Abstract
The hypolipidemic effects of the poorly soluble ayurvedic resin guggul, especially the molecular targets and mechanism, have not been well investigated to date. In the present study, we have formulated a liquid product, GU-MCT810, composed of a proprietary Commiphora mukul gum resin extract and medium-chain triglyceride and investigated its hypolipidemic effects in vitro. Treatment of HepG2 cells significantly reduced low-density lipoprotein cholesterol and increased the high-density lipoprotein/low-density lipoprotein ratio. GU-MCT810 showed direct inhibition of HMG-CoA reductase activity in a dose-dependent manner and compared very well with the inhibitory effect of statins like Pravastatin and Mevastatin. The adipocyte differentiation was also inhibited by GU-MCT810 treatment. GU-MCT810 increased the AMPKα phosphorylation and AMPK kinase activity and inhibited the phosphorylated form of mTOR expression, indicating the molecular targets affected by the nutraceutical compound. The preparation also highly upregulated the expression of LXR and PPARα genes and moderately upregulated BABP and SHP genes.
The worldwide obesity epidemic has brought about dramatic increases in the prevalence of chronic comorbid conditions, including insulin resistance, type 2 diabetes, the metabolic syndrome, atherosclerosis, and increased risk of cardiovascular disease. In many countries, such as India and China where there exist long-standing traditional medicine systems, botanical-based medicines and other traditional modalities are used to treat these conditions. While there is considerable ethnobotanical knowledge concerning the efficacy and safety of these traditional medicines, frequently there is little rigorous scientific evidence establishing their efficacy and little understanding about their mechanisms of action.
The ancient Ayurvedic literature Sushruta Samhitha written by Sushrata 2500 years ago refers to the utility of the exudates guggul, a gum resin of the tree Commiphora mukul, for treating obesity and lipid disorders. 1 The ethyl acetate extract of this gum resin, designated Gugulipid, has since been shown to exhibit lipid-lowering activity in normal and hyperlipidemic rats, rabbits, and monkeys.1–3 It has also been shown to raise high-density lipoprotein levels in rabbits accompanied by a lowering of low-density lipoprotein, resulting in regression of atheromatous lesions induced in rabbits on a high-fat diet. Two pregnane derivatives of guggul, Z-guggulsterone and E-guggulsterone, were identified as the key active ingredients responsible for the hypolipidemic activity of Gugulipid.4,5 Both isomers of guggulsterone possess similar hypolipidemic activity, and an 80:20 mixture of the Z- and E-isomers has been shown to lower cholesterol and triglycerides in normal and high-fat-fed rats.2,6,7 Furthermore, guggulsterone and a close analog, 80-574, have also been shown to raise high-density lipoprotein in normal rats accompanied with a lowering of very low-density lipoprotein and low-density lipoprotein.1,8
The hypolipidemic effects of guggul and its ethyl acetate- and ether-soluble fractions have been evaluated previously in human clinical trials. Most of these clinical studies performed in India demonstrated a significant degree of individual variation in response to guggul treatment; however, some response to guggul treatment was observed in 70% to 80% of patients. 9 In the United States, a study was designed to evaluate the short-term safety and efficacy of guggul extracts in Western populations. 10 In 103 ambulatory, community-dwelling, otherwise healthy adults with hypercholesterolemia, treatment with Gugulipid at concentrations of 1000 or 2000 mg showed that 18% of patients responded favorably to Gugulipid treatment, with a decrease in low-density lipoprotein level of more than 5%.
The frequently noticed substantial variations in outcomes of clinical studies have been attributed to differences in ethnic and genetic backgrounds, dietary restraints, and lifestyle. However, the variability and poor solubility affecting bioavailability of guggul preparations likely represents a significant contributing factor. Consequently, the development of standardized guggul-based preparations is an important step for further preclinical and clinical research with this traditional medicinal. To this end, we have formulated a nutraceutical complex that includes an extract of guggul prepared with a supercritical CO2 cosolvent extraction with ethanol with a medium-chain triglyceride oil composed of C8 and C10 fatty acids standardized to 2.0% active guggulsterones (GU-MCT810). GU-MCT810 was studied for its effect on 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase activity, high-density lipoprotein and low-density lipoprotein levels, 5′ adenosine monophosphate-activated protein kinase α (AMPKα) phosphorylation and AMPK kinase activity, mammalian target of rapamycin (mTOR) phosphorylation in HepG2 liver cells, as well as adipose differentiation of 3T3L1 pre-adipocytes. The effect of GU-MCT810 on expression of genes associated with cholesterol synthesis and metabolism were also investigated in HepG2 cells.
Materials and Methods
GU-MCT810 Preparation
Guggul (Commiphora mukul) gum resin was obtained from India. After cryo-milling, the powder was extracted by a cosolvent blend of CO2 + ethanol (95 + 5) under supercritical conditions of 30 MPa/323 K. This separates among others cembrene-type diterpenes, polypodane-type triterpenes, pregnane-type sterones, as well as sterols from the starting material but no glycosides, polysaccharides, and other gum-like components. The yield of extract was approximately 20% referred to starting material. Depending on raw material quality, the resulting extract, a viscose yellow liquid, has 3.5% to 5.0% of guggulsterones Z + E analyzed by high-performance liquid chromatography. The genuine extract was standardized to a content of 2% Z + E sterones by addition of MCT oil giving a homogenous liquid product that was used for efficacy testing. A typical high-performance liquid chromatography fingerprint of guggul sterones is shown in Figure 1.

