Abstract
Background
External nasal dilator strips are used as nonpharmacological therapy to reduce snoring and daytime sleepiness. In a product improvement initiative, a marketed strip (BRNS) and 2 prototype nasal strips were evaluated.
Objective
To compare the nasal patency and multiple-use dermal tolerability of the BRNS and prototype nasal strips using both objective and subject-reported outcome measures.
Methods
Two studies were conducted separately in healthy volunteers ≥18 years of age. A single-day nasal patency randomized crossover study assessed minimal cross-sectional area (MCA; second restriction) and nasal volume (using acoustic rhinometry); nasal inspiratory flow and resistance (using posterior rhinomanometry); and subject-reported evaluations of the BRNS compared with the butterfly strip and teardrop strip prototypes. A single-center, randomized, controlled, parallel-group, evaluator-blinded study assessed dermal tolerability of the BRNS and the butterfly strip over 7 consecutive nights of use, using the Dermal Response Scale (DRS) and subject-reported comfort and ease of removal.
Results
In the Patency study (N = 50), all 3 strips demonstrated significant improvement from baseline in MCA, nasal volume, and nasal flow parameters (resistance and peak flow). The prototype strips demonstrated significantly more improvement in nasal volume than the BRNS, and the butterfly strip showed significantly more improvement in MCA than the BRNS; all strips were similar with respect to nasal flow and subject-reported nasal breathing outcomes. In the Dermal Tolerability study (N = 82), all subjects scored 0 (no evidence of irritation) on the DRS at all 7 morning assessments; the BRNS was numerically, but not significantly, superior to the butterfly strip on subject-reported outcomes.
Conclusion
The Patency study demonstrated significant improvement from baseline in nasal dimensions and flow for all 3 evaluated strips; between-strip differences were confined to nasal dimensions. Both the BRNS and butterfly strip were generally well tolerated, with no evidence of dermal response over 7 consecutive nights of use.
Keywords
Introduction
External nasal dilator strips, a nonpharmacological therapy for chronic nasal congestion and snoring, are also used by athletes to improve respiration and exercise capacity. 1 One example is the Class I medical device Breathe Right® Nasal Strip (BRNS; GlaxoSmithKline Consumer Healthcare [GSKCH]; Warren, NJ), which consists of medical tape coated with medical-grade acrylate adhesive for skin application, two plastic springs, and a top coating that is applied across the bridge extending to the alar creases. The BRNS is marketed over the counter for the relief of nighttime nasal congestion and snoring.
The BRNS has been found to reduce snoring and associated symptoms (including daytime sleepiness and morning dry mouth) 2 and to increase minimum cross-sectional area (MCA) and nasal volume in the anterior nasal cavity in patients with chronic nasal congestion, compared with the decongestant nasal spray xylometazoline. 3 In contrast to xylometazoline, the BRNS had little effect on the cross-sectional area and volume of the posterior nasal cavity; however, both treatments significantly increased peak nasal inspiratory flow (PNIF). 3
Studies in healthy volunteers have documented improvements in functional nasal breathing parameters, including increased MCA and volume of the nasal passage, increased inspiratory and expiratory nasal airflow, and reduced nasal resistance.4–6
As part of a continuing product improvement and development program, 2 new prototype nasal dilator strips were developed. One prototype has an asymmetric butterfly shape that adheres to the cheek instead of the nose flare; this strip was expected to pull outward on multiple areas of the nose rather than in a straight line across the nose like the BRNS. The second prototype was shaped like a teardrop. Here, we report on 2 studies that evaluated nasal breathing functional parameters (nasal patency) and dermal tolerability, using objective and subject-rated outcome assessments, with the developmental prototype strips compared with the marketed BRNS.
