Abstract
This study examined the role of expectancy in the placebo effect of a sham dietary supplement for weight loss in 114 obese adults with metabolic syndrome. All participants received lifestyle education and were randomized to 1 of 3 conditions: (1) a daily placebo capsule and told that they were taking an active weight loss supplement, (2) daily placebo and told they had a 50% random chance of receiving either the active or placebo, or (3) no capsules. At 12 weeks, weight loss and metabolic outcomes were similar among the 3 groups. Participants in both groups that took capsules showed decreased weight loss self-efficacy and increased expectations of benefit from dietary supplements. Participants not taking capsules showed the opposite. Adverse events were more frequently reported in groups taking capsules than those who were not. These findings suggest that supplements without weight loss effects may have nocebo effects through diminished self-efficacy.
Introduction
A significant proportion of the benefit from drugs and medical procedures has been attributed to the placebo effect. 1 Placebos have been reported to improve both subjective and objective outcomes in a broad range of conditions. 2,3 A few studies have reported on placebo effects in pharmacologic therapy for obesity 4,5 and cardiovascular diseases. 6,7 However, mechanisms of placebo effects have not been fully elucidated. If present, placebo effects may be due to altered expectancies regarding treatment efficacy, altered behavior leading to improved lifestyle, or, more directly, altered physiology as a result of exposure to the placebo. 1,8 –10 Expectancy of benefit can be altered by information about a medication during a trial, and has been shown to influence outcomes. 11 –13 Expectancy can also lead to negative (or nocebo) effects of inactive treatment 14 resulting in real treatments having diminished effects. 15,16
Expectancies may be separated into 2 types: outcome and response. Outcome expectancy is the opinion that the treatment will lead to a particular outcome. 17 Response expectancy refers to a subject’s own response to the intervention. Better response expectancy suggests higher self-efficacy. Perceived self-efficacy has been shown to predict a number of health behaviors, including regular exercise and weight loss.18(pp259-318),19 With regard to weight loss, self-efficacy refers to a person’s belief about being able to resist temptations (ie, eat less and make better food choices) and exercise more. 20 Bernier and Avard 21 reported that pretreatment self-efficacy assessed with a 10-item scale was significantly related to maintenance of weight loss at 6 weeks’ follow-up. Despite these findings, the contribution of expectancy in the placebo response with regard to domains and magnitude of effects on weight loss or metabolic outcomes remains unclear.
The aim of the present study was to examine the size, character, and mechanisms of the placebo effect of a dietary supplement for weight loss in an at-risk population. Obesity and metabolic syndrome feature behavioral components and both conditions have objective physiological outcomes. 22 –24 Expectancy was primarily modeled by the perceived probability of having received an active agent, with expectancies about weight loss agents and perceived self-efficacy as contributing factors.
Methods
Participants
A total of 834 respondents for a weight loss study were solicited from the Portland, Oregon area and screened by telephone. In all, 243 were assessed in a qualifying visit. One hundred fourteen met the study criteria: aged 21 to 60 years old, obese (body mass index ≥30 and ≤45 kg/m2) with metabolic syndrome. Metabolic syndrome was defined as having ≥3 of 5 National Cholesterol Education Program risk criteria of blood pressure ≥135 mm Hg systolic or ≥85 mm Hg diastolic blood pressure, fasting blood glucose ≥100 mg/dL, triglycerides ≥150 mg/dL, high-density lipoprotein cholesterol <40 mg/dL for men or <50 mg/dL for women, and waist circumference >35 inches for women or >40 inches for men. 22 Exclusion criteria included the following: diabetes, congestive heart failure, a history of stroke, coronary artery disease, cancer, psychiatric hospitalization within the past year, use of weight loss medications currently or within the previous 6 months, a body weight change of greater than 5% within 6 months prior to study entry, current weight <90% of their lifetime maximum, more than 21 alcoholic drinks per week, current or planned pregnancy or breast-feeding, or currently exercising more than 30 minutes 3 times a week. Concomitant medications needed to be stable for the duration of the 12-week trial and for at least 3 months prior to study initiation. This study was approved by the institutional review board at Oregon Health & Science University and all participants gave signed consent before enrollment.
