Abstract
Iron is a trace element found in the diet and is essential for cognition and energy metabolism. Poor iron status is a concern for female athletes, who might not consume adequate levels of dietary iron and could experience elevated iron loss from physiologic processes associated with physical activity. Physical performance and cognitive decrements have been demonstrated in female athletes with iron deficiency anemia; both improve with increased iron intake. Two approaches for increasing iron intake in female athletes are the consumption of iron supplements and increasing dietary intake of foods high in iron to include iron-fortified foods. Both of these approaches have advantages and disadvantages that should be considered when treating female athletes with poor iron status.
Keywords
Iron is a trace element essential for many critical biological processes, including those involved in cognition and physical activity. Iron confers function through incorporation into proteins and enzymes. Many of these proteins and enzymes, such as myoglobin, cytochrome c, and hemoglobin are critical for the maintenance of human performance. 1 Poor iron status remains a worldwide public health concern and affects billions of people; in fact, iron deficiency is the most prevalent micronutrient deficiency disease in the world. 2 In the United States, iron deficiency and iron deficiency anemia, respectively, occur in up to 12% and 4% of women between the ages of 20 and 49 years. 3 Iron deficiency is defined clinically using cutoff values for a series of iron status indicators such as transferrin saturation and serum ferritin level. Iron deficiency anemia is clinically defined when individuals present with iron deficiency as well as hemoglobin values below a cutoff value.
Diminished iron status in athletes, sometimes referred to as “sports anemia,” was first reported more than 40 years ago. 4 Although the first studies reporting reductions in iron status in athletes were conducted using male volunteers,5,6 more recent work has described declines in indicators of iron status in female athletes following periods of physical training. 7 Similarly, research from our laboratory has detailed reductions in iron status in female military personnel during basic combat training, an 8- to 10-week course that includes a series of aerobic and muscle strength training activities.8–10
Poor iron status is of particular concern for female athletes. First, the prevalence of iron deficiency and iron deficiency anemia is greater in female athletes when compared to other demographic groups, 11 as they have difficulty maintaining iron balance because of insufficient dietary iron intake coupled with increased iron losses through mechanisms associated with heavy physical activity. 8 Second, iron deficiency and iron deficiency anemia are known to affect cognitive and physical function, which are paramount to athletic performance. 9 Iron supplementation and the consumption of iron-fortified foods could be useful for health professionals when recommending countermeasures for preventing poor iron status in female athletes. This review will detail recent studies of iron supplementation and fortification in physically active women, and compare and contrast the efficacy of these methods for maintaining iron balance.
Iron Balance in Female Athletes
