Abstract
Background
Aspirin is frequently utilized for antiplatelet therapy in children with congenital heart disease (CHD). Patients who are unresponsive to aspirin, as measured by aspirin reaction units (ARU), are at higher risk for thrombotic events. It is undetermined if dose modification of aspirin results in adequate responsiveness in these patients. This study evaluates the prevalence and risk factors for aspirin nonresponsiveness and the results of dose escalation in this population.
Methods
This is a retrospective review of patients cared for in the cardiac care unit at a quaternary care academic congenital heart center who received aspirin and had responsiveness evaluated between January 2018 and January 2023. Patient demographics and clinical characteristics were extracted from the medical record. Descriptive, parametric, and nonparametric univariate analysis were employed.
Results
A total of 142 patients (69 [49%] female, 45 [32%]Non-Hispanic White, and 63 [44%] Hispanic]) were identified. Median age at first aspirin responsiveness assessment was 54 [interquartile range, IQR: 23.3-411.5] days with a median weight of 5.2 [IQR: 3.64-9.29] kg. Of these, 32/142 (22.5%) were nonresponsive on their initial testing. Of these patients, 23/32 (72%) had follow-up testing with 19/23 (83%) subsequently becoming therapeutic. This was achieved with an increased dose in 12/19 (63%) patients and increased duration of therapy in 7/19 (37%) patients. Seventeen of 142 (12%) patients experienced a thrombotic event, 13/17 (77%) of which were therapeutic on initial responsiveness assessment.
Conclusions
It is common for CHD patients to be aspirin nonresponsive with initial weight-based dosing. If aspirin is used in this population, it is necessary to evaluate ARUs on all patients as underdosing is not uncommon with current weight-based dosing methods.
Introduction
Aspirin is commonly administered to congenital heart disease (CHD) patients because they have an elevated risk for thrombosis, particularly in the perioperative period.1,2 Risk factors for thrombosis in this population include central venous catheters, younger age, cyanosis, shunt-dependent single-ventricle circulation, and duct-dependent pulmonary circulation.3–7 Thrombosis carries an increased risk of mortality, longer length of stay, and other complications. 8 Heparin and its derivatives, in conjunction with aspirin, are the most frequently utilized agents for thrombosis prevention. While aspirin doses of 1 to 5 mg/kg/d are suggested in clinical practice, the dosing of aspirin for optimal thrombosis prevention in children remains unclear. 9
There is significant variability in interpatient response to aspirin, with nonresponsiveness reported in 10% to 26% of patients with CHD.7,10,11 Patients found to be nonresponsive are at higher risk for thrombotic complications, including loss of venous access, limb swelling, pain, and discoloration, superior vena cava syndrome, and pulmonary embolism. 10 It is unknown whether dose escalation or increased treatment duration may impact responsivity to aspirin in the CHD population. This study seeks to assess prevalence of, and risk factors for, aspirin nonresponsiveness as well as response of initial nonresponders to interventions such as increased aspirin dose and/or duration in a CHD population being cared for in a cardiac intensive care unit.
Material and Methods
This was a retrospective review of all CHD patients cared for in the cardiac intensive care unit at Dell Children's Medical Center from January 7, 2018, through January 11, 2023. All patients who received aspirin for antiplatelet therapy and underwent a test for aspirin responsiveness were included. Patients who were receiving other forms of antiplatelet therapy (eg, Clopidogrel) or received aspirin for reasons other than antiplatelet therapy were excluded from analysis. In addition to information on aspirin dosing, patient demographics, congenital heart diagnoses, surgeries, and thrombotic events were extracted from patient charts. The Institutional Review Board at the University of Texas at Austin Dell Medical School approved this retrospective chart review, “Aspirin Response Units in the Critical Care Unit” (IRB#: STUDY00001543), on January 27, 2023. Procedures were followed in accordance with the ethical standards of the responsible committee on human experimentation and with the Helsinki Declaration of 1975.