Design
Participants attended a baseline visit within a month after qualifying and returned for an outcome visit 12 weeks later. All study participants were told that they were being recruited for a clinical trial of a natural product plus lifestyle education for weight loss. Consent forms presented the purpose of the study as being “to see if the effect of medication for these conditions changes when one’s expectancy of receiving the medication changes.” Participants were informed of the potential of receiving placebo, but the true purpose of the study—that only expectancy was being evaluated and that all supplements were placebo—was withheld. All participants received education and support for diet and exercise and were computer-randomized in blocks of 3 to 1 of 3 groups. One group was told they would be given an active weight loss supplement (100% expectancy of receiving the active supplement). Those in the second group were told they were in a double-blind, placebo-controlled trial wherein they would be randomly assigned to either active or placebo supplement (50% expectancy of an active supplement). Those in the third group received lifestyle education alone without capsules (0% expectancy of an active supplement). As part of the placebo capsules intervention, an unproven rationale for a nonexistent but defensible nutraceutical combination (green tea extract and phosphatidyl serine) was modeled in purpose and formulation on some currently popular supplements for weight loss. In this rationale, participants were told that the supplement would enhance weight loss by moderating cortisol secretion, increasing fat “burning and energy,” and reducing cravings, thereby making it easier to adhere to diet and exercise recommendations. The supplement was, in fact, 600 mg of brown-tinted rice-flour in clear pullulan (vegetarian polysaccharide) capsules (Oregon’s Wild Harvest, Sandy, OR). Participants were instructed to take 1 capsule 3 times a day before meals. The project coordinator, who was responsible for randomization, dispensing, and compliance verification by pill count, was not blinded to group assignment. The weight loss counselor assessed pill compliance and adverse events during weekly phone calls, was aware of which subjects were taking capsules and which were not, but was blind as to capsule takers’ group assignment.
Weight Loss Counseling and Support Program
Educational group sessions were led by a trained counselor during weeks 2 and 6 of the 12-week study period. Diet quality and appropriate portion size were emphasized. During week one, subjects were given an analysis of their dietary intake from data obtained by a 3-day food diary (analyzed by the ESHA Research Food Processor, Salem, OR) in relation to healthy intake for their weight, height, and age. Subjects were given a goal for daily caloric intake that would allow them to lose approximately 1 pound per week and were instructed to keep food diaries 3 days a week. They were provided with pedometers to record daily steps taken. They were directed to increase their baseline steps by a modest 50%. The weight loss counselor called participants weekly during the 12 weeks to encourage adherence, respond to participant concerns, and evaluate for adverse events.
Measures
At baseline and follow-up visits, all subjects underwent anthropometric (weight, body mass index, waist circumference), psychometric (self-efficacy, personality factors, anxiety, depression, stress, expectations of dietary supplement efficacy, motivation to lose weight), and physiological (blood pressure, metabolic syndrome score, fasting blood glucose, glycated hemoglobin [HbA1c], lipids) measures. Nursing staff blinded to treatment assignment performed anthropometric and physiological measures, except waist circumference which was assessed by the study coordinator. Adherence to the interventions were assessed by pill count, change in reported caloric intake from baseline to outcome based on diet records, and a diary of daily pedometer steps.
Psychometric assessments were completed by participants. The primary physiological outcome of interest was change in body mass index between baseline and week 12. Adverse events were recorded during study visits and weekly phone calls. Three expectancy outcomes were measured: (1) probability of getting active agent (primary experimental condition), (2) self-efficacy, and (3) beliefs about the efficacy of dietary supplements. Self-efficacy and beliefs of benefit from dietary supplements were considered expectancies. Four validated self-efficacy instruments were used: the General Perceived Self-Efficacy Scale, 25 the Eating Self-Efficacy Scale, 26 the Weight Efficacy Life-Style Questionnaire, 27 and the Exercise Self-Efficacy Scale. 28 Motivation, considered a modifying factor, was assessed by questions regarding how much participants believe their behavior affects their risk related to metabolic syndrome and obesity, how “worth it” it is for them to make changes, and how much weight they would like to lose by the end of the study and to reach their ideal weight.