Methods
The studies of nasal patency (Patency study) and of multiple-use dermal tolerability (Dermal Tolerability study) were conducted separately; each study enrolled healthy volunteers and were conducted in accordance with the International Council for Harmonisation Guideline for Good Clinical Practice and the ethical principles specified in the Declaration of Helsinki. The study protocols were approved by an Independent Ethics Committee before initiation, and all subjects provided written informed consent.
Patency Study
The Patency study (ClinicalTrials.gov identifier NCT01105949; conducted from September to October 2009 in Cardiff, UK) was a single-center, 1-day, randomized, single-blind, crossover study; the primary objective was an acoustic rhinometry comparison of the effects on MCA (second restriction) and total nasal volume during the use of the different nasal dilator strips. Treatments included the marketed BRNS (tan), a prototype asymmetric butterfly design nasal strip (butterfly strip, marketed in the United States as the Breathe Right Advanced Strip from 2010 to 2014), and a prototype teardrop design nasal strip (teardrop strip), all supplied by GSKCH. The nasal strips were applied by a member of the study site staff and worn for a maximum of 2 hours for each evaluation.
Secondary objectives were to compare the effects of the 3 nasal strips on 2 dynamic measures of nasal airway breathing (NAB) and on subject-reported measures of NAB, described below.
Study design
The single-day evaluation comprised 3 periods, each with a baseline and treatment phase; subjects were randomized with respect to the order of treatments (nasal strips) applied during the 3 periods according to a sponsor-provided randomization schedule. During the baseline phase of each period (with no nasal strip applied), objective measurements were performed in this order: acoustic rhinometry, posterior rhinomanometry, and PNIF. The treatment phase commenced immediately with the application of the nasal strip, followed by the performance of the 3 objective measurements in the same order. The second and third periods followed per the randomized treatment sequence, using the same procedure, with a 30- to 60-minute washout period between periods. Subject-reported NAB assessments were conducted during each period, before and after the application of the indicated nasal strip.
Study population
Subjects aged ≥18 years, in good general health, and capable of understanding and completing study procedures and providing informed consent were eligible. Potential subjects were excluded if they had a current respiratory infection (cold or flu); a history of perennial/allergic rhinitis or rhinitis medicamentosa; evidence of nasal polyps, significant nasal tract malformation (eg, severe deviated septum, where surgery is indicated, or concha bullosa documented by anterior rhinoplasty), skin irritation, allergic contact dermatitis, active immunologic skin disease (eg, psoriasis), skin cancer, or other skin conditions; bacterial sinusitis within 2 weeks prior to baseline; use of menthol-containing oral products within 2 hours prior to any study assessment; use of antibiotics or α-adrenergic agents within 1 week prior to baseline; or use of glucocorticosteroids within 1 month prior to baseline.
Study outcomes and assessments
The primary efficacy end points were change from baseline for MCA (second restriction) and total nasal volume (static measurements) of the nose using acoustic rhinometry. Acoustic rhinometry, conducted separately on each nostril using a GM Instruments (Kilwinning, UK) Model A1 Acoustic Rhinometer, captured MCA and location (distance behind nares) of the first restriction (0–3 cm of nasal cavity behind nostril), MCA and location of the second restriction (0–5 cm behind nostril), and nasal volume of the first 3 cm of the anterior nasal cavity behind the nostril.
Secondary end points included changes from baseline for NAB using posterior rhinomanometry and PNIF, and changes from baseline for subjective questions about NAB.
Posterior rhinomanometry measured the combined inspiratory resistance of both nasal passages using a GM Instruments Model NR6-2 rhinomanometer. The average of mean nasal inspiratory nasal resistance (Pa/cm3 sec) over 4 breaths was calculated, followed by a repeat measurement over 4 breaths. If the coefficient of variation (CV) of those 2 values was ≤10%, the mean of those measures was used; if the CV was >10%, another 2 measurements were taken; this procedure was repeated until a pair of measurements produced a CV ≤10%.
