Abstract
Objective
Endoscopic skull base surgery is advancing, and it is important to have reliable methods to repair the resulting defect. The objective of this study was to determine the failure pressures of 2 commonly used methods to repair large dural defects: collagen matrix underlay with fibrin glue and collagen matrix underlay with polyethylene glue, as well as a novel repair method: fascia lata with nonpenetrating titanium vascular clips.
Methods
The failure pressure of the 3 dural repairs was determined in a closed testing apparatus. Defects in porcine dura were created and collagen matrix grafts were used as an underlay followed by either fibrin glue (FG/CMG) or polyethylene glycol glue (PEG/CMG). A third condition using a segment of fascia lata was positioned flush with the edges of the dural defect and secured with titanium clips (TC/FL). Saline was infused to simulate increasing intracranial pressure (ICP) applied to the undersurface of the grafts until the repairs failed.
Results
The mean failure pressure of the PEG/CMG repair was 34.506 ± 14.822 cm H2O, FG/CMG was 12.413 ± 5.114 cm H2O, and TC/FL was 8.330 ± 3.483 cm H2O. There were statistically significant differences in mean failure pressures among the 3 repair methods.
Conclusion
In this ex vivo model comparing skull base repairs’ ability to withstand cerebrospinal fluid leak, the repairs that utilized PEG/CMG tolerated the greatest amount of pressure and was the only repair that exceeded normal physiologic ICP’s. Repair methods utilizing glues generally tolerated higher pressures compared to the novel repair using clips alone.
Background
Cerebrospinal fluid (CSF) leaks occur in various ways, but large defects are often caused after tumor resection. 1 Especially for situations of large defect and high flow CSF leaks, it is important to have reliable methods to close these leaks to prevent complications such as headache and meningitis.2,3 Multiple repair techniques have been described, and the selection of repair technique is often dictated by the size, location, defect etiology, and surgeon preference. 4 In general, however, most repair methods use some combination of multilayer reconstruction with grafts, flaps, glues, and temporary reconstruction support. 4
A previous study in an ex vivo model suggested that a combination of collagen matrix underlay with fibrin glue created a more effective seal than other tested repair methods when used in a simulated CSF leak repair model. 5 However, the findings suggested that even the most robust repair tested could not withstand physiologic causes of increased intracranial pressure (ICP) such as straining or sneezing. Therefore, it is important to develop more robust methods of reconstructing the skull base and preventing CSF leaks after surgery. Recently, titanium clips were shown to be effective in repairing the carotid artery and achieving hemostasis in a major vascular injury model. 6 As a result, this study sought to determine whether the use of these titanium clips to standard dural repairs would augment the pressure at which they leaked when compared to more common repair methods. Three conditions of dural repair for large skull base defects were evaluated: collagen matrix graft with fibrin glue (FG/CMG), collagen matrix graft with polyethylene glycol glue (PEG/CMG), and titanium clips with fascia lata (TC/FL). The null hypothesis was that there was no difference between the repair methods.
Materials and Methods
Testing Apparatus
The test design was a modification of an apparatus used in a previous study.5,7 The device was fabricated from 2″ polyvinyl chloride (PVC) piping as depicted in Figure 1. A small hole was drilled on the side of the end cap, with a 3-way stop cock used to infuse saline solution and monitor chamber pressure, simulating increasing ICP.

Apparatus constructed in CAD software. Displayed in exploded view to show all parts used for construction. PVC, polyvinyl chloride.
A silicone brain was positioned under the simulated cribriform plate within a cylindrical tube. A section of the cribriform plate with 30 mm × 25 mm opening was modeled according to a real computed tomography scan of the skull base (Able, Lexington, MA), imported into CAD (computer-aided design) software (3D Systems, Rockhill, SC), and printed in polycarbonate (Airwolf, Costa Mesa, CA). A second dural support disk was prepared with an identical opening and positioned flushed to the opening of the simulated cribriform plate resection.
The cavity pressure was monitored with a pressure transducer (AMTEK, Inc., Ajman, UAE), and its output was transcribed directly onto an excel spreadsheet using WindaqXL (DATAQ, Akron, OH). The transducer was calibrated in mm Hg, and all measurements were converted to centimeters of water (cm H2O).
Dura and Repair Material
Porcine dura was used because of its similar mechanical properties to human dura. 8 Porcine dura mater and fascia lata were harvested from euthanized pigs and placed in saline and stored at 4°C. All experimentation was conducted within 5 days of retrieval to avoid degradation of the dura. The repair material tested included collagen matrix, fibrin glue, titanium clips, and polyethylene glycol glue. The dural defects were all uniformly cut to 24 mm × 19 mm dimensions. Then, a 40 mm × 34 mm collagen matrix was positioned as a dural underlay graft with overlap over the edges of the dural defect, which mimics actual reconstructions. The novel repair with titanium clips was performed with a 24 mm × 19 mm piece of fascia lata rather than collagen matrix. This decision to use fascia lata with these dimensions was used to imitate the standard fashion of vessel repair, where the vessel edges are end-to-end without overlap.