Following completion of the 12-week study visit, groups taking capsules were asked whether they believed they were taking a real active or a placebo, then debriefed as to the true purpose of the study, and given the opportunities for counseling and to withdraw their data from analysis in compliance with standards for deception in research. 29,30
Statistical Analysis
Attrition by group was tested using a 2 × 3 χ2 analysis. Mixed analysis of variance using a 3 (between: group assignment) by 2 (within: baseline vs outcome) design was used to test differences in weight loss outcomes by group, with the time × group interaction representing between-group differences in changes due to treatment. The 4 self-efficacy measures were analyzed using a 3 (between: group assignment) by 2 (within: baseline vs outcome) mixed multivariate analysis of variance.
All P values presented are unadjusted; outcomes beyond the primary measures of weight loss are considered exploratory. In order to assess simple differences between the most extreme groups (0% and 100% expectancy) and between groups who did or did not receive supplements (0% group vs combined 50% + 100% groups), post hoc comparisons between these groups were performed for the weight loss and self-efficacy outcomes.
Results
Of 114 participants enrolled, there were 35 (30.7%) dropouts (Figure 1). Baseline characteristics of participants are presented in Table 1. Seventy women and 9 men completed the study; their ages ranging from 32 to 60 years (mean 49.4 years, standard deviation 7.6). Twenty-five women and 2 men completed in the 100% expectancy group, 27 women and 1 man completed in the 50% expectancy group, and 18 women and 6 men completed in the 0% expectancy group. Last-observation-carried-forward imputation was performed for all participants who had baseline data, yielding a total of 106 participants for an intention-to-treat analysis. One participant who had extreme recorded weight loss of more than 65 lbs (more than 3 standard deviations from the group mean) during the study period was dropped from analyses of affected changes in weight and body mass index. A 2 × 3 χ2 analysis for attrition by group was not statistically significant, χ2(2) = 1.07, P = .585.
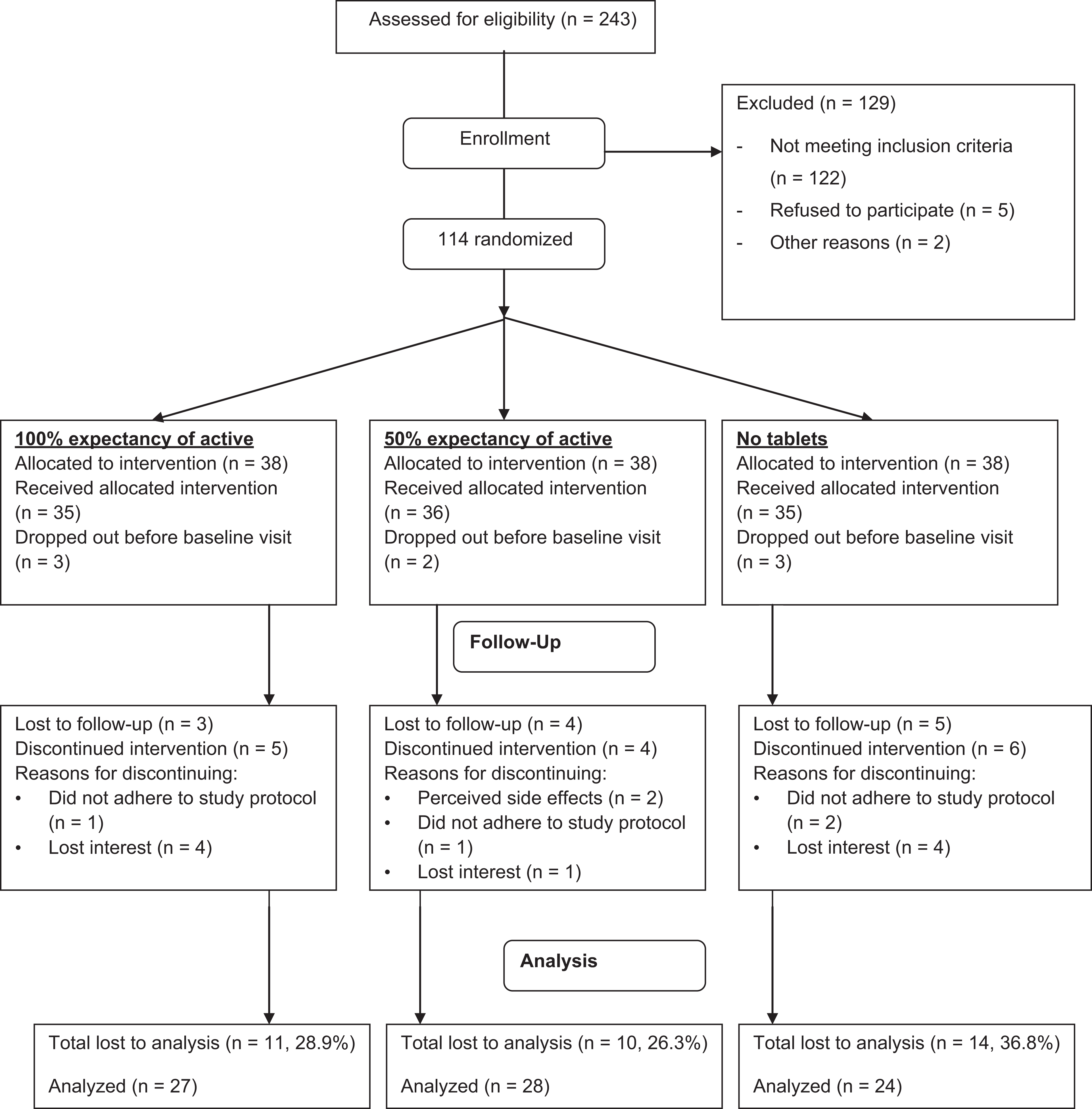
Flow sheet of study design.
Baseline Characteristics of Participants.

Weight Loss, Waist Circumference, and Blood Pressure
