Abstract
Postoperative pain continues to be an intense area of research in pediatric cardiac surgery, and there is a scarcity of information regarding the use of methadone in this population. We performed a single center retrospective descriptive cohort analysis of all pediatric cardiac surgery patients who received intraoperative methadone between January 1, 2020 and December 21, 2024. In total 111 congenital cardiac surgeries were completed in patients aged 0-18 years during the study period, of which 45 (41%) received intraoperative methadone. In the cohort receiving methadone, there were 29 male patients (64%), with an average age of 10.3 (SD±5.3) year. The average dose of methadone was 0.16mg/kg (SD±0.05mg/kg). With respect to post-operative analgesia, average pain scores at 6, 12, and 24 hours of 1.8 (SD±2.3), 1.95 (SD±2.7), and 1.44 (SD±2.2), respectively. The median 24-hour opioid requirement was 0.59 Morphine Milligram Equivalents(MME)/kg (IQR 0.25-0.96). In total, 32 of our 45 patients (71%) were able to be extubated in the operating room with no reintubations or need for rescue non-invasive positive pressure ventilation (NIPPV). Our study demonstrates that even a modest dose of intraoperative methadone in pediatric cardiac surgery can be safe and effective for intra and postoperative analgesia. Additional randomized controlled trials are necessary to further evaluate its effectiveness.
Keywords
Introduction
In the United States, approximately 40,000 children per year undergo congenital heart surgery, with a large variation in intraoperative and postoperative analgesic regimens.1,2 Postoperative pain continues to be an intense area of research in pediatric cardiac surgery, as children with congenital heart disease are prone to significant hemodynamic consequences related to inadequate analgesia. 3 Although protocols and guidelines have been created with the goal of improving postoperative analgesia, they have relatively poor adherence and do not consistently demonstrate improved pain control in the literature.4,5 Recently, methadone has become a drug of interest for intra- and postoperative analgesia. Methadone's pharmacokinetics make it appealing for postoperative analgesia owing to its long duration of analgesia, relatively brief peak respiratory depressant effects, and its antagonism at the N-methyl-D-aspartate (NMDA) receptor. 6
Initially studied in complex spine surgery, methadone has been shown to reduce the rates of postoperative pain and postoperative opioid use in cardiac surgery, bariatric surgery, and same-day surgeries.7–10 Subsequently, methadone has been introduced as part of a multimodal analgesic strategy for pediatric cardiac surgery, although there is a paucity of literature describing its dosing and safety in this population.11–13 The aim of this retrospective descriptive cohort analysis was to add to the limited body of knowledge surrounding the use of intraoperative methadone in pediatric cardiac surgery, specifically as it pertains to postoperative ventilator time, opioid requirements, and pain scores. Herein, we present a multiyear study in an older cohort which demonstrates the safe and effective use of relatively low dose methadone to facilitate intraoperative extubation with safe postoperative outcomes.
Methods
Following Institutional Review Board approval (IRB #26835), a retrospective analysis of all pediatric cardiac surgeries completed between January 1, 2020 and December 21, 2024 was performed. Informed consent was waived for this study. This time frame was chosen due to the introduction of methadone into the pediatric formulary for intraoperative use in 2020. At this time, methadone was added to the intraoperative analgesia bundle which included fentanyl, morphine, hydromorphone, and acetaminophen. The administration and dosing of methadone during this study period were at the discretion of the pediatric cardiac anesthesiologist. Subsequently, all anesthetic records of patients who were 0-18 years of age and underwent cardiac surgery were reviewed for the use of intraoperative methadone, and those patients were included for further review. Patients were excluded from further analysis if they did not have a complete data set, if their surgery was cancelled, or if they were found to require mechanical ventilation at baseline. Demographic information, surgical procedures and times, and postoperative outcomes including pain scores, ventilator times, reintubations, postoperative opioid requirements, time until rescue opioid administration, and length of stay were collected and analyzed using Microsoft Excel (Version 2503). Pain scores were taken from the patient's chart either as a Numerical Pain Score (NRS) or The Face, Legs, Activity, Cry, and Consolability (FLACC) scale. Milligrams of morphine equivalency (MME) was calculated using the formula MME = (micrograms of fentanyl/1000) + (milligrams of hydromorphone×20). Morphine equivalents of methadone were not calculated due to the unpredictable nature of this conversion. Subsequently, a descriptive cohort analysis of all pediatric cardiac surgery patients who received intraoperative methadone was performed in Microsoft Excel and Minitab. An extensive literature review of methadone in pediatric cardiac surgery was conducted.