High-performance liquid chromatogram of guggulsterones in supercritical Commifora mukul extract.
A certain difference in the Z/E sterones distribution became obvious during analytical work. Three raw material batches for GU-MCT810 production were analyzed to have a Z/E ratio of 3.2-3.7/1. The ratio remained unchanged after supercritical extraction and extract standardization. In comparison, 2 commercial lots of guggul ethyl acetate extract revealed a Z/E ratio of 1.2-1.6/1 only, which might be due to the different production method. The supercritical extract was also characterized by higher sterol content relative to the sterone amount.
High-Performance Liquid Chromatography Analysis of Guggul Sterones
Analysis was performed with a Merck Hitachi LaChrom Elite system consisting of DAD L-2455, column oven L-2350, autosampler L-2200, and pump L-2130 with EZChrom Elite 3.3.2 software (VWR International GmbH). Separation was achieved on a LiChrosphere 100 RP-18 (5 μm) endcapped column, 250-4 mm (Merck KG). Mobil phase was A 40% water, B 30% methanol, C 30% acetonitrile + tetrahydrofuran (4 + 1) with gradient profile (0-20 minutes) A (40% to 0%), B (30% to 50%), C (30% to 50%). Solvent flow was 1.0 mL/min, column temperature 40°C, detector wavelength 248 nm, injection volume 10 μL of 2 to 4 mg/mL extract dissolved in methanol and passed through a 0.45 μm polytetrafluoroethylene filter. Guggul sterones were calculated as guggulsterone Z available as standard from Sigma-Aldrich (St Louis, MO). Elution sequence (tR) was 8.0 minutes for guggulsterone E and 9.3 minutes for guggulsterone Z.
Cell Lines
Human liver cell line (HepG2) and mouse pre-adipocyte cell line (3T3L1) were grown in Eagle’s minimum essential medium and Dulbecco’s modified Eagle medium, respectively, supplemented with 10% fetal bovine serum, 100 U/mL of streptomycin, and 100 μg/mL of penicillin in a 5% CO2 humidified incubator maintained at 37°C.
Cytotoxicity Assay
GU-MCT810 cytotoxicity of HepG2 and 3T3L1 cells were analyzed by MTT assay using the cell proliferation kit. 11
High-Density Lipoprotein and Low-Density Lipoprotein Analysis
HepG2 (5 × 106/10 mL) cells were plated in 100 × 20 mm tissue culture dishes and treated with increasing concentrations (0-100 μg/mL) of GU-MCT810 for 72 hours at 37°C in a CO2 incubator. After treatment, cells were scraped off, washed with phosphate-buffered saline once, and homogenized in a sonicator with 60 strokes on ice. The cell extract was centrifuged at 13 000 rpm for 10 minutes and the supernatant was used for high-density lipoprotein and low-density lipoprotein estimation. The cell extract (100 μL) was mixed with equal volume of 2× Precipitation buffer (MBL International, Woburn, MA) and incubated at room temperature for 10 minutes. The tubes were centrifuged at 5000 rpm for 10 minutes and the supernatant was used for high-density lipoprotein estimation. The precipitate was dissolved in 200 μL of phosphate-buffered saline and used for low-density lipoprotein estimation. Both high-density lipoprotein and low-density lipoprotein were quantified by the high-density lipoprotein and low-density lipoprotein/very-low-density lipoprotein cholesterol quantification kit (MBL International, Woburn, MA) according to the manufacturer’s protocol. The percentage change in cholesterol was calculated based on the levels in the untreated control sample.
HMG-CoA Reductase Activity
The direct inhibition of HMG-CoA reductase activity by GU-MCT810 was analyzed using the HMG-CoA reductase activity assay kit (Sigma-Aldrich, St Louis, MO). The reaction mixture (200 μL) containing 1× assay buffer, increasing concentrations (0.01-0.1 μg) of GU-MCT810, NADPH, HMG-CoA, and HMG-CoA reductase were dispensed into 96-well plates along with 3 separate wells, 1 with reaction mixture containing no inhibitor (100% activity) and 2 other wells with the positive inhibitors, Pravastatin and Mevastatin, instead of GU-MCT810. The plates were placed in the Bio-Rad Benchtop microplate reader maintained at 37°C and the kinetic program was used to collect the absorbance at every 20 second for a total of 10 minutes. The microplate reader plots the kinetic curves using the 30 readings it collected. The specific activity of HMG-CoA reductase was calculated (units/mg protein) based on the formula given in the Sigma-Aldrich protocol. The kinetic curves were used to calculate the relative activity of enzyme at each concentration of GU-MCT810 or positive control. The inhibition percentages for each GU-MCT810 concentration and positive control sample (Pravastatin and Mevastatin) were calculated on the basis of full activity of enzyme without any inhibitor.
