from Tabas region (Yazd province, Iran) during the summer and its dried powder was dissolved in distilled water overnight at room temperature and the yielded suspension was used intraperitoneally. Concentrations and dosages of the extract were expressed as crude amount of the dried oleo gum resin used in preparing the stock solution.
Drugs Administration
Morphine sulfate (8 mg/kg) and sodium diclofenac (30 mg/kg) were used as positive control drugs. Intraperitoneal injection of naloxone (5 mg/kg), theophylline (5 mg/kg), yohimbine (5 mg/kg), methysergide (5 mg/kg), haloperidol (1 mg/kg), and gelibenclamaide (2 mg/kg) were used for evaluating the mechanism action of asafetida. Intradermal injection of carrageenin was used to induce paw edema.
Hot-Plate Test
The hot-plate test was carried out according to the method previously described.
19
Briefly, before the initiation of experiment, mice were habituated to a Plexiglas cylinder for 5 minutes. In these experiments, the hot-plate apparatus was maintained at 54°C ± 0.1°C. Before any intervention, each animal was placed into an acrylic cylinder (20 cm in diameter) on the heated surface, and the time (in seconds) between placement and licking of their hind paws or jumping (whichever occurred first), was recorded as the response latency (reaction time) and served as control latency for each mouse. A 45-second cutoff was used to prevent tissue damage. After baseline behavior tests, each animal was immediately administered with its assigned drug. The animals intraperitoneally received either vehicle (distilled water, 10 mL/kg) or asafetida dissolved in distilled water at 3 doses (2.5, 5, 10, and 20 mg/kg) and morphine sulfate 15 minutes before recording the test latencies. The reaction time of each mouse was again evaluated at 15, 30, 45, and 60 minutes after treatment as the test latencies. Control and test latencies for each mouse in each treatment group were used to determine the percentage of maximum potent effect (%MPE) by applying the following formula:
Acetic Acid–Induced Writhing Test
The abdominal constriction test described by Morucci et al
20
was used to measure the analgesic activity of asafetida. Male mice pretreated with asafetida (2.5, 5, 10, and 20 mg/kg) and diclofenac sodium (30 mg/kg) as positive control. Fifteen minutes later, all mice were treated with intraperitoneal injection of 0.6% acetic acid to cause a typical stretching response. Five minutes after acetic acid injection, mice were kept in individual cages and writhing or stretching of each mouse was counted for a period of 30 minutes by a blinded individual. The analgesic effect was measured by calculating the mean reduction in the number of abdominal constrictions for each drug as compared with saline control. Percentage inhibition of writhing was calculated by using the following formula:
Assessment of Some Possible Mechanisms Involved in Antinociceptive Activity
To investigate the possible mechanisms by which asafetida inhibits acetic acid–induced nociception, mice were pretreated with different drugs include naloxone (5 mg/kg intraperitoneally), theophylline (5 mg/kg intraperitoneally), yohimbine (5 mg/kg intraperitoneally), methysergide (5 mg/kg intraperitoneally), haloperidol (1 mg/kg intraperitoneally), and glibenclamide (2 mg/kg orally). 21 After 30 minutes, the animals received asafetida (10 mg/kg intraperitoneally) and 15 minutes later acetic acid was injected and the number of writhes was counted for 30 minutes.