PNIF was conducted using the In-Check portable nasal flow meter (Clement Clarke International Limited, Essex, UK); the same numbered meter was used for all measurements in each subject. Three measurements were taken for each subject, with the third measurement used for data analysis.
With respect to subject-reported outcomes, each subject rated the ease of nasal breathing before and after nasal strip application using a 0 to 100 mm visual analog scale (VAS; range: 0 = extremely difficult; 100 = extremely easy). In addition, each subject rated the change in breathing after nasal strip application (compared with no strip) on an 11-point numerical rating scale (range: −5 = much worse; 0 = no change; 5 = much better).
Safety was assessed based on the tabulation of adverse events (AEs) and serious adverse events (SAEs).
Statistical methods
A previously completed study of the marketed BRNS led to the estimation that 50 subjects would provide 90% power to detect a difference of 0.0625 cm2 in second restriction cross-sectional area, with a within-subject standard deviation (SD) of 0.133 cm2 (data on file, GSKCH); sufficient subjects were screened to provide 50 subjects who completed all study measurements.
The safety population comprised all subjects who received nasal strip application, and the intent-to-treat (ITT) population, used for efficacy analysis, comprised all subjects who completed at least 1 objective measurement in both the baseline and the treatment phases.
All efficacy analyses were based on change from baseline in both objective and subjective (subject-reported) measures. Summary statistics were calculated for all 3 baseline and treatment phase values for each objective measurement. SAS (SAS Institute Inc., Cary, NC) mixed models were used for analysis, with treatment and period as fixed effects, subject as a random effect, and baseline as a covariate. Between-product comparisons employed the pairwise Dunnett Multiple comparison procedure; all comparisons were performed at the 5% significance level, and 95% confidence intervals were calculated. Subject-level changes from baseline were calculated as the mean of repeat treatment measurements minus the mean of repeat baseline measurements. For the subject-reported assessments, summary statistics were developed for all 3 baseline and treatment phases. Subject-level changes from baseline were calculated as the treatment measurement minus the baseline measurement; analysis methods were similar to those used for the objective measurements. Summary statistics were tabulated for demographic and baseline characteristics.
Dermal Tolerability Study
The Dermal Tolerability study (study #B3560692; ClinicalTrials.gov identifier NCT01495494) was a single-center, randomized, parallel-group, evaluator-blinded study of the local tolerability of the marketed BRNS (clear strip), compared with the asymmetric butterfly strip (marketed in the United States as the Breathe Right Advanced Strip from 2010–2014), conducted in November 2009 in Verona, NJ. The primary objective was to compare the safety and tolerability of the marketed BRNS and the butterfly strip over 7 nights of continuous use; secondary objectives were to evaluate both strips with respect to comfort of wear, overnight adhesion, and ease of removal.
Study design and treatments
The Dermal Tolerability study included objective evaluations of the skin application site as well as subject-reported assessments (via diary cards) and included 3 visits to the study site. During visit 1, the subject’s application site and skin type were determined before the application of the nasal strip. During visits 2 and 3 (the mornings following application nights 3 and 7), each subject’s application site was assessed and scored using the Dermal Response Scale (DRS), and a questionnaire was used to assess comfort and ease of removal.
Subjects were randomized to treatment with either the marketed BRNS (clear) or the butterfly strip. The subjects received study product application and removal instructions at the baseline visit and were instructed to apply the test products to the nose at bedtime for 7 consecutive nights. All nasal strips were supplied by GSKCH. Treatment allocation was determined according to a computer-generated randomization schedule provided by the sponsor.
Study population
Subjects ≥18 years of age in good general health, who were free of lesions/skin conditions capable of affecting assessments/results and able to understand/complete study procedures and provide informed consent were eligible. Potential subjects were excluded if they had any disease or condition possibly affecting skin response or nasal breathing; allergy, intolerance, or hypersensitivity to any component of the test products or to latex; had a history of skin cancer; had recent (<30 days) allergic or contact dermatitis reaction at the test site, allergy or atopy, reactions to adhesive bandages, or skin or other immunologic disease; had baseline DRS score >1; or had used a menthol-containing oral product within 2 hours of any study assessment, glucocorticosteroids within 1 month prior to baseline, or any treatment that might affect nasal congestion.
