Abstract
Background
Mahaleb is an aromatic spice prepared from the fruit stone of the St. Lucie Cherry that is used as a flavoring agent in traditional Turkish and Middle Eastern baking. Immunodiagnostic kits for almond, which are based on polyclonal almond-specific IgG antibodies, have been shown to demonstrate considerable cross-reactivity with mahaleb as was incidentally discovered during a cluster of allergen-related food recalls in 2015.
Objective
Though acute allergy to almond is somewhat common, allergies to mahaleb have not been previously documented. However, based on antigenic similarity observed with almond-specific IgG, it is predicted that mahaleb nut proteins would exhibit some level of cross-reactivity with almond-specific IgE and may therefore potentiate acute allergic symptoms in individuals with food allergy to almond.
Methods and Results
Skin-prick testing using extracts compounded from pistachio, almond, and mahaleb revealed positive wheals measuring 8, 3, and 7 mm respectfully. Indirect enzyme-linked immunosorbent assay (ELISA) using plate-bound antigens prepared from pistachio, almond, and mahaleb revealed IgG positive responses to all three targets. ELISA and Western blot analysis performed using goat anti-almond polyclonal IgG demonstrated significant cross-reactivity between almond and mahaleb, but not to pistachio.
Conclusion
This is the first documented case of acute allergy to mahaleb, co-occurring in the context of plural tree nut allergies, providing novel evidence that mahaleb may pose a risk to nut-allergic individuals and indicating a need for awareness of spice contamination with nut and mahaleb residues.
Background
Allergies to tree nuts are relatively common, with prevalence estimates based on sensitization parameters ranging from 0.8-12.2%, depending largely on factors such as age and geological location. 1 Amongst 5,149 tree nut allergic patients registered with the Peanut and Tree Nut Allergy Registry, reported frequency of allergies to individual nuts in the US are walnut, 34%; cashew, 20%; almond, 15%; pecan, 9%; pistachio, 7%; hazel, Brazil, pine, macadamia, and hickory, less than 5% each, 2 values that are differ considerably with reported frequencies abroad. 1 Almond, a member of the Rosaceae family, is not a real tree nut, though it is customarily included in the general tree nut scheme. A number of allergens have been characterized in almonds, including Pru du 1 (PR-10, a defense protein), Thaumatin-like protein (TLP or Pru du 2), prolamins Pru du 2S and Pru du 3, profilin Pru du 4, an acidic ribosomal protein 60sRP Pru du 5, as well as cupins Pru du 6 (amandin) and Pru du γ-conglutin, 3 each of which belong to protein families that are relatively conserved amongst nuts. Accordingly, individuals with almond allergies frequently co-present with hypersensitivity towards distantly related tree nuts as well.4–6
Almonds (Prunus amygdalus var. dulcis) are members of the Prunus genus which consists of such fruits as peaches, plums, apricots, and cherries, including the St. Lucie cherry, (Prunus mahaleb). Mahaleb is the seed portion of the St. Lucie cherry and is used as a flavoring agent in baked goods of Turkish and Middle Eastern origin. Since the edible portions of almond and mahaleb are analogous tissues within the endocarps and the plants demonstrate close phylogenetic association, strong antigenic similarity is predicted for the two drupes. Indeed, commercial enzyme-linked immunosorbent assay (ELISA) kits based on almond-specific IgG antibodies configured in a sandwich format exhibit significant levels of cross-reactivity with mahaleb proteins, a property that has resulted in several errant food recalls in the UK and Canada.7,8 Despite these findings, there are no clinical reports documenting allergy to mahaleb in subjects with tree nut allergies. As food allergy symptoms are mediated by allergen-specific IgE antibodies, it stands to reason that IgE from an almond allergic individual might be expected to cross-react with homologous mahaleb proteins so as to induce allergic symptoms following exposure to mahaleb.