Aspirin Responsiveness
Per protocol, weight-based dosing of 5 mg/kg/d rounded for convenient administration determined the initial dose. Aspirin responsiveness was evaluated via standard laboratory practices with the VerifyNow Aspirin assay (Instrumentation Laboratory Company), resulting in a value in aspirin responsiveness units (ARU). VerifyNow is a point-of-care device, utilizing arachidonic acid, that assesses platelet dysfunction secondary to aspirin and can be administered as soon as 2 h following at least one aspirin dose. 12 A value of 550 ARUs or greater indicated aspirin nonresponsiveness, while a value less than 550 ARUs indicated aspirin responsiveness. 10 While the VerifyNow Aspirin assay has only been validated in adults, this assay is commonly utilized in the pediatric population with a value of 550 ARUs or greater indicating aspirin nonresponsiveness.10,11,13,14 Decisions on timing of aspirin responsiveness testing and changes to aspirin dosing as a result of the responsiveness testing outcome were made by the multidisciplinary care team consisting of cardiac intensivists, cardiologists, cardiac surgeons, and hematologists.
Thrombosis Monitoring
As part of our Society of Thoracic Surgeons data collection and reporting system, all patients underwent prospective follow-up and data collection for all thrombotic events. Per the Society of Thoracic Surgeons data collection and reporting system, thrombotic events were defined as a mass of platelets, fibrin, or other blood elements occurring in one or more of the following locations: intracardiac, central vein, pulmonary artery, peripheral deep vein, systemic to pulmonary artery shunt, systemic artery in situ (central or peripheral), and systemic artery embolic. 15 Thrombotic events were further defined as occurring prior to discharge within the surgical hospital stay or through the 30th postoperative day if discharged before 30 days.
