Abstract
Purpose:
Various surgical methods have been reported for Kummell’s disease with neurologic deficits. The aim of this study was to introduce long-segmental posterior fusion (LPF) combined with vertebroplasty (VP) and wiring as an alternative surgical technique.
Material and Methods:
We retrospectively analyzed 10 patients undergoing posterior decompression and LPF combined with VP and wiring for Kummell’s disease with neurologic deficits from January 2011 to December 2014. The radiologic outcomes included the local kyphotic angle (LKA) and vertebral wedge angle (VWA). Clinical outcomes, including the visual analog scale (VAS), the Oswestry Disability Index (ODI) and the Frankel grade were assessed. Surgery-related complications were also evaluated.
Results:
The mean age of the included patients was 77 ± 8 years with a mean follow-up period of 31.4 ± 4.9 months and a mean bone mineral density of −3.5 ± 0.7 (T-score). The mean operation time was 220 ± 32.3 minutes with a mean blood loss of 555 ± 125.7 mL. The preoperative LKA and VWA were significantly corrected postoperatively (37.9 ± 8.7° vs. 15.3 ± 5.3°, p = 0.005 for LKA; 21.3 ± 5.1° vs. 7.6 ± 2.8°, p = 0.005 for VWA) without a loss of correction at the last follow-up. The VAS and ODI were also significantly improved (7.7 ± 1.1 vs. 3.0 ± 1.6, p = 0.007 for VAS; 90.3 ± 8.9 vs. 49.6 ± 22.7, p = 0.007 for ODI). The Frankel grade of all patients was improved by at least 1 or 2 grades at the last follow-up. Surgery-related complications such as intraoperative cement leakage and implant loosening during the follow-up were not observed.
Conclusions:
LPF combined with VP and wiring might be an effective surgical option for Kummell’s disease with neurologic deficits, especially for the elderly patients with morbidities.
Level of Evidence:
level IV.
Introduction
Kummell’s disease, first reported by Hermann Kummell in 1894, is a clinical condition in which patients develop a vertebral collapse and kyphosis in the thoracolumbar region in a delayed fashion after minor spinal trauma. 1 Although the pathogenesis is unclear, Kummell’s disease typically manifests as an intravertebral vacuum cleft (IVC) and failure of the fracture healing process. 2,3 Kummell’s disease is most commonly observed at the thoracolumbar junction with greater motion, which increases the occurrence of IVC. 4 Abnormal motion due to IVC leads to spinal column instability and local kyphosis aggravated by the patient’s position. 5,6 The aggravation of kyphosis and neural compression by bone fragments can produce neurologic deficit. 1
Because the optimal surgical technique for Kummell’s disease has not been established, treatments should be tailored according to the individual case. Vertebroplasty (VP) and kyphoplasty have been shown to be effective for pain management in cases without neurologic deficits. 4,7,8 However, more extensive surgical treatment is usually required for neural decompression, kyphosis correction, and stabilization of the spinal column for Kummell’s disease with neurologic deficits. Some studies have recommended anterior reconstructive surgeries such as corpectomy with anterior fusion or circumferential fusion. 9 -11 Anterior reconstructive surgery has yielded favorable results including a good fusion rate and kyphosis correction, but the anterior approach may cause pulmonary, intra-abdominal and retroperitoneal complications. Moreover, long operative time and greater blood loss may be a drawback considering patient age and condition. 9 -11
Only posterior-approach surgery is considered a surgical option because of lower morbidity and fewer complications, especially for the elderly patients. VP combined with short-segment posterior fixation showed favorable results in short-term follow-up. 12 However, the long-term failure rate due to severe osteoporosis is high. 13,14 In the current study, we reported the efficacy of long segmental posterior fusion (LPF) combined with VP and wiring as an alternative surgical technique for patients with Kummell’s disease with neurologic deficits. The purposes of this retrospective study were as follows: (i) to investigate the initial kyphosis correction by evaluation of radiologic parameters, (ii) to report the advantages of our surgical technique in terms of clinical outcomes including pain control and neurologic improvement and (iii) to evaluate whether the radiologic and clinical improvements maintain in long-term follow-up.
