Abstract
The current work assessed a pharmaceutical dosage form of Myrtus communis L. (myrtle) in reflux disease compared with omeprazol via a 6-week double-blind randomized controlled clinical trial. Forty-five participants were assigned randomly to 3 groups as A (myrtle berries freeze-dried aqueous extract, 1000 mg/d), B (omeprazol capsules, 20 mg/d), and C (A and B). The assessment at the beginning and the end of the study was done by using a standardized questionnaire of frequency scale for the symptoms of gastroesophageal reflux disease (FSSG). In all groups, both reflux and dyspeptic scores significantly decreased in comparison with the respective baselines. Concerning each group, significant changes were found in FSSG, dysmotility-like symptoms and acid reflux related scores. No significant differences were observed between all groups in final FSSG total scores (FSSG2). Further studies with more precise design and larger sample size may lead to a better outcome to suggest the preparation as an alternative intervention.
Gastroesophageal reflux disease (GERD) is known as a chronic and recurrent problem, which is defined via the regurgitation of the stomach contents into the esophagus. This disease may cause various anatomical and pathological complications. 1 Reports revealed that the disease prevalence in North America and Europe is up to 27.8% and 25.9%, respectively. 2 In Iran, GERD has shown an increase during recent decades. With reference to the reports, that prevalence rate of this disorder is estimated more than 20% among Iranian people. 3
The pathophysiology of this disorder is not well diagnosed. However, some mechanisms such as reduction of salivation, defect in esophageal clearance, decrease in lower esophageal sphincter pressure at rest, and increased acidity as well as transient lower esophageal sphincter relaxation are suggested. 4 Diagnostic methods are usually including esophageal pH-metry test, endoscopy, and barium esophagogram as well as inhibition of gastric acid secretion. 5
Treatment lines are defined as proton pump inhibitors and H2 receptor antagonists as well as prokinetic agents that are usually applied as adjuvant therapy. 5
In addition to conventional intervention, numerous approaches have been titled by complementary systems of medicine for such disorders. 6 As a complementary and integrative medicine, traditional Persian medicine offers many pharmacological and therapeutic approaches to the various disorders. 7 Traditional Persian medicine is a school of holistic medicine, which is not only a summation of previous experiments but also a collection of indigenous information and findings of early Persian practitioners in diagnosis, prevention, and elimination. 8 Outstanding scholars of Persian medicine have dedicated their fruitful medical and pharmaceutical wisdom to the medical knowledge during medieval era. 9 Large part of Persian medical manuscript encompasses gastrointestinal diseases. GERD is not a new disease, but a disease that has been defined since long time ago. This disorder is a collection of various unpleasant symptoms mainly complained as heartburn (mostly called as herqat-al me’deh in Persian medical manuscript). On the other hand, many medicinal herbs in various pharmaceutical dosage forms have been mentioned for gastrointestinal complications. 10,11 Among those, Myrtus communis L. (commonly known as myrtle) from the family Myrtaceae was reported as a potent medicinal herb in the management of these disorders. 12 The anti–Helicobacter pylori and anti-inflammatory activities of this herb in gastrointestinal disorders have been proved in previous studies. 13 In addition, this medicament is also introduced as a main part in a traditional medicine for herqat-al me’deh in Persian medical manuscripts. 10 Accordingly, the current work aimed to assess a relevant pharmaceutical dosage form of myrtle in reflux disease in comparison with omeprazol via a double-blind randomized controlled clinical trial.
Materials and Methods
Study Design
The current study was a 6-week, outpatient, and double-blind randomized controlled clinical trial in adult aged from 18 to 60 years. Patients with confirmed symptomatic GERD were enrolled to the study. The study consisted of a 2-week screening period before intervention followed by a 4-week treatment period. The follow-up procedure was performed by telephone contact and visits. Visits occurred 2 times, at the first and at the end of the intervention period.
The study was conducted according to the precepts of Declaration of Helsinki (Hong Kong revisions, 1983) as well as good clinical practice. It was approved by the institutional review board of Shiraz University of Medical Sciences (Code number: CT-90-5900). This clinical trial was also registered at Iranian Registry of Clinical Trials database (http://www.irct.ir) with a registration ID of IRCT2012072710410N1.
All patients signed and filled out the informed institutional review board–approved consent form before being involved in the evaluation.