Statistical Analysis
Categorical variables are displayed as n (%) while normally distributed continuous data are displayed in mean ± SD; non-normally distributed continuous data are displayed in median [interquartile range, IQR]. Descriptive statistics, parametric (χ2 and t tests) and nonparametric (Fisher exact test and Kruskal-Wallis) analyses were utilized to compare variables. All statistical tests are two-tailed, and a P < .05 was considered statistically significant. All statistical analyses were performed using R and RStudio. 16
Results
Patient Characteristics
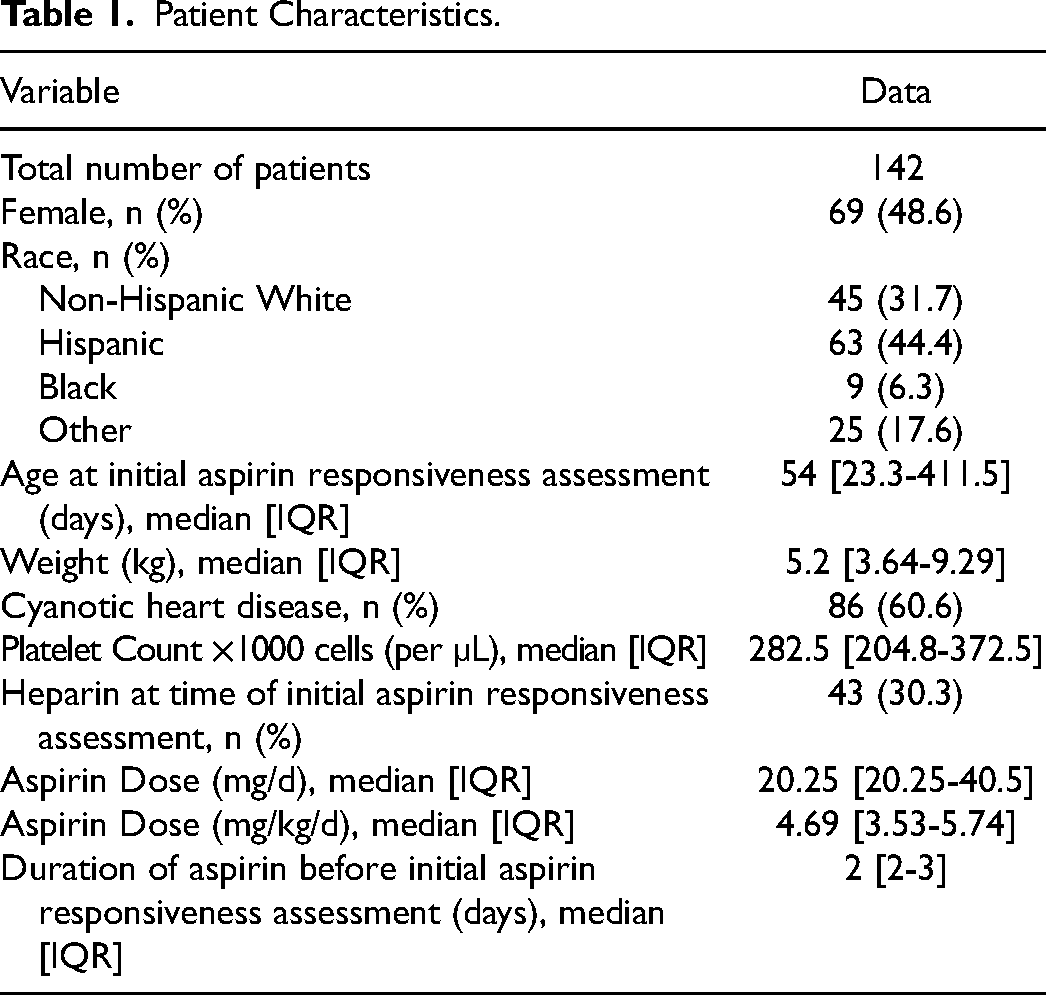
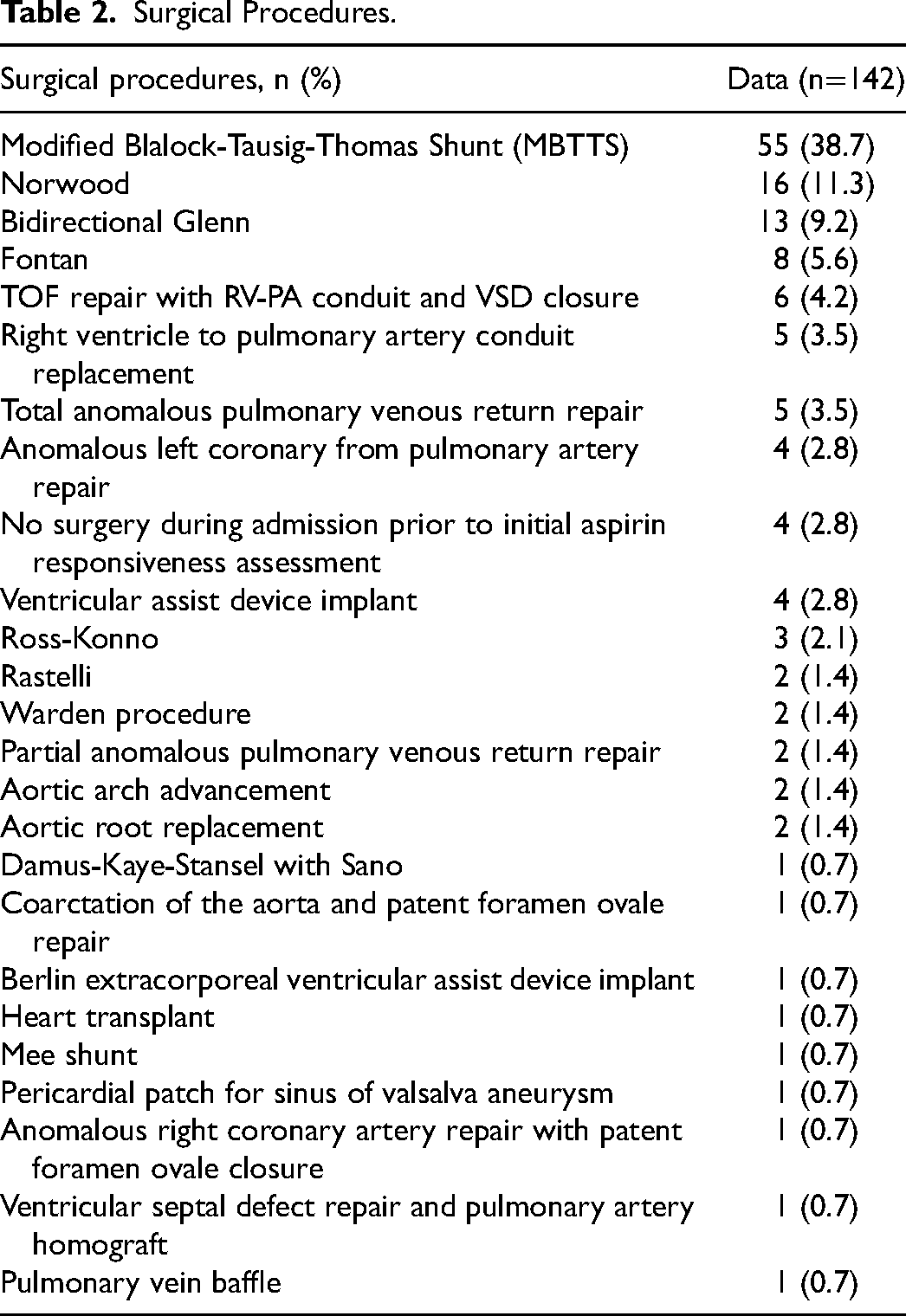
A total of 142 patients were identified for inclusion in the analysis. A total of 235 aspirin responsiveness assessments were done with a range of one to nine assessments per patient. Median age at first aspirin responsiveness assessment was 54 [IQR: 23.3-411.5] days with a median weight of 5.2 [IQR: 3.64-9.29] kg; 86/142 (60.6%) had cyanotic heart disease, 69/142 (48.6%) patients were females, 45/142 (31.7%) were White non-Hispanic, and 63/142 (44.4%) were Hispanic (Table 1). The three most common surgical interventions performed during the admission prior to initial aspirin responsiveness assessment were (1) Modified Blalock-Tausig-Thomas shunt (MBTTS) in 55/142 (38.7%) patients, (2) Norwood procedure in 16/142 (11.3%), and (3) Bidirectional Glenn procedure in 13/142 (9.2%) (Table 2). No patients had Factor V Leiden, protein C, protein S, or antithrombin III deficiency. No patients experienced a bleeding event while on aspirin therapy.