Total body iron has been estimated at approximately 3.8 g in males and 2.3 g in females, although these estimates can shift based on body weight. 12 Although consuming adequate levels of iron through the diet is most important for maintaining positive balance, factors affecting iron loss also have a major impact on iron status (Figure 1). Athletes can have increased iron losses from gastrointestinal bleeding, which can occur following physical activity. In one report, median fecal hemoglobin concentrations increased by nearly 40% following marathon running; reported peak losses were 1.5 mg/g of feces. 13 Although the finite cause of increased gastrointestinal bleeding following physical activity remains unknown, contributors could include gut ischemia or aggravation of preexisting gut lesions. Sweat loss, a marginal, yet consistent factor for athletes, can affect iron status; estimates of iron loss from sweat range from 22 μg/L of sweat up to 6 mg/L but vary widely based on sweat collection techniques. 14 Furthermore, menstrual blood loss has been demonstrated to be a strong predictor of iron status, and female athletes prone to large menstrual losses can be at greater risk of iron deficiency or iron deficiency anemia. Although estimates of iron loss through menstruation vary and are largely driven by total menstrual blood loss, one recent report indicates mean daily iron losses ranging from 0.23 to 0.4 mg/d in a population of young women. 15
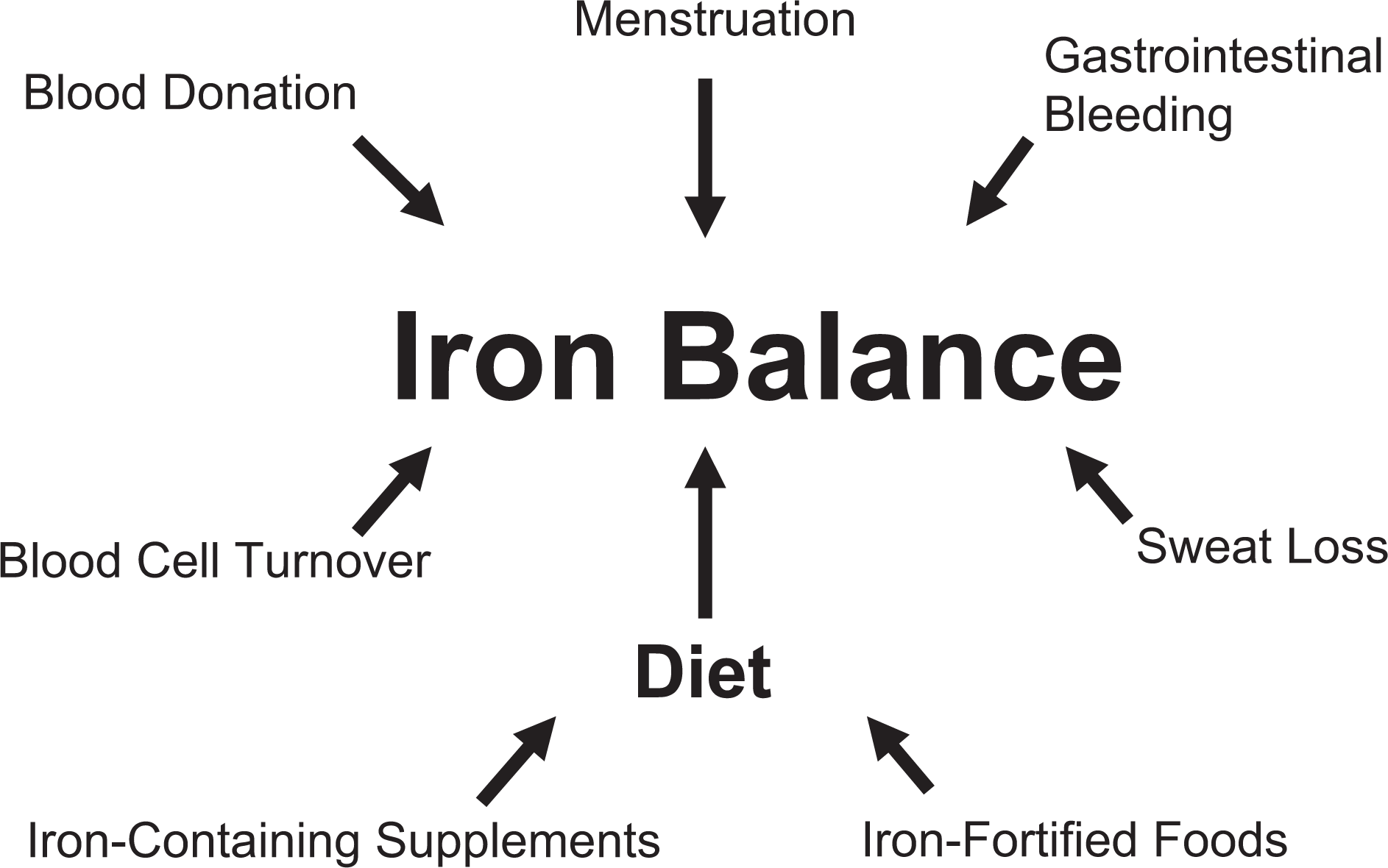
Factors contributing to iron balance
Hepcidin, a recently discovered hormone regulator of iron homeostasis, can provide another mechanism affecting iron balance in athletes. Hepcidin regulates iron balance by degrading ferroportin, a protein responsible for iron transfer from the enterocyte and other cell types, thereby reducing the biologically available iron supply. 16 Although the expression of hepcidin is affected by iron status, the synthesis of this protein is also affected by the presence of proinflammatory cytokines, such as interleukin-6. As interleukin-6 can rise following acute bouts of physical activity, 17 it is possible that heavy bouts of exercise could stimulate hepcidin synthesis, with subsequent effects on iron balance in athletes. The observation that both male and female athletes exhibit an elevation in inflammatory markers, including interleukin-6, and that urinary hepcidin levels rise following exercise supports the link between physical activity, inflammation, and diminished iron status.17–19
Current recommendations for dietary iron intake are 15 mg/d for girls between the ages of 14 and 18 years and 18 mg/d for women 19 to 50 years old. 20 The recommendation for pregnant women is higher, 27 mg/d, because of gestational needs of the fetus and blood losses that occur during delivery. The bioavailability of dietary iron is largely dependent on the composition of the diet. Heme iron, predominantly found in animal protein, is better absorbed at 10% to 35% than nonheme iron, found mainly in plant foods. Nonheme iron is generally more abundant than heme iron in the diet; however, less than 10% of nonheme iron is absorbed. 21 A number of dietary factors affect nonheme iron absorption. Ascorbic acid, fermented foods, and factors in meat, poultry, and fish all enhance absorption, 22 whereas phytate, polyphenols, and other minerals inhibit iron absorption. 23 A strong predictor of both heme and nonheme iron absorption is iron status. Both heme and nonheme iron absorption are tightly regulated at the enterocyte 23 resulting in the efficient recycling of body iron. Iron bioavailability affects an individual’s ability to obtain required levels of iron and could ultimately place certain populations, such as vegetarians, at higher risk for iron deficiency and iron deficiency anemia because of a lack of heme iron in the diet.
Supplementation for the Prevention of Iron Deficiency and Iron Deficiency Anemia in Female Athletes
