Abstract
Seonghyangjeongkisan has been used as a therapeutic agent for cerebral disease in Korea, but its effectiveness in Alzheimer’s disease is not well known. In this study, we examined whether Seonghyangjeongkisan could protect against amyloid β–induced cytotoxicity in neuroblastoma cells and the brain. Seonghyangjeongkisan rescued amyloid β–induced cytotoxicity dose dependently and reduced amyloid β–induced apoptosis and reactive oxygen species. Injection of mice with amyloid β impaired performance on the passive avoidance task, but Seonghyangjeongkisan markedly improved memory impairment in mice, with it being more effective than tacrine treatment in mice. Moreover, the activation of stress-related kinases such as extracellular signal-regulated kinase, c-Jun NH2-terminal kinase, and p38 was suppressed, and the phosphorylation of τ protein, which is known as a marker of Alzheimer’s disease, was also suppressed by Seonghyangjeongkisan treatment in the hippocampus. These results demonstrate that Seonghyangjeongkisan reduces amyloid β-induced toxicity in the brain, suggesting that it may be a useful complementary therapy against Alzheimer’s disease.
Alzheimer’s disease is a devastating neurodegenerative disorder affecting approximately 26 million people worldwide, and there are no effective treatments for this disease. 1 Two important pathological features of the brains of people with Alzheimer’s disease are the presence of senile plaques of amyloid β in the extracellular space and the occurrence of abundant neurofibrillary tangles in neurons. 2 Subsequent to the characterization and purification of cerebral amyloid and neurofibrillary tangles, the amino acid sequences of amyloid β 3 and the τ protein in the neurofibrillary tangles 4 were identified.
In vitro studies have shown that amyloid β increases the phosphorylation of τ protein, 5,6 which reveals that extracellular accumulation of amyloid β precedes that of τ hyperphosphorylation in neurons. Molecular genetic studies have shown that abnormal gene mutations either in amyloid precursor protein or in familial Alzheimer’s disease accelerate amyloid β generation and aggregation. 7,8 Further mutations of the familial Alzheimer’s disease causative gene presenilin 1 enhanced the secretion of the longer form of amyloid β protein called amyloid β42, 9 leading to an increased amyloid β42/amyloid β40 ratio to cause the induction of toxic amyloid β aggregation. 10 Amyloid β42 is known to form seeds that accelerate amyloid β aggregation. 11 The β-amyloid hypothesis that amyloid β accumulation is one of the primary causes of neuronal degeneration and induces accumulation of τ protein in the brains of people with Alzheimer’s disease was therefore proposed. Thus, on the basis of this hypothesis, various types of immunomodulation interventions have been studied, which include anti–amyloid β aggregation agents and secretase inhibitors/modulators blocking amyloid β production. 12
The Korean medicine Seonghyangjeongkisan has traditionally been used as a therapeutic agent for cerebral infarction. A previous study has suggested that Seonghyangjeongkisan could have regulatory effects on inflammatory reactions and immune responses, which play a role in the pathogenesis of cerebral infarction. 13 However, few studies have evaluated its anti-Alzheimer’s disease effects. The antiamyloid β effects of Seonghyangjeongkisan are currently unknown. Therefore, in the present study, we investigated the neuroprotective effects of Seonghyangjeongkisan against amyloid β–induced cytotoxicity in SH-SY5Y neuroblastoma cells and mice. A MTT (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide) assay was conducted to determine whether Seonghyangjeongkisan would protect SH-SY5Y cells against amyloid β. We also explored its antioxidant effect by studying the inhibition of reactive oxygen species and the phosphorylation activity profiles of stress-related kinases and τ protein to understand the underlying mechanisms responsible for its effects.