Patient Characteristics.
Surgical Procedures.
Initial Aspirin Responsiveness
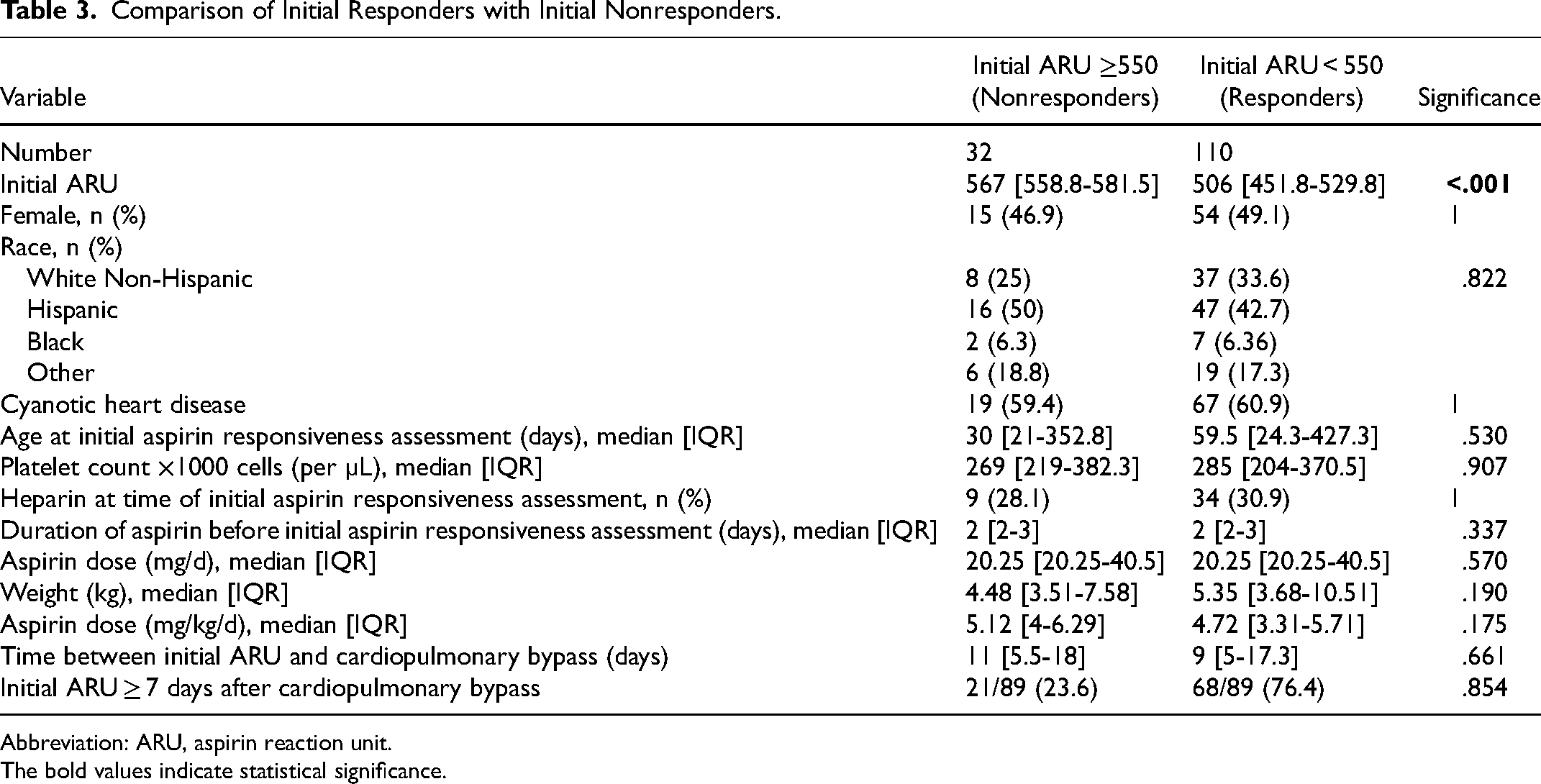
Median aspirin dose was 20.25 [IQR: 20.25-40.5] mg/d and 4.69 [IQR: 3.53-5.74] mg/kg/d, and patients were taking aspirin for a median of 2 [IQR: 2-3] days prior to responsiveness assessment (Table 1). Initial aspirin responsiveness was collected a median of 9 [IQR: 5-17.5] days after surgery requiring cardiopulmonary bypass. Median platelet count at the time of initial responsiveness assessment was 282.5 [IQR: 204.8-372.5] ×1000 cells (per µL). Forty-three (30.3%) patients were on heparin at the time of responsiveness assessment. Of those on heparin at time of responsiveness assessment, 9 (20.9%) were aspirin nonresponsive and 34 (79.1%) were aspirin responsive (P = 1) (Table 3). Of those on heparin at time of responsiveness assessment, aspirin nonresponsive patients had a median platelet count of 259 [IQR: 190-425], and aspirin responsive patients had a median platelet count of 288 [IQR: 248.8-398.5] (P = .57).
Comparison of Initial Responders with Initial Nonresponders.
Abbreviation: ARU, aspirin reaction unit.
The bold values indicate statistical significance.
Initial Aspirin Nonresponsiveness
During the initial aspirin responsiveness assessments, 32/142 (22.5%) had ≥550 ARU and were deemed nonresponsive. Between initial aspirin nonresponders and responders, no differences were found in gender (15/32 [46.9%] vs 54/110 [49.1%] female, P = 1), race (8/32 [25%] vs 37/110 [33.6%] white non-Hispanic, P = .677), cyanotic heart disease (19/32 [59.4%] vs 