Providing additional iron in the form of a dietary supplement is one method for achieving and maintaining optimal iron status in female athletes at risk for iron deficiency and iron deficiency anemia, and many studies have investigated this approach for preventing poor iron status. McClung et al 10 recently studied the effects of daily 100 mg ferrous sulfate supplementation on iron status and performance in female soldiers during basic combat training. In this randomized, placebo-controlled trial, those women who began basic combat training with iron deficiency anemia and who received the iron supplement exhibited a mean increase in hemoglobin concentration of 1.2 g/dL, which raised the group mean to 12.2 g/dL, a value above most clinical definitions of anemia. 10 These women also demonstrated significantly faster 2-mile run times when compared with the women with iron deficiency anemia who consumed the placebo. A 2-mile run has been demonstrated to correlate well with maximal oxygen uptake as a measure of endurance capacity. 24 Therefore, in this study, iron supplementation effectively increased hemoglobin concentrations above an anemic value, and presumably facilitated systemic oxygen transport, thereby contributing to improved performance in women who began the study with iron deficiency anemia. 10 In women who began basic combat training with normal iron status at baseline, decrements in serum ferritin and soluble transferrin receptor were observed during basic combat training, but these decrements were attenuated for those receiving the iron treatment, although running performance was not affected. 10 Attenuating declines in serum ferritin (an iron storage protein) and decreasing soluble transferrin receptor (an indicator of functional iron status) indicates improved maintenance of body iron stores. 25 Thus, iron supplementation provided a clear advantage for iron status and performance for females with iron deficiency anemia and although it prevented declines in iron status in females with normal iron status, a clear performance benefit was not apparent.
Although benefits of iron supplementation for female athletes with iron deficiency anemia have been well documented,1,26 advantages are less clear in nonanemic women with iron deficiency. Some studies have demonstrated that supplementation of the iron deficient population can affect iron status and improve maximal oxygen uptake 27 ; however, in female athletes with iron deficiency, iron supplementation has not always been demonstrated to improve physical performance. Recent studies have aimed to determine clinically relevant iron status biomarkers, other than hemoglobin concentration, that can provide a biochemical definition for the degree of iron deficiency that could affect performance in female athletes. For example, Brownlie et al28,29 demonstrated that soluble transferrin receptor can be a useful biomarker to assess female athletes with iron deficiency who would benefit from iron supplementation for improved performance. In one supplementation trial, 100 mg of ferrous sulfate or placebo was provided daily for 6 weeks along with a prescribed exercise regimen. When supplemented, women with initial soluble transferrin receptor values greater than 8 mg/L, indicating tissue iron deficiency, were able to improve time to finish during a 15-km cycling trial. However, supplementation was not associated with performance changes in women with baseline soluble transferrin receptor values less than 8 mg/L.28,29 Similarly, another recent study demonstrated that longitudinal increases in soluble transferrin receptor were associated with decreased performance on a 2-mile run. 9 Together these findings suggest that soluble transferrin receptor is a useful biomarker for identifying iron deficient, nonanemic, female athletes whose diminished tissue iron stores could be affecting performance.