Experimental Technique
A prepared dural segment with the defect was positioned on the dural support disk and centered and then covered by a second disk (Figure 1). Studs secured the disks to prevent rotational displacement of the dural specimen. The positive control was dura with a 24 mm × 19 mm hole, and the negative control of the study was intact dura.
Repair Tests
Three rhinological repairs were tested: PEG/CMG, FG/CMG, and TC/FL. Each repair was performed 5 times, and dura and repair materials were replaced after each experiment. The saline solution was pumped into the side port at a rate of 30 mL/h, to mimic the normal production of intracranial CSF. 9 Infusion continued throughout the cavity and applied pressure to the inferior surface of the graft until the repair failed, simulating a CSF leak. Repair failure was defined as the inability of the repair to maintain pressure or a visible fluid leak around the defect edges.
Glue repair
FG and PEG were prepared and reconstituted according to manufacturer’s instructions. Collagen matrix grafts were placed through the dural defect in an underlay fashion with at least 3 to 5 mm overlap with the defect edges. FG or PEG glue was applied liberally across the surface of the graft and over the defect edges and was left to cure for 2 minutes.
Titanium clip repair
A segment of fascia lata was cut 24 mm × 19 mm and positioned underlay to the edges of the dural defect. The edges of the defect and fascia lata graft edges were brought together with everting forceps and were then secured with titanium clips. The clips were placed 1 mm apart around the periphery of the defect. No additional glue was used over the edges, as seen in Figure 2.

Dural repair technique with fascia lata and titanium clips.
Results
The mean failure pressures are summarized in Table 1. The mean failure pressure of the negative control was 207.411 ± 79.259 cm H2O, while the positive control with a large defect yielded a mean failure pressure of 0 cm H2O. In descending order, the mean failure pressures of the dural repairs were as follows: PEG/CMG was 34.506 ± 14.822 cm H2O, FG/CMG was 12.413 ± 5.114 cm H2O, and TC/FL was 8.330 ± 3.483 cm H2O.
Summary of Failure Pressures in cm H2O for Each Trial.

Abbreviations: CI, confidence interval; FG/CMG, fibrin glue/collagen matrix graft; PEG/CMG, polyethylene glycol glue/collagen matrix graft; SD, standard deviation; TC/FL, titanium clip/fascia lata.
Analysis of variance between groups yielded an F-Ratio of 27.7 (P < .000001). Thus, the null hypothesis (that there is no difference between failure pressures) was rejected. A post hoc Tukey’s test (Figure 3) demonstrated that the negative control was significantly different from all 3 repair techniques (TC/FL: P < .0005, PEG/CMG: P < .0014, and FG/CMG: P < .0006). Among repair methods, only PEG/CMG proved to be significantly different from the other 2 repair methods (TC/FL: P < .0049 and FG/CMG: P < .0136). Again, the null hypothesis (that there is no difference between the PEG/CMG and both the TC/FL or FG/CMG repairs) was rejected. There was no statistically significant difference between the TC/FL and FG/CMG repairs (P < .1784).