Materials and Methods
Patient Selection
From January 2011 to December 2014, 10 consecutive patients treated with posterior decompression and LPF combined with VP and wiring for Kummell’s disease with progressive neurologic deficits were included in the study. The inclusion criteria were i) Kummell’s disease diagnosed by IVC signs on computed tomography scans and osteonecrosis with a fluid signal on magnetic resonance imaging, ii) fracture instability due to IVC measured by anterior vertebral height (AVH) collapse in the standing/sitting position compared to the supine position (Figure 1), iii) severe osteoporosis with bone mineral density (BMD) of T-score ≤ −3.0, iv) inability to ambulate due to progressive neurological deficits and v) follow-up more than 2 years. The exclusion criteria were i) pathologic fracture due to metastatic spine disease, ii) osteoporotic compression fracture without formation of IVC, iii) traumatic burst fracture and unstable fracture with three-column injury or posterior ligamentous complex injury and iv) refusal to participate in the study. Perioperative parameters such as blood loss, the duration of surgery, and fusion levels were collected. All patients were treated by 2 surgeons at a single center. This study was approved by the Institutional Review Board of the Catholic University of Korea (SC15RISI0153).

A, Preoperative magnetic resonance imaging showing Kummell’s disease with an intravertebral vacuum cleft (IVC, white arrow) at L1. Preoperative lateral radiographs showing fracture instability. B, In the Supine position. C, In the Standing position.
Principle of LPF Combined With VP and Wiring
The principle of LPF combined with VP and wiring is based on achieving initial kyphosis correction and stabilization by LPF with anterior support using VP at the vertebral collapse due to IVC. Posterior instrumentation was performed at the vertebrae 3 or 4 levels above and 2 or 3 levels below the collapsed vertebra with IVC. In addition, to prevent mechanical failure such as the pullout of pedicle screws in osteoporotic spines, cement-augmented pedicle screws and additional wirings were performed. Specifically, wiring was performed between the base of the spinous process above the upper instrumented vertebra and the bilateral rods just below the screw heads. In the case of severe osteoporosis, additional wiring was performed at the next proximal level and/or the distal level in the same manner (Figure 2).
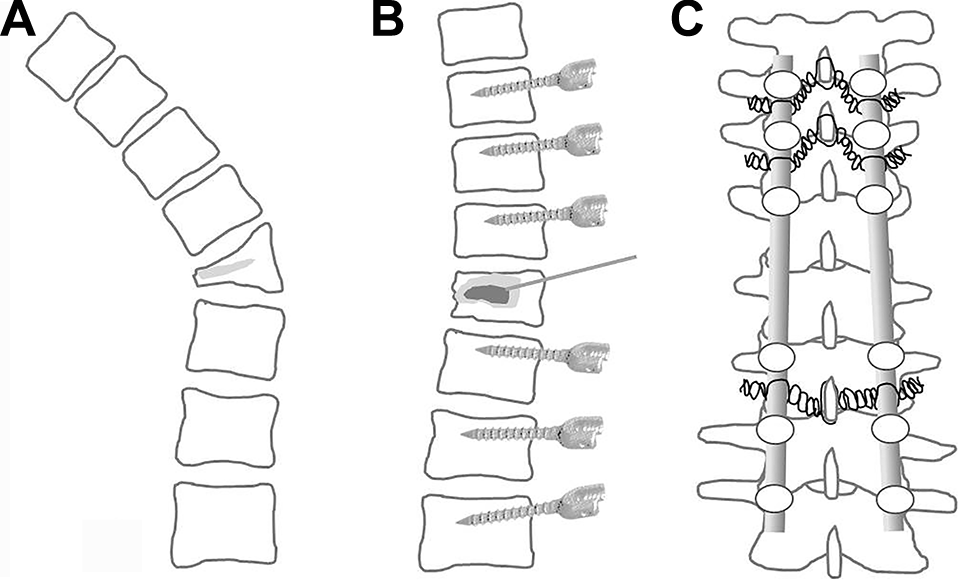
Schematic illustration of long-segmental posterior fusion combined with vertebroplasty (VP) and wiring. A, Kummell’s disease with an intravertebral vacuum cleft (IVC, bright gray). B, After kyphosis correction and stabilization, polymethyl methacrylate (the dark gray) was injected into the IVC. C, Wiring between the above spinous process and the 2 rods was used for rigid fixation.