Adipocyte Differentiation
3T3L1 pre-adipocytes (American Type Culture Collection, Rockville, MD) were plated in Dulbecco’s modified Eagle medium supplemented with 10% (v/v) fetal bovine serum, 1% (v/v) penicillin–streptomycin (100 U/mL penicillin and 100 μg/mL streptomycin) in 24-well BIOCOAT plates (BD Biosciences, San Jose, CA) at a cell density of 105 cells/well in 2 mL of medium. On the next day, when cells were attached well, medium was replaced with differentiation medium containing 0.5 mM 3-isobutyl-1-methylxanthine, 1 μM dexamethasone, 0.125 mM indomethacin, and 1 μM insulin (MDI) in the 10% fetal bovine serum–supplemented Dulbecco’s modified Eagle medium along with varying concentrations of GU-MCT810 (0-400 μg/mL). The cells were incubated for 72 hours in a CO2 incubator maintained at 37°C. Subsequently, the differentiation medium was replaced with Dulbecco’s modified Eagle medium containing 1 μM insulin and varying concentrations of GU-MCT810 for 48 hours, and on the fifth day, insulin medium was aspirated out and replaced with fresh insulin medium. 12 On the seventh day, medium was removed from the plate and 500 μL of 10% formalin was added into each well. The plate was incubated for 5 minutes and later replaced with fresh formalin (500 μL) for 1 hour. The wells were washed with 500 μL 60% isopropanol and the wells allowed to dry completely. The cells were stained with 200 μL 0.6% Oil Red O solution for 10 minutes and washed 4 times with 2 mL of water each time. The plates were allowed to dry completely and Oil Red O was eluted from each well in 750 μL of 100% isopropanol for 10 minutes. The eluted solution was transferred to 1.5 mL spectrophotometric cuvettes and the absorbance was measured at 500 nm wavelength in a spectrophotometer. The decrease in absorbance compared to untreated sample indicates the inhibition of adipocyte differentiation.
AMPK Kinase Activity
HepG2 cells were treated with increasing concentrations of GU-MCT810 in a CO2 incubator for 72 hours. Cells were scraped off and cellular protein was extracted first using Invitrogen (Carlsbad, CA) extraction buffer containing 100 mM Tris pH 7.4, 100 mM NaCl, 1 mM ethylenediaminetetraacetic acid, 1 mM NaF, 20 mM Na4P2O7, 2 mM Na3VO4, 1% Triton X-100, 10% glycerol, 0.1% sodium dodecyl sulfate, and 0.5% sodium deoxycholate. This buffer keeps the phosphorylated protein without any degradation. The protein concentration of the cell extract was determined by Lowry’s method before its storage in the −80°C freezer. Cell extracts (equivalent to 100 μg protein) were analyzed for AMPK kinase activity using the AMPK kinase assay kit (Cyclex, Nagano, Japan) according to the manufacturer’s protocol.
AMPKα Phosphorylation
HepG2 cells were treated with increasing concentrations of GU-MCT810 for 72 hours. Cells were scraped off and cellular protein was extracted first using Invitrogen extraction buffer. Cell extract equivalent to 100 μg protein was analyzed for the levels of phosphorylated form of AMPKα (pT172) protein expression using the ELISA kit from Invitrogen. Generally, the level of phosphorylation correlated with the activity of AMPKα.
mTOR Expression
The cell extracts from GU-MCT810-treated HepG2 cells were used to analyze the level of human phospho-TOR expression using the ELISA kit from R&D Systems (Minneapolis, MN). Cell extracts equivalent to 100 μg protein was analyzed for mTOR expression, and rapamycin-treated cells were used as a positive control.
Gene Expression Studies
mRNA expression of genes associated with cholesterol and fatty acid synthesis such as liver X receptor (LXR), farnesoid X receptor (FXR), peroxisome proliferator-activated receptor alpha (PPARα), small heterodimer partner (SHP), and bile acid binding protein (BABP) were analyzed by the reverse transcriptase-polymerase chain reaction assay.11,13 Total RNA was extracted from GU-MCT810-treated HepG2 cells using the TriZol reagent (Invitrogen) and 5 μg of total RNA was reverse transcribed with the first strand cDNA synthesis system. The first strand cDNA (0.25 μg) was amplified with Platinum Taq DNA polymerase and gene-specific forward and reverse primers. The housekeeping gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was also amplified as a control. Reverse transcriptase-polymerase chain reaction products were separated on a 2% agarose gel along with Hae III digested φX174 DNA marker and photographed. The relative expression of each gene was quantified from the gel pictures using the UN-SCAN-IT gel software (Silk Scientific Corporation, Orem, UT). 13
Statistical Analysis
Mean and standard deviation estimates were calculated using Excel software. One-way analysis of variance with Dunnet’s multiple comparison test was used to compare different treatments using the GraphPad Prism software (La Jolla, CA) and statistical significance was determined.
Results
Cytotoxicity
GU-MCT810 does not induce any cytotoxicity even at the highest concentration of 200 μg/mL in both HepG2 and 3T3L1 cell lines (data not shown).
Effect of GU-MCT810 on Low-Density Lipoprotein and High-Density Lipoprotein Content
Treatment of HepG2 cells with GU-MCT810 caused a significant inhibition of low-density lipoprotein cholesterol. A concentration-dependent inhibition of low-density lipoprotein cholesterol was quite evident, and the inhibitory effect was very significant (P < .01) at 50 and 100 μg/mL GU-MCT810 concentrations compared to untreated cells (Figure 2). GU-MCT810 treatment of HepG2 cells also induced significant changes in the synthesis of high-density lipoprotein cholesterol (Figure 3). Even though there was a decreased synthesis of low-density lipoprotein cholesterol at 50 and 100 μg/mL GU-MCT810, a significant increase in the ratio of high-density lipoprotein/low-density lipoprotein cholesterol levels was evident (Figure 4).