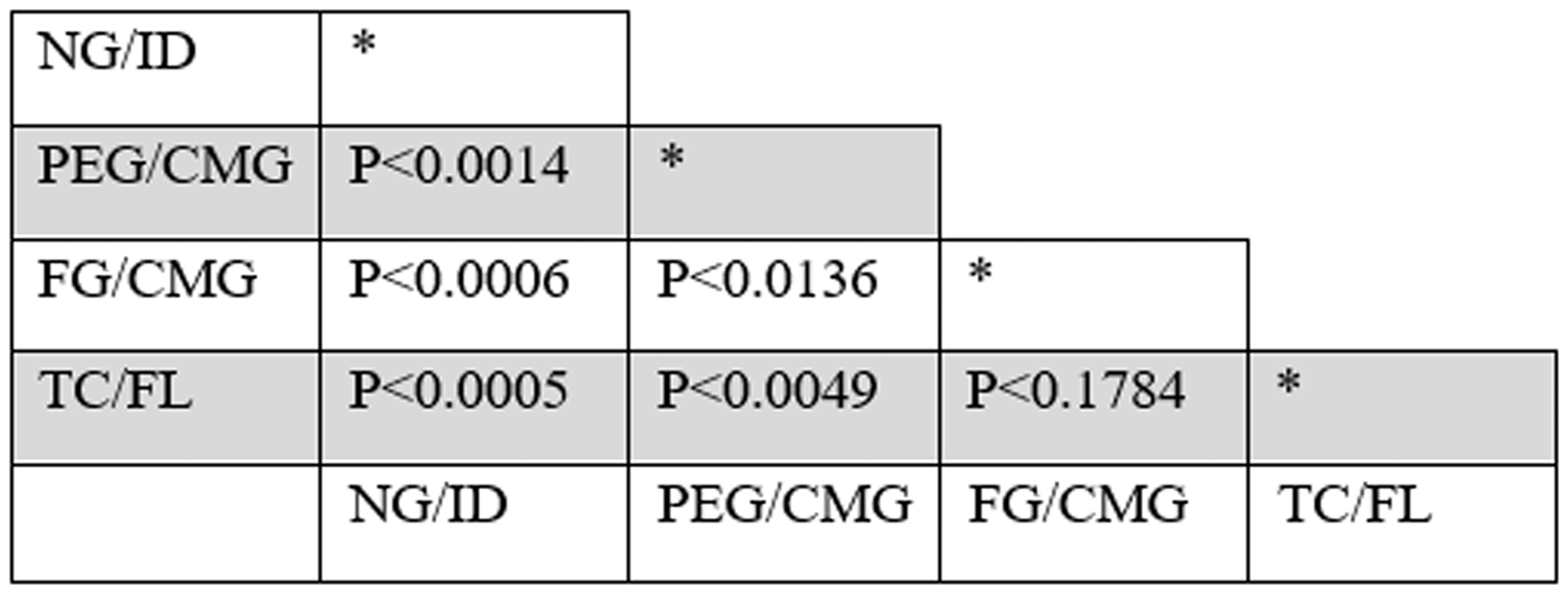
Post hoc Tukey’s test P values of dural repair failure pressures. FG/CMG, fibrin glue/collagen matrix graft; NG/ID, no glue intact dura (negative control); PEG/CMG, polyethylene glycol glue/collagen matrix graft; TC/FL, titanium clip/fascia lata.
Discussion
There are numerous endoscopic techniques utilized for the repair of large skull base defects. 10 Many small defects are caused by trauma and are usually self-limited; however, conservative measures are inadequate for large dural defects resulting from tumor resection.10,11 Most importantly, these large skull base repairs should be able to withstand the normal adult ICP in supine position (9.5–20.4 cm H2O) and pressure changes during physiologic situations such as coughing or straining. 12
Endoscopic management of small CSF leaks and encephaloceles has a high degree of success, exceeding 90% after the first repair attempt. 13 However, large dural defects created during tumor removal with an associated high CSF leak flow rate can prove more difficult to repair and have higher rates of recurrent CSF leak. 14 Larger grafts are necessary to reconstruct these skull base defects and to tolerate normal spikes or increases in ICP until healing occurs.
Efforts are made to help keep repairs in place, such as removal of a rim of mucosa surrounding these skull base defects, ensuring sufficient overlap of the graft over defect edges. Furthermore, a variety of different materials can augment the reconstruction including autologous mucosal grafts, flaps, fat plugs, fascia, and artificial materials like collagen matrix, hydroxyapatite, and so on.15–20 Once the graft has been placed, different sealants can be used such a PEG and FG. There is a paucity of literature and clinical studies assessing the efficacy of sealants and in fact, there are conflicting results. More commonly, it is believed that fibrin sealants lead to a reduction in overall postoperative CSF leak complications and length of postoperative hospitalization, though the need of sealants with nasoseptal flaps has been challenged.21,22 The use of retrospective studies to analyze the strength of these dural repairs are subject to shortcomings such as selection bias, thus ex vivo experimentation offers the opportunity for studies in a controlled and reproducible setting.
Fibrin sealants are commercially available and contain 2 key ingredients: fibrinogen and thrombin.23,24 When applied on the surface of an overlay graft, it can activate the clotting cascade, stimulate angiogenesis, and mitigate hematoma formation.23,24 Fibrin glue facilitates generation of a fibrin clot to adhere to tissue, with additional hemostatic and adhesive abilities. Previous work reported that collagen matrix and fibrin glue performed better than nonglue repairs in an ex vivo model. 5
Polyethylene glycol is composed of completely synthetic, resorbable material and has been approved for achieving watertight closure when applied to the surface of skull base repairs. 25 Clinical studies have demonstrated that it is both safe and effective in practice and may be preferred by some clinicians because it requires very little preparation and forms a watertight hydrogel layer within seconds of dispensing.25–29 This seal is reported to be robust and can tolerate irrigation and gentle suction maneuvers with less concerns of graft dislodgment than in the absence of a sealant. As a result of the described favorable profile, it has been widely used in efforts of minimizing postoperative CSF leak. 30
The nonpenetrating titanium clip system permits reapproximation of vascular edges without penetrating the vessel wall.31–34 It has demonstrated efficacy for use in major vascular injuries in skull base surgery and has recently been approved for dural reconstruction for its potential in preventing postoperative CSF leaks.6,35 Thus far, there is evidence that the clips can be used to repair leaks in the spinal dura,36–39 as well as one case report that demonstrated the use of titanium clips in repairing a CSF leak at the skull base. 40 Skull base repairs often fail at the leading edge of the graft. Therefore, these nonpenetrating clips have theoretical potential to secure repairs in place while they heal and enable dural closure without creating any holes.