Sample Size, Randomization, and Criteria
The participants were both women and men ranging in age from 18 through 60 years with symptomatic GERD. The disorder was confirmed by positive endoscopic or histologic evidence of reflux-related esophagitis within 2 weeks of screening. Patients with history or presence of clinically significant gastroesophageal reflux symptoms that were treated unsuccessfully for at least 1 year were enrolled. On the other hand, Patients were excluded from the study if there was a history or presence of clinically significant upper gastrointestinal anatomic or motor disorders (including strictures, webs, or diverticula), Barrett esophagus, peptic ulcer disease, or erosive gastroduodenitis. History of gastrointestinal surgery (except appendectomy and herniorrhaphy), suggested or confirmed malignancy, and known history of human immunodeficiency virus (HIV/AIDS) were also causes for exclusion, as were clinically significant abnormalities during the prestudy physical examinations (including electrocardiography), clinically significant medical or surgical disorders, significant laboratory abnormalities, and acute childhood illness. Pregnant or breastfeeding mothers and women at the childbearing period without any contraception were also excluded.
Patients treated with proton pump inhibitors within 28 days, using histamine-2 receptor antagonists, or prokinetic agents within 3 days, or participating in another investigational drug trial or experimental medical trial within 30 days before randomization were also excluded.
The sample size was determined according to a previous study. 14 A total of 45 patients were randomly enrolled into three groups via block randomization.
Intervention
A total of 45 participants were assigned randomly to 3 groups as A, B, and C. The first group (A) received myrtle berries extract in capsules (1000 mg once a day). The second group (B) received omeprazol (Dr Abidi Pharmaceuticals) capsules (20 mg once a day), and the last group (C) received myrtle fruit (1000 mg once a day) in addition to omeprazol (20 mg once a day) for 4 weeks.
Myrtle berries were purchased from a medicinal plants market in Rasht (north of Iran) and authenticated by a botanist at Department of Phytopharmaceuticals, School of Pharmacy, Shiraz University of Medical Sciences, Shiraz, Iran. A voucher number was assigned to the sample. Berries were then coarsely grinded and boiled (1:10) in distilled water for 3 to 5 minutes. The extract was subsequently concentrated using a rotary evaporator and freeze-dried at −50°C in a freeze dryer for 72 hours. The dried powder was weighed and filled in capsules. According to the traditional dose of administration, 2 capsules of dry extract (1000 mg/d) were considered.
At the beginning, patients were screened and visited by a gastroenterologist. Then they were referred them to PhD candidates of traditional Persian medicine who used a standardized questionnaire of Frequency Scale for the Symptoms of GERD (FSSG). The FSSG questionnaire consists of 12 questions which are compromised 7 acid reflux–related symptoms (RS: questions 1, 4, 6, 7, 9, 10, and 12) and 5 dysmotility-like symptoms (DS: questions 2, 3, 5, 8, and 11). The maximum scores of FSSG total score, RS and DS are 48, 28, and 20, respectively. In line with randomization, patients were then guided toward the traditional pharmacy in the same clinic to receive the respective interventions. At the end of the protocol, same questionnaire were completed by same physician.
Outcomes
The study was performed using the FSSG questionnaire. 15,16 Heart burn, bloated stomach, gastric heaviness after meals, chest rubbing, sick feeling after meals, heartburn after meals, burning sensation in throat, fullness while having meals, difficulty while swallowing, regurgitation into throat, burping a lot, and heartburn while bending were considered as primary outcomes.
Statistical Analysis
Using SPSS 16, results were derived and compared with the baseline data. Data were presented as mean ± standard deviation. With regard to the changes in FSSG parameters, analysis between groups was carried out by 1-way analysis of variance and least significant differences methods. Changes in FSSG within groups and associated symptoms were statistically analyzed by McNemar’s chi-square (binomial test) and paired-sample t tests.
Results
Out of 45 patients equally divided between 3 groups, 3 of them did not complete the protocol. Therefore, 42 patients with mean age of 35.26 ± 1.20 years finished the trial. The disorder was chronic and participants with GERD were suffering for 1 to 21 years (mean = 6.70 ± 0.88 years) (Figure 1). Additional demographic information is represented in Table 1.