Inhibition of the Carrageenin-Induced Edema
Edema was induced in the right hind paw of mice by intradermal injection of 0.1 mL of 2% carrageenin. Asafetida was dissolved in water and was given intraperitoneally 15 minutes before the carrageenin injection. The mice were euthanized 3.5 hours after carrageenin injection. The difference between the weight of the injected and uninjected paws was calculated for each animal. The change in paw weight was compared with that in control animals and expressed as the percent inhibition of the carrageenin-induced paw edema (%CPE). 22
Lipoxygenase Inhibition Assay
The soybean 15-lipoxygenase was used to test the 15-lipoxygenase inhibitory activity of asafetida. For this purpose, 50 mL of extract solution was added to test solution containing 3 mL of phosphate buffer (0.1 M, pH 8), 50 mL enzyme solution (final concentration 167 U/mL) to achieve the enzyme inhibition between 20% and 80%. After 4 minutes of incubation of test solution, the substrate (linoleic acid, final concentration 134 mM) was added and the change in absorbance was measured for 60 seconds at 234 nm. The IC50 (half maximal inhibitory concentration) value was calculated graphically using the slopes of absorbance curves. The enzyme solution was kept in ice and tested at intervals to ensure that the enzyme activity was constant. All experiments were performed by UV/Vis Unico Double Beam Spectrophotometer at 25°C in triplicate. 23
Antioxidant Activity Assay for DPPH Free Radical Scavenging Activity
The antioxidant activity of asafetida was evaluated spectrophotometrically following the 2,2-diphenyl-1-picrylhydrazyl (DPPH) method. Asafetida was evaluated at 100 mg/L, by mixing 0.75 mL with 1.5 mL of a freshly prepared DPPH solution (20 mg/L); then, sample was mixed thoroughly and kept in the dark for 30 minutes, at room temperature. After that, each mixture was tested for the DPPH radical–scavenging activity by reading the absorbance at 517 nm on a spectrophotometer. Blank solution was prepared by mixing 0.75 mL of ultrapure water with 1.5 mL of the DPPH solution (20 mg/L) and reading at the same wavelength. The antioxidant activity percentage was calculated using the following formula:
Data Analysis
All data are expressed as the mean ± standard error of the means. GraphPad Prism 5 was used for data analysis. Statistically significant differences were determined using 1-way analysis of variance with the Tukey Kramer posttest for multiple comparisons. The values of P < .05 were regarded as statistically significant.
Results
Hot-Plate Test
Latency responses for animals in different groups are shown in Table 1. The latencies for time 0 (baseline latency) were statistically analyzed by 1-way analysis of variance and there was no significant difference between the groups. Despite the similarity in baseline latencies in different groups, for more accuracy the %MPE against thermal stimulus was calculated for each animal in each time point after the treatment. The most effective dose of asafetida was 10 mg/kg and its maximum effect was observed 15 minutes after asafetida injection (Figure 1).
Hot Plate Latency Responses of Animals in Different Groups (n = 6).

a Indicates the significant difference (P < .05) between the latency times of each group and the baseline as control time, using 1-way analysis of variance followed by Tukey’s posttest.

Central antinociceptive activity of asafetida at different doses. %MPE was measured 15 minutes after asafetida injection. %MPE, % maximal potent effect. *P < .05 compared with control.
Acetic Acid–Induced Writhing Test and Assessment of Some Possible Antinociceptive Mechanisms
The effect of different drugs treatments on acetic acid induced writhing is presented in Figure 2. The number of writhes at dose 10 mg/kg of asafetida was significantly less than control group (P < .05). We also investigated some possible mechanisms involved in asafetida-induced antinociception. As shown in Figure 2, pretreatment of animals with the different antagonists include naloxone, glibenclamide, theophylline, yohimbine, methysergide, and haloperidol could not reverse the antinociceptive effect of asafetida.

Effect of intraperitoneal injection of naloxone, glibenclamide, theophylline, yohimbine, methysergide, and haloperidol on antinociceptive effect of asafetida 10 mg/kg. *P < .05 compared the number of writhes in different groups with control.
Effect of Asafetida on Carrageenan-Induced Mice Paw Edema
The result was expressed as percentage of inhibition of weight increase at the right hind paw in comparison to the uninjected left hind paw. In this test, asafetida showed a significant anti-inflammatory effect only at dose of 2.5 mg/kg (P < .05; Figure 3).
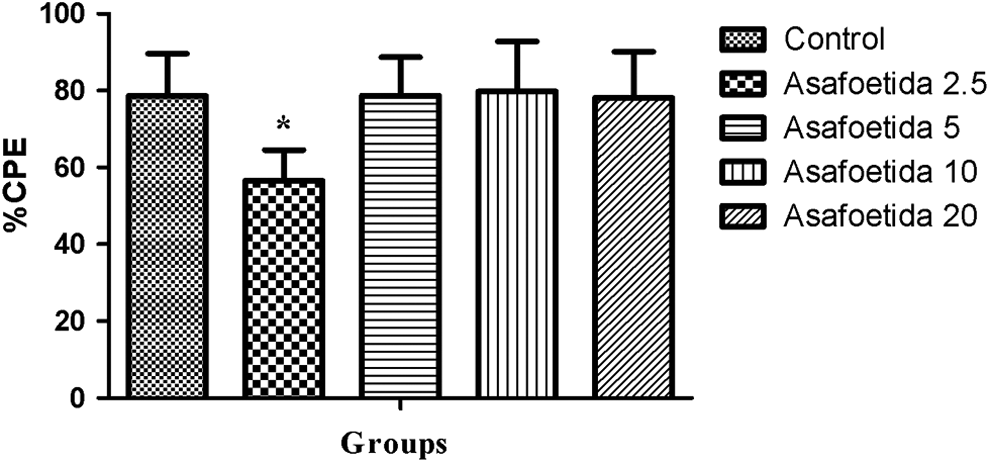
Effect of asafetida on inhibition of carrageenin paw edema (%CPE) in different groups. *P < .05 compared with control. % CPE, % carrageenin paw edema
Lipoxygenase Inhibitory and Radical Scavenging Activity
The lipoxygenase activity was measured as an increase in the absorbance at 234 nm, which reflects the formation of hydroperoxy linoleic acid. The IC50 for asafetida highest inhibitory effect obtained at 39 μg/mL. In this work, our results also showed that antioxidant activity of asafetida was 42% (Table 2).