Methods
Preparation of Seonghyangjeongkisan
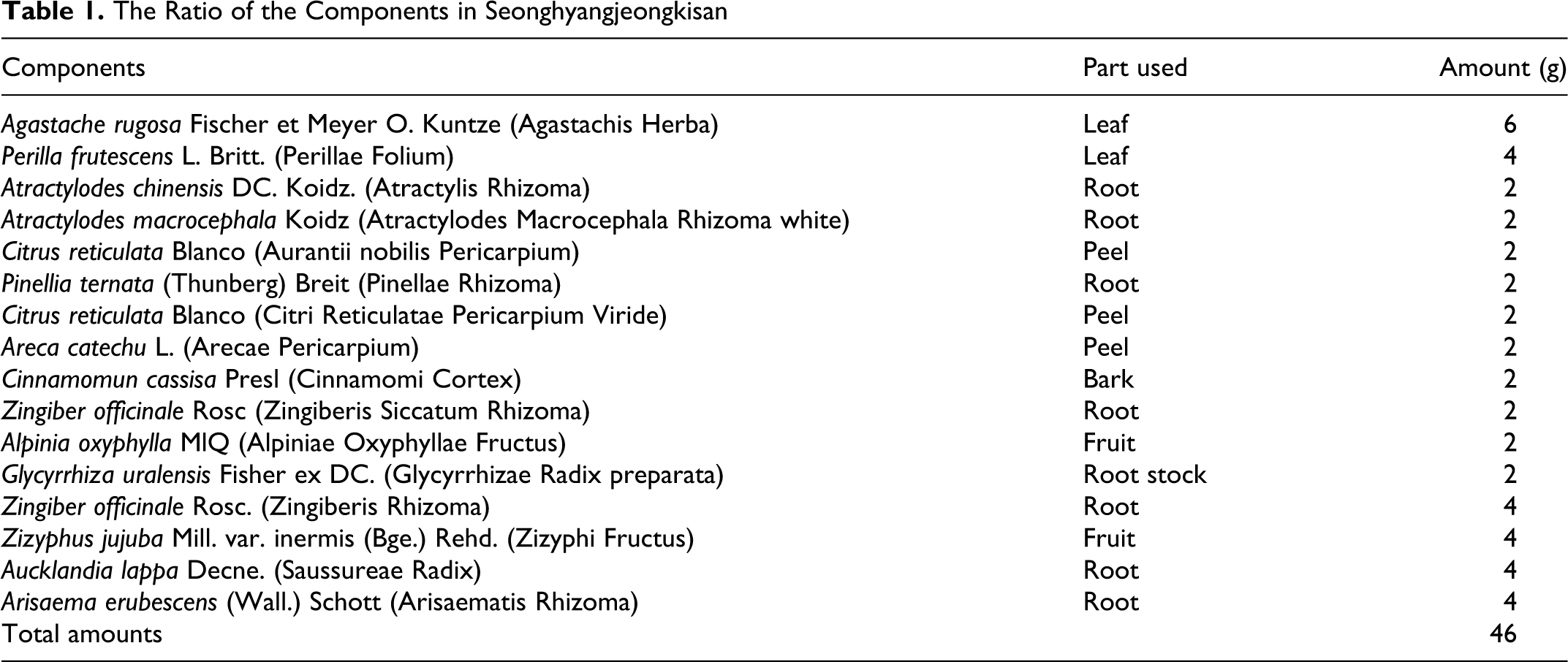
Seonghyangjeongkisan, which is a mixture of 16 traditional herbs, as shown in Table 1 , was obtained from the College of Oriental Medicine of Dongguk University. These herbs are available in China, Korea, Vietnam, and India. The extract of Seonghyangjeongkisan was prepared by decocting the dried prescription of herbs with boiling distilled water (46 g/230 mL). The duration of the decocting process was about 2 hours. The decoction was then filtered, lyophilized, and kept at 4°C.
The Ratio of the Components in Seonghyangjeongkisan
Cell Culture
Human neuroblastoma SH-SY5Y cells were obtained from American Type Culture Collection (Rockville, MD) and grown in Dulbecco’s modified Eagle’s medium (Sigma, USA) containing 10% fetal bovine serum, 100 unit/mL penicillin, and 100 μg/mL streptomycin (Gibco, Carlsbad, CA). The cells were maintained at 37°C in 5% CO2 and plated at a density of 1 × 105 cells/mL. Cells were pretreated with the indicated concentrations of Seonghyangjeongkisan for 30 minutes before exposure to amyloid β1-42 (25 μM) for 24 hours.