Serum ferritin is another iron status biomarker that could distinguish iron deficient females who would benefit from iron supplementation. Murray-Kolb and Beard 30 conducted a randomized, placebo-controlled trial, and studied the effects of 160 mg ferrous sulfate daily for 16 weeks on iron status and cognitive function in a group of women (n = 113), aged 18 to 35 years. This study documented diminished performance on cognitive tests in women with poor iron status at the beginning of the study, when compared with women with normal iron stores at baseline. Longitudinal comparisons demonstrated that women with a significant increase in serum ferritin, following the treatment period, also exhibited improved scores on cognitive tests, when compared with those women with no change in serum ferritin, suggesting that improving iron stores could prove beneficial for maintaining or improving cognitive performance. 30 However, caution is warranted when using serum ferritin as a biomarker to determine iron status since other factors, such as inflammation, can confound this measure.
Although clear benefits of providing supplemental iron to the female athlete with iron deficiency are not always apparent, future research should investigate relationships between biomarkers of iron status and performance benefits with iron supplementation. It would be advantageous to define a profile to identify those female athletes with iron deficiency that could benefit from iron supplementation and to avoid iron supplementation for those who would most likely obtain no benefit. Adverse effects of iron supplementation have not been reported in many published trials, however, it should be noted that the intake of high doses of iron, often provided in supplements, can be associated with oxidant stress and free radical cellular damage. 31 Clearly, high doses of iron should be avoided in those with hemochromatosis or other iron overload disorders. Iron is not readily excreted by men and postmenopausal women, thereby increasing the chance for overload if excess iron is consumed. 32 Women of childbearing age with regular menses are less at risk, yet it is not uncommon for female athletes to suffer menstrual cycle disruptions. Iron supplements should be provided with caution in an effort to prevent negative outcomes associated with iron overload.
Fortification for the Prevention of Iron Deficiency and Iron Deficiency Anemia in Female Athletes
As compared with supplementation, fortification of foods with iron can provide a tolerable and more sustainable method of delivery to the female athlete. Many successful public health initiatives using food fortification efforts have improved micronutrient status, including iron status in various populations. For example, iron-fortified fish sauce significantly reduced iron deficiency and iron deficiency anemia among Vietnamese people, and in South Africa, the fortification of curry powder reduced iron deficiency anemia in women from 22% to 5%. 33 It is hypothesized that the prevalence of iron deficiency and iron deficiency anemia in the United States is lower than other nations in part because of the fortification of wheat flour with iron, which started in the 1940s. 34 Consistent with that observation, when Sweden stopped fortifying wheat flour with iron in 1994, there was a decline in iron intake, and consequently an increase in the prevalence of iron deficiency was observed. 21 These data suggest that fortification is an effective long-term approach for improving and maintaining optimal iron status in large populations. In addition, obtaining nutrients from food sources provides enjoyment for many people, 35 which can enhance compliance, compared with nutrient consumption from supplements. For example, compliance with consumption of iron-fortified food bars was more than 90%, compared with less than 60% for iron supplements in a group of female soldiers during basic combat training (J. P. McClung, unpublished data, 2010), suggesting a preference for the fortified food. Increasing iron intake could be required for the duration of a female athletic career because of dietary habits and repeated exposure to the physiologic stressors associated with exercise. As such, iron fortification of food products could be an effective solution to assure sustained adequate intake.