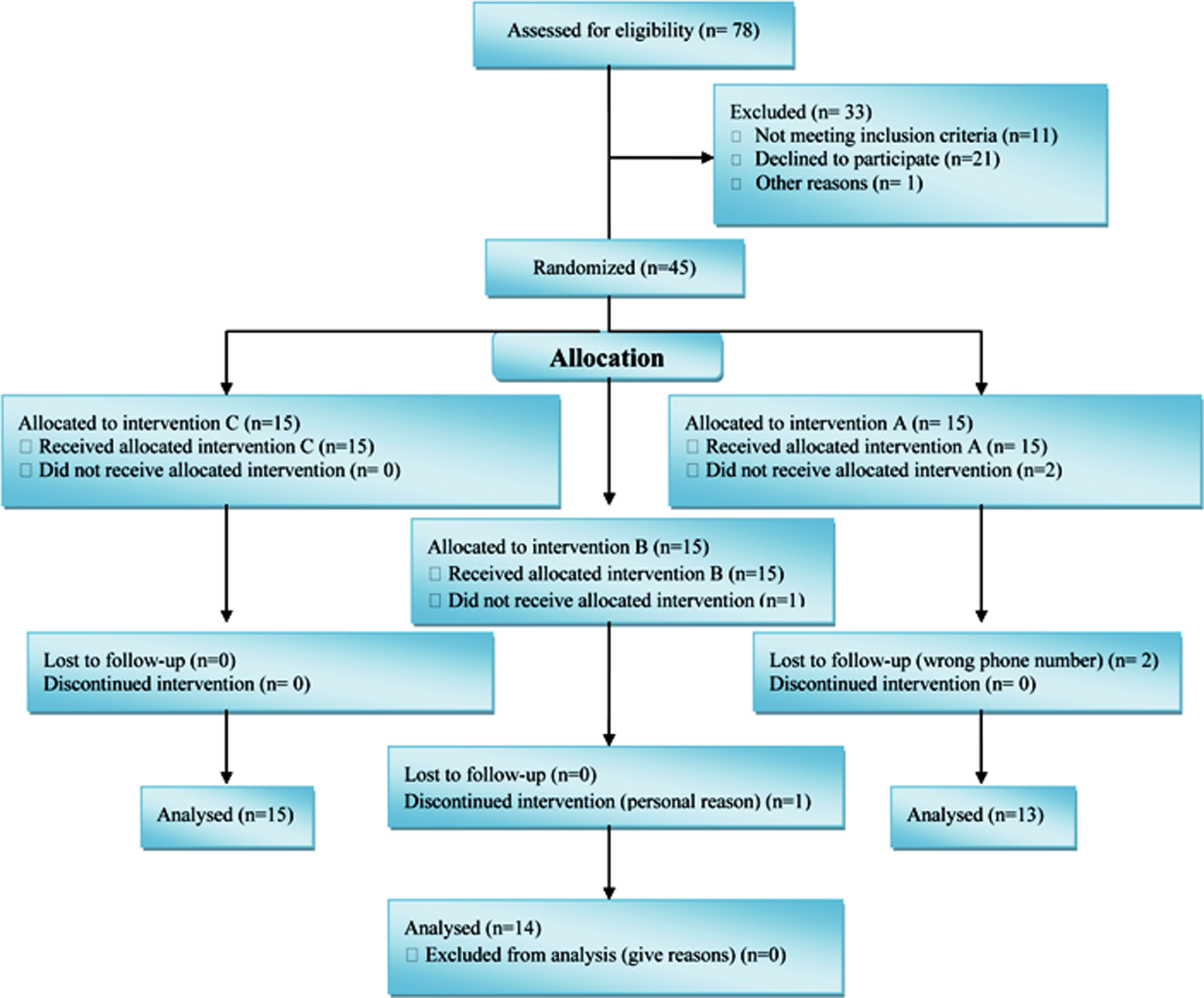
CONSORT (Consolidated Standards of Reporting Trials) flow diagram.
Demographic Information.

All patients represented typical symptoms of GERD and were referred by a gastroenterologist. There were no demographic differences between the studied groups. Also no significant differences were found in FSSG total scores baseline (FSSG1) between all groups (Table 2).
Data Related to the Pretreatment Scores of FSSG (FSSG1).

Abbreviation: FSSG, Frequency Scale for the Symptoms of Gastroesophageal Reflux Disease.
a P values ≤.05 were considered significant.