Cytotoxicity Assay
Cell viability was determined using the MTT (Sigma, USA) assay. In brief, SH-SY5Y cells were seeded on 12-well plates at a density of 5 × 103 cells/well and treated with various concentrations of Seonghyangjeongkisan for 24 hours. The medium was removed, and the cells were incubated with 2 mg/mL of MTT solution. After incubation for 4 hours at 37°C and 5% CO2, the supernatants were removed and dimethyl sulfoxide (Sigma, USA) was added. The reactants were measured in terms of optical density at 580 nm with a microplate reader (UV max, Molecular Devices, USA). The optical densities were converted into percentages using the following formula: Cell viability (%) = (Optical density sample/Optical density of negative control) × 100. Negative control cells were treated with complete Dulbecco’s modified Eagle’s medium alone.
Apoptosis Assay
To evaluate the effect of Seonghyangjeongkisan on amyloid β–induced apoptosis in human neuroblastoma SH-SY5Y, cells were incubated overnight on sterile cover glasses before transfection and fixed by 3.7% formaldehyde for 10 minutes at room temperature. The fixed cells were washed 3 times with phosphate-buffered saline. Hoechst 33342 staining solution was added to the cell suspension to obtain a final concentration of 10 μg/mL, and the cell–dye mixture was incubated at 37°C for 60 minutes. The stained nuclei were observed using a fluorescence microscope (S46; Microscopes Inc, MO) with an ultraviolet filter, at 360 nm.
Reactive Oxygen Species Assay
Intracellular reactive oxygen species generation was detected using a fluorometer. The reactive oxygen species–sensitive dye, 2,7-dichlorodihydrofluorescein diacetate (Sigma, USA), enters cells passively, is converted to dichlorofluorescein diacetate, reacts with reactive oxygen species, and ultimately forms the fluorescent product, dichlorofluorescein. The treated cells were incubated with 20 μM 2,7-dichlorodihydrofluorescein diacetate for 30 minutes. The fluorescence intensity was measured at 480 nm excitation and 530 nm emission using a fluorescence microplate reader (SpectraMax Gemini EM, Molecular Device, Sunnyvale, CA).
Experimental Animals and Schedule
Protocols for animal use were reviewed and approved by the Institutional Animal Care and Use Committee at the Dongguk University Ilsan Hospital and were in accordance with National Institute of Health guidelines. Healthy imprinting control region male mice (20-26 g, 6 weeks old) were obtained from OrientBio (Seoul, Korea) and were allowed 1 week for quarantine and acclimatization. Animals were housed under conditions of constant temperature (22°C ± 1°C) and relative humidity (55% ± 1%) and a 12-hour light/dark cycle (light on at 7:00
Passive Avoidance Task
The passive avoidance task was carried out as described previously, 17 with further modifications. The apparatus (AP model; O’Hara Co, Tokyo, Japan) for this learning task consisted of a white illuminated compartment and a dark compartment equipped with electrical wires; the 2 chambers were separated by a guillotine door. For the acquisition trial, each mouse was placed singly in the light chamber and allowed to explore by opening the door before returning to its home cage. After repeating the exploration twice, the mouse received an electrical shock of 0.3 mA for 3 s on entering the dark chamber. After moving back to the light chamber, the door was closed, and the mouse was returned to its home cage. The initial latency to enter the dark chamber was recorded. Mice remaining in the light chamber for more than 300 s during the acquisition trial were excluded from the following retention test. The memory retention test was performed the next day without any electrical shock. The mice were again placed in the light chamber, and the time taken to enter the dark chamber was recorded as the step-through latency, which was measured for up to 300 s. Animals that do not remember the electrical shock will cross over earlier than those that remember.