In 2014 and 2015, a number of food recalls occurred worldwide due to contamination of spices with ground nut and tree nut residues. However, a subset of the recalls associated with putative almond contamination of cumin spice in Canada and the UK.7,8 Initial routine immunodiagnostic-based and PCR-based methods performed by the UK Food Standards Agency (FSA) and the Canadian Food Inspection Agency (CFIA) for almond determination were unable to discriminate mahaleb residues from almond allergens, due to high a high degree of similarity of proteins and DNA sequences. However, subsequent confirmatory analysis of proteins and DNA using liquid chromatography-tandem mass spectrometry (LC-MS/MS)-based and PCR-based techniques were later advanced that uniquely identified mahaleb-specific markers.8,9 Interestingly, the same year, a number of unrelated spice related food recalls across Europe were shown to be definitively caused by almond contamination of paprika, 10 indicating that mahaleb contamination of spice was singularly limited to cumin products to date. Despite the antigenic similarity, no ensuing considerations have been made in connection to the mahaleb contamination, presumably due to the lack of clinical data demonstrating allergy to mahaleb. Herein, we report the first case of allergy to mahaleb co-occurring in the context of plural tree nut allergies.
Case Presentation
A 40-year old Caucasian female of French Canadian ethnicity presented with moderate pruritus on the thorax and oropharyngeal swelling shortly following ingestion of roughly 250 mg of mahaleb seed kernels. The symptoms resolved following self-treatment with diphenhydramine.
The kernels were not ground and not mixed with other ingredients, though it remains unknown whether it was raw or lightly roasted. The subject had not knowingly consumed mahaleb predating the reaction. The subject’s clinical history included 20+ years of pollinosis to birch and late onset food allergy to several types of nuts starting at age 19 which were later clinically confirmed by skin prick testing at age 24 and again at age 29. In particular, the subject had previously experienced anaphylaxis to walnut, pistachio, pecan, hazelnut, cashew, and Brazil nut marked by systemic hives, oropharyngeal swelling, facial swelling, and hypotension, and necessitating intervention with combined epinephrine and diphenhydramine, and in some instances, oral prednisone. The subject had additionally manifested food-dependent exercise induced-anaphylaxis following consumption of peanut and almond marked by oropharyngeal swelling, facial swelling and hives on the arms and chest that resolved with administration of diphenhydramine. There was no history of food allergies among immediate family members, though one immediate family member had a positive history for seasonal pollinosis.
At the time of presentation, the subject had been observing a strictly nut-free diet for many years, including elimination of all tree nuts and peanuts. The most recent prior episode of anaphylaxis (food-dependent, exercise-induced) had occurred roughly six and a half years earlier at age 34 following accidental ingestion of almond in a prepared food necessitating intervention with epinephrine and diphenhydramine. On account of the history of almond allergy and exposure to mahaleb, the subject was assessed for responsiveness to mahaleb proteins four days following the mahaleb exposure.
Allergen Extracts and Skin Prick Testing
Due to lack of diagnostic extracts for mahaleb testing, U.S. Pharmacopeia (USP) grade allergen extracts were prepared at IEH according to USP <795> Pharmaceutical Compounding—nonsterile preparations using raw pistachio, raw almond, and mahaleb kernels purchased from local grocery stores. Pistachio and almond extracts where prepared in house as a process control. The mahaleb kernels used in compounding were raw in appearance but may have been lightly roasted –no information was provided on the packaging material. Seed/nut material was finely ground and extracted with a Tris-buffered saline solution to isolate soluble seed/nut proteins. Pistachio was selected for this study based on severity of past responses and due to genetic dissimilarity to almond. Soluble protein concentrations were determined using the bicinchoninic acid (BCA) assay (Thermo Fisher Scientific, Wilmington, DE) with bovine serum albumin (BSA) as a reference standard. Approximately 5 µg of extracted protein was delivered to the volar surface of the subject’s lower arm using DUOTIP-TEST® II (ALK-Abello, Port Washington, NY) and applied using the rotation technique. The results of skin prick tests were scored according to the interpretation scheme supplied by the manufacturer, with wheal measurements taken at 30 min (Figure 1(A)). The wheal measured 8 mm for pistachio, 7 mm for mahaleb, and 3 mm for almond. In comparison, the vehicle control (Tris-buffered saline) measured less than 2 mm in diameter. At 45 min post-induction (Figure 1(B)), erythema was observed for both the pistachio and the mahaleb, and slight pseudopodia was observed for mahaleb. Almond responsiveness remained moderate, and vehicle control remained negative.