Although none of the repair techniques sustained failure pressures close to that of intact dura, the PEG/CMG repair withstood considerably higher failure pressures than the other repair techniques with a mean failure pressure of 34.506 ± 14.822 cm H2O. From a clinical perspective, the PEG/CMG repair was the only repair in this study that reasonably withstood adult supine ICP. In comparison, the TC/FL and FG/CMG repairs were not statistically significantly different from each other with failure pressures that would have marginally tolerated adult supine ICP.
The improved performance by PEG/CM is most likely explained by its ability to form a hermetic, water tight hydrogel layer immediately upon dispensing in the ex vivo model. Conversely, FG depends on the intrinsic coagulation cascade to create a tight adhesive closure, which could not be recreated in this ex vivo model. There are no head-to-head in vivo studies that compare the efficacy of these glues in anterior skulls base reconstructions; however, for lateral skull base repairs, it is notable that the present findings are consistent with the observations seen by Than et al. who demonstrated that the incidence of CSF leakage was less when dural closures were augmented with PEG compared to FG. 41
Although the titanium clips have been used to repair CSF leaks in the spine successfully, there are some differences between the thicker dura in the spine and thinner dura at the skull base, especially in the cribriform region. Previous case studies using spinal dura describe an end-to-end approximation of dura before closure with titanium clips, but it is conceivable that an underlay closure could have helped seal the repair. Furthermore, once the clips are deployed they cannot be repositioned or reused, otherwise there is the risk of dural laceration and graft dislodgment. 36 In this study, it is also possible that the clips were not placed correctly. In fact, there are no data for the number of clips or distance between clips that optimize dural repairs and prevent CSF leaks. It is also possible that use of the clips may be more effective in repairing small dural defects or when combined with a sealant to improve the ability to withstand leaking. Further studies are needed to elucidate the role of these clips in skull base repair.
This study has several limitations. Even though this was a controlled setting with standardized defects, there was still some variability among the individual trials for each glue and clip repair. Although there were efforts to apply consistent and even amounts of glue, this was not able to be measured quantitatively. Dura mater and fascia lata used were obtained from different pigs, so specific mechanical properties of each specimen may have varied among trials, although the investigators were careful to ensure standardization of handling and preparation of the specimens. Although the failure pressures using titanium clips and fascia lata tolerated lower pressures than those using glues with collagen matrix grafts, it is possible that these findings could be attributed to the differences in repair material used. An end-to-end closure was used in the clip repair group in accord with the spinal dural closure literature, though it is possible that titanium clips would have improved performance using an underlay closure, which was not studied and is a potential aim of future research. Recent studies have demonstrated a decreased risk of CSF leaks with pedicled nasoseptal and fibrin sealant; 42 however, this ex vivo model could not assess the added strength that vascularized tissue provide. Finally, it is likely that in vivo blood coagulation and wound healing would bolster the strength of all the repairs, but the degree to which it would affect the repairs remains unknown.
Conclusions
This study compared the mean failure pressures of 3 rhinological repairs for CSF leaks: collagen matrix with fibrin glue, collagen matrix with polyethylene glycol glue, and a novel use of titanium clips with fascia lata. Repairs that used PEG/CMG tolerated the greatest amount of pressures in this ex vivo model and was the only repair method in this study to withstand adult supine ICP. TC/FL tolerated the lowest pressures compared to repairs utilizing glues. The direct clinical significance of these results remains unclear, and future studies should aim to assess these different repair techniques in in vivo settings.
Footnotes
Authors’ Note
This study was presented as an oral presentation at the Combined Otolaryngology Spring Meeting through the Triological Society, May 1–5, 2019, Austin, TX.
Acknowledgments
The authors thank 59th Clinical Research Division, Lackland Airforce Base for supplying porcine dura.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Philip G. Chen, MD, is a consultant for Stryker Corporation and Medtronic. Commercial products mentioned in this presentation are not intended to constitute an endorsement by the U.S. Air Force or any other federal government entity.
Ethical Approval
This study was deemed exempt from direct Institutional Review Board or Institutional Animal Care and Use Committee (IACUC) review; however, IACUC sanction was obtained.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