Western Blot Analysis
The treated cells were lysed with a triple-detergent lysis buffer to detect various proteins related to apoptosis and reactive oxygen species generation. For the detection of Bax and Bcl-2, the cells were fractionated into mitochondria and cytosol using a mitochondria/cytosol fractionation kit (Thermo Fisher Scientific, USA) according to the manufacturer’s instructions. The nuclear fractionation of each lysate was used for nuclear factor-erythroid 2 p45-related factor 2 translocation. Cell lysates were separated by 15% SDS-polyacrylamide gel electrophoresis and then transferred to nitrocellulose membranes. Membranes were incubated with 5% skim milk in Tris-buffered saline with 0.1% of Tween 20 (TBS-T) for 1 hour and then with the anti-Bax, Bcl-2, heme oxygenase-1, or nuclear factor-erythroid 2 p45-related factor 2 antibody (Santa-Cruz Biotechnology, Inc, CA) overnight at 4°C, followed by incubation with horse radish peroxidase–conjugated secondary antibodies for 1 hour. Immunoreactive bands were detected using Supersignal West Pico Chemiluminescent Substrate (Thermo Fisher Scientific, USA). The animals were killed at 17 days after amyloid β1-42 or saline injection, and the brains were immediately removed and stored at −80°C until use. The hippocampus was homogenized in lysis buffer containing 50 mM Tris–base (pH 7.5), 150 mM NaCl, 2 mM ethylenediaminetetraacetic acid, 1% glycerol, 10 mM NaF, 10 mM Na-pyrophosphate, 1% NP-40, and protease inhibitors (0.1 mM phenylmethylsulfonylfluoride, 5 μg/mL aprotinin, and 5 μg/mL leupeptin). Also, 30 μg of tissue lysate was electrophoresed using SDS-polyacrylamide gels and transferred to nitrocellulose membranes, which were then incubated with anti-phospho-c-Jun N-terminal kinase, anti-c-Jun N-terminal kinase, anti-phospho-p38, anti-p38 (Cell signaling technology, Beverly, MA), anti-phospho-Tau (S262), and anti-Tau antibody (BioSource International, Camarillo, CA) for 16 hours at 4°C. After washing with TBS-T (0.05%), the blots were incubated with horseradish peroxidase–conjugated antirabbit or antimouse IgG, and the bands were visualized using the ECL system (Thermo Fisher Scientific, USA). Band images were obtained using a Molecular Imager ChemiDoc XRS+ (Bio-Rad, Hercules, CA), and band intensity was analyzed using Image Lab software version 2.0.1 (Bio-Rad).
Statistical Analysis
All values are expressed as mean ± standard error of the mean. Data were analyzed by 1-way ANOVA, and then differences among means were analyzed using Dunnett’s test or Tukey–Kramer’s multiple comparison test. Differences were considered significant at P < .05.
Results
Cell Cytotoxicity of SH-SY5Y Cells
To determine the cytotoxic effect of various concentrations of the Seonghyangjeongkisan extract, the viability of SH-SY5Y cells was determined by MTT assay. As shown in Figure 1A, Seonghyangjeongkisan had no cytotoxicity at any of the concentrations tested. Figure 1B shows the cell viability at various Seonghyangjeongkisan concentrations and cotreatment of amyloid β for 24 hours. Our results showed that amyloid β had cytotoxicity at concentrations of 25 μM examined with 50% cell viability compared with control. The viabilities of cells treated with 10, 100, and 1000 μg/mL Seonghyangjeongkisan were 68% ± 1.52%, 76.2% ± 2.09%, and 93.5% ± 1.72%, respectively, compared with control, and cell viability increased in a dose-dependent manner with Seonghyangjeongkisan. These data indicate that amyloid β caused a decrease in the viability of SH-SY5Y cells, and Seonghyangjeongkisan alleviated amyloid β-induced cytotoxicity of human neuroblastoma cells.

Effect of Seonghyangjeongkisan (SHJKS) on the cell viability measured by MTT assay: A. The human neuroblastoma cells were incubated with various concentrations of SHJKS for 24 hours. B. SH-SY5Y cells were pretreated with SHJKS (10, 100, and 1000 μg/mL) for 30 minutes before exposure to amyloid β (25 μM) for 24 hours. Values are presented as mean ± standard error of the mean of 6 measurements.

Effect of Seonghyangjeongkisan (SHJKS) on the amyloid β (Aβ)–induced apoptosis: A. Morphological changes of SH-SY5Y cells identified by observation under fluorescence microscopy (×20). Arrows are indicative of apoptotic cells. At least 15 fields were analyzed per slide, and 3 slides were prepared per group. Bar = 100 μm. B. SHJKS increased the Bcl-2/Bax protein ratio in SH-SY5Y cells. Each protein was normalized to β-actin. The results are expressed as mean ± standard error of the mean of 3 experiments performed in triplicate.
