Abstract
Inflammatory myofibroblastic tumor (IMT) of the heart is a very rare tumor, constituting less than 5% of primary heart tumors. This case report describes the unexpected finding of IMT of the heart causing pulmonary artery obstruction in an asymptomatic toddler.
During a well-child visit, a healthy 15-month-old girl weighing 11 kg was found to have a new grade 2 to 3 systolic murmur. Transthoracic echocardiography revealed a mass obstructing the main pulmonary artery (PA) with a right ventricle-PA (RV-PA) gradient of 55 mm Hg. Surprisingly, right ventricular function was normal (Figure 1). Cardiac magnetic resonance imaging (Figure 2) showed that the mass was hypointense, well-demarcated, and attached to the anterior cusp of the pulmonary valve, reducing the main PA lumen by at least 60%. The mass could not be further characterized by imaging.
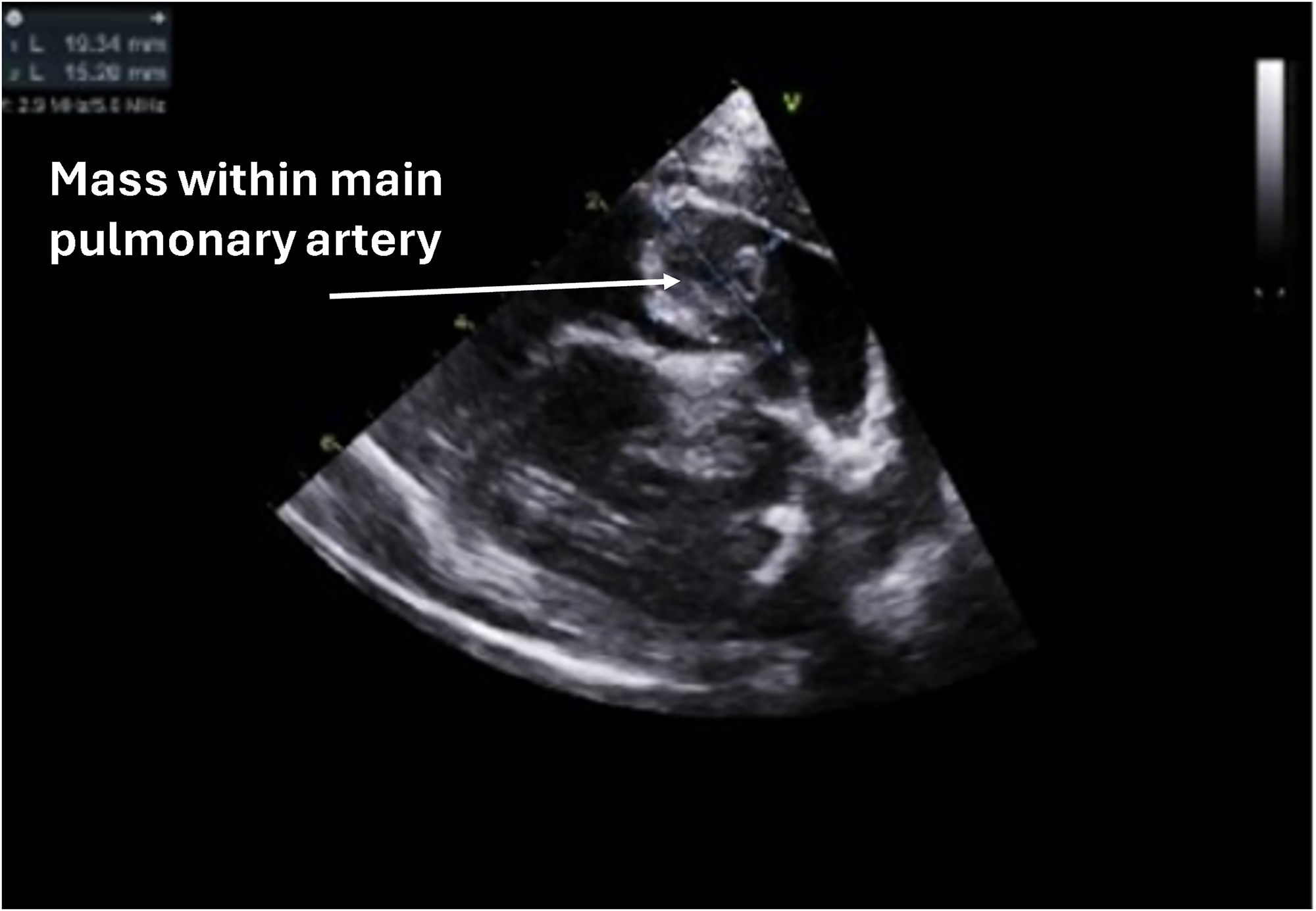
2D echocardiographic imaging displaying the mass obstructing right ventricular outflow through the main pulmonary artery. The attachment to the pulmonary valve cusp is not visible.