67/110 [60.9%], P = 1), age at assessment (30 [IQR: 21-352.8] vs 59.5 [IQR: 24.3-427.3] days, P = .53), heparin use (9/32 [28.1%] vs 34/110 [30.9%], P = 1), days of aspirin (2 [IQR: 2-3] vs 2 [IQR: 2-3], P = .337), aspirin dose (5.12 [IQR: 4-6.29] vs 4.72 [IQR: 3.31-5.71] mg/kg/d, P = .175), and duration since cardiopulmonary bypass (11 [IQR: 5.5-18] vs 9 [IQR: 5-17.3] days, P = .661) (Table 3). A subgroup analysis of patients who had an initial aspirin responsiveness assessment greater than or equal to seven days after cardiopulmonary bypass was conducted and demonstrated that 21 (23.6%) patients were aspirin nonresponders and 68 (76.4%) patients were aspirin responders (P = .854) (Table 3).
Aspirin Dose and Duration Escalation
Of the 32 patients who were initially aspirin nonresponsive, 23 (71.9%) had at least one follow-up aspirin responsiveness assessment. Of the 9/32 (28.1%) who did not have a follow-up assessment, 2/9 (22.2%) were discharged from the hospital within 48 h after initial assessment and thus did not undergo repeat assessment. An additional 3/9 (33.3%) continued on aspirin therapy and were inpatient for 12 to 14 days after initial aspirin response assessment but repeat assessments were not performed per the care team's decision. Aspirin dose was increased in 4/9 (44.4%) patients whoexperienced an increase in aspirin dose upon discharge, where 2/4 (50.0%) patient's dose was increased from 20.25 to 40.5 mg, 1/4 (25.0%) patient's dose increased from 40.5 to 81 mg, and 1/4 (25.0%) patients dose increased from 81 to 162.5 mg with no repeat assessment. None of the nine patients without follow-up assessments were readmitted to the hospital with a thrombotic event.