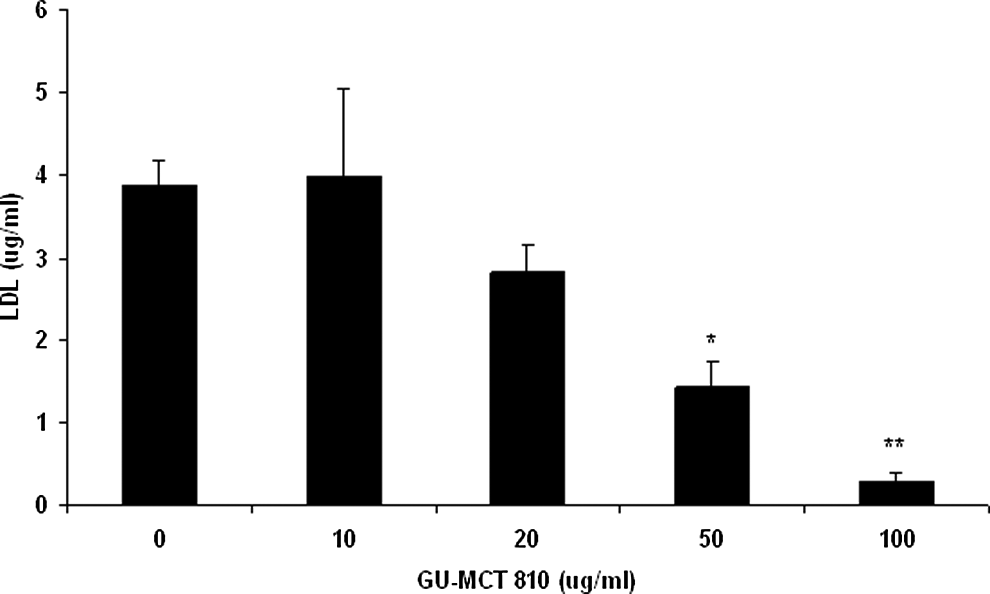
Inhibitory effect of GU-MCT810 on low-density lipoprotein cholesterol in HepG2 cells.

Effect of GU-MCT810 on high-density lipoprotein cholesterol in HepG2 cells (**P < .01; ***P < .001).
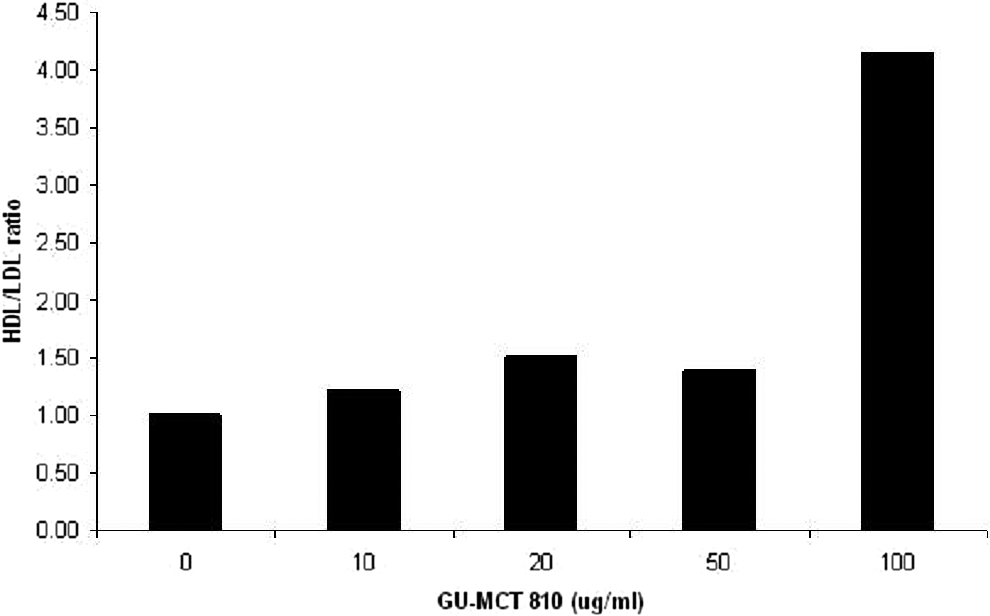
Modulation of high-density lipoprotein/low-density lipoprotein ratio in HepG2 cells.
Inhibition of HMG-CoA Reductase Activity
The results of kinetic assay presented in Figure 5 indicated that both prescription statin drugs, Pravastatin and Mevastatin, significantly inhibited HMG-CoA reductase activity. Pravastatin (0.04 μg) and Mevastatin (0.04 μg) inhibited HMG-CoA reductase activity by 83.7% and 63.7%, respectively. GU-MCT810 also inhibited HMG-CoA reductase activity in a dose-dependent manner. GU-MCT810 at a 0.1 μg concentration showed a 77.6% inhibition (P < .001) of HMG-CoA reductase activity.

Inhibitory effect of GU-MCT810 on HMG-CoA reductase activity.