Study outcomes and assessments
Each treatment arm included ≥25% of subjects with Fitzpatrick skin types I and II (combined), requiring assessment during screening; based on unprotected skin response to 30 to 45 minutes of sun exposure, the Fitzpatrick scale ranges from type I (always burns easily and never tans) to type VI (never burns and deeply pigmented).
The primary efficacy outcome was DRS-assessed skin irritation. The application site was assessed using the DRS at the screening visit, at baseline, and at study visits 2 (day after the third use of the nasal strip) and 3 (day after the seventh use of the nasal strip). The DRS, an 8-point scale for skin irritation status (from 0 [no evidence of irritation] to 7 [strong reaction beyond test site]), was used to assess the left side, right side, and bridge of the nose. Any subject with a DRS score ≥3 was instructed to discontinue nasal strip use, and the application site was reassessed at the next visit and continually until symptom resolution; any DRS score ≥4 (4 = definite edema) was recorded as an AE. The safety profile was considered acceptable if <30% of subjects had a DRS score ≥3 on any nasal region.
Secondary efficacy outcomes were product comfort and ease of morning removal (from a product acceptability survey) and level of attachment upon awakening. At study visits 2 and 3, subjects completed a 2-question product acceptability survey on product comfort (graded on a 5-point scale; 1 = very comfortable, 5 = very uncomfortable) and ease of morning removal (graded on a 6-point scale; 1 = very easy, 5 = very difficult, 6 = completely detached strip upon awakening). Subjects recorded times of nasal strip application and removal, as well as the level of strip attachment upon awakening, scored on a 5-point scale (1 = completely attached, 2 = 1 of 4 sides lifted, 3 = 2 of 4 sides lifted, 4 = 3 of 4 sides lifted, 5 = completely detached), on daily diary cards.
Safety was assessed based on the tabulation of AEs and SAEs. Irritation scores by the region of evaluation at each visit, discontinuation rates due to skin irritation or some other reason related to the study product, and the proportion of patients reporting AEs were also evaluated.
Statistical methods
As an exploratory study, no formal sample size calculations were conducted; a sample size of 80 (40 per treatment group) was considered adequate to detect a safety/tolerability signal.
The safety population comprised all randomized subjects who used the test product at least once with ≥1 safety assessment. DRS analysis was conducted on the safety completer population (all subjects completing the visit 3 assessment).
In the primary analysis, DRS measurements were summarized using frequency distributions for each nose region and visit; summary statistics were derived for each treatment and DRS score. Analyses were performed using the mixed-model approach, with treatment and period as independent variables and change from baseline as the dependent variable. Dropouts/discontinuations were evaluated using Kaplan-Meier survival curves for each treatment, and AEs/SAEs were tabulated. Responses to the product acceptability and level of attachment questions were summarized using frequency distributions; one sample t test was used to determine whether mean scores were different from 3 (ie, neither comfortable nor uncomfortable). Descriptive statistics were used for demographic and baseline variables.
Results
Patency Study
Study population
A total of 56 subjects were screened, of whom 50 were enrolled. All completed the study; both the safety and ITT populations comprised the 50 enrolled subjects. Study population demographics are summarized in Table 1.
Study Population Baseline Demographics for the Patency and Dermal Tolerability Studies. a

Abbreviations: BRNS, Breathe Right nasal strip; NA, not available; SD, standard deviation.
aSafety populations.
bFitzpatrick skin phototype information was not collected in the Patency study.