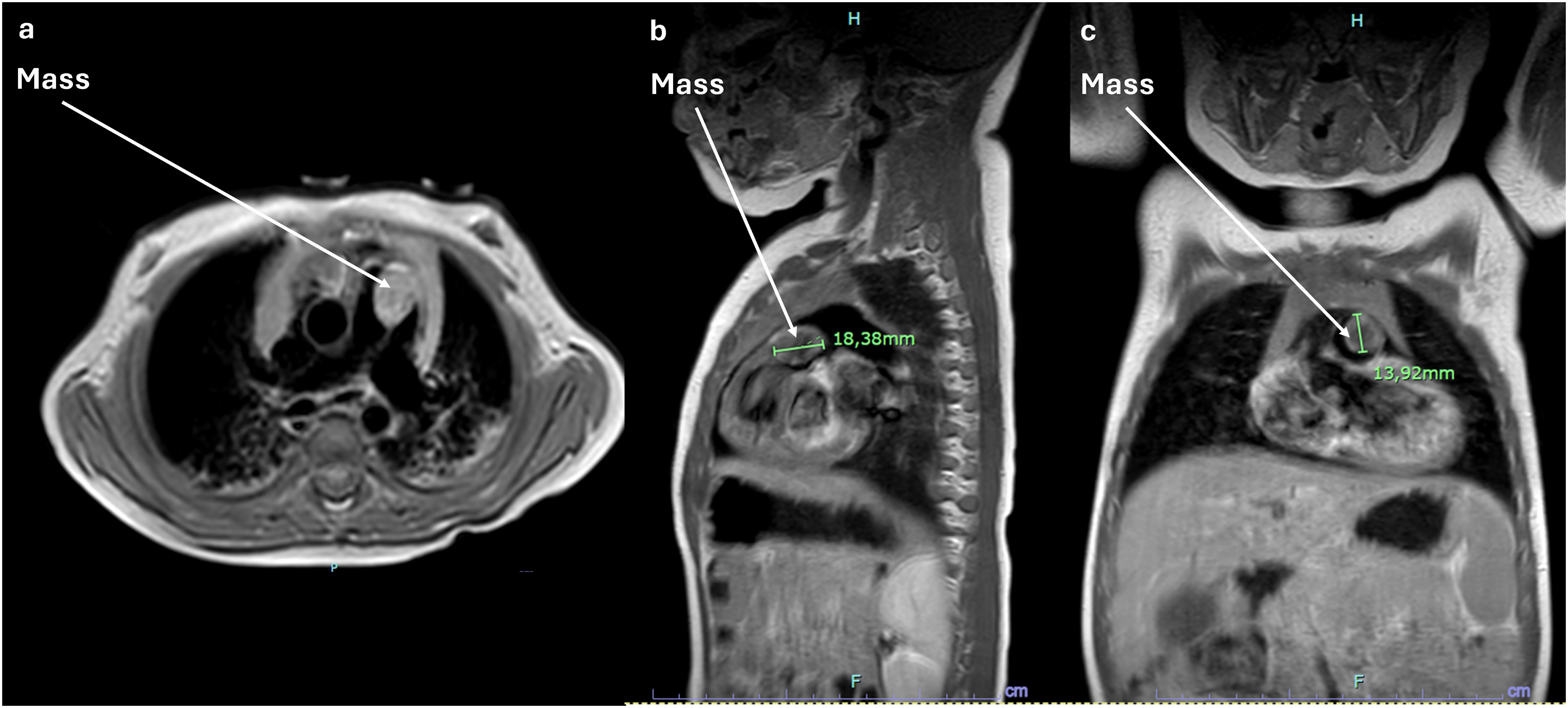
Mass viewed on magnetic resonance imaging (MRI) in (a) transverse, (b) sagittal, and (c) coronal sections.