Effect of GU-MCT810 on Adipocyte Differentiation
Figure 6 shows the inhibition of adipocyte differentiation by GU-MCT810 in 3T3L1 pre-adipocytes. GU-MCT810 treatment inhibited adipocyte differentiation of 3T3L1 cells in a dose-dependent manner, with more than 50% inhibition at 400 μg/mL concentration.

Inhibition of adipocyte differentiation in 3T3L1 cells by GU-MCT810 (*P < .05; **P < .01).
Effect on AMPKα Phosphorylation
GU-MCT810 treatment of HepG2 cells significantly upregulated the phosphorylation of AMPKα enzyme (Figure 7). GU-MCT810 increased the AMPKα phosphorylation by 162.8% (P < .001) at 20 μg/mL concentration, and even the modulation of AMPKα phosphorylation at 5 μg/mL GU-MCT810 was significantly different than the untreated sample (P < .05).

Increase in AMPKα phosphorylation by GU-MCT810 in HepG2 cells (*P < .05; **P < .01; ***P < .001).
Effect of GU-MCT810 on AMPK Kinase Activity
A significant increase in the AMPK kinase activity was induced by GU-MCT810 treatment of HepG2 cells (Figure 8). The increase in the activity was significant at 20 μg/mL of GU-MCT810 concentration, and the maximum increase in activity was noticed at 50 μg/mL concentration.
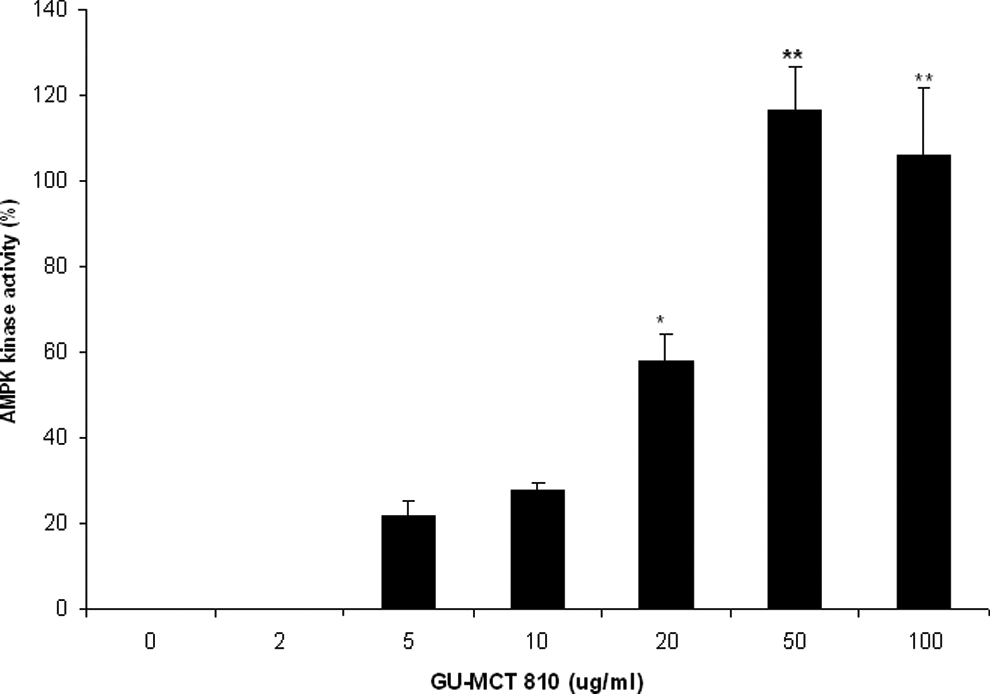
Increase in AMPK kinase activity by GU-MCT810 in HepG2 cells (*P < .05; **P < .01).
Inhibition of mTOR Phosphorylation
Higher concentrations of GU-MCT810 (10-50 μg/mL) demonstrated a significant inhibitory effect on mTOR phosphorylation (Figure 9). GU-MCT810 reduced the level of phospho-mTOR expression from 1917 pg/mL to 1262 pg/mL at 10 μg/mL dose (P < .01) and to 957 pg/mL at 50 μg/mL dose (P < .001) in HepG2 cells.
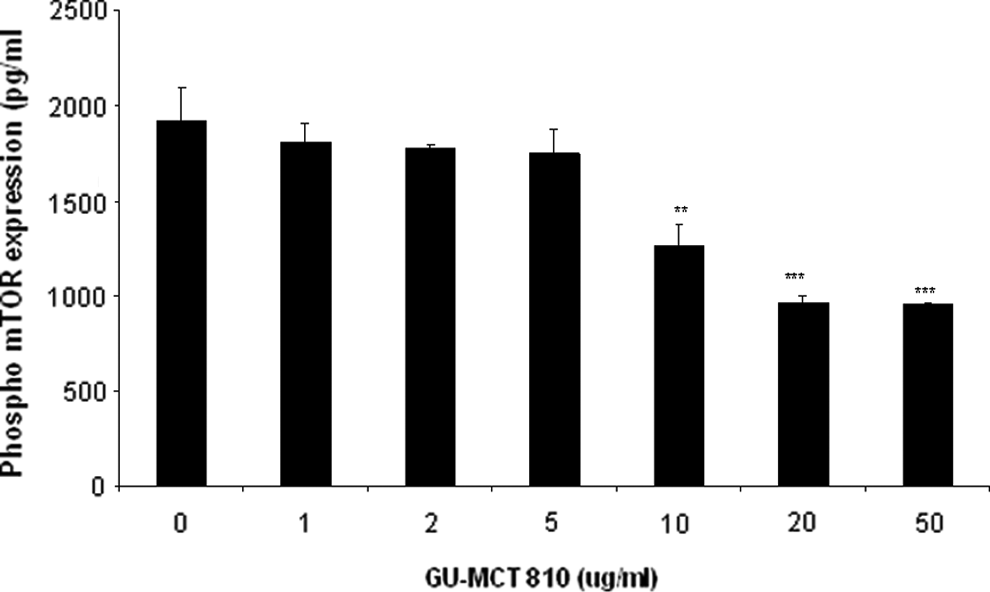
Inhibition of phosporylated form of mTOR expression by GU-MCT810 in HepG2 cells (**P < .01; ***P < .001).