Efficacy results
Results for the primary efficacy measurements, MCA (second restriction) and nasal volume (0–3 cm), are summarized in Figure 1. All 3 tested strips produced highly significant increases (improvements) in both MCA and nasal volume from baseline (P < .0001 for all 3). The butterfly strip demonstrated significantly greater improvement in MCA than the marketed BRNS (P = .0030). Both the butterfly strip and the teardrop strip demonstrated significantly greater improvement in nasal volume than the marketed BRNS (P < .0001 for both).

Acoustic Rhinometry Results From the Patency Study (ITT Population). BRNS, Breathe Right nasal strip; ITT, intent-to-treat; MCA, minimum cross-sectional area. aMean change was adjusted for baseline, with baseline used as a covariate in the model. Baseline values for MCA (second restriction) were 0.89, 0.88, and 0.91 cm2 for the BRNS, asymmetric butterfly, and teardrop strip, respectively, and for volume of the first 3 cm were 4.27, 4.29, and 4.26 cm3, respectively. bP < .0001 versus baseline. cP = .003 versus BRNS. dP < .0001 versus BRNS.
With respect to posterior rhinomanometry and PNIF (data not shown), all 3 strips demonstrated significant improvement after strip application compared with baseline (all P < .05). There were no significant differences between the marketed and prototype strips with respect to change from baseline in nasal resistance (posterior rhinomanometry) or PNIF results.
Results for subject-reported outcomes are summarized in Figure 2. For the “ease of breathing” question, all 3 strips demonstrated highly significant improvement from baseline (P < .0001). For the “change in breathing” question, all 3 strips demonstrated highly significant improvement from baseline (P < .0001). There were no significant differences between the marketed and prototype strips with respect to change from baseline on either of the 2 subject-reported measures.

Subject-Reported Measures From the Patency Study (ITT Population): (A) Adjusted Mean Percentage Change in Ease of Breathing VAS Rating and (B) Mean Numerical Rating of How Breathing Feels With the Strip Applied. BRNS, Breathe Right nasal strip; ITT, intent-to-treat; VAS, visual analog scale. aMean change was adjusted for baseline, with baseline values used as a covariate in the model. bP < .0001 versus baseline; neither prototype was statistically significantly different versus the BRNS on either the VAS or the numerical rating scale. cResponses on a 0 to 100 mm visual analog scale (0 = extremely difficult, 100 = extremely easy). dResponses on an 11-point numerical rating scale (−5 = much worse, 0 = no difference, 5 = much better).
Safety results
No AEs were reported during this study; all 3 tested strips were generally well tolerated.
Dermal Tolerability Study
Study population
A total of 82 subjects were screened; all were enrolled and randomized to the marketed BRNS (n = 41) or the butterfly strip (n = 41) groups. All 82 screened and enrolled subjects completed the study. Study population demographics are summarized in Table 1. The mean (SD) age was 48.5 (16.1) years (range, 18–79 years) and most subjects were female (70 of the 82, 85.4%).
Safety/efficacy results
No AEs were reported during the study (primary end point); all subjects had DRS scores of 0 (no evidence of irritation) for all nose regions at baseline and all postbaseline assessments.
With respect to subject-reported outcome questions (how does the strip feel on the nose and ease of removal), both tested strips were rated positively (mean scores <3 on 5- and 6-point scales, respectively) throughout the study period (Table 2). Mean scores for both test strips on both questions were significantly lower (better) than the neutral response (P < .0001) at each time point. Scores for the marketed BRNS were numerically lower (indicating better comfort/removability) than the butterfly strip for both questions at each time point, but between-group differences were not statistically significant.
Subject-Reported Assessments From the Dermal Tolerability Study (Safety Population).
