Abstract
The inhibition of the growth of Mycobacterium tuberculosis by the extracts of Andrographis paniculata has been studied using intracellular and axenic hypoxic conditions. The inhibition (confirmed using the gold standard colony forming unit assay) was found to increase with “double stimuli” or higher concentration of the extract. Organic solvent extracts were found to inhibit bacterial growth more than the aqueous extracts under microaerophilic conditions mimicked through axenic and intracellular assays. This could be further explored to evaluate the potential of the plant to be used against nonreplicating/dormant bacilli.
Introduction
Andrographis paniculata Nees. (Family Acanthaceae) has been widely used in traditional Asian medicines. Its application as an antibacterial, 1 antimalarial, 2 analgesic, 3 and antihepatoxic 4 agent has been documented. Additionally, the immune stimulatory properties 5,6 of Andrographis paniculata and its efficacy in treating gastrointestinal and upper respiratory tract infections have also been demonstrated. 7,8 The broad antimicrobial spectrum of Andrographis paniculata, its ability to clear uncomplicated respiratory tract infections, and its immune modulatory properties 9 makes it a potential candidate for testing against Mycobacterium tuberculosis.
Tuberculosis has a global presence in low-, middle-, and high-income countries. Despite extensive tuberculosis-control efforts on the part of the World Health Organization and local health departments, the tuberculosis epidemic continues to ravage the developing world, affecting susceptible individuals. 10 The problems in tuberculosis are further compounded by the rapid emergence of drug resistance, the burden of drug resistance being much higher in low-income countries like India and South Africa. 11
What further affects its incidence is the transmission of the bacterium from one person to other, creating secondary cases that may remain latently infected. These deep-seated latent tuberculosis bacilli are not presented to the immune system and their lower metabolic rate makes them poor drug targets. These bacteria, under conditions of immune suppression, may replicate giving rise to an active tuberculosis case. This suggests a state of homeostasis between the pathogen and host, which is used by the pathogen to its own advantage.
Use of immune stimulants that could help bacterial clearance by creating a homeostatic imbalance may be a plausible treatment option for tuberculosis. This approach, when combined with minimal side effects, enhances the utility of medicinal plants for antituberculosis therapy.
The aim of the current study was to assess the antimycobacterial potential of Andrographis paniculata. While most studies to date have focused on testing the antimycobacterial activity against standard laboratory strains using axenic assays, our study uses 3 drug-susceptible and resistant clinical strains in addition to H37Rv for testing. The assays include both intracellular and axenic hypoxic environments representing the different metabolic states of the pathogen to mimic different diseased states.
Material and Methods
Plant Material
Leaves of Andrographis paniculata (Herbarium No. BSI 131744) were procured from Shindewadi, Satara district, Maharashtra, India. The plant was selected based on its broad antibacterial properties along with immune modulatory characteristics. 9 The plants were collected and authenticated by Dr P. Tetali, Naoroji Godrej Centre for Plant Research. Voucher specimens of the plant were deposited at the Botanical Survey of India, Western Center, Pune, India. Postprocurement leaves were air dried and stored at 4°C to 8°C.
Extract Preparation
Coarsely powdered plant material was subjected to sequential extraction 12 using acetone, ethanol, and distilled water using the Soxhlet apparatus. A total of 300 mL of respective solvent was continuously refluxed with 25 g of plant material for a period of 24 to 30 hours for efficient extraction of the phytoconstituents. After ethanol extraction and evaporation of the solvent, the aqueous extract was prepared by boiling the plant material until the volume of water was reduced to 25%. The aqueous extract was lyophilized (Thermo Fisher Scientific, Waltham, MA) and the acetone and ethanol extracts were allowed to air dry. For the assays, the extracts were reconstituted at 20 mg/mL concentration in dimethyl sulfoxide, filtered through 0.2 μm, 25 mm dimethyl sulfoxide–resistant Acrodisc syringe filters (Pall Corporation, Port Washington, NY) and stored at −20°C for up to 2 weeks. The extraction efficiency measured in terms of the yield is represented in Table 1.
Yield of Aqueous, Acetone, and Ethanolic Extracts of Andrographis paniculata and the R f Values of Andrographolide as Obtained Using HPTLC Fingerprinting.

Abbreviation: HPTLC, high-performance thin layer chromatography.
Phytochemistry
The leaves of Andrographis paniculata are characterized by a number of active constituents, namely, diterpenoid lactones, flavonoids, and polyphenols. However, the therapeutic properties of the plant extract have been attributed to its prime constituent, Andrographolide. 13 The phytochemical fingerprint of the extract was thus ascertained through the presence of andrographolide (a marker compound) using high-performance thin layer chromatography.