The mean ± standard deviation weight change for all groups was −1.48 ± 2.46 kg (range −9.2 to 3.6 kg) from baseline to 12 weeks. Mean ± standard deviation proportional reduction in body mass index was 2.4% ± 9.0% (Table 2). Mixed analysis of variance showed no significant differences between the study groups in either weight loss, F(2, 102) = 0.584, P = .56, or proportional reduction in body mass index, F(2, 102) = .539; P = .59. Post hoc comparisons between the 0% and 100% groups remained insignificant. In addition to performing the primary analysis using data imputed by last observation carried forward, we also checked sensitivity of the analyses of variance by reanalyzing with missing values imputed by mixed modeling and using only those participants with no missing follow-up data. The results are highly similar across the 3 methods, with only minor variations in significance levels.
Baseline and Change in Body Mass Index and Waist Circumference by Group.a

a Results are given as mean (standard deviation).
b Outlier removed from body mass index calculation.
A small change in weight (mean ± standard deviation −.20 ± 1.64 kg for all participants) occurred between screening and baseline, was not different between study groups, F(2, 103) = .039; P = .96, and did not affect the results of the weight loss primary outcome (P = .93 for difference in weight loss between groups, after adjusting for run-in weight change).
Chemistries
Changes in glucose, HbA1c, total cholesterol, triglycerides, low-density lipoprotein, and high-density lipoprotein levels are presented in Table 3. Comparisons adjusted for baseline values showed no significant differences in these outcomes between groups.
Measures of Glucose and Lipid Metabolism in Groups.a

a Results are given as mean change (standard deviation) from baseline to outcome visit.
Self-Efficacy
We hypothesized that placebo-taking groups (50% or 100% expectancy) would exhibit increases in all 4 self-efficacy measures (general, eating, exercise, and weight self-efficacy) and, secondarily, that baseline self-efficacy would be associated with body mass index proportion lost. Mixed multivariate analysis of variance failed to show a significant group by time interaction for any of the self-efficacy measures, however, indicating that there is not sufficient evidence for changes in self-efficacy due to group assignment (general self-efficacy, P = .28; eating self-efficacy, P = .33; exercise self-efficacy, P = .12; weight self-efficacy, P = .10). There were larger differences between groups for exercise and weight self-efficacy (Figures 2 and 3). Planned post hoc comparisons between the 0% and 100% groups showed marginally significant differences in changes in exercise self-efficacy (mean difference = −.66; P = .05) and weight self-efficacy (mean difference = −.657; P = .04). Comparisons between the 0% expectancy group (no supplement) and the pooled 50% and 100% groups, who were given a supplement, showed similar differences in change of exercise self-efficacy (mean difference = −.61; P = .03) and weight self-efficacy (mean difference = −.45; P = .09).

