Abstract
Coral reef aorta (CRA) is a rare, calcified aortic lesion observed primarily in adults and elderly patients. This condition can lead to resistant hypertension and impaired organ perfusion. Although surgical treatment remains the predominant approach, endovascular intervention is emerging as an alternative for suitable cases. We present a 16-year-old male patient with a history of kidney transplantation who was admitted due to hypertension. Imaging revealed severe stenosis at the isthmus level caused by a calcific lesion characteristic of CRA. Given the complexity of the lesion and the surgical challenges, an endovascular approach was chosen. A covered stent was successfully implanted, resulting in significant hemodynamic improvement. At the sixth month follow-up, the patient remained normotensive, and antihypertensive medications were discontinued. To our knowledge, this case represents the first pediatric patient reported to have undergone endovascular treatment for severe aortic coarctation associated with CRA. Our findings suggest that endovascular intervention using a covered stent may provide a viable alternative to surgery in selected pediatric cases, minimizing morbidity and mortality. Further studies are needed to assess the long-term outcomes of this approach.
Introduction
The coral reef aorta (CRA) is a rock-hard, eccentric, patchy calcific lesion of the infrarenal and suprarenal aorta, primarily observed in adult patients.1,2 Unlike calcific lesions that develop due to atherosclerosis, endophytic lesions follow an intraluminal course. Although predominantly located in the visceral aorta, they can occur throughout. Depending on the location of the endophytic lesion, findings such as resistant hypertension, claudication, and impaired organ perfusion may be initial symptoms prompting presentation. 3 Treatment is planned depending on the location and extent of the calcific lesion, the severity of stenosis, and the patient's general condition. Thromboendarterectomy or bypass is the most commonly used surgical treatment options. Endovascular treatments are not widely used due to concerns about aortic rupture and dissection. 4 Very few cases of CRA localized in the aortic arch have been reported in the literature, and endovascular treatment in this regard is limited to rare case reports.
Case Presentation
A 16-year-old male patient, who was being followed after a kidney transplant, was admitted to the ward due to hypertension. His medical history revealed that he was treated for hemolytic anemia between the ages of 2 and 4. A transvenous dual pacemaker was placed due to complete atrioventricular block at the age of 10, and he underwent peritoneal dialysis for six months due to chronic kidney disease that developed as a result of antitubular basement membrane disease at the age of 11, followed by a living-related kidney transplant from his mother. During the physical examination, blood pressure was measured at 138/74 mm Hg in the right arm and 97/57 mm Hg in the right foot. A 3/6 systolic ejection murmur was noted in the aortic area, and femoral pulses were weakly palpable.
Cardiomegaly and pulmonary congestion findings were evident on chest x-ray, and a calcification line compatible with the border of the aorta wall was noted (Figure 1A). Echocardiographic examination revealed hypertrophy of the left ventricle, mild aortic and mitral regurgitation, and stenosis in the proximal brachiocephalic trunk. The descending aorta flow could not be visualized. Computed tomography (CT) angiography revealed extensive calcification in the vessel walls from the ascending aorta to the thoracic aorta, with significant stenosis, particularly at the isthmus level of the descending aorta, resulting in lumen stenosis (Figure 1B and C). In an effort to determine the timing of the onset of calcific lesions, prior imaging studies were reviewed. A CT angiography performed four years earlier, as part of the prerenal transplant workup, demonstrated subtle calcific changes at the level of the aortic isthmus (Figure 1D).