Briefly, high-performance thin layer chromatography was carried out using chloroform–methanol (7:1) solvent system for the organic extracts. For the aqueous extracts the solvent system used was ethyl acetate–methanol–water–acetic acid (10:1.35:1:0.0.1). The extracts were spotted on precoated silica gel G60 F254 thin-layer chromatography plates (product of Merck, Darmstadt, Germany) along with the reference standard, that is, andrographolide (a generous gift from Professor K. S. Laddha, Institute of Chemical Technology, Mumbai, India) using Linomat V Automatic Sample Spotter (CAMAG, Muttenz, Switzerland), run in a “twin trough thin-layer chromatography chamber,” dried and visualized in “CAMAG TLC visualizer” before and after derivatization with anisaldehyde–sulfuric acid.
Bacterial Strains
Standard laboratory strain H37Rv and 6 clinical strains (3 drug susceptible and 3 drug resistant) were chosen for the assay. Drug susceptibility was assessed against all first-line drugs, isoniazid, rifampicin, ethambutol, and pyrazinamide, using the Buddemeyers and mycobacterial growth indicator tube 960 assay. 14 The strain characteristics are described in Table 2.
Characteristics of the Strains Selected for Testing the Intracellular Efficacy of Medicinal Plants.

Abbreviations: HERZ, H–isoniazid, E–Ethambutol, R–Rifampicin, Z–Pyrazinamide; Cs, drug susceptible strain; Cr, drug resistant strain.
aPhenotypic drug resistance was ascertained using MGIT TB 960 assay.
bGenotypic drug resistance was ascertained using MTB DR plus genotype HAINS assay kit.
Intracellular Assays
The human lung carcinoma epithelial cell line A549 (NCCS, Pune, India) was used for the assay. Cells were grown in Dulbecco’s modified Eagle medium supplemented with 10% fetal calf serum and 4 μg/mL of gentamycin. In a 96-well plate the A549 cells (106/well) were infected with Mycobacterium tuberculosis at a multiplicity of infection of 1:1 for 6 hours. Six hours post infection, extracellular bacteria were washed and A549 cells were treated with amikacin (50 μg/mL) for 2 hours to kill the remaining extracellular bacteria. After amikacin treatment, the Mycobacterium tuberculosis–infected cells were incubated overnight at 37°C with the plant extracts at a concentration of 25 μg/ml. On the 0, 3rd, 5th, 7th, and 10th day postinfection, the cells were washed and lysed with 0.1% sodium dodecyl sulfate for 7 minutes to release the intracellular bacteria. The lysate was 3-fold serially diluted with phosphate-buffered saline and 10 μL of the highest 2 dilutions was spotted onto Middle Brook 7H11 (MB7H11) agar plates supplemented with Oleic Albumin Dextrose Catalase (Becton Dickinson, Franklin Lakes, NJ) and 0.5% glycerol. The plates were incubated at 37°C for 3 weeks and the colony forming units were enumerated. The activity of the plant extracts was then interpreted. The inhibition of bacterial growth was represented as percent inhibition calculated using the following formula:

Double Stimuli/Higher Concentration
Intracellular bactericidial activity of the plant extracts on Mycobacterium tuberculosis strains was also tested at a higher dose (100 μg/mL) and double stimulus (25 μg/mL) repeated on day 5 (96 hours post infection). The rest of the experimental setup remained unchanged for the assessment. The activity of the plant extracts was interpreted as percent inhibition of growth.
Axenic Assays
Ten milliliters per tube of Middle Brook 7H9 broth supplemented with ADC and 0.5% glycerol was aliquoted and H37Rv suspension containing 104 CFU/mL was inoculated into the tubes. The plant extracts at a concentration of 100 μg/ml were added to the respective tubes. Positive (viable Mycobacterium tuberculosis) and medium controls (MB7H9) along with a rifampicin (1 μg/mL) control were maintained. The aforementioned setup in triplicate was subjected to differentially reducing oxygen concentration, viz, aerobic, microaerophilic, and anaerobic conditions. The microaerophilic conditions were obtained using the candle jar method. 15 The anaerobic conditions were achieved in an anaerobic jar with a gas pack (Hi Media, Mumbai, India) and confirmed using the indicator tablets provided by the manufacturer, which changed color from pink to purple under anaerobic conditions. The sets were incubated for a period of 10 days at 37°C to ensure that the bacterium has completed sufficient number of replication cycles at the end of the incubation period. After incubation each of the tubes were vortex mixed, serially diluted 10-fold, and 10 μL of this dilution was spotted on MB7H11 agar plate supplemented with Oleic Albumin Dextrose Catalase and 0.5% glycerol. The experiment was performed thrice to ensure reproducibility of results. The activity of the plant extracts was interpreted as percent inhibition of growth in comparison to the control group in the absence of plant extract under identical growth conditions.