Changes in exercise efficacy over time by group.
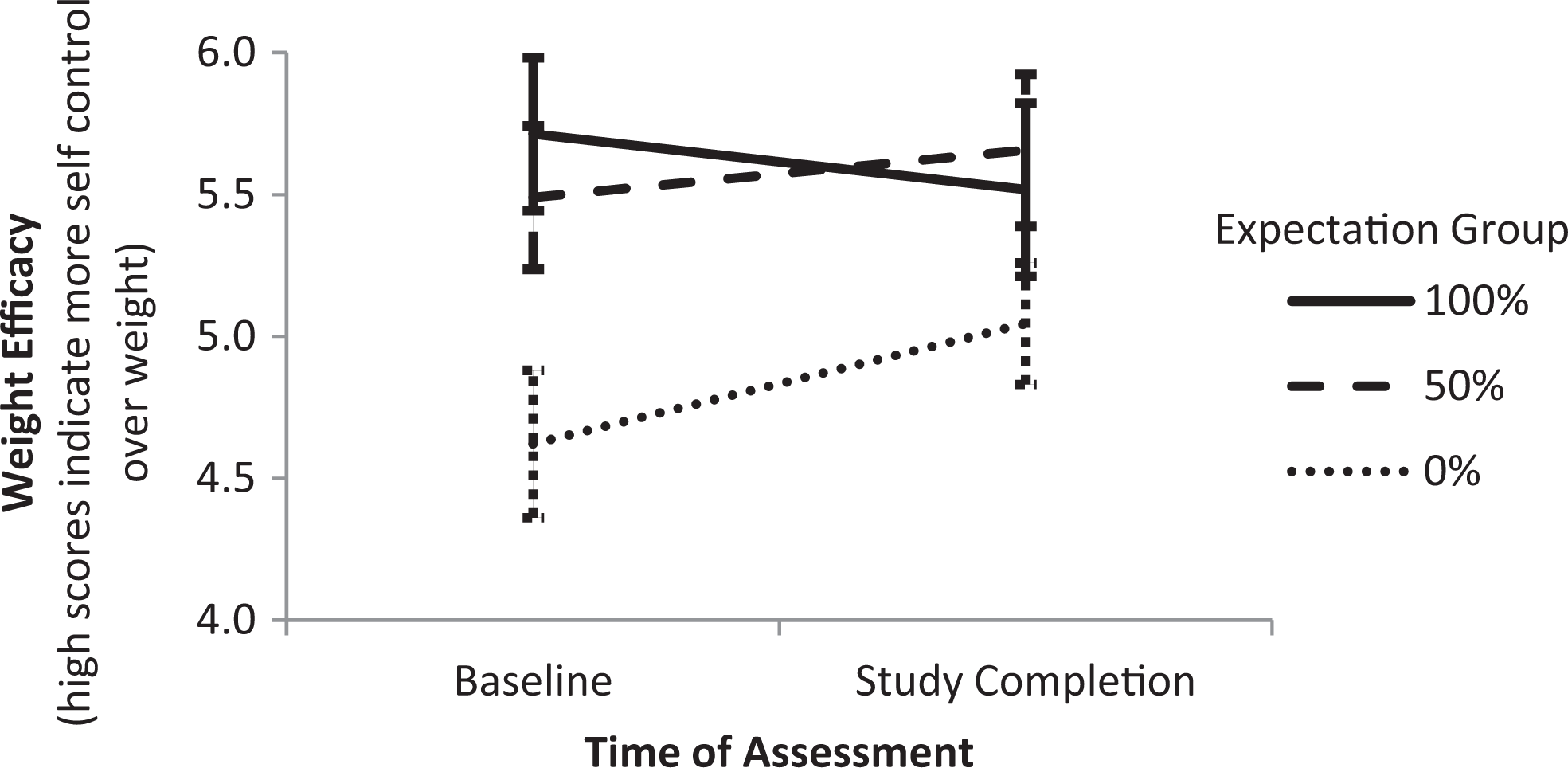
Changes in weight efficacy over time by group.