Effect of GU-MCT810 on Gene Expression
The mRNA expression of LXR, FXR, PPARα, SHP, and BABP genes as well as the housekeeping gene GAPDH gene are presented in Figure 10, and the relative estimates are given in Figure 11A-E. GU-MCT810 upregulated the expression of LXR, FXR, and PPARα genes with no apparent change in the housekeeping gene GAPDH. A clear and significant dose-dependent upregulation was evident in the expression of LXR, PPARα, and SHP genes and a low to moderate but steady upregulation in the expression of FXR gene. Although BABP gene expression was altered with GU-MCT810 treatment, the change appeared to be bimodal.

Gene expression (mRNA) analysis of cholesterol synthesis associated genes (LXR, PPARα, FXR, SHP, and BABP) along with housekeeping gene GAPDH in GU-MCT810-treated HepG2 cells.
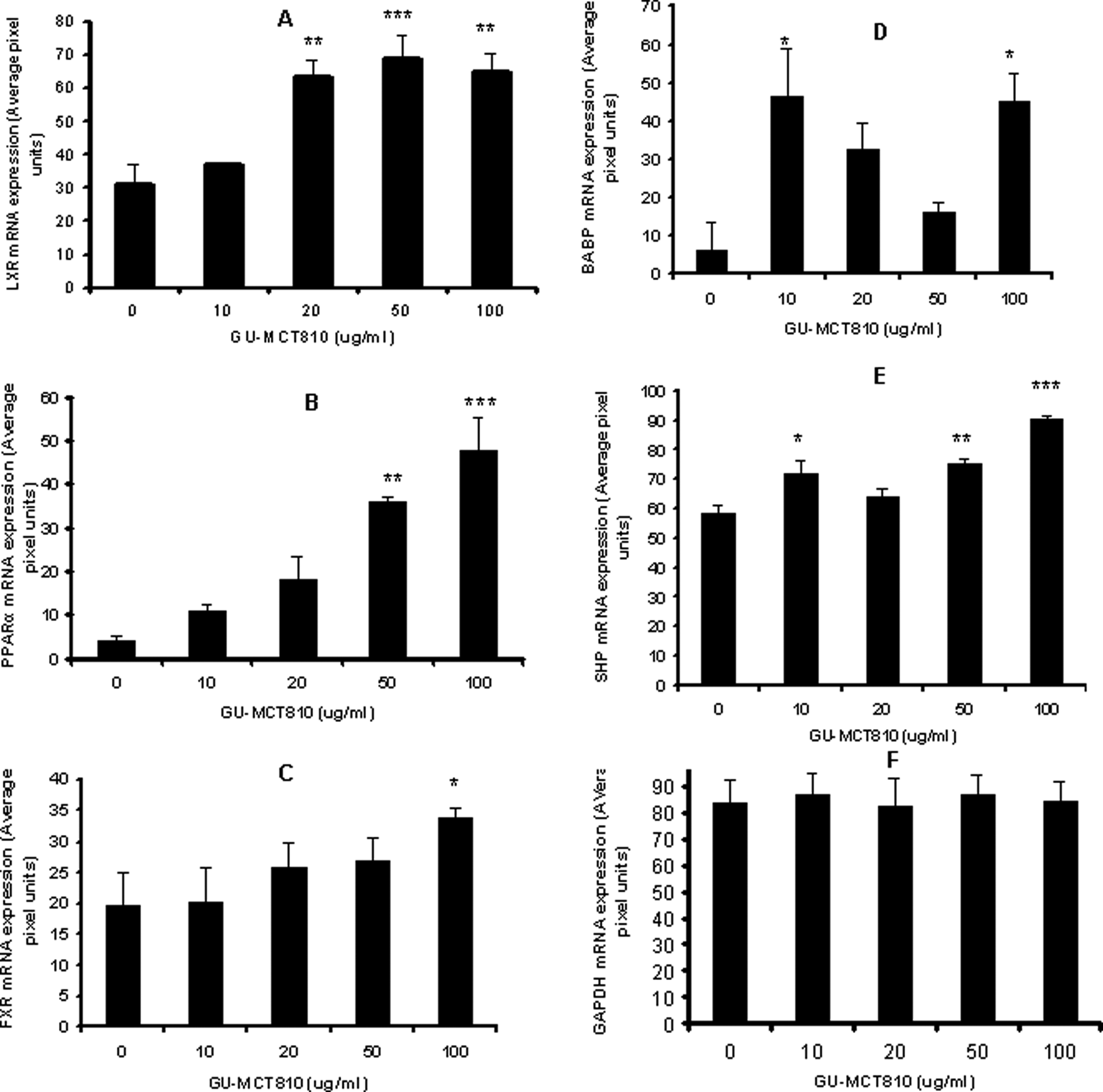
(A-F) Quantification of reverse transcriptase-polymerase chain reaction gels by UN-SCAN-IT software and the relative expression of genes (average pixel units) are plotted against GU-MCT810 concentrations.