Statistical Analysis
Intracellular Assays
Significant inhibition was considered as ≥80%, as it was the maximum inhibition recorded for H37Rv at the minimum concentration (25 μg/mL) for each of the 3 plant extracts.
Axenic Assays
Difference between the means of inhibition of bacterial growth were analyzed using a paired t test with 95% confidence interval.
Results
The 3 plant extracts were preliminarily screened using the intracellular assays at a single concentration (25 μg/mL). The efficacious extracts were further screened to assess enhancement in their bactericidal ability by either increasing the concentration of the extract (100 μg/mL) in a single dose or providing a double stimulus at the same concentration (25 μg/mL) at a later time interval. The extracts showing activity under intracellular conditions were selected for further screening of its efficacy axenically under reducing oxygen concentration.
Phytochemistry
The R f value of the 3 extracts and the reference standard andrographolide are presented in Table 1. The chromatographic profile visualized at 254 nm for aqueous extract and under visible light after derivatization with anisaldehyde–sulfuric acid for the organic extracts is depicted in Figure 1. As seen from the figure, andrographolide was found to be present in all the 3 extracts.

High-performance thin layer chromatography profile of Andrographis paniculata.
Intracellular Assay
It was found that the aqueous extracts were noninhibitory against both standard and clinical strains. Although the acetone extract was found to be inactive against H37Rv, significant activity was observed against 2 drug susceptible and 2 resistant clinical strains. It was found that higher concentrations of the acetone extract were significantly active against all tested bacterial strains (standard and clinical). Additionally, double stimulus at 25 μg/mL also seemed to have enhanced bactericidal activity against all tested strains in comparison to a single dose at the same concentration.
The ethanolic extract was found to be active against H37Rv and 2/3 drug susceptible and 1/3 drug resistant clinical strains. The activity was enhanced at the higher concentration (100 μg/mL) and on providing a double stimulus (25 μg/mL). The extract was found to be active against 2/3 drug resistant strains using both the approaches. Both the extracts were less efficacious against the same drug resistant strain (Cr3; Table 3).
Percent Inhibition of Mycobacterial (Standard and Clinical) Growth Under Intracellular Condition Using a CFU Assay Method.a
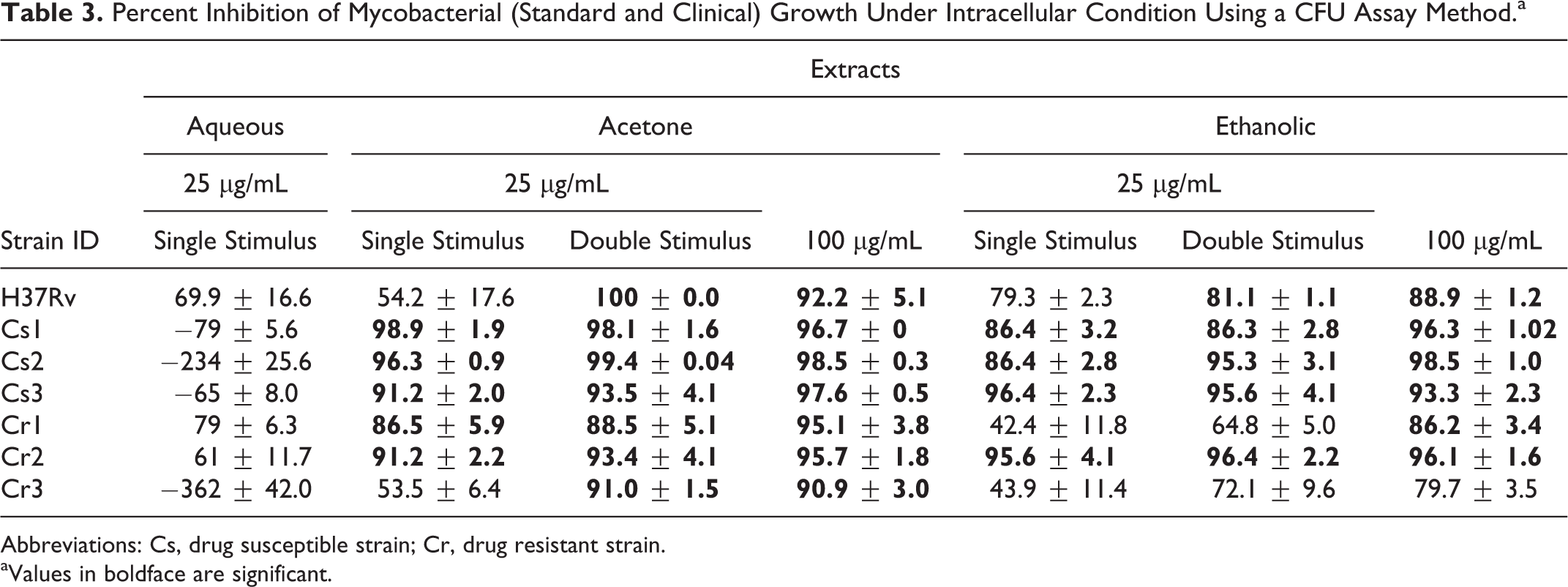
Abbreviations: Cs, drug susceptible strain; Cr, drug resistant strain.