Groups displayed a pattern of a decline in self-efficacy over time for the 100% expectancy of active group, relatively stable values for the 50% active expectancy group, and increasing self-efficacy for the 0 expectancy (no capsules) group. Although between-group differences in change in general self-efficacy were not significant (P = .28), the pattern of means for this measure was similar to those observed for exercise and weight self-efficacy (Figures 2 and 3). No correlation between the 4 baseline self-efficacy measures and body mass index proportion lost reached statistical significance (all |r| < .1).
Expectancies Regarding Supplements
Participants rated, on a visual analog scale, their belief that supplements in general would help them lose weight, as well as belief that the particular supplement used in this study would do so. The group by time interaction for supplements in general was not significant, F(2, 101) = 1.49, P = .23; nor was that for the particular supplement in the study, F(2, 101) = 2.14, P = .12. Figure 4 displays the pattern of mean confidence in the supplement, which shows opposite trends to the efficacy measures. That is, the group not taking capsules exhibited decreasing belief in the particular supplement over time, as did the 50% group, whereas the 100% expectancy group showed increasing belief. The baseline general supplement effectiveness belief measure showed some negative (but statistically insignificant) correlation with body mass index proportion lost, Spearman’s r s(103) = −.174, P = .08. Participants with higher initial belief lost a smaller proportion of body mass index. The measure of belief in the specific study supplement did not correlate significantly with body mass index proportion lost (r s = −.088, P = .38).

Changes in belief in the supplement over time by group.
Adherence
No significant differences were detected between groups on adherence to the weight loss program, whether considering increase in steps taken or change in calories consumed per day (both Ps > .35). No group differences were found in adherence to the research protocol, including completion of the food diary or of pedometer records (P > .35).
Adverse Events
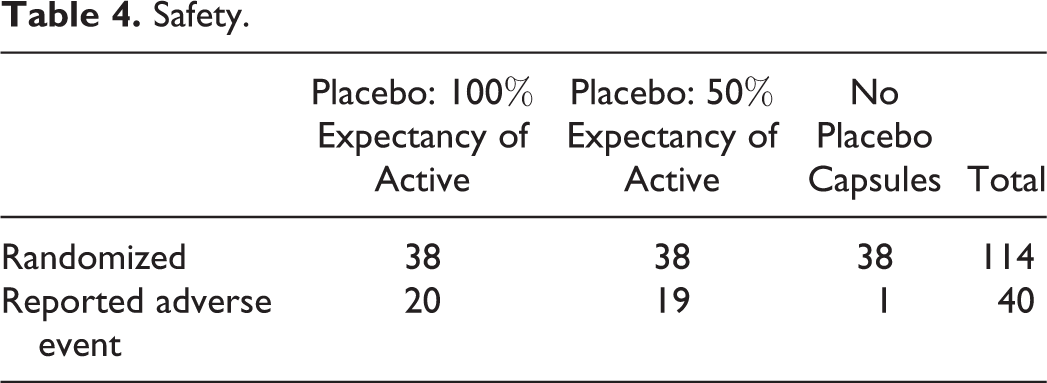
Although no participants received active treatment, headache, nausea, and faster heart rate were listed as potential side effects in the consent form, partially in order to make the placebo supplement appear credible as a medication. In subsequent reporting of adverse effects, headache was the most commonly reported adverse event (n = 16), followed by gastrointestinal problems (n = 13). Others included nausea, faster heart rate, increased thirst, anxiety, acne, and memory loss. Four cases were reported to Oregon Health & Science University’s Institutional Review Board involving arm pain, mouth pain, palpitations, fungal infection, rash with breathing problems, and chest pain, all occurring in one of the pill-taking groups. Capsule takers reported markedly greater numbers of adverse events than the lifestyle alone group, χ2(2) = 26.4, P < .001; Table 4).
Safety.
Assessment of Blinding and Deception
Investigators called all subjects in capsule-taking groups at study completion. Seventy percent of them (those responding within 3 attempts to reach them) were interviewed. On debriefing, none suspected the true purpose of the study. In the 100% expectancy group, 19% (3 of 16) believed they had received a placebo. In the 50% expectancy group, 47% (9 of 19) believed they had received a placebo. On being informed of the true design of the study and that all capsules were placebo, none of the participants were notably distressed and none withdrew their data from analysis.