In all groups, both reflux and dyspeptic scores significantly decreased in comparison with the respective baselines. Concerning each group, significant changes were found in FSSG (P values of .002, <.001, and <.001 for groups A, B, and C, respectively; Figure 2), DS (P values of .016, <.001, and <.001 for groups A, B, and C, respectively; Figure 3), and RS (P values of .005, <.001, and <.001 for groups A, B, and C, respectively; Figure 4) total scores at the end of the protocol as compared with the baseline (Table 3). Moreover, at the end of the study, no significant differences in final FSSG total scores (FSSG2) were observed between all groups.

The Frequency Scale for the Symptoms of Gastroesophageal Reflux Disease (FSSG) total scores before (1) and after (2) the treatment within each group.
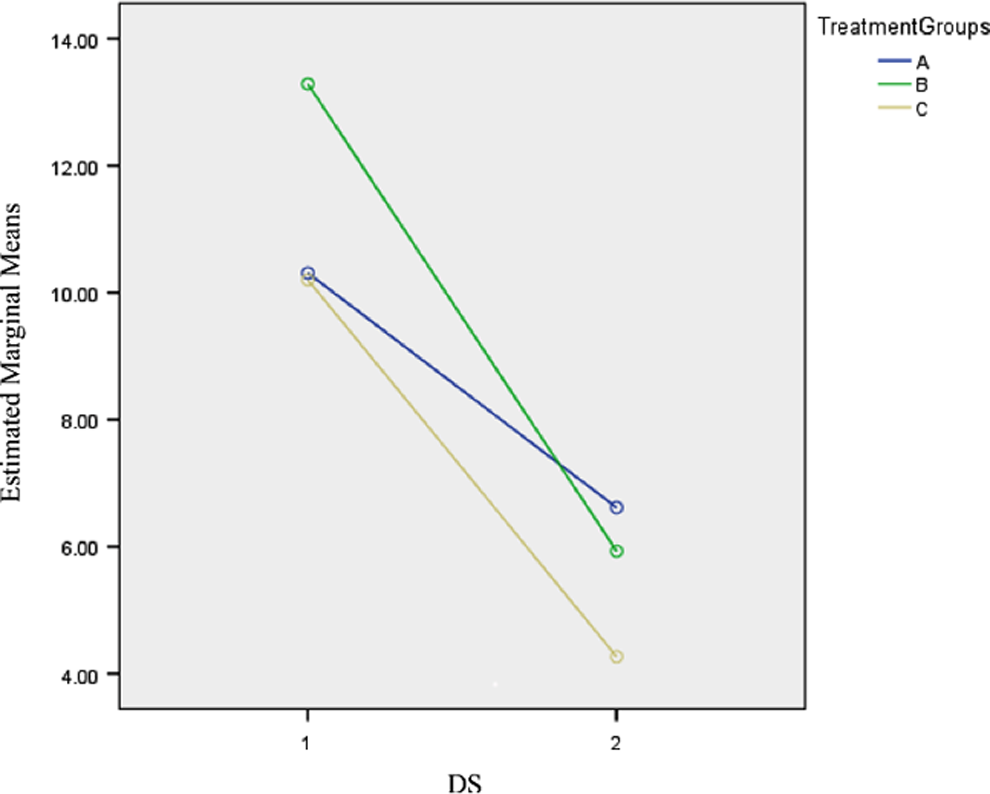
The dysmotility-like symptoms (DS) total scores before (1) and after (2) the treatment within each group.

The acid reflux–related symptoms (RS) total scores before (1) and after (2) the treatment within each group.
Pre- and Posttreatment FSSG, RS, and DS Total Scores (FSSG1, DS1, and RS1 as Pretreatment and FSSG2, DS2 and RS2 as Posttreatment Scores).

Abbreviations: FSSG, Frequency Scale for the Symptoms of Gastroesophageal Reflux Disease; DS, dysmotility-like symptoms; RS, acid reflux–related symptoms.