Recently, the provision of iron through an iron-fortified sports bar was studied in a randomized, placebo-controlled study. In the study, 36 female soldiers consumed the bar throughout basic combat training, and performance and biological outcomes were assessed. Bars were consumed twice daily and each contained 100 mg ferrous sulfate in an encapsulated form. Consumption of fortified bars improved iron status in females with iron deficiency anemia, significantly increasing hemoglobin concentration and mitigating the increase in soluble transferrin receptor observed in the placebo group. 36 Whereas females with iron deficiency anemia experienced improvements in iron status, declines in iron status were not prevented in women with normal iron status or iron deficiency when consuming the iron-fortified bar. Previous studies have demonstrated that consumption of 100 mg of ferrous sulfate daily in supplement form was able to attenuate declines in iron status during military training in females with normal iron status, 10 which could indicate that iron bioavailability might not have been optimal in the iron-fortified bars, especially as the amount of elemental iron provided in the bars was more than double that provided in supplement form in a similar study.10,36 Although ferrous sulfate is less bioavailable in food products than in capsule form, this source of iron is more bioavailable when compared with other iron fortification compounds, 37 making ferrous sulfate the form of iron preferred for both supplement and fortification use. It is often used in iron supplements but poses difficulties when incorporating it into food items as it decreases the shelf life of foods and can create flavors and color changes undesirable to the consumer. 37 Food scientists should strive to determine methods for including highly bioavailable iron during fortification while maintaining the integrity of the food product.
Consuming a well-balanced diet to include enhancers of iron absorption (ie, meat, poultry, fish, and foods high in ascorbic acid) increases the bioavailability of iron and improves absorption of iron from fortified foods. 33 Lyle et al 38 found that consumption of 4 to 6 ounces of meat per day with other foods high in iron was more successful in protecting iron stores when compared with daily supplementation with 2 levels of ferrous sulfate coupled with a diet low in iron. In this study, 38 groups were composed of women participating in a 12-week exercise program with normal iron status, iron deficiency, and iron deficiency anemia. Hemoglobin concentration increased significantly after 12 weeks in the group consuming meat and high iron foods, but not in the iron supplemented groups, suggesting the quality of iron ingested in foods was superior and better absorbed despite a lower quantity of total iron consumed. 38 A daily intake of 4 to 6 ounces of lean muscle foods (considered part of a healthy and balanced diet as stated by the food guide pyramid) 39 effectively maintained and even improved some indicators of iron status in females participating in moderate exercise in this study, demonstrating that iron balance can be achieved for those who are physically active through the consumption of highly bioavailable iron food sources.
Best Practices for Obtaining Recommended Iron Intakes
Athletes who are known to be at risk for iron deficiency and iron deficiency anemia should be educated regarding which foods they should consume for high iron bioavailability. These include lean meats, poultry, and fish. Although beans contain nonheme iron and phytate, an iron absorption inhibitor, they are a source of iron for vegetarians. Enhancers of iron absorption, such as tomatoes and citrus fruit, which contain high amounts of ascorbic acid, should be encouraged with these foods as part of a complete meal, whereas inhibitors, such as products high in phytate, should be limited and consumed at other times. Incorporation of iron-fortified foods, such as fortified cereal products, into a diet can be a safe and cost-effective method of increasing dietary iron intake. These foods can be of particular benefit for female athletes who abstain from eating meat. Although iron-fortified foods have been successful for the general population for improving iron status, further studies should aim to determine if this is a viable approach for reducing iron deficiency and iron deficiency anemia in female athletes.
Female athletes and military personnel with iron deficiency anemia have shown physical performance and cognitive decrements, both of which were ameliorated with the provision of iron supplements.10,30 For female athletes with iron deficiency anemia, supplementation can swiftly replete hemoglobin concentration, normalize iron status biomarkers, improve V
Footnotes
The opinions or assertions contained herein are the private views of the authors and are not to be construed as official or as reflecting the views of the United States Army or the Department of Defense. Any citations of commercial organizations and trade names in this report do not constitute an official Department of the Army endorsement of approval of the products or services of these organizations.
Acknowledgments
The authors acknowledge Holly L. McClung for her editorial review of this article.
Laura J. Bass and James P. McClung contributed to the conception and design of this article.
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The authors disclosed receipt of the following financial support for the research and/or authorship of this article: Financial support for the production of this manuscript was provided by the United States Army Medical Research and Materiel Command (USAMRMC).