Results
A total of 111 congenital cardiac surgeries were completed in patients who were 0-18 years of age during the study period. A total of 45/111 (41%) received methadone. In the cohort receiving methadone, there were 29/45 male patients (64%) and an average age of 10.3 (SD ± 5.3) years. The procedures performed and additional demographic information are available in Table 1. The average dose of methadone was 0.16 mg/kg (SD ± 0.05) and patients received an additional 0.36MME/kg (IQR 0.17-0.51MME/kg) dose of supplemental opioids intraoperatively. Thirty-five of 45 patients (78%) of patients received intraoperative acetaminophen. The median time to first rescue opioid administration was 101.5 (IQR 56-220) minutes, with a median first 24-h postoperative supplemental opioid requirement of 0.59MME/kg (IQR 0.25-0.96). The average pain scores at 6, 12, and 24 h were 1.8 (SD ± 2.3), 1.95 (SD ± 2.7), and 1.44 (SD ± 2.2), respectively. A total of 32/45 patients (71%) were extubated intraoperatively. For those patients not extubated intraoperatively, the initial ventilator time was 119 min (IQR 69-212.5). There were no reintubations in either group. Patients extubated intraoperatively had an average 24-h supplemental opioid dose of 0.57 MME/kg (95% CI 0.41-0.79) compared with 1.3 MME/kg (95% CI 0.23-2.4) for those not extubated intraoperatively (p = 0.16). The median length of stay in our cohort was 2.35 days (IQR 2.2-4.2 days). A summary of opioid administration and pain scores is available in Table 2.
Characteristics of Patients and Cases Included in This Study. Abbv. Values are Reported as Means ± Standard Deviation, Median (Interquartile Range) and Total Sums.

ASD, atrial septal defect; CPB, cardiopulmonary bypass; IQR, interquartile range; LOS, length of stay; VSD, ventricular septal defect
Opioid Dosing and Pain Scores. Data are Reported as Mean ± SD or Median (Interquartile Range).
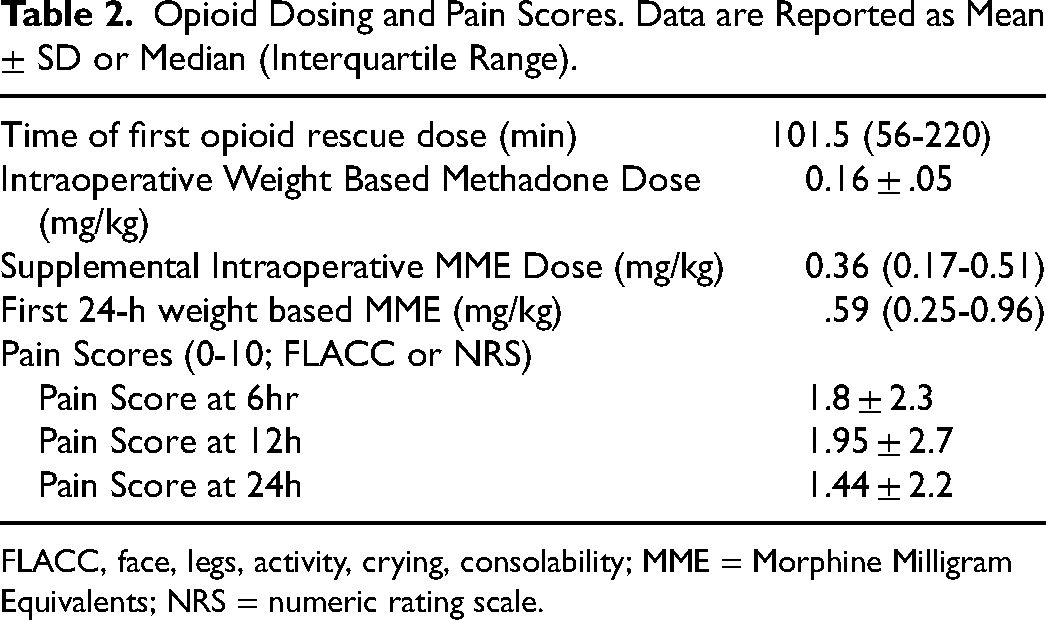
FLACC, face, legs, activity, crying, consolability; MME = Morphine Milligram Equivalents; NRS = numeric rating scale.
Discussion
Adequate control of postoperative pain is of the utmost importance, especially in a vulnerable population such as pediatric congenital cardiac patients. Identifying the ideal analgesic regimen in this population has proven challenging, owing in part to the critical nature of these patients’ cardiopulmonary systems and the known respiratory depression and subsequent hypercarbia that can occur with opioid analgesics. In our own institution, we have introduced methadone as part of a multimodal analgesic strategy to facilitate early extubation and improved analgesia for this patient population.