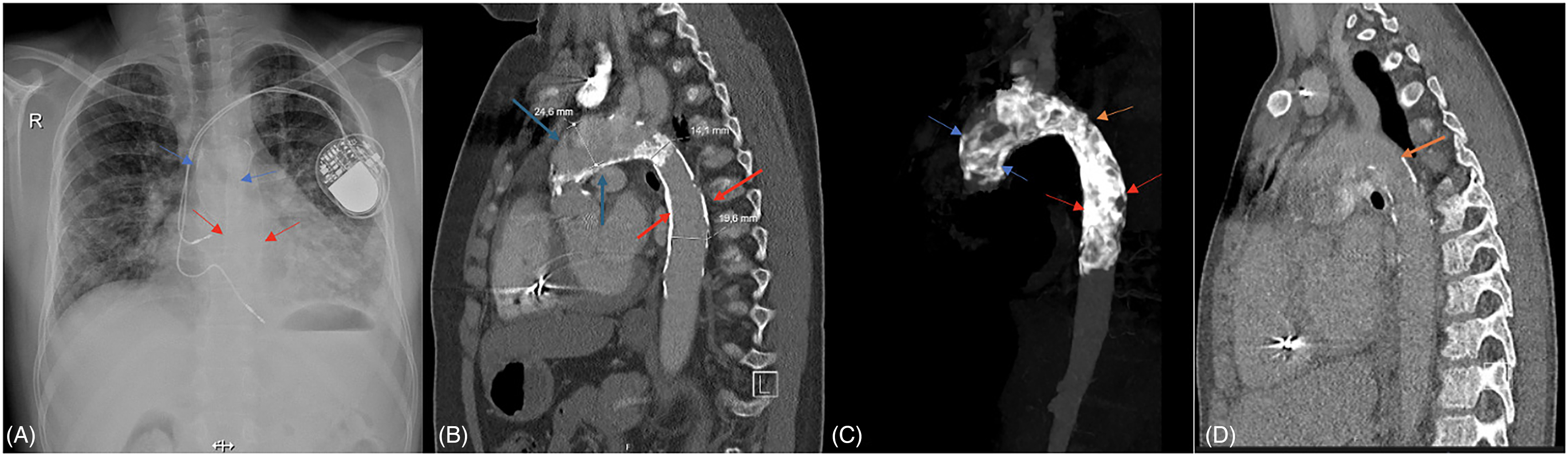
(A) The chest x-ray indicates signs of cardiomegaly and pulmonary congestion. The calcifications affecting the walls of the ascending aorta, aortic arch, and descending aorta are highlighted with arrows. (B) Calcific lesions causing stenosis at the isthmus level in the descending aorta are observable in the sagittal section of the computed tomography (CT) angiography. (C) A widespread calcified lesion extending from the ascending aorta to the thoracic aorta is presented in the MIP range format obtained from the CT angiography image. (D) Subtle calcific changes were noted along with the wall of the proximal thoracic aorta on the sagittal image of the CT angiography performed four years ago. (The arrows point to calcified lesions in the ascending aorta (A), the calcified lesions in the descending aorta ( B, C), and the calcified lesions at the isthmus (D) level.).
Considering the patient's ongoing hypertensive course despite medical treatment, the possibility of organ perfusion being affected due to kidney transplantation, and the possibility of affecting allograft perfusion, it was decided that intervention should be performed in the coarctation area. Thus, endovascular treatment using a covered stent was decided.
The procedure was performed under general anesthesia using the right femoral artery. Imaging revealed dense calcification starting from the ascending aorta and involving the aortic arch and thoracic aortic vessel wall. In the hemodynamic evaluation, the ascending aorta pressure was measured as 135/80 mm Hg; the descending aorta pressure was measured as 90/70 mm Hg; and a gradient of 45 mm Hg was observed. Due to the calcific lesion, the narrowest area at the isthmus level was measured as 6 mm. After stenosis, the diameter of the descending aorta was measured as 21 mm. It was decided to implant a 39 mm Covered CP stent by loading it onto an 18 × 4 cm Z med balloon (Figure 2).