Seonghyangjeongkisan Inhibits the Amyloid β–Induced Apoptosis of SH-SY5Y Cells
Figure 2A presents the apoptotic morphology of SH-SY5Y cells under Seonghyangjeongkisan treatment in the presence and absence of amyloid β (25 μM). As compared with the control group, amyloid β–activated SH-SY5Y cells showed chromatin condensation and nuclei fragmentation. The cotreatment of amyloid β with Seonghyangjeongkisan reduced apparent apoptotic morphological changes, such as shrunken or fragmented nuclei. Bcl-2 inhibits apoptosis by preventing mitochondrial membrane depolarization, whereas Bax promotes apoptosis by inducing mitochondrial membrane depolarization and cytochrome c release. Therefore, we investigated the effect of Seonghyangjeongkisan on Bcl-2/Bax expression in amyloid β–induced SH-SY5Y cells by Western blotting (Figure 2B ). Compared with the amyloid β–treated group, the Seonghyangjeongkisan-treated group exhibited significantly increased Bcl-2 expression, with decreased expression of Bax. When the cells were treated with Seonghyangjeongkisan only, there was no change in the Bcl-2/Bax expression (data not shown). This suggests that Seonghyangjeongkisan inhibited the amyloid β–induced apoptosis of SH-SY5Y cells by upregulating Bcl-2 expression.

Effect of Seonghyangjeongkisan (SHJKS) on oxidative stress: A. SH-SY5Y cells were incubated with SHJKS (1 mg/mL) for 30 minutes before 25 μM of amyloid β (Aβ) administration for 1 hour. Reactive oxygen species generation was measured by the fluorescence intensity of dichlorofluorescein in RFU (relative fluorescence units). Values are indicated as mean ± standard error of the mean. B. Representative Western blot and corresponding densitometric analyses for heme oxygenase-1 (HO-1) and nuclear factor-erythroid 2 p45-related factor 2 (Nrf2) that were normalized versus β-actin. Cells were cotreated with 100 μg/mL of SHJKS and 25 μM of Aβ for 16 hours. The data are expressed as mean ± standard error of the mean of 3 experiments.

Effect of Seonghyangjeongkisan (SHJKS) on amyloid β (Aβ)–induced learning and memory deficits in the passive avoidance task. Daily administration of tacrine (5 mg/kg) and SHJKS (1 g/kg) for 14 days was followed by Aβ injection. Mice were subjected to the acquisition trial on day 6 after the Aβ1-42 injection. After 24 hours, the step-through latency time was measured during a cut-off period of 300 s. The data are expressed as mean ± standard error of the mean, with 8 mice per group.
Seonghyangjeongkisan Inhibits Reactive Oxygen Species Generation in SH-SY5Y Cells
Because amyloid β–induced oxidative stress is related to neuron damage and memory loss, the antioxidant effect of Seonghyangjeongkisan was analyzed. As shown in Figure 3A , reactive oxygen species levels in SH-SY5Y cells was significantly increased by 3 times with amyloid β treatment when compared with the control group (P < .05). Seonghyangjeongkisan alone also increased reactive oxygen species levels compared with the control group. However, pretreatment of Seonghyangjeongkisan with amyloid β significantly decreased reactive oxygen species levels compared with the group treated with amyloid β alone. The involvement of reactive oxygen species in amyloid β–induced oxidative damage was further assessed by examining the expression of heme oxygenase-1 and nuclear factor-erythroid 2 p45-related factor 2. Because heme oxygenase-1, which is regulated by nuclear factor-erythroid 2 p45-related factor 2, is induced as a defense against oxidative stress, we assessed whether Seonghyangjeongkisan could induce heme oxygenase-1 expression as well as nuclear nuclear factor-erythroid 2 p45-related factor 2 expression (Figure 3B). The expression levels of heme oxygenase-1 and nuclear factor-erythroid 2 p45-related factor 2 were significantly increased in SH-SY5Y cells treated with amyloid β compared with control. Seonghyangjeongkisan alone increased heme oxygenase-1 but not nuclear factor-erythroid 2 p45-related factor 2 levels as compared with control. This upregulation of heme oxygenase-1 was further increased by cotreatment of Seonghyangjeongkisan with amyloid β in the human neuroblastoma cells. Thus, it is possible that Seonghyangjeongkisan actually acts as a cell irritant, milder than amyloid β that just induces cell protective mechanisms. These data imply that Seonghyangjeongkisan may exert an antioxidative effect through the upregulation of heme oxygenase-1.