aValues in boldface are significant.
Axenic Assay
The activity of the plant extracts was tested under reduced oxygen concentrations to mimic conditions for studying the effect of plant extracts on latent bacteria and to dissect the intracellular environment. Hence, the assay was restricted to H37Rv only. The active acetone and ethanolic extracts were tested at 100 μg/mL for their activity under reducing oxygen concentrations in axenic conditions. It was found that neither acetone (P = .3) nor ethanolic extracts (P = .28) had significant antibacterial activity under aerobic conditions, which is concordant with the MABA results reported earlier. 16 The ethanolic extract was found to be significantly active (P = .0008) under microaerophilic conditions only. Though not statistically significant, the acetone extract demonstrated partial activity (P = .07) under microaerophilic conditions. However, under anaerobic conditions, the inhibition was minimal (P = .13; Figure 2).
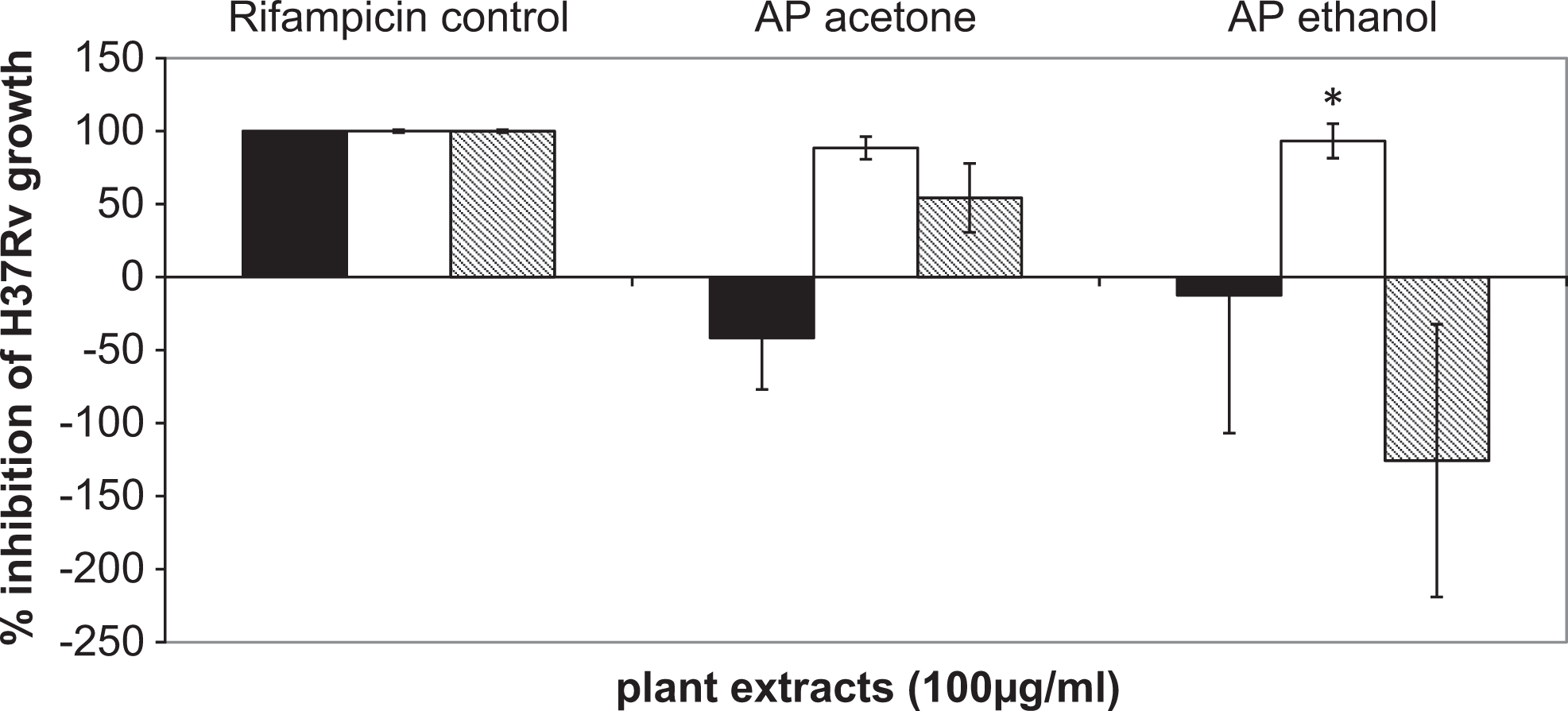
Testing of plant extracts on H37Rv under reducing oxygen conditions.