Skin prick testing: the ventral aspect of the subject’s forearm was challenged with allergen extracts prepared from pistachio (P), almond (A), mahaleb (M), and vehicle control (V) using the DUOTIP-TEST® II applied using the rotation technique. The results of the testing we assessed at (A) 30 min and (B) 45 min for responsiveness to epidermal challenge.
Western Blot Analysis
Proteins (50, 25, and 12.5 µg) from the raw pistachio, almond, and mahaleb extracts used in the skin prick tests were denatured and electrophoretically separated using 12% acrylamide gel and a mini protean II Apparatus (Bio-Rad Laboratories, Hercules, CA). Protein profiling as determined by 2 D sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) demonstrated numerous bands for all three extracts ranging in molecular size of 12-90 kDa, with shared banding patterns seen for almond and mahaleb (Figure 2). A duplicate gel was transferred to a 0.45 µm pore size nitrocellulose membrane (Bio-Rad Laboratories) using a Mini-Trans Blot® Cell (Bio-Rad Laboratories) then immunoblotted with goat-anti-almond IgG (Microbiologique Inc, Seattle, WA), a reagent raised against raw and roasted almond protein extracts as well as mouse anti-goat IgG HRP secondary (Microbiologique). The blot was resolved using the chromogen Tetramethylbenzidine (KP, Gaithersburg, MD) for ∼1-2 min. Whereas numerous bands were observed for the almond lanes (ranging from 14-100 kDa) and the mahaleb lanes (ranging from14-100 kDa), no blotting was observed for the 3 pistachio lanes using the polyclonal goat anti-almond IgG (Figure 3), thereby confirming shared antigenicity between mahaleb and almond, and no cross-reactivity with pistachio. Interestingly, a band corresponding to the molecular weight of amandine (42-46 kDa), a highly conserved legumin-type protein and major allergen recognized by almond-allergic patients, 11 was detected in both the almond and mahaleb lanes. However, follow up blastp analysis of amandine did not establish any positive hits for mahaleb, though at the time that this manuscript was being prepared, only 5 mahaleb protein sequences had yet been banked at NCBI. Interestingly, polyclonal goat anti-almond IgG reacted more intensely against mahaleb than almond (Figure 3), implying that the mahaleb extract was more antigenic than the almond extract -both of which were compounded at the same time and using identical protocols.

SDS-PAGE analysis of soluble protein extracts prepared using Tris-buffered saline solution from pistachio, almond, and mahaleb: 12.5, 25, and 50 µg of each target, respectively were denatured and resolved by electrophoresis using a 12% acrylamide gel.
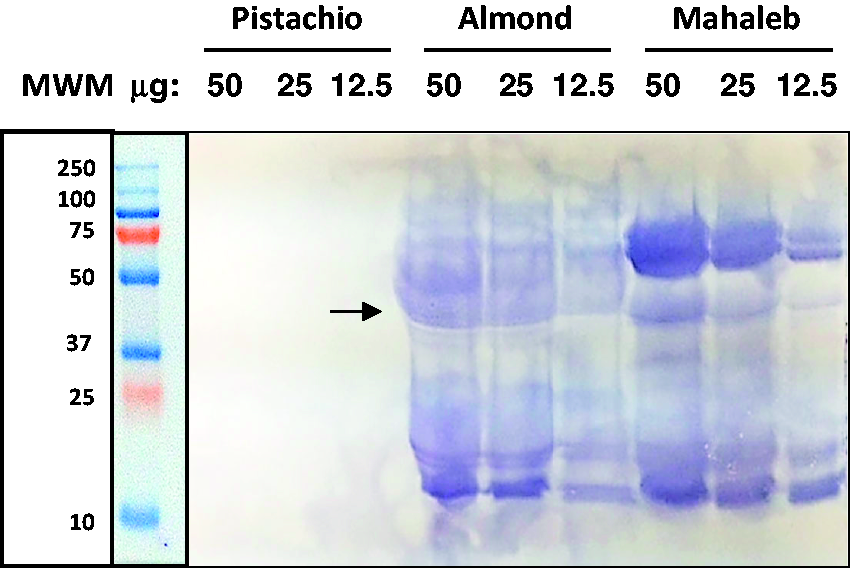
Western blot analysis of soluble protein extracts prepared using Tris-buffered saline solution from pistachio, almond, and mahaleb: 12.5, 25, and 50 µg of each target, respectively were denatured and resolved by electrophoresis using a 12% acrylamide gel then transferred to a nitrocellulose membrane and immunoblotted using goat anti-almond IgG (10 µg/mL). Arrow indicates putative band for amandin, the major allergen recognized by almond-allergic patients.