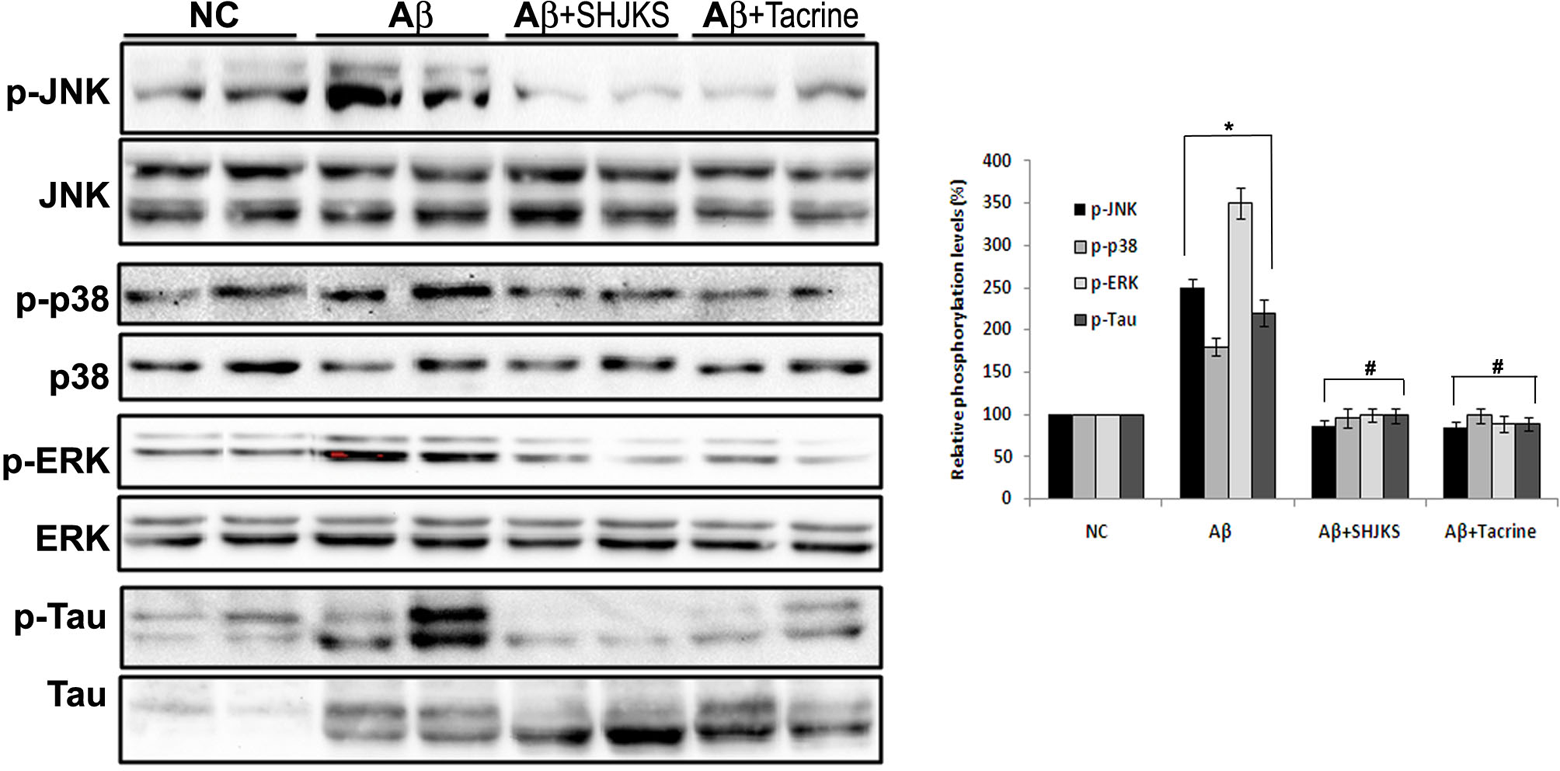
Seonghyangjeongkisan (SHJKS) decreased activities of p38, extracellular signal-regulated protein kinase (ERK), c-Jun N-terminal kinase (JNK), and τ phosphorylation by amyloid β (Aβ) injection in the mouse hippocampus. The hippocampal lysates were electrophoresed using 10% SDS-PAGE and inmmunoblotted with each antibody. The intensity of the protein bands was quantitated by densitometry, and the phosphorylated form was normalized versus the total form. The data are expressed as mean ± standard error of the mean with 8 mice per group.