Surgical Technique
Surgical procedures were performed under general anesthesia. The patient was placed in the prone position with the abdomen suspended to avoid excessive epidural bleeding. Placing the patient in the prone position on a Wilson frame induced spinal extension of the thoracolumbar junction and thus allowed elevation of the collapsed vertebral height in a vertebral body with IVC. A midline longitudinal skin incision and paravertebral muscle dissection were performed by posterior approach. After the entry points for screw insertion were identified, under-tapping by 0.5 to 1 mm was performed to prevent screw loosening. Moreover, 1 to 2 cc of polymethyl methacrylate (PMMA; Confidence Spinal Cement System, DePuy Spine Inc., Raynham, MA, USA) was instilled into the vertebral bodies through the tapping hole before the insertion of each screw. Posterior decompressive laminectomy was performed at the collapsed vertebral body level. After adequate decompression at the collapsed vertebral body level, pre-bent rods were placed into the screws. Greater reduction of the collapsed vertebral height and kyphosis correction were obtained by pushing the t-handle driver gently on the screw heads above and below the collapsed vertebral body with IVC while tightening the rods and screws.
The degree of reduction in the anterior vertebral height and the correction of kyphosis at the level of the IVC were checked by C-arm fluoroscopy during instrumentation. Then, using a standard transpedicular or parapedicular vertebroplasty technique, 4 to 6 cc of PMMA was injected into the IVC under fluoroscopic guidance (Figure 3). 15 Kyphoplasty was not performed in any case. After the VP, the holes for wiring were made carefully using a burr and a towel clip at the spino-laminar junction. Proximal wiring was performed between the spinous process above the upper instrumented vertebra and the rods just below the proximal screw heads. Auto- and allogenic chip bone grafts were added onto the posterior surfaces of the involved vertebrae and the posterolateral surfaces of the laminectomy level. On postoperative day 2, the patients were sent to a rehabilitation center for standing and walking exercises. The patients were required to wear a thoracolumbosacral orthosis for 3 months and discharged when they could walk.
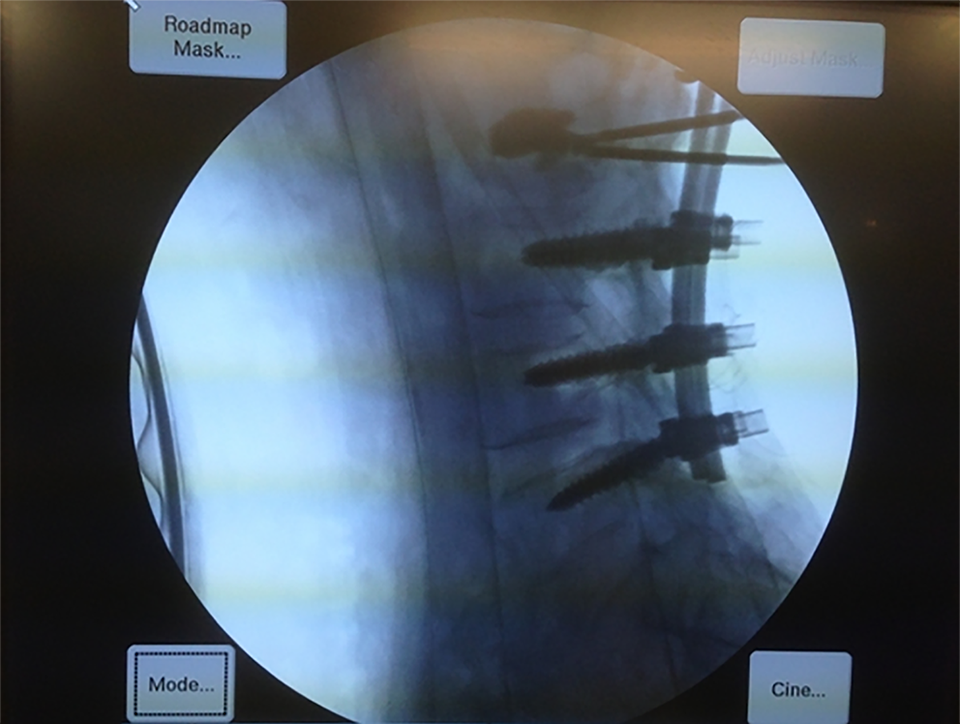
Intra-operative fluoroscopic image of vertebroplasty for the intravertebral vacuum cleft of the L2 vertebral body.