Discussion
Andrographis paniculata is known to possess broad-spectrum pharmacological activities. The antimycobacterial efficacy of ethanolic extract of leaves of Andrographis paniculata has been reported. 17 Andrographolide, an active constituent of the plant, is known to be active against pulmonary tuberculosis, tuberculous meningitis, and pyelonephritis. 7 A randomized double blind placebo controlled clinical evaluation of extract of Andrographis paniculata (KalmCold) reported its efficacy in patients with uncomplicated upper respiratory tract infection. 18
The plant is believed to boost immune system functions, that is, production of white blood cells, release of interferon, and so on, 19,20 have demonstrated that incubation of monocytes-derived macrophages of healthy participants with the extract of Andrographis paniculata and propolis (adjuvant) activated bactericidal activity against intracellular Mycobacterium tuberculosis. Ethanolic extract of Andrographis paniculata is known to enhance both antigen-specific and nonspecific responses. 21 Thus in the present study, the activity observed under intracellular conditions could be due to the immune stimulatory potential of the plant. The extracts also could act on the bacterium residing intracellularly. This could be dissected further for the potential of the plant to serve as an adjunct to conventional therapy for latent tuberculosis.
Andrographis paniculata is rich in terpenoids. 8 A study by Mishra et al 22 has reported the antibacterial activity of terpenoids extracted in the methanolic extract of Andrographis paniculata leaves against gram positive bacteria including Mycobacterium tuberculosis. Terpenoids form an integral component of essential oils. An important characteristic of essential oils and their components is their hydrophobicity, which enables them to partition in the lipids of the bacterial cell membrane and mitochondria, disturbing the structures and rendering them more permeable. 23 Additionally, it has been suggested that low oxygen levels can enhance the activity of essential oils. 23 Thus, the significant activity of ethanolic extracts observed under microaerophilic conditions could be accredited to the better penetrance of the extract.
The need of alternative therapeutic approaches for tuberculosis treatment has been outlined earlier. 24 New therapeutics under development aim to reduce emergence of drug resistance, shorten treatment regimens, and approach different bacterial targets. This study demonstrates the antimycobacterial activity of Andrographis paniculata observed under axenic microaerophilic conditions and in the intracellular assay system. This suggests the potential of using the Andrographis paniculata extracts in treatment of latent tuberculosis. Our study also highlights the use of multiple ways to test for efficacy of a product. While axenic assays are important for a preliminary screen, methods mimicking the in vivo environment should be tested to assess the potential of the product under consideration. Additionally, the study also demonstrates that standard strains that are acclimatized to laboratory conditions may not represent the complete antimicrobial profile of a natural product. Testing against clinical drug susceptible and resistant strains may be indicative of the utility of the plant in the evolving drug resistance scenario.
Footnotes
Authors’ Note
The funding agency had no role in the study design; in the collection, analysis, and interpretation of data; in the writing of the article; and in the decision to submit the article for publication.
Acknowledgments
Contribution of Mr Dipen Desai for the intracellular assays is acknowledged. We also thank Anchrom Enterprises (India) Pvt Ltd for assistance in HPTLC analysis.
Author Contributions
PB was involved in designing the experiments, conducting them, analyzing them, and writing the article. PG was involved in designing the experiments, conducting them, analyzing them, and writing the article. PD was involved in the phytochemistry of the plant. PT was responsible for collection and authentication of the plant material. TB was involved in designing the assays and editing the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: We gratefully acknowledge the support of Department of Biotechnology, Government of India, through Grant No. BT/01/COE/05/06/01.
Ethical Approval
Approval for this study was obtained from the ethics committee of Foundation for Medical Research (July 20, 2001).