Seonghyangjeongkisan Protects Cognitive Function in Amyloid β–Treated Mice
The effect of Seonghyangjeongkisan on long-term memory was investigated in the passive avoidance task. A significant group effect was observed on step-through latency during the test trial (Figure 4). The amyloid β–treated group had lower latencies than the normal control group (P < .05). However, tacrine and Seonghyangjeongkisan reversed this effect (P < .05 and P < .001, respectively). It suggested that Seonghyangjeongkisan attenuated cognitive deficits in amyloid β–injected mice.
Seonghyangjeongkisan Suppresses Phosphorylation of p 38, Extracellular Signal-Regulated Protein Kinase, c-Jun N-Terminal Kinase, and τ in the Hippocampus of Amyloid β–Treated Mice
Both the sample immunoblot and the mean data obtained from densitometric analysis indicate that the activities of these 3 kinase phosphorylations were enhanced in hippocampal tissues prepared from amyloid β–treated mice (Figure 5). This upregulation of p38, extracellular signal-regulated protein kinase, and c-Jun N-terminal kinase phosphorylation was reverted by administration of Seonghyangjeongkisan and tacrine. In the case of τ activity, the increase and decrease of its phosphorylation were similar to those of the 3 kinases. The suppressed τ phosphorylation from Seonghyangjeongkisan treatment was further confirmed by immunohistochemistry (figure not shown).
Discussion
Numerous medicinal herbs have been used in the form of decoctions because their effects can be seen in terms of reactions to their constituent compounds when administered to humans. Seonghyangjeongkisan is such an example, and it consists of 16 different herbs. Seonghyangjeongkisan has been used as a therapeutic agent for cerebral disease, especially in Korea in conjunction with Sasang Constitutional Medicine, which takes a typological constitution approach to holistic medicine by balancing an individual’s psychological, social, and physical aspects to achieve wellness and increased longevity. The only previous study of Seonghyangjeongkisan reported that it may have regulatory effects on the pathogenesis of cerebral infarction. 13
The present study demonstrated that Seonghyangjeongkisan alleviated amyloid β–induced learning and memory deficits for mice, demonstrated by the mice remaining in the well-lit side of a 2-compartment apparatus and not entering the dark, where it received the electrical stimulus. Because a mouse innately gravitates to darkness, the animal has to suppress this instinct through pairing the aversive stimulus (electrical stimulus) with the desired compartment (dark chamber). Animals that do not remember the aversive stimulus will cross over earlier than animals that do remember. 18
We then investigated the downstream consequences of an increase of amyloid β in the hippocampus, with the objective of increasing our understanding of the mechanisms that might contribute to hyperphosphorylation of τ protein. Because abnormal τ phosphorylation in Alzheimer’s disease can be seen at the level of cytoplasmic neurons, and τ proteins contain phosphorylation sites for several protein kinases, we measured the phosphorylation level of protein kinase of 38 kDa (p38), extracellular signal-regulated protein kinase, and c-Jun N-terminal kinase in the hippocampus. All these kinases have the capacity to phosphorylate τ protein, 19 and they are activated in response to UV irradiation, proinflammatory cytokines, and environmental stress, including oxidative stress, which leads to cell death. 20 In fact, interleukin-1β as a proinflammatory cytokine stimulated activity of such kinases through increased production of reactive oxygen species, 20 which concurs with observations from our current study. Our data confirm that the phosphorylation of p38/extracellular signal-regulated protein kinase/c-Jun N-terminal kinase is a response to cellular stress from amyloid β injection with substantial production of nitric oxide in the serum (data not shown). As expected, Seonghyangjeongkisan decreased the phosphorylation of extracellular signal-regulated protein kinase, c-Jun N-terminal kinase, p38 kinase, and τ protein, implying that changes in their phosphorylation state into hyperphosphorylation exert a neurotoxic effect.