Radiologic Parameters
Anterior vertebral height (AVH)
The AVH was defined as the length between the most anterior point of the superior and inferior endplates in the lateral radiographs. Preoperative plain radiographs in the standing or sitting position, as well as the supine position, were taken to assess fracture instability by comparing the AVH in the standing/sitting position to that in the supine position. The plain radiographs of the thoracolumbar spine were taken in the standing or sitting positions 2 weeks after surgery and at the last follow-up.
Local kyphotic angle (LKA) and vertebral wedge angle (VWA)
We measured the LKA and VWA to assess the correction of kyphosis. The LKA was measured by drawing lines parallel to the superior endplate of the intact adjacent upper vertebra and the inferior endplate of the intact adjacent lower vertebrae. The VWA was measured by drawing lines parallel to the superior endplate and the inferior endplate of the vertebrae with the IVC (Figure 4). The radiologic parameters were measured with DICOM PACS software (Marosis, Marotech, Seoul, Korea).

Measurement of the local kyphotic angle (line c and line d) and vertebral wedge angle (line a and line b) in Kummell’s disease with an intravertebral vacuum cleft.
Clinical Parameters
Visual analog scale (VAS)
VAS is a subjective assessment tool used to measure the intensity of back pain. VAS pain score ranges from 0 to 10 points. A VAS score of 0 was defined as no pain and a score of 10 was defined as the worst pain.
Oswestry disability index (ODI)
ODI is a principal outcome measure designed to evaluate the management of spinal disorders. It is a self-administered questionnaire divided into 10 sections, which section is scored on a zero to 5 point (the greatest disability): pain intensity, personal care, lifting, walking, sitting, standing, sleeping, sex life, social life, and traveling. The index is calculated by dividing the total score by the total possible score, and then multiplying the results by 100. The intervals of 0%-20%, 21%-40%, 41%-60%, 61%-80%, and 81%-100% were considered as mild dysfunction, moderate dysfunction, severe dysfunction, disability, and long-term bedridden, respectively. 16
Frankel grade
Frankel score classification provided an assessment of spinal cord injury, which was divided into 5 grades of A, B, C, D, and E. Grade A meant complete neurological injury without motor and sensory function, and grade B meant preserved sensation only. Grade C and D meant preserved nonfunctional motor and functional motor, respectively. Grade E meant normal motor and sensory function. 17
Clinical outcomes including the VAS for back pain and the ODI for disability were evaluated before surgery, 2 weeks after surgery, and at the last follow-up. The Frankel scale was assessed before surgery and at the last follow-up. The surgery-related complications including cement leakage, instrumental failure and wound infections were also collected.
Statistical Analyses
Wilcoxon signed rank tests were used to evaluate the difference between the pre- and postoperative radiologic and clinical parameters. Statistical analyzes were performed with SPSS 21.0 software (SPSS Inc., Chicago, IL, USA). All data are presented as the mean ± standard deviation. P-values of < 0.05 were considered statistically significant.