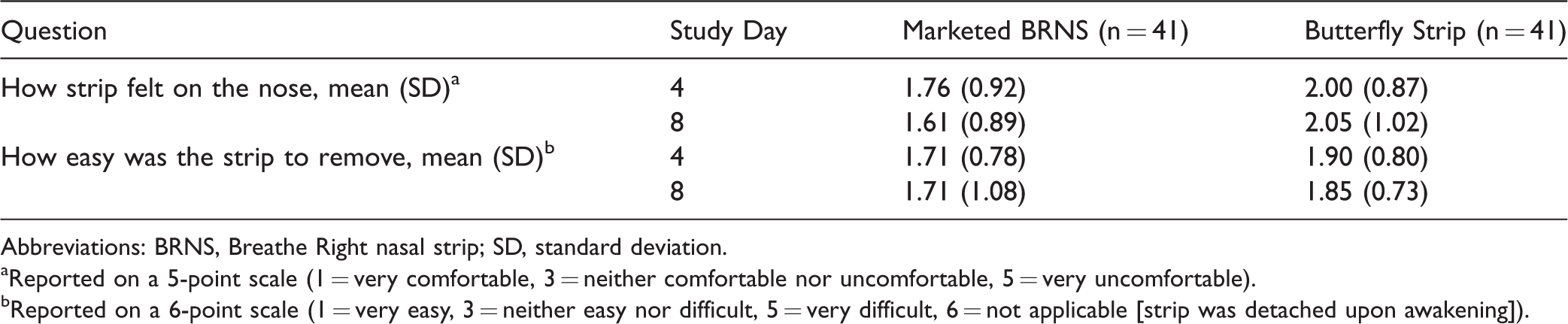
Abbreviations: BRNS, Breathe Right nasal strip; SD, standard deviation.
aReported on a 5-point scale (1 = very comfortable, 3 = neither comfortable nor uncomfortable, 5 = very uncomfortable).
bReported on a 6-point scale (1 = very easy, 3 = neither easy nor difficult, 5 = very difficult, 6 = not applicable [strip was detached upon awakening]).
Mean daily nasal strip attachment scores upon awakening were between 1 (completely attached) and 2 (1 of 4 sides lifted) for both the marketed BRNS (range, 1.41–1.68) and the butterfly strip (range, 1.54–1.98) across all 7 assessments; mean scores were generally lower (indicating better attachment) in the marketed BRNS group. The distributions of mean daily attachment scores for each group on days 1 to 3 and 4 to 7 are illustrated in Figure 3. As shown, the percentage of subjects with significant strip detachment (≥2 of 4 sides lifted) was higher in the butterfly strip group overall but was similar between treatment groups when comparing results from days 4 to 7 (Figure 3); attachment of the butterfly strip appeared to improve over time.

Overnight Nasal Strip Detachment (≥2 of 4 Sides Lifted), Dermal Tolerability Study. Percentages of subjects who reported various degrees of nasal strip detachment (2 of 4 sides lifted [blue], 3 of 4 sides lifted [red], or completely detached [green]) upon awakening throughout the 7-day study (left side) and during study days 4 to 7 (right side). The remaining subjects in each group reported complete nasal strip attachment (4 of 4 sides attached) upon awakening. BRNS, Breathe Right nasal strip.
Discussion
In the Patency study comparison of the marketed BRNS with prototype designs, all evaluated nasal strips produced highly significant improvements from baseline in objective nasal valve parameters—including MCA (second restriction), nasal volume, and PNIF—as well as reductions (improvements) in nasal resistance; comparable improvements from baseline were demonstrated for each product in the subject-reported assessments regarding nasal breathing.
The Dermal Tolerability study demonstrated good dermal tolerability for both the marketed BRNS and butterfly strips, with no evidence of skin irritation, during 7 consecutive nights (574 subject-nights) of wear. These observations are particularly encouraging given that >50% of the subjects were of Fitzpatrick skin types I and II with the highest risk of contact reactivity.