Enzyme-Linked Immunosorbent Assay
To further assess antigenicity, proteins (10 µg/mL) from raw pistachio, almond, and mahaleb extracts were plate-bound to polystyrene microwell plates using carbonate buffer. As a procedural control, goat-anti-almond IgG (Microbiologique) was used as a primary antibody at the indicated concentrations and resolved with mouse anti-goat IgG HRP conjugate (Microbiologique) at 1:1,000. The subject’s serum was obtained following routine phlebotomy and serially titered to enable assessment of IgG reactivity against all three targets, using anti-human IgG alkaline phosphatase (Sigma-Aldrich, St. Louis, MO) as secondary antibodies which were used at 1:2,000 dilution. Microwells (Costar 9017, Corning Life Sciences, Tewksbury, MA) were incubated 30 min at 37 °C with primary antibody/serum and 30 min at 37 °C with labeled secondary. Human IgG titers were resolved using phosphatase substrate (Sigma-Aldrich) dissolved in diethanolamine solution and 1 M sodium hydroxide as stop solution. Human IgG binding was determined using a Tecan spectrophotometer (Maennedorf, Switzerland) and 405/650 nm filter settings (Figure 4). The goat-anti almond IgG reactivity was resolved using 100 µL of TMB substrate (Microbiologique) and 1 M phosphoric acid as a stop solution and read using the 450/650 nm filter settings (Figure 5). Determination of specific IgE levels against target proteins was attempted but the values were too low to detect using the available enzyme-conjugated detection reagents, thus allergen specific IgE levels could not be reported. Though sero-reactivity for all three targets was observed, positive IgG signal was seen at serum dilutions >1:100 for pistachio and mahaleb, whereas serum IgG reactivity to almond proteins was roughly 3-fold lower. It is critical to note that while there are diagnostic tests for food sensitivities based on the detection of allergen-specific IgG moieties, the use of these tests is not supported by the American Academic of Allergy, Asthma, and Immunology due to the lack of supporting evidence to support its use. Paralleling the results of the Western blot, goat-anti-almond IgG equally detected almond and mahaleb proteins (Figure 5), while low cross-reactivity using the anti-almond IgG was detected against the pistachio proteins.

Indirect ELISA analysis of human IgG against almond, pistachio, and mahaleb proteins using the subject’s serum diluted at the indicated concentrations. Error bars refer to the standard error of mean (SEM) from triplicate wells.

Indirect ELISA analysis of goat-anti-almond IgG against almond, pistachio, and mahaleb proteins using antibody diluted at the indicated concentrations. Error bars refer to the standard error of mean (SEM) from triplicate wells.