A decision was made to proceed with surgical excision. The distal ascending aorta and both vena cavae were cannulated. After cardioplegia was delivered the right atrium was opened, and a small interatrial communication was created to decompress the left heart. A transverse incision was then made in the main PA, and the mass was visualized. It was smooth, firm, whitish, and measured 2.5 × 1.5 × 0.8 cm. It was pedunculated to the remnant ductus arteriosus on the PA wall and attached to the base of the anterior pulmonary valve cusp and the muscular base in the right ventricular outflow tract. The mass was excised with its attachment sites, along with the cusp that was totally incorporated into the mass, and tissue samples were sent for analysis (Figure 3). The two other leaflets remained intact. The PA and right atrium incisions were closed with 6-0 Prolene sutures, while the interatrial communication was closed with 5-0 Prolene. Although the pulmonary valve was not repaired, there was normal right ventricular function postoperatively, with an RV-PA gradient of 9 mm Hg and mild pulmonary regurgitation (PR). Cardiopulmonary bypass and cross-clamp times were 67 and 44 min, respectively.
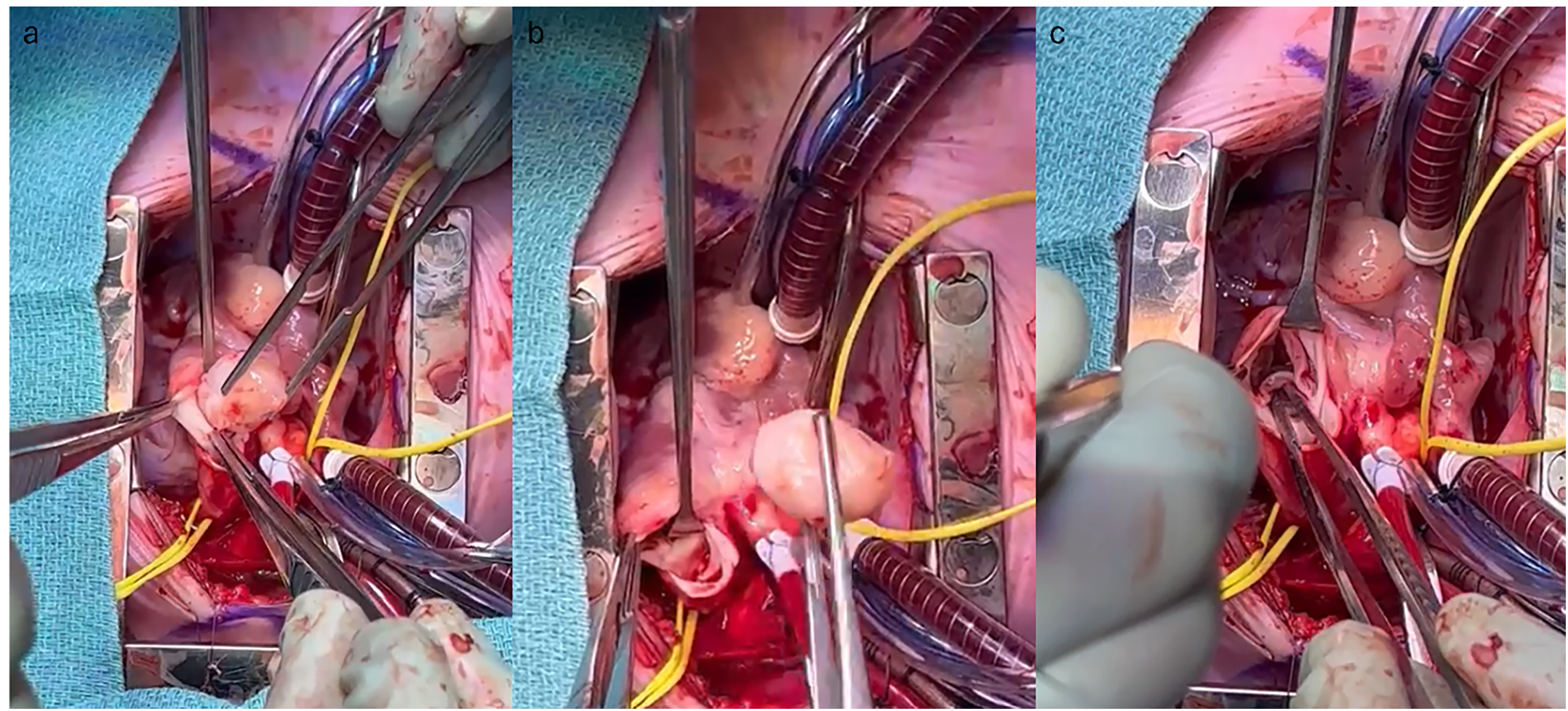