Results
Patient Demographics
Table 1 summarizes the baseline characteristics of the patients. The included 10 patients were 9 women and 1 man with a mean age of 77 ± 8 years. The mean BMD was −3.5 ± 0.7 (T-score). The mean interval between the fracture and surgery was 7.5 ± 1.9 months with a mean follow-up period of 31.4 ± 4.9 months. The mean operation time was 220 ± 32.3 minutes with a mean blood loss of 555 ± 125.7 mL.
Patient Demographics and Surgical Parameters.
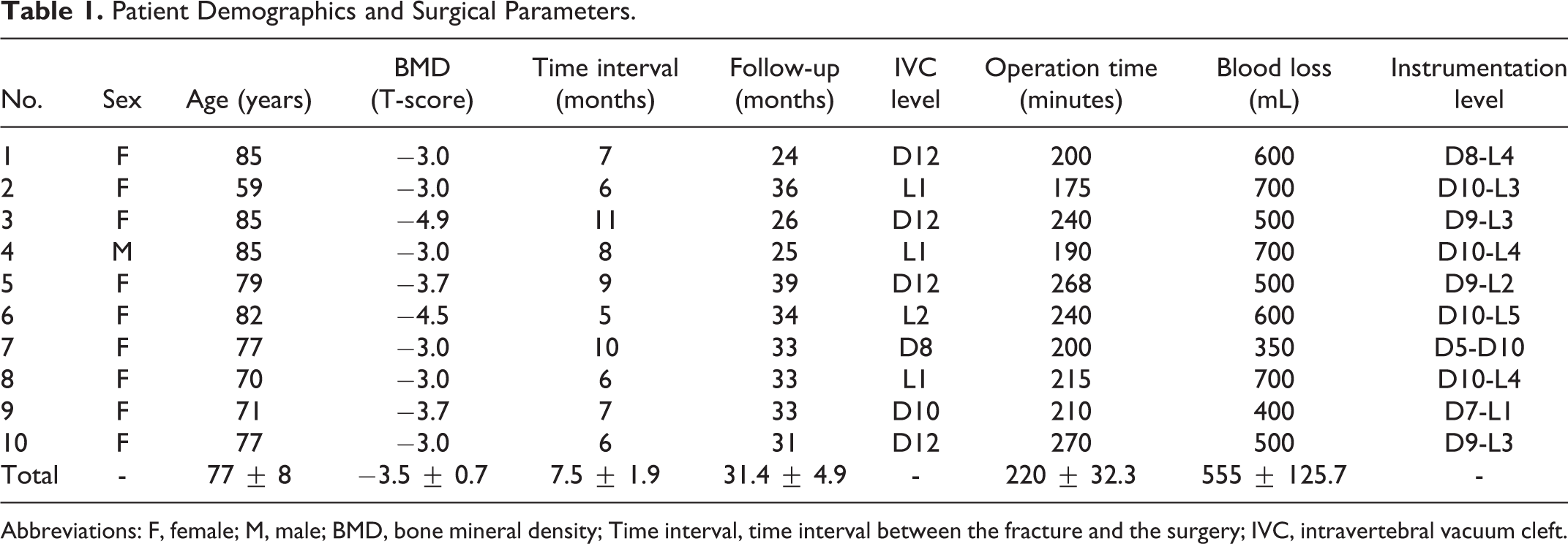
Abbreviations: F, female; M, male; BMD, bone mineral density; Time interval, time interval between the fracture and the surgery; IVC, intravertebral vacuum cleft.