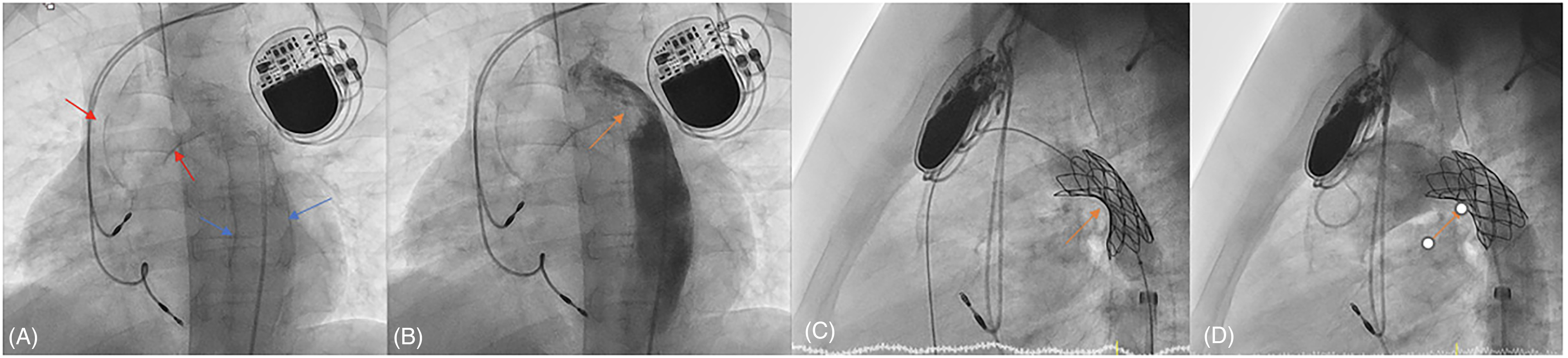
(A) In the catheter angiography laboratory, viewed under the scope in the anterior-posterior 30° left anterior oblique (LAO) position, calcific lesions in the ascending aorta extending to the thoracic aorta are visible (arrows). (B) The injection of contrast material performed in the anteroposterior 30° LAO position reveals a calcific lesion that is causing stenosis, accompanied by a distinct filling defect at the level of the isthmus (arrow). (C) In the lateral position, recoil due to the calcific mass effect (arrow)is noted following the dilatation of the 39 mm covered CP stent positioned in the coarctation region with the Z-med II 18 × 4 cm balloon. (D) After being inflated twice using the same balloon in the indentation area, the stent is observed to have opened and be in the correct position.
After the implantation of the stent, the patient was transferred to the pediatric intensive care unit (PICU) for observation. While the patient was being monitored in the PICU, a swelling in the groin was noted, and an ultrasound revealed a pseudoaneurysm. Due to its broad base, the pseudoaneurysm was considered unsuitable for interventional treatment. Surgical repair of the pseudoaneurysm was successfully performed. An echocardiographic assessment indicated that the descending aorta was patent, with minimal flow from the proximal layers of the stent to the left subclavian artery. The control CT angiography, performed before discharge, showed that the stent was correctly positioned, with no signs of an aneurysm developing in the vessel wall (Figure 3B and C). At the sixth-month follow-up, the patient remained normotensive, allowing for the gradual tapering and eventual discontinuation of antihypertensive therapy. A lateral chest radiograph confirmed the stent's appropriate positioning and structural integrity, with no evidence of fracture or migration (Figure 3D).
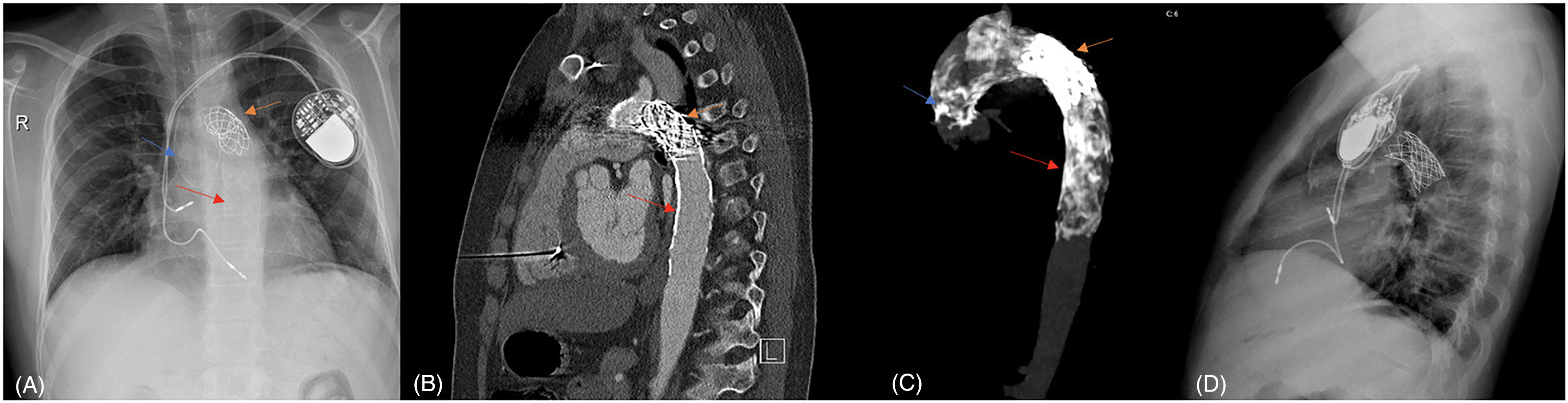
(A) A chest radiograph taken one month later demonstrates that the findings of cardiomegaly and pulmonary congestion have regressed. The calcification affecting the vessel wall in both the ascending and descending aorta and the stent placed percutaneously is indicated with arrows. (B) In the sagittal section of the computed tomography (CT) angiography, it can be observed that the stent placed at the isthmus level is in the appropriate position. (C) The CT angiography image displays a widespread calcific lesion extending from the ascending aorta to the thoracic aorta and a stent positioned at the isthmus level in MIP range format. (D) A lateral chest radiograph confirmed appropriate positioning and stability of the stent at the six-month follow-up.