There are a handful of studies in the pediatric cardiac surgery population demonstrating that the use of intraoperative methadone decreases the amount of postoperative opioids required without significant adverse effects.11–13 In our cohort of 45 patients, the average dose of methadone was 0.16 mg/kg (SD ± 0.05 mg/kg). Dose finding studies have found that a dose range between 0.15 mg/kg and 0.3 mg/kg (ideal body weight) are safe and appropriate, with a potential signal towards an optimal dose of approximately 0.2 mg/kg of ideal body weight.7,14 Iguidbashian et al. utilized a methadone dose of 0.26 mg/kg total body weight, which was higher than our average dose of 0.16 mg/kg. Their group also found a longer length of time to first supplemental opioid administration of 306 min, compared with 101.5 min in our study. This difference can likely be explained by the higher average dose of methadone used. 11 While the dose range of methadone utilized by Wang, et al, was 0.1 to 0.4 mg/kg, 86.5% of patients utilized doses of 0.15-0.2 mg/kg, which was comparable to our cohort. 15 It is pertinent to note that 64% (29/45) of patients in our study received a methadone dose of <0.2 mg/kg, which likely represents a cautious initiation of this medication given the novelty of its use in our institution. It is possible that an increased methadone dose could further decrease postoperative opioid use and result in a further reduction in pain scores. However, caution should be used when extrapolating dose-finding studies from noncardiac surgery, as postbypass plasma concentration of methadone can be significantly altered due to hemodilution from the pump prime and sequestration from the bypass components. 16
The additional intraoperative analgesic requirement identified in our methadone cohort was 0.36 MME/kg (IQR 0.17-0.51MME/kg). With respect to postoperative analgesia, we observed excellent pain control with average pain scores at 6, 12, and 24 h of 1.8 (SD ± 2.3), 1.95 (SD ± 2.7), and 1.44 (SD ± 2.2), respectively. The median 24-h opioid requirement was 0.59MME/kg (IQR 0.25-0.96). These results are similar to those in the study by Barnett et al, who found that patients in their methadone cohort had a postoperative opioid requirement of 0.4 MME/Kg (IQR 0.2-2). 12 These findings are also reflected in studies by Blasiole et al. and Wang et al. who found a significant decrease in the use of short-acting opioids intraoperatively, overall decreased opioid consumption, as well as decreased pain scores.15,17
The initiation of intraoperative methadone within the multimodal bundle has also facilitated the ongoing effort to support on-table extubation in this patient population. In total, 32 of our 45 patients (71%) were able to be extubated in the operating room with no reintubations or need for rescue Noninvasive Positive Pressure Ventilation (NIPPV), which leads the national average which was found to be 53%-58% in recent multicenter studies. 18 Several studies have demonstrated that intraoperative extubation can be a crucial factor in decreasing overall hospital length of stay and cost in adult cardiac surgery patients, with some studies reporting up to a 30% reduction.19,20 In addition to reducing length of stay, other benefits include better hemodynamics, less need for inotropic support, reduced incidence of ventilator acquired pneumonia, and reduced complications from prolonged sedatives. 21
In our study, patients extubated intraoperatively had a 24-h postoperative MME requirement of 0.57 MME/kg. Patients not extubated in the operating room had a 24-h postoperative MME requirement of 1.30MME/kg (p = 0.16). Seventy-eight percent (35/45) of patients also received acetaminophen intraoperatively, which was weight-based and administered at the end of the case. Although this difference was not statistically significantly different, this study was not powered to assess this specifically, nor is intraoperative extubation likely causal in its relationship to decreased opioid consumption. It is likely that this difference reflects the common use of intravenous opioids for sedation in mechanically ventilated patients or the increased complexity of patients not extubated intraoperatively. This result should therefore be interpreted as hypothesis-generating only, but may be pertinent as reduction in postoperative opioids has been shown to reduce the incidence of hyperalgesia, central sensitization and potential long term dependence and neurobehavioral changes. 22
The current study adds to a growing body of literature demonstrating the safe and effective introduction of intravenous methadone for intra- and postoperative analgesia in pediatric cardiac surgery patients. Specifically, we present an older cohort of patients who received a relatively modest dose of intraoperative methadone and still demonstrated excellent analgesia. Additionally, the use of intraoperative methadone in a multimodal regimen allowed for frequent intraoperative extubation and short postoperative ventilator times without the need for reintubations or rescue NIPPV. It is worth addressing concerns regarding the safety profile of methadone, especially as it relates to QT prolongation and patients with renal or hepatic dysfunction. Fortunately, QT prolongation and resulting arrhythmias appear to occur primarily at extremely high doses of methadone and with chronic admistration.23,24 Additionally, only 20% of methadone is eliminated by renal excretion, and liver fibrosis and cirrhosis do not significantly alter the concentration-to-dose ratio of methadone.25,26
Future large, randomized controlled trials are needed to compare traditional analgesic strategies with those including methadone and determine if it should be considered a staple in a multimodalanalgesic bundle. Additional dose finding studies in pediatric cardiac surgery should be undertaken to determine the ideal dose of intraoperative methadone in this patient population. While questions certainly remain regarding methadone use in this patient population, recent shifts prioritizing early extubation, minimization of opioid administration, and the use of adjuvant analgesics create a void that methadone may well fill.
Conclusion
The addition of a modest methadone dose to our pediatric cardiac surgery analgesia bundle was safe and effective, allowing for excellent intra- and postoperative pain control and expedient extubation, while maintaining a promising safety profile. We believe the future of methadone as an intraoperative analgesic in this population is promising and future studies are warranted to investigate the optimal dosing to maximize its benefits.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