Radiologic Outcomes
The radiologic outcomes are presented in Table 2. The AVH in the supine position was 14.9 ± 2.2 mm and decreased to 6.35 ± 2.1 mm in the standing or sitting position with a mean percentage of decrease in AVH of 57.3 ± 12.5%. The mean LKA and VWA were significantly corrected from 37.9 ± 8.7° and 21.3 ± 5.1° preoperatively to 15.3 ± 5.3° and 7.6 ± 2.8° postoperatively, respectively (p = 0.005 for both). No significant loss of correction of LKA and VWA was observed at the last follow-up compared to the postoperative values (all p > 0.05).
Radiologic Outcomes of Long-Segmental Posterior Fusion With Vertebroplasty and Wiring for Kummell’s Disease.
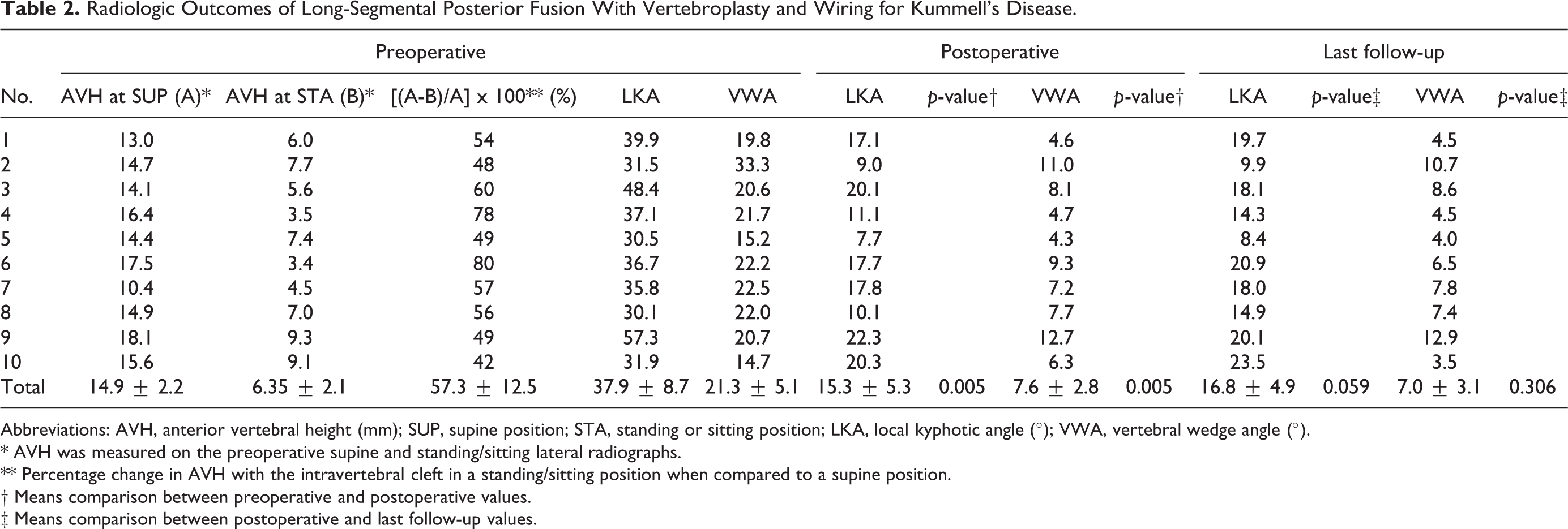
Abbreviations: AVH, anterior vertebral height (mm); SUP, supine position; STA, standing or sitting position; LKA, local kyphotic angle (°); VWA, vertebral wedge angle (°).
* AVH was measured on the preoperative supine and standing/sitting lateral radiographs.
** Percentage change in AVH with the intravertebral cleft in a standing/sitting position when compared to a supine position.
† Means comparison between preoperative and postoperative values.
‡ Means comparison between postoperative and last follow-up values.