Antioxidant and Lipoxygenase Inhibitory Activities of Asafetida.

Discussion
In this study, antinociceptive and anti-inflammatory effects of asafetida were evaluated in experimental models of pain and inflammation. The results showed that a dose of 10 mg/kg asafetida has the highest antinociceptive effect. Therefore, we also investigated the some possible mechanism of analgesic activity of asafetida by using several antagonists. Naloxone, glibenclamid, theophylline, yohimbine, methysergide, or haloperidol could not significantly affect the antinociceptive effect of asafetida. These results showed that asafetida did not act via the receptors antagonizing by these drugs. The fact that asafetida produced its analgesic action in both tonic and phasic nociceptive models is indicative that it may possess both central and peripheral antinociception. One possible mechanism of action for the active constituents of this oleo gum resin could be related to lipoxygenase and/or cyclooxygenase in the arachidonic acid cascade at the peripheral route. 25 Interestingly in this study, asafetida showed a remarkable inhibitory activity against lipoxygenase. According to these results, the IC50 of lipoxygenase activity of asafetida was 39 μg/mL. The antinociceptive activity of asafetida partly may be due to the phenolic compounds, such as ferulic acid, that are present in asafetida. 5 Several reports showed that some therapeutic effects of plant extract such as analgesic and anti-inflammatory exerted by presence of phenolic compounds.16,26,27 We also examined anti-inflammatory activity of asafetida using the carrageenan in mouse paw edema in which asafetida showed an anti-inflammation effect only at its minimum dose (2.5 mg/kg). The activity of asafetida against inflammation and edema is concordant with previous reports about anti-inflammatory potential of several Ferula species 6 or derivatives isolated from this genus. 28,29 This anti-inflammatory effect aroused from the results can possibly be due to the plants monoterpenes which showed anti-inflammatory effect in different plant species. 30 Umbelliprenin, which is one of the sesquiterpene coumarins of asafetida, can inhibit the activity of 5-lipoxygenase and shows the anti-inflammatory action. 22 Appendino et al 31 showed that sesquiterpene dienones include fetidones A and B and sesquiterpene coumarin ethers such as 8-acetoxy-5-hydroxyumbelliprenin have potent nuclear factor-κB–inhibiting properties. Free radicals are strongly involved in several pathological and physiological processes, including cancer, cell death, inflammation, and pain. 32,33 Ferulic acid, which is an important asafetida component, has a potent antioxidant activity 3 and may act as an analgesic and anti-inflammatory component of asafetida. In addition, flavonoid compounds with antioxidant and anti-inflammatory properties are among the fractions of Ferula assa foetida 34 and could be another candidate that acts as an analgesic and anti-inflammatory component of asafetida.
Conclusions
Asafetida induces potent anti-inflammatory and analgesic activity, probably via suppression of the peripheral inflammatory processes and/or through modulation of the nociceptive neural pathways in the central nervous system. The exact mechanism and the bioactive principles responsible for these actions remain to be explained. Further investigations are needed to help obtain more information about these items.
Footnotes
Acknowledgments
The authors thank all people who have assisted the experimental procedure.
Author Contributions
SMB: Corresponding and writing. STH: Research. AM: Research. MHD-R: Editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from Shahid Sadoghi University of Medical Sciences.
Ethical Approval
Ethical approval for this study was obtained from the Ethics Committee of the Shahid Sadoghi University of Medical Sciences (Approval number: 7244295).