(a) Smooth, white-colored mass obstructing the right ventricular outflow tract and the main pulmonary artery. (b) Resected mass held by pick-ups. (c) Status-post mass excision with the two remaining pulmonary valve cusps.
Pathological analysis revealed a fascicular architecture of fusiform cells without cytonuclear atypia, mitoses, or necrosis. Cells had an elongated eosinophilic cytoplasm and ovoid vesicular nucleus. There was evidence of peripheral neovascularization and myxoid changes, as well as a lymphoplasmacytic infiltrative process. No sign of neoplasia was found at the attachment sites. Immunohistochemistry showed fusiform cells positive for smooth muscle actin (SMA), CD34, and factor XIII, but negative for CD31, keratin, AE1/AE3, desmin, S-100, myogenin, ALK-1, STAT6, and panTRK (Figure 4). Fluorescence in situ hybridization was negative for ALK or ETV6 rearrangements. Genetic testing revealed a PDGFRA fusion combining exons 1 to 22 from PDGFRA and exons 5 to 20 from USP8. Overall, this was indicative of an inflammatory myofibroblastic tumor (IMT). Postoperatively, the patient had a swift recovery and was discharged home three days later. She remains asymptomatic and free of recurrence 18 months later, with her last echocardiogram showing mild PR. She continues to be followed yearly by cardiology with an echocardiogram.