Clinical Outcomes
The clinical outcomes are presented in Table 3. The mean VAS and ODI were significantly improved from 7.7 ± 1.1 and 90.3 ± 8.9° preoperatively to 3.0 ± 1.6 and 49.6 ± 22.7 postoperatively, respectively (p = 0.007 for both). In addition, VAS was improved without statistical significance and the ODI scores were significantly improved at the last follow-up compared to the postoperative scores (3.0 ± 1.6 vs. 2.3 ± 1.4, p = 0.13 for VAS; 49.6 ± 22.7 vs. 49.6 ± 22.7, p = 0.024 for ODI).
Clinical Outcomes of Long-Segmental Posterior Fusion With Vertebroplasty and Wiring for Kummell’s Disease.

Abbreviations: VAS, visual analog scale; ODI, Oswestry Disability Index.
* Means comparison between preoperative and postoperative values.
* Means comparison between postoperative and last follow-up values.
Neurologic recovery at the last follow-up was obtained in all patients who had preoperative neurologic deficits, from Frankel grade C to D in 2 patients, from Frankel grade C to E in 4 patients, and Frankel grade D to E in 4 patients (Table 4). A descriptive case of LPF combined with VP and wiring for Kummell’s disease with neurologic deficits and kyphosis is presented in Figure 5.

An 85-year-old woman treated with long-segmental posterior fusion with vertebroplasty and wiring. A-E, Kummell’s disease with intravertebral vacuum cleft (IVC, white arrow) and cord compression at T12. F-H, Plain radiographs showing kyphosis correction and maintenance preoperatively, postoperatively and 26 months after surgery.
Patient Numbers Before and After Surgery by Frankel Classification.

Two patients presented with paralytic ileus and a superficial wound infection was observed in 1 patient, which resolved with conservative treatments including oral intake restriction and intravenous antibiotics. Obvious loosening of the internal fixation and complications related to cement were not observed during follow-up.
Discussion
Patients with Kummell’s disease with neurologic deficits are often older and may have severe osteoporosis and a variety of medical diseases. It is well known that spinal instrumentation failures occur more often in osteoporotic spines. 18,19 In the case of posterior fixation with pedicle screws, failure is a result of decreased fixation strength in poor bone quality. 20 This insufficient fixation leads to increased instrument failure, such as screw pullout and loosening, warranting revision surgery that can lead to additional complications. 20 Moreover, the condition of the patients may not be sufficient to undergo reoperation for surgery failure at a later date. It is important to obtain optimal results with a one-stage operation. Therefore, it is necessary to consider a surgical method with a short operation time, less blood loss, relatively few complications, and less possibility of reoperation due to osteoporosis or fixation failure.
Some studies reported that anterior reconstructive surgery was better because of advantages including reconstruction of the stable spinal column and the correction of local kyphosis. 21,22 However, the anterior approach can be very risky, considering that most patients with Kummell’s disease are elderly individuals who may have several cardiopulmonary problems. A study on the complications of the anterior approach reported a high complication rate of 31% (140 of the 447 patients). 23 Additionally, anterior-posterior combined surgery requires a long operative time and is associated with greater blood loss. 24,25
For these reasons, we performed VP as an alternative method for anterior reconstruction. VP enables surgeons to achieve anterior strength of the vertebral body without the complications caused by the anterior approach. Indeed, many previous studies have reported that VP could increase stiffness by 107.8% and strength by 90.5% in the vertebral body. 26,27 Shorter operative time and less blood loss were distinct advantages of the present study. The mean duration of surgery was 220 minutes (range, 175-270 minutes), and the mean blood loss was 555 mL (range, 350-700 mL). This result is contrary to a previous study reporting a mean operative time of 351 minutes (range, 200-580 minutes) and a mean blood loss of 2892 mL (range, 900-5850 mL) in the anterior-posterior combined surgery for osteoporotic vertebral fracture. 24
Moreover, VP-related complications such as cement leakage were not found in our study, although serious complications related to VP have been reported. It appears that the reason for the lack of cement leakage was the restoration of the height of the collapsed vertebrae with IVC by posterior instrumentation before VP. When a patient was placed prone on the operating frame, the height of the vertebral body containing the IVC increased. We could then restore the height of the collapsed vertebral body by posterior stabilization using instruments. Restoring the vertebral height could reduce the intravertebral pressure and lead to a favorable environment for VP, decreasing the risk of cement leakage. In addition, rigid long-segmental posterior fusion combined with wiring might prevent injected cement from being migrated and dislodged.