In the Patency study, the prototype and marketed strips were similar with respect to changes from baseline in PNIF and nasal resistance. Although in the Patency study the butterfly and teardrop strips produced significantly larger increases in MCA and nasal volume than the marketed BRNS, between-group differences in the subject-reported measures of breathing ease and comfort were small in comparison to within-group variability and not statistically significant. In addition, the marketed BRNS scored higher than the butterfly strip with respect to comfort, ease of removal, and level of overnight attachment in the Dermal Tolerability study (the teardrop strip was not evaluated in this study).
The Patency study demonstrated the ability of all tested strips to produce highly significant improvements in all objective nasal breathing parameters. These results lend further support to those of previous studies in which nasal dilator strips have been found to increase the volume and cross-sectional area of the nasal valve region7–11 and improve PNIF,12,13 thereby relieving sensations of nasal congestion. 8 These findings are consistent with the mechanism of action, in which the strips pull outward on the exterior lateral walls of the nasal vestibule, thereby increasing the diameter and cross-sectional area of the internal nasal valve area.8,14 The 2 new prototype strips were designed with the hope of improving upon the success of the BRNS, and they did indeed result in statistically significantly greater improvements in dimensional measures (MCA and nasal volume). However, similar between-group differences were not observed in flow parameters (nasal resistance and PNIF). Moreover, there were no significant between-group differences after nasal strip application in subjective measures of nasal breathing ease. These findings suggest that there is a complex relationship between changes in nasal valve dimensions and changes in nasal airflow; and that perceptions of nasal breathing ease may be driven more by flow parameters than by dimensional parameters. Although this study was not designed to explore the magnitude of minimal clinically important differences in either dimensional or flow parameters, it is clearly a potential topic for future studies.
The butterfly strip (also known as asymmetric butterfly) was marketed as the Breathe Right Advanced Strip from 2010 to 2014 but is no longer on the U.S. market.
Study limitations included the relatively small sample sizes, which may have constrained the ability to discern subtle differences in the performance of the tested strips. In addition, the use of healthy volunteers as subjects did not permit evaluation with regard to the BRNS indication, relief of nighttime nasal congestion, and snoring. Another limitation is that while statistically significant improvements in nasal patency were observed, it is unknown what level of nasal patency or what degree of change in nasal patency is clinically meaningful. Subjective patient-reported outcomes were based on VAS and numerical rating scales of breathing, which have not been validated. In addition, subjects had relatively high VAS scores at baseline (77–80 of 100), whereas in real-world settings, the strips may be used by subjects with more nasal patency complaints. Finally, the 7-night duration of the Dermal Tolerability study limited the ability to detect adverse dermal effects with longer term use.
Conclusion
Results from these 2 studies demonstrated the ability of the marketed BRNS and the tested prototype nasal strips to improve multiple measures of nasal breathing with little risk of skin irritation when used nightly for a 1-week period. Although the prototype strips produced significantly larger improvements in nasal dimensions than the marketed strip, these differences were not reflected in nasal airflow parameters and were offset to some extent by similar, and in some cases superior, ratings for the marketed strip in subject-rated assessments.
Footnotes
Acknowledgments
Medical writing and editorial assistance was provided by Stephen Collins, MS, and Diane M. Sloan, PharmD, at Peloton Advantage.
Ethical Approval
These studies were conducted in accordance with the International Council for Harmonisation Guideline for Good Clinical Practice and the ethical principles specified in the Declaration of Helsinki. The study protocols were approved by an Independent Ethics Committee before initiation, and all subjects provided written informed consent.
Statement of Human and Animal Rights
This article does not contain any studies with animal subjects.
Statement of Informed Consent
All subjects provided written informed consent.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: John Ward is an employee of AbbVie Ltd, Maidenhead, Berkshire, UK, and was an employee of GlaxoSmithKline Consumer Healthcare at the time of study conduct. Renee Ciesla, William Becker, and Gilbert Marava Shanga are employees of GlaxoSmithKline Consumer Healthcare, Warren, NJ, USA.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by GlaxoSmithKline Consumer Healthcare, Parsippany, NJ, USA.