Discussion
Despite a carefully constructed rationale for a weight loss supplement and controlled study conditions, a placebo effect of dietary supplement superimposed on lifestyle education was not apparent on weight loss or metabolic outcomes. In addition, we found that the expectancy effects of taking such oral agents might be disadvantageous in conditions where the first line of treatment is behavioral.
While some short-term studies in humans as well as animal studies have documented placebo effects, 31 –36 others have cast doubt on a substantial role for placebo. 37 –39 In a retrospective review of clinical trials performed between 1948 and 1998 containing both placebo and no treatment arms, the placebo effect size was negligible in comparison to no treatment in most studies. 4 This is in contrast to more recent mechanistic studies that demonstrate a placebo effect on objective measures of brain function or other outcomes. 40,41 Among the 9 conditions for which there were data, only pain studies showed a significant effect of placebo. In a 2004 update of the review that included an additional three studies in obesity, a negative effect was found on desired weight loss (subjects lost less weight with a placebo pill than with no pill) though the difference was not statistically significant. 42 However, there has been little study of placebo in the induction of salutary behavior, which may be influenced through altered expectancies. One study suggests that an illusory invulnerability may be created by taking supplements. 16
In our study, participants with stronger beliefs in supplements tended to lose a smaller proportion of weight. Participants with 100% expectancy of receiving the active supplement tended to report a decline in self-efficacy over the course of the study and to increase their beliefs in the supplement’s effects. The group with 50% expectancy of active supplement remained somewhat stable in self-efficacy scores and did not change in their level of belief in the supplements, while those taking no capsules increased in self-efficacy and tended to reduce their belief in the value of supplements.
During the 3-month follow-up, despite the finding that measures of adherence were not different between the three groups, our data suggest the possibility that taking oral agents may reduce salutary behavior changes through a negative effect on self-efficacy for appropriate long-term weight loss behaviors. In addition, adverse events in those taking placebo supplements in this study were reported at about twenty times the rate of those not taking supplements. Since one third of those undertaking a serious weight loss attempt also try dietary supplements, 43 our findings suggest a possible detrimental effect of inefficacious weight loss agents—that usage may lead some patients to become less adherent to beneficial lifestyle recommendations. 16 The implications of this finding may extend to prescription drugs to lower body weight in that the act of taking a medication may impair salutary behavior toward weight loss, irrespective of pharmacological efficacy, potentially undermining effectiveness of combined drug and lifestyle interventions. If these findings are confirmed, another implication for this research is that a nocebo effect of oral agents in a placebo-controlled trial may falsely increase the apparent effect of a test agent beyond no oral agent at all. This would argue that “no agent” arms should be included in weight loss clinical trials and, concomitantly, the absence of such an arm should be considered a design weakness in interpreting outcomes.
Limitations of our study are the relatively small sample size and short intervention period, and the measures of adherence are based mostly on participant self-report. Having participants take their pills before meals in the 2 pill groups may have added additional attention to eating in the pill groups but this did not have a clear effect. Evaluating this expectancy–dose model in a larger cohort over a longer period would confirm or refute whether the changes in attitude seen here would influence weight loss and maintenance.
Footnotes
Author Contributions
BO, JQP, and CC were responsible for the study design, experiment, and analyses. CC and EC conducted the experiments and collected data. KMT participated in data collection and led education sessions. WLG and DH conducted statistical analyses. KMT and CC were responsible for leading manuscript preparation. All authors read, edited, and approved the final manuscript.
Authors’ Note
Research was performed at the Oregon Health & Science University and the National College of Natural Medicine in Portland, Oregon. Study findings were presented at the following conferences in 2007: American Association of Naturopathic Physicians (AANP) Annual Meeting, Palm Springs, California; Symposium for Portland Area Research in CAM (SPARC), Portland, Oregon; International Congress on Complementary Medicine Research in Munich, Germany; NIH NCCAM Center Directors Meetings, Bethesda, Maryland.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was funded by National Institutes of Health (NIH) Grant No. (1U19) AT002656-01, by support from the Oregon Clinical and Translational Research Institute (OCTRI), Grant No. UL1 RR024140 from the National Center for Research Resources (NCRR), a component of the NIH, and NIH Roadmap for Medical Research, and by the Helfgott Research Institute.
Ethical Approval
This study was approved by the Institutional Review Board of the Oregon Health & Science University, Portland, Oregon (IRB00000673).