Discussion
Coral reef aorta is a calcific aortic lesion primarily seen in adult and elderly patients. While predominantly located in the supra- and infrarenal regions, CRA can affect various segments of the aorta. In the literature, rare case reports show that CRA can rarely be located in the ascending aorta, aortic arch, and thoracic aorta.
Although the etiopathogenesis of CRA is not fully understood, chronic atherosclerosis, inflammation, and thrombus-related calcification have all been proposed as potential contributors. Chronic kidney disease, while not directly implicated in CRA pathogenesis, is a recognized risk factor for vascular calcification. In patients with extensive circumferential aortic calcification, where cross-clamping is challenging or surgical risk is elevated, endovascular approaches may provide a viable alternative to traditional surgical methods such as thromboendarterectomy or anatomical/extra-anatomical bypass. A limited number of cases have been reported in the literature regarding stenosis in the aortic arch due to CRA. In a 26-year-old patient followed up with diabetes and hyperlipidemia by Sonawane et al, CRA, which caused significant stenosis at the isthmus level, was detected in the examinations performed due to hypertension. In this case, successful endovascular treatment was performed with a balloon-expandable covered stent. 5 Le Bars et al found that a 64-year-old patient with severe heart failure and low ejection fraction (EF: 10%) had CRA, which resulted in severe stenosis in the aortic arch; successful endovascular treatment was achieved using a covered stent. 6
One of the most significant challenges in the endovascular treatment of CRA in the aortic arch is the risk of aortic rupture and dissection. While self-expanding stents are commonly employed in the management of aortic aneurysms or dissections, where gradual radial force and conformability are advantageous, they may be insufficient in treating heavily calcified, intraluminal stenotic lesions. 7 Therefore, a balloon-expandable covered stent was preferred in this case to provide immediate and controlled radial expansion against the rigid, calcified aortic wall. To minimize the risk of rupture, the stent was deliberately under-dilated using a balloon smaller than the reference diameter of the descending aorta.
While endovascular approaches have been explored in selected high-risk adult patients, reports of such interventions in pediatric populations are exceedingly rare. Jha et al described CRA-related stenosis in the ascending aorta of a 12-year-old, which was surgically managed. 8 In contrast, to our knowledge, our case appears to be the first in which endovascular stent implantation was successfully performed for severe aortic coarctation due to CRA in an adolescent.
Conclusion
Although the Coral reef aorta is primarily located in the visceral part of the aorta, it can manifest in any region. Therefore, endovascular treatments may be considered an option for the Coral reef aorta in more complex locations, such as the aortic arch. The use of covered and balloon-expandable stents for endovascular treatment, as well as the dilation of the coarctation area using a balloon smaller in diameter than the descending aorta, can prevent the development of fatal complications such as aortic rupture and dissection.
Footnotes
Authors’ Statement
Each of the patients in this case series and/or their parents were fully informed and gave written consent for publication of this report.
Authors’ Contributions
Mete Han Kızılkaya contributed to patient diagnosis and follow-up, conceptualization and design of the work, manuscript writing, and final approval. Cemile Pehlivanoğlu contributed to patient diagnosis and follow-up. Batıgül Taş contributed to conceptualization and design of the work, data collection, manuscript writing, and literature search. Terman Gümüş contributed to patient diagnosis and follow-up. Mehmet Biçer contributed to patient diagnosis and follow-up. Ilmay Bilge contributed to manuscript revision and final approval of the manuscript. Ender Ödemiş contributed to conceptualization and design of the work, manuscript revision, and final approval of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