Conclusions
In January 2015, IEH participated in a market-wide analysis of numerous spice samples following an unprecedented number of voluntary food recalls in the US originating from ground paprika and cumin contaminated with peanut and tree nut residues. 12 Confirmation testing performed at IEH on “almond”-positive spice samples provided by the FDA using mahaleb-specific PCR did not reveal contamination with mahaleb (data not shown), consistent with confirmation testing performed on recalled spice across Europe and the UK.9,10 This in contrast to the testing performed in Canada and the UK where mahaleb rather than almond was found to be the true source of contamination in recalled spice samples, which were then later rescinded.7,8
In November of 2015, we identified a subject with a putative mahaleb allergy. The subject’s history of tree nut allergies, including almond allergy, was indication to pursue testing. Though oral challenge was declined, positive skin prick testing and ELISA performed using the subject’s serum IgG revealed significant reactivity to mahaleb that compared in intensity to that of pistachio, a tree nut unrelated to almond or mahaleb for which the subject had a history of severe allergy. In comparison, reactivity to almond protein was modest, both in the skin prick testing and the almond-specific IgG levels as determined by indirect ELISA. Given the subject’s earlier response to ingesting mahaleb, it appears that the subject was previously sensitized to mahaleb before the testing was performed. . Based on the skin prick test results, it remains possible that the reactivity directed against mahaleb proteins was specific for mahaleb and not cross-reactive with almond proteins, implying a possible oral allergy syndrome related to the subject’s birch pollinosis. Alternatively, it remains possible that mahaleb proteins, especially those ranging in size of 55–65 kDa, may simply be more antigenic than almond proteins, as supported by the data depicted in Figure 3 using polyclonal goat antibodies directed against almond proteins. This later interpretation is not incongruous with the findings of Noble et al., who have shown that IgE present in the serum of an almond allergic individual exhibited reactivity with mahaleb, both in ELISA and in Western Blot, though in their analysis, the authors were able to show competitive binding of antibody against both targets. 13
However, cross-reactivity with nut allergens is frequently observed in the context of birch pollen allergy, 1 and elevated serum levels of birch pollen-specific immunoglobulin IgE are reported for individuals with oral-allergy syndrome towards plant-derived proteins. 14 Importantly, the results of this study do not preclude the possibility of cross-reactivity as it is important to consider that the nut extracts used for the SPTs were prepared from apparent raw nut materials, and it remains plausible that roasted nut extracts would have elicited different outcomes. Moreover, there is some uncertainty as to the degree of thermal processing that the mahaleb kernels were subjected to prior to purchase. In this line, roasting is known to enhance the antigenicity of nut allergens through the formation of glycation end products through the Maillard reaction and possible generation of neo-epitopes, enabling improved IgE-binding. 15
To better understand the scope of antigenic similarity between almond and mahaleb, we assessed the ability of polyclonal IgG raised against almond protein (both raw and roasted proteins) that is used in immunodiagnostic kits for the detection of almond residues in food. The antibody was assessed both in indirect ELISA and Western blot analysis. Reactivity towards almond and mahaleb proteins was essentially identical using this reagent, whereas little (∼5 times less) or no activity was seen towards pistachio proteins, thereby indicating that the polyclonal anti-almond IgG was highly specific for seed material derived from the Prunus genus.
Importantly, extensive cross-reactivity among the different members of the Rosaceae family, in particular, within the same Prunus genus, should be expected based on genetic similarity 16 and therefore potential clinical allergy to other members of the family should not be overlooked, especially if the reported reaction is confirmed. 17 Due to the significant level of cross-reactivity with respect to the almond specific polyclonal IgG as well as the subject’s responsiveness to mahaleb in the context of a known allergy to tree nuts including almond, we bring attention to clinicians, regulatory bodies, and food manufacturers the need to address the concerns of potential mahaleb contamination in the food supply as well as the need to develop food testing tools that can selectively identify the presence of mahaleb residues in food. 12
Footnotes
Authors’ Note
This work was presented at the Conference: International Association for Food Protection (IAFP)’s European Symposium on Food Safety, May 2016, Athens, Greece.
Acknowledgment
We thank Dr Walter Hill for his assistance with manuscript preparation.
Author Contributions
L. B., J. M., and H. J. performed the experiments. S. M. G. oversaw the research, and the work was performed in the research facilities owned and operated by M. S.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The subject provided informed consent for participation in this study and proof of consent can be provided on request.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: IEH and MEI are a privately owned company, no external funding or government contract funds were used in this study.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Consent to participate and for publication was obtained from the study subject. A copy of the written consent is available for review on request. Procedures followed were in accordance with the Helsinki Declaration as revised in 2013.