Discussion
Current study was a double-blind randomized controlled clinical trial assessing a monoingredient herbal preparation on reflux disease in comparison with omeprazol, as a conventional treatment. 17,18 It is well accepted that GERD is a chronic complication that is accompanied by typical or atypical clinical symptoms like chest pain, dysphagia, heartburn, hoarseness, and laryngitis. 19 Currently, proton pump inhibitors and H2 receptor antagonists are applied for the management of GERD. 20,21
Numerous clinical trials have been performed on the effectiveness of these medicaments in GERD. 22 A study on omeprazol (20 mg/d) versus omeprazol (20 mg/d) plus sustained release baclofen demonstrated that the combination may be more effective in the alleviation of heartburn and regurgitation in patients with GERD. 23 A randomized crossover study compared the effectiveness of rabeprazole with omeprazol in noncoronary chest pain diagnosed patients with GERD. Outcomes of that study revealed that the newer proton pump inhibitor could reduce the symptoms more quickly. 24
In addition to the drug-drug comparison, other studies have assessed the effectiveness of a certain operation versus proton pump inhibitors. In a randomized open, parallel-group trial, esomeprazole was evaluated against standardized laparoscopic antireflux surgery in patients with chronic GERD. Results showed that most patients in both groups have achieved and remained in remission at 5 years of evaluation. 25
Apart from the assessment of conventional treatment approaches, unconventional forms of treatment such as acupuncture, herbal medicines, and dietary interventions have also been taken into consideration. 26 Studies have shown that low-carbohydrate diets may improve the disorder symptoms. 27 Acupuncture against doubling the proton pump inhibitor dose has exerted more effective response in reducing the symptoms in GERD patients over a period of 4 weeks. 28 The adjuvant application of melatonin with common pump inhibitors has shown successfulness in resolving the symptoms in GERD patients. 29 In an animal study, treatment of 500, 250, and 125 mg/kg of Lonicera japonica Thunb extract could dose-dependently inhibit gross esophageal and gastric mucosa lesion with the underlying mechanism, antioxidant activity. 30 A multi-ingredient herbal preparation containing peppermint, chamomile, and iberis was symptomatically found more effective than placebo in patients with functional dyspepsia. 31 In an experimental study, flavonoids and α-tocopherol showed preventive effects against esophageal mucosal damage in rats with reflux esophagitis. The impact of these medicaments was found via antioxidant activity. 32 Against all these reports, an investigation revealed that small groups of GERD patients have had trend and attitude to use supplemental complementary and alternative medicine. 33
In our study, a 4-week intake of myrtle berries extract could significantly reduce reflux and dyspeptic scores as compared with the baseline. The extract was also found to be as effective as omeprazol at the end of the study. Myrtle berries are very rich in tannins and polyphenols. 34 The protective effects of 2 aqueous and 2 methanol extracts of myrtle berries were proved in rat gastric ulcer induced by ethanol, indomethacin, and pyloric ligation. Aqueous extracts reduced the ulcer index. On the other hand, gastric juice volume and total acidity were decreased by all doses both aqueous and methanol extracts, as compared with omeprazol. 35
Traditionally, it is believed that herbal medicaments with astringent properties have tonic impacts on the function and condition of the gastrointestinal system. 11 In line with this implication, this current study was designed and performed to evaluate the effectiveness of myrtle extract. According to the results, it can be concluded that presence of tannins and polyphenols in berries extract could be responsible for the studied effects. 36
Limitations
Although myrtle berries capsules were effective as compared to the respective baseline in group, no significant differences were found between those studied groups at the end of the protocol. This could be due to the smaller number of the participants and short duration of the study. In addition, this study was accompanied by no intervention-free follow-up. It is considerable that GERD is usually a chronic disease and discontinuing the intervention may lead to recurrence of the symptoms.
Conclusion
The current study attempted to comparatively evaluate a natural preparation with a conventional treatment. Despite the effectiveness of this medicament in the related studied group, no significant differences were observed in comparison to the control and group of combination. Further studies with more precise design and bigger sample size may lead to a better outcome to suggest the assessed herbal preparation as an alternative intervention.
Footnotes
Acknowledgments
The authors wish to express their appreciation to Dr Mehrdad Karimi, RCC department of Shiraz University of Medical Sciences as well as Prof. Peyman Jafari for their kind guidance during design and conduction of this investigation. They would also like to appreciate Shiraz University of Medical Sciences, Shiraz, Iran.
Author Contributions
The work presented here was carried out through collaboration between all authors. MEZ and RA defined the research theme and designed the methods and experiments. MMZ prepared the herbal formulation. MMZ, MEZ, MN and RA carried out the laboratory experiments. ARS analyzed the data. MEZ and MKH interpreted the results. MEZ, RA, and MMZ wrote the article. All authors have contributed to, reviewed, and confirmed the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Ethical Committee of Shiraz University of Medical Sciences, Shiraz, Iran (Code number: CT-90-5900).