Recently, short-segmental posterior instrumentation combined with VP was reported for kyphosis correction in patients with osteoporotic vertebral collapse with IVC. 12,28 However, this procedure may not lead to sufficient stability of the spinal constructs in the osteoporotic vertebrae. 13 Despite the favorable outcomes observed in short-segmental posterior instrumentation combined with VP, instrumental loosening at the bone-screw interface and the loss of kyphosis correction are major concerns. 13 Particularly, the correction loss rate of kyphosis after short-segmental posterior fusion combined with VP increased by 88% after 2 years of follow-up in osteoporosis patients. 14 In this study, the correction loss of kyphosis was only 1.9 ± 1.7° and screw loosening was not observed. These results suggest that our technique using LPF and wiring may increase fixation strength and prevent screw loosening and the loss of kyphosis correction.
In the osteoporotic spine, the minimization of loads at individual fixation points is important, and the use of multiple levels of fixation is considered for instrumentation. 18,20 In this respect, our surgical method has several advantages. First, LPF and augmented pedicle screws reduce the probability of instrument failure and correction loss by providing load distribution and strong fixation to vertebrae with poor bone quality. The injected cement into the IVC level also provides anterior support to minimize posterior pedicle screw stress. Previous studies have reported the techniques of spinous process wiring 29 -31 or sub-laminar wiring 20,32 for posterior spinal stabilization. We modified these wiring techniques and applied them to surgery for strong fixation. 20,29 -32
The radiologic and clinical outcomes were also significantly improved after surgery. The postoperative correction of local kyphosis and segmental kyphosis was 21° and 14°, respectively. Although a slight loss of correction was observed at the last follow-up of more than 30 months, this difference was not statistically significant. Severe osteoporosis in all patients may have resulted in this correction loss during follow-up. Regarding the clinical outcomes, the VAS and ODI were also significantly improved after surgery and maintained well until the last follow-up. Neurologic function was improved in all patients by at least 1 or 2 Frankel grades at the last follow-up. Thus, posterior decompression and LPF combined with VP is an effective treatment method for patients with Kummell’s disease with neurologic deficits.
This study had several limitations. First, this study was a retrospective case series with a small number of patients. Selection bias might have affected the results and the strengths and weaknesses of our surgical technique were not directly compared to other surgical techniques. Thus, further comparative studies are required to verify the efficacy of the surgical method for patients with Kummell’s disease with neurologic deficits. Second, solid bony union in posterior instrumented fusion was not evaluated by CT scans. However, posterior fusion could be obtained because instrument failures such as rod failures and screw pullout were not observed during more than 2-years of follow-up. Finally, kyphosis correction and height restoration of the collapsed vertebrae could be limited in chronic fracture without formation of IVC. Despite these limitations, this study demonstrated the radiologic and clinical outcomes of LPF with VP and wiring for patients with Kummell’s disease with severe osteoporosis with an average T-score of −3.5.
Conclusion
LPF combined with VP and wiring could significantly correct local and segmental kyphosis, and the correction maintained to the last follow-up. VAS and ODI were also significantly improved without major complications after surgery. Neurologic deficits in all patients were successfully recovered by at least 1 or 2 Frankel grades at the last follow-up. Thus, LPF combined with VP and wiring might be an effective surgical option for Kummell’s disease with neurologic deficits, especially for the elderly patients with morbidities.
Footnotes
Authors’ Note
Hyung-Youl Park and Ki-Won Kim contributed equally to this study as co-first authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Small Grant for Exploratory Research (SGER) through the Ministry of Education of the Republic of Korea and The Catholic University of Korea Songeui (2018R1D1A1A02049202).