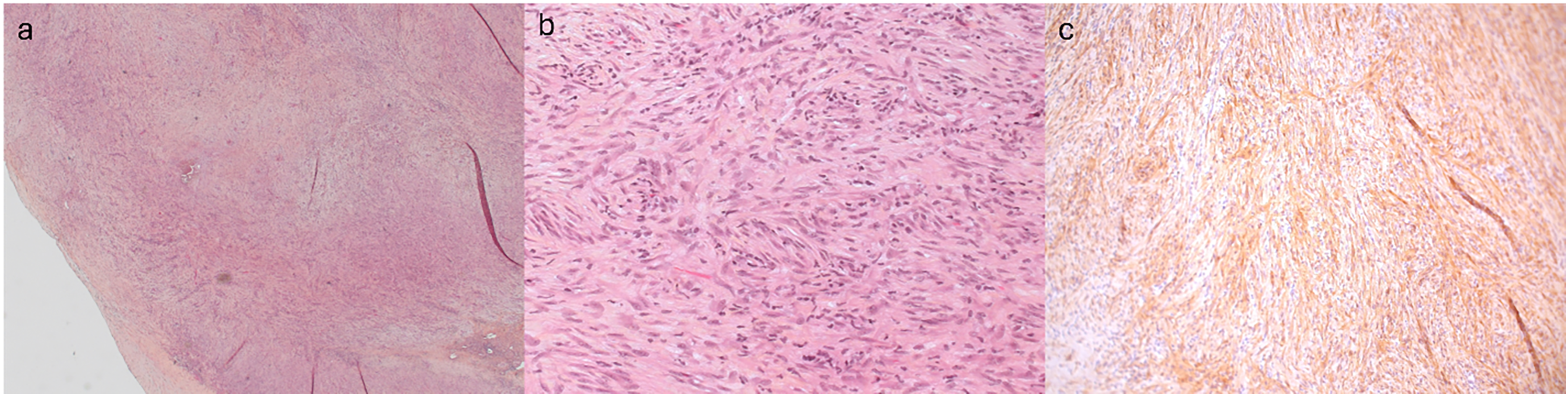
Histology of the excised tissue. (a) Homogeneous architecture with fascicles of spindle cells originating from the pulmonary artery (H&E staining ×25). (b) Spindle cells without cytologic atypia associated with few interspersed lymphocytes (H&E staining ×200). (c) Spindle cells are positive for smooth muscle actin (SMA) immunohistochemistry staining.
Discussion
Inflammatory myofibroblastic tumor of the heart is an extremely rare tumor representing around 5% of primary heart tumors, with fewer than 100 cases documented. 1 Most pediatric cardiac tumors are benign, with cardiac rhabdomyomas being the most common (33%), followed by fibromas (18%), myxomas (18%), and teratomas (8%). 2 Inflammatory myofibroblastic tumor rarely develops in the heart, but it has been reported in the lung, spleen, liver, and gallbladder. 3 In the heart, IMT is typically endocardial, most commonly in the right atrium or RV. 1 Pulmonary artery involvement is extremely rare. While some patients are asymptomatic, others experience dyspnea, cyanosis, pallor, and chest pain. 4 About a third of documented cases are in patients under one-year of age. 1
The etiology is unclear. It is sometimes considered a primary immunologic lesion, shrinking with steroid use, while others classify it as a neoplasm. While usually benign, post-resection recurrence has been documented. Its prognosis is related to location rather than neoplastic potential. Interestingly, atypical and aggressive cases of extracardiac IMT have been reported. 3 Magnetic resonance imaging findings are not well described due to its rarity.
The differential diagnosis of a cardiac soft tissue mass in children is broad. In IMT of the heart, the mass is typically pedunculated and composed of spindle cells with little atypia or mitosis, along with inflammatory cells in myxoid stroma. These spindle cells may be associated with small blood vessels. 4 Although cardiac myxomas may resemble IMTs, IMT is usually SMA-positive, whereas myxomas are typically not. While myxomas are often CD34-positive, IMT is usually CD34-negative, contrary to our patient. 5 Around 50% of IMT cases express ALK-1, with ALK negativity indicating a less favorable outcome. 4 In ten documented cases of infants with IMT of the heart, ALK expression was negative. 1
This patient had a fusion of PDGFRA, combining exons 1 to 22 from PDGFRA and exons 5 to 20 from USP8. This fusion has been reported in four cases of calcified chondroid mesenchymal neoplasms 6 and in a pediatric cardiac spindle cell neoplasm. 7
This report describes an unusual finding of IMT of the heart in an asymptomatic female toddler, causing PA obstruction. Despite the obstruction, the right ventricular function was preserved. To our knowledge, this is the second such documented case, highlighting its rarity. 8
Supplemental Material
Supplemental Material
Footnotes
Authors' Statement
Verbal and written permission were granted by the parent of the patient to publish the case report.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