Discussion
Cholesterol is an essential material in all eukaryotic organisms. It serves as a precursor for steroids such as corticosteroids, sex hormones, vitamin D, as well as bile acids and serves as an essential structural component of cell membranes. 14 Hyperlipidemia is generally characterized by increased fasting concentrations of total cholesterol, low-density lipoprotein cholesterol, and triglyceride in conjunction with decreased concentrations of high-density lipoprotein cholesterol. Evidence from human clinical and epidemiological studies suggests that hyperlipidemia is one of the most important modifiable risk factors for coronary heart disease.15,16 Hyperlipidemia is strongly associated with cardiovascular disease because it promotes atherosclerosis development in arteries. This disease process is a major risk factor for myocardial infarction, stroke, and peripheral vascular disease. Since higher blood low-density lipoprotein contributes to this process more than the cholesterol content of the high-density lipoprotein particles, 17 low-density lipoprotein particles are often termed bad cholesterol because they have been linked to atheroma formation. On the other hand, high concentrations of functional high-density lipoprotein, which can remove cholesterol from cells and atheroma, offer protection and are sometimes referred to as good cholesterol. Plants manufacture phytosterols (substances chemically similar to cholesterol), which can compete with cholesterol for reabsorption in the intestinal tract, thus potentially reducing cholesterol reabsorption. 18 Several plant products contain phytosterols and are reported to prevent and/or ameliorate hyperlipidemia.19–21 In the present study, GU-MCT810 treatment of liver cells resulted in the reduction in low-density lipoprotein cholesterol and increase in the high-density lipoprotein/low-density lipoprotein ratio. GU-MCT810 is formulated with 2% guggulsterones content. Two pregnane derivatives, Z-guggulsterone and E-guggulsterone,1,4,5,8 were identified as the key active components contributing to the hypolipidemic activity of Gugulipid. Both isomers have similar hypolipidemic activity, and an 80:20 mixture of the Z- and E-isomers has been shown to lower cholesterol and triglycerides in normal and high-fat-fed rats.1,6
HMG-CoA reductase, the pharmacological target of statins, is considered to be the rate-limiting enzyme for the synthesis of cholesterol from acetyl-CoA. In humans, most of the cholesterol biosynthesis occurs in the liver. Statins such as Pravastatin and Mevastatin are referred to as a very potent class of drugs that reduce serum cholesterol levels and prevent cardiovascular diseases. Statins were originally designed to compete with HMG-CoA for binding at the catalytic site of HMG-CoA reductase and thereby reduce the synthesis of mevalonate. They inhibit HMG-CoA reductase activity with Ki values in the nanomolar range, whereas the natural substrate HMG-CoA binds only at micromolar concentrations and is therefore easily replaced by drugs. A decrease in cholesterol synthesis in cells leads to a homoeostatic response, involving upregulation of cell surface receptors that bind atherogenic low-density lipoprotein and very-low-density lipoprotein. 22 The statin drugs like Pravastatin and Mevastatin significantly inhibited the HMG-CoA reductase activity, the latter being less effective than the former in the present investigation. GU-MCT810 (50-100 μg/mL) also showed a significant and dose-dependent inhibition of HMG-CoA reductase activity in the in vitro assay. Eighty percent inhibition of HMG-CoA reductase activity was induced with 0.1 μg GU-MCT810 in the direct kinetic assay. Unlike statins, the natural supplement GU-MCT810 may not possess serious side-effects that need to be evaluated further in clinical trials.
AMPK regulates lipid, cholesterol, and glucose metabolism in specialized metabolic tissues, such as liver, muscle, and adipose tissue. AMPK has been proposed to act as a fuel gauge in mammalian cells, and it plays a key role in the regulation of lipid metabolism. 23 Among the large number of AMPK protein targets, HMG-CoA and Acetyl-CoA carboxylase are well known24,25 because of their role in cholesterol synthesis. These 2 enzymes, involved in cholesterol and fatty acids synthesis, are inactivated by AMPK-mediated phosphorylation, leading to cholesterol and triglyceride inhibition. AMPK has conserved threonine residue (Thr1-172), whose phosphorylation by upstream kinase is absolutely required for its activity. By inhibiting fatty acid and triglyceride synthesis and biogenesis, AMPK activation has the potential to reduce hypertriglyceridemia, as well as elevated storage of triglycerides in muscle and liver. GU-MCT810 treatment of HepG2 cells has significantly increased the phosphorylation of AMPK and also caused significant increase in AMPK kinase activity. A dose-dependent increase in both phosphorylation and kinase activity was quite evident. We have also studied the effect of GU-MCT810 treatment on reducing the formation and storage of fat droplets by quantifying the stained fat droplets with Oil Red O stain in 3T3L1 cells. 3T3L1 cells on differentiation by chemical treatment (MDI) showed large amounts of droplets that would be stained with Oil Red O stain. GU-MCT810 treatment reduced the adipocyte differentiation in a dose-dependent manner with higher concentrations (200 and 400 μg/mL) being significant compared to untreated cells. Adipocyte differentiation is very much involved in the storage of excess energy in the form of triglyceride droplets in order to maintain lipid homeostasis, thereby promoting the survival of human body. Obesity is also associated with excess adipose tissue differentiation overloaded with high-energy fat droplets. Adipocytes also release inflammatory adipokines like TNF-α, IL-6, adiponectin, and adipsin, which can contribute to major obesity-associated complications including type 2 diabetes, cardiovascular disease, metabolic syndrome, and insulin resistance.26–29 Therefore, inhibition of adipocyte differentiation by GU-MCT810 can be useful to control obesity-related complications.