In addition, Seonghyangjeongkisan reduced amyloid β–induced apoptotic cell death and reactive oxygen species generation in vitro in SH-SY5Y cells. Apoptosis or programmed cell death is an extremely fine-tuned regulatory event consisting of proapoptotic and antiapoptotic factors. 21 Bcl-2 family proteins are critical regulators of apoptosis. Whereas Bcl-2 is one of the antiapoptotic members of the family, Bax is a proapoptotic member. Our results clearly demonstrated that amyloid β had a significant apoptotic effect on SH-SY5Y cells in vitro. However, the inhibitory effect of Seonghyangjeongkisan against apoptosis has been well proven by the increase of Bcl-2/Bax expression. These findings are in accordance with previous publication showing that an altered Bcl-2/Bax ratio induced the release of cytochrome c for the activation of the terminal cascade of apoptosis by inducing mitochondrial membrane depolarization. 22
As is well known, oxidative stress is implicated in the pathogenesis of Alzheimer’s disease. In humans, the antioxidant response element regulates the expression of a number of cytoprotective antioxidant enzymes and scavengers, which contribute to the endogenous defense against oxidative stress. 23 Genes regulated by the antioxidant response element include heme oxygenase-1. This protein can be regulated through various transcription factors, which include AP-1 (through extracellular signal-regulated protein kinase and/or c-Jun N-terminal kinase), 24 nuclear factor-kB, 25 and nuclear factor-erythroid 2 p45-related factor 2. 23 In the present experiment, reactive oxygen species and heme oxygenase-1 expressions were induced by only Seonghyangjeongkisan treatment, suggesting that Seonghyangjeongkisan may act as a minor cell irritant, milder than amyloid β, that just induces a cell protective response. However, nuclear factor-erythroid 2 p45-related factor 2 was not induced by Seonghyangjeongkisan. Thus, these results imply that Seonghyangjeongkisan induces heme oxygenase-1 expression through AP-1 or nuclear factor-κB except nuclear factor-erythroid 2 p45-related factor 2. This agrees with results from previously published reports. For example, heme oxygenase-1 induction by sevoflurane is associated with extracellular signal–regulated protein kinase1/2 and AP-1 activation in the rat liver, and c-Jun N-terminal kinase and p38 signaling pathways are involved in arsenite-induced heme oxygenase-1 expression in hepatocytes. 26,27 Thus, which signaling pathway may be involved in the Seonghyangjeongkisan-induced heme oxygenase-1 expression remains to be elucidated.
Currently, there are no effective treatments and no known means to prevent Alzheimer’s disease. 1 The treatment for Alzheimer’s disease is a daily struggle for physicians, and the development of effective treatment for Alzheimer’s disease represents one of the greatest challenges to modern psychopharmacological research. 28 The important role of the cholinergic system in Alzheimer’s disease is well documented even though the usefulness of cholinergic agents that act to boost cholinergic transmission to improve attentional function in Alzheimer’s disease still remains well justified. 28 Thus, natural herbal compounds that inhibit acetylcholinesterase activity can be potential candidates for Alzheimer’s disease treatment. 29 Tacrine is such an acetylcholinesterase inhibitor for the treatment of Alzheimer’s disease. Our current study showed that tacrine was as effective as Seonghyangjeongkisan for memory function. Future research on Seonghyangjeongkisan should include acetylcholinesterase activity assays.
In conclusion, Seonghyangjeongkisan administration ameliorates learning and memory deficits in the amyloid β–treated mouse model and leads to downregulation of extracellular signal–regulated protein kinase, c-Jun N-terminal kinase, p38 kinase, and τ protein, including Bax, and upregulation of heme oxygenase-1 and Bcl-2. These beneficial actions of Seonghyangjeongkisan may be, at least in part, caused by the protective effect of Seonghyangjeongkisan against oxidative stress induced by amyloid β. Consequently, Seonghyangjeongkisan may have significant therapeutic potency as complementary therapy for delaying or improving the cognitive impairments associated with Alzheimer’s disease.
Footnotes
Acknowledgments
The authors thank Mr Ha Jin Jeong and Miss Sujin Lee for their help with this study.
SJ and B-SK were responsible for the study design, experiment, and manuscript writing. J-HK conducted the experiments, and SCP collected and analyzed data. All authors read and approved the final manuscript.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant of the Oriental Medicine R&D Project, Ministry for Health & Welfare & Family Affairs, Republic of Korea (B090068) and Graduate school of Oriental Medicine, Dongguk University, Seoul.
This study received permission from the Animal Ethics Committee of Dongguk University, Korea (No 2010-0738).