Mammalian target of rapamycin (mTOR), one of the downstream targets of AMPK, functions as an intracellular nutrient sensor to control protein synthesis, cell growth, and metabolism. Possible interplay between mTOR and AMPK signaling pathways has been shown in recent studies. 30 These 2 pathways serve as a signaling nexus for regulating cellular metabolism, energy homeostasis, and cell growth, and dysregulation of each pathway can contribute to the development of metabolic disorders such as obesity, type 2 diabetes, and cancer. 31 AMPK responds to energy stress by suppressing cell growth and biosynthetic processes, in part through its inhibition of the rapamycin-sensitive mTOR (mTORC1) pathway.32,33 In the present study, GU-MCT810 treatment has increased the phosphorylation and activity of AMPK in HepG2 liver cells while demonstrating significant inhibition of the phosphorylated form of mTOR expression.
Several nuclear receptors play an important role in regulating fatty acid, cholesterol, and bile acid synthesis and are quite critical for the maintenance of lipid homeostasis in vivo. These include liver X receptor (LXR), Farensoid X receptor (FXR), and peroxisome proliferation-activated receptors (PPAR).34–40 Additionally, 2 FXR target genes, orphan receptor SHP and bile acid binding protein (BABP), have also been implicated in the productive regulation of bile acid from cholesterol.20,35,41 The liver X receptors are nuclear receptors that act as cholesterol sensors and regulate cholesterol metabolism. Bile acids are the major metabolites of cholesterol in the body and they are essential for the solubilization and absorption of dietary cholesterol and fat-soluble vitamins. 42 The nuclear hormone receptor farnesoid X receptor plays a critical role in the regulation of bile acid, triglyceride, and cholesterol homeostasis. The liver X receptor and farnesoid X receptor function as intracellular sensors for sterols and bile acids, respectively. In response to their ligands, these receptors induce transcriptional responses that maintain a balanced, finely tuned regulation of cholesterol and bile acid metabolism. Liver X receptor also permit the efficient storage of carbohydrate- and fat-derived energy, whereas farnesoid X receptor activation results in an overall decrease in triglyceride levels and modulation of glucose metabolism. 43 Emerging evidence suggests that the tissue-specific action of these receptors is also crucial for the proper function of the cardiovascular, immune, reproductive, endocrine pancreas, renal, and central nervous systems. Together, liver X receptors and farnesoid X receptors represent potential therapeutic targets for the treatment and prevention of numerous metabolic and lipid-related diseases. In the present investigation, GU-MCT810 treatment of HepG2 cells was found to clearly up regulate the mRNA expression of genes associated with cholesterol synthesis and homeostasis such as LXR, PPARα, and SHP to a large extent and FXR and BABP genes regulating triglyceride, bile acid, and cholesterol homeostasis to a lesser extent. It is reported that LXR, FXR, and PPARα agonists lower both serum triglycerols and cholesterol levels,44–48 and therefore, they are used as targets for drug development. The function of PPARα is to increases the fatty acid oxidation and its activation decreases triglyceride concentrations by increasing the expression of lipoprotein lipase and decreasing apo C-III concentration. PPARα activation also decreases thrombosis and vascular inflammation. 49 The modulation of these biomarkers at the transcriptional level would be molecular changes underlying the hypolipidemic effect of GU-MCT810.
The present investigation was undertaken to evaluate the in vitro properties of GU-MCT810 on pharmaceutical, biochemical, and molecular targets of clinical conditions associated with hyperlipidemia, obesity, and potentially hyperglycemia. Consequently, GU-MCT810 can also play a role in addressing metabolic syndrome. However, while the observations we have reported are suggestive of potential clinical value, further investigation with in vivo and clinical studies is required, particularly with regard to bioavailability and pharmacokinetics.
Footnotes
Author Contributions
Drs Cheppail Ramachandran, Smitha M. Nair, and Steven J. Melnick were responsible for study design, experimentation, and article preparation. Dr Karl-W. Quirrin formulated and prepared GU-MCT for the study and performed chemical analysis. All authors have read and approved the article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Steven J. Melnick is the founder and Chief Operating Officer of Dharma Biomedical LLC, which is an evidence-based ethnobotanical and evochemical drug discovery company operating on for-profit basis. Karl-W. Quirrin is the Chief Executive Officer of Flavex Naturextrakte GmbH, a company producing specialty botanical extracts for cosmetics and food supplements on the basis of supercritical CO2 extraction. Dr Cheppail Ramachandran and Dr Smitha M. Nair are employees of Dharma Biomedical LLC.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