For the 23 patients who were initially nonresponsive and had follow-up assessment, 19 (82.6%) subsequently became responsive. Responsiveness was achieved with increased aspirin dose in 12/19 (63.2%) patients with 11/12 (91.7%) patients' dose increased from 20.25 to 40.5 mg and 1/12 (8.3%) patient's dose increased from 81 to 121.5 mg. In the other 7/19 (36.8%) patients, responsiveness was achieved at the same dose of aspirin with longer duration of therapy, where 4/7 (57.1%) patients were responsive one day after initial nonresponsiveness and 3/7 (42.9%) patients were responsive two or three days after initial nonresponsiveness. Four of 23 (17.4%) patients never achieved aspirin responsiveness despite increases in aspirin dose and repeat assessments after the initial nonresponsive assessment. Two of 4 (50%) of these patients were placed on Lovenox and 2/4 (50%) were continued on the same dose of aspirin. After dose and duration escalation, 138 (97.2%) of 142 patients achieved aspirin responsiveness.
Thrombotic Event Characteristics
Seventeen (12%) of the 142 patients experienced a thrombotic event within 30 days of the initial aspirin responsiveness assessment. Thrombotic events had a higher incidence in patients of younger age at initial ARU (30 [IQR: 15-56] vs 63 [IQR: 25-513] days of age, P = .008), patients who weighed less at initial ARU (3.71 [IQR: 3.55-5.2] vs 5 [IQR: 3.67-11.3] kg, P = .022), patients with cyanotic heart disease (16 [94.1%] vs 70 [56%], P = .003), patients with MBTTS as the primary operation (10/17 [58.8%] vs 45/125 [36%], P = .034), and patients with greater time between cardiopulmonary bypass and initial ARU (17 [IQR: 9-29] vs 9 [IQR: 5-16.8] days, P = .010) (Table 4). In addition to the thrombotic events occurring after MBTTS, Norwood, and Glenn operations, thrombotic events also occurred in one patient after right ventricle to pulmonary artery conduit replacement and in one patient after total anomalous pulmonary venous return repair.
Comparison of Patients With Thrombotic Events and Without Thrombotic Events.

Abbreviations: ARU, aspirin reaction unit; MBTTS, Modified Blalock-Tausig-Thomas shunt.
The bold values indicate statistical significance.
On univariate logistic model, patients with cyanotic heart disease were 12 times more likely to experience a thrombotic event (OR: 12.57, 95% CI: 1.61-97.71, P = .015). On univariate logistic model, there was no significant difference in risk for thrombotic event among patients of younger age at initial ARU (OR: 0.99, 95% CI: 0.99-1.00, P = .173), lower weight at initial ARU (OR: 0.81, 95% CI: 0.63-1.03, P = .08), patients with MBTTS (OR: 2.53, 95% CI: 0.90-7.13, P = .07), and patients with greater time between cardiopulmonary bypass and initial ARU (OR: 2.09, 95% CI: 0.64-6.79, P = .218).
Thrombotic event location varied with 6 (35.3%) thromboses occurring in the inferior vena cava, 1 (5.9%) extending from the inferior vena cava to a common femoral vein, 3 (17.6%) extending from an external iliac vein to a common femoral and saphenous vein, 3 (17.6%) occurring in a common femoral vein, 1 (5.9%) occurring in a common femoral artery, 2 (11.8%) occurring in a cephalic vein, and 1 (5.9%) occurring in a basilic vein (Table 5).
Thrombotic Event Locations.

Thrombotic Events
Of the 17 patients who experienced a thrombotic event, 13/110 (11.8%) were initial aspirin responders and 4/1712.5%) were initial nonresponders. Of the four initial nonresponders experiencing a thrombotic event, 2/4 (50%) became aspirin responsive on subsequent ARU assessments prior to the thrombotic event (Figure 1). There was no difference in thrombotic event occurrence comparing initial aspirin responders, initial aspirin nonresponders who became responsive, and initial aspirin nonresponders who remained nonresponsive (13 [11.8%] vs 2 [10.5%] vs 2 [50%], P = .252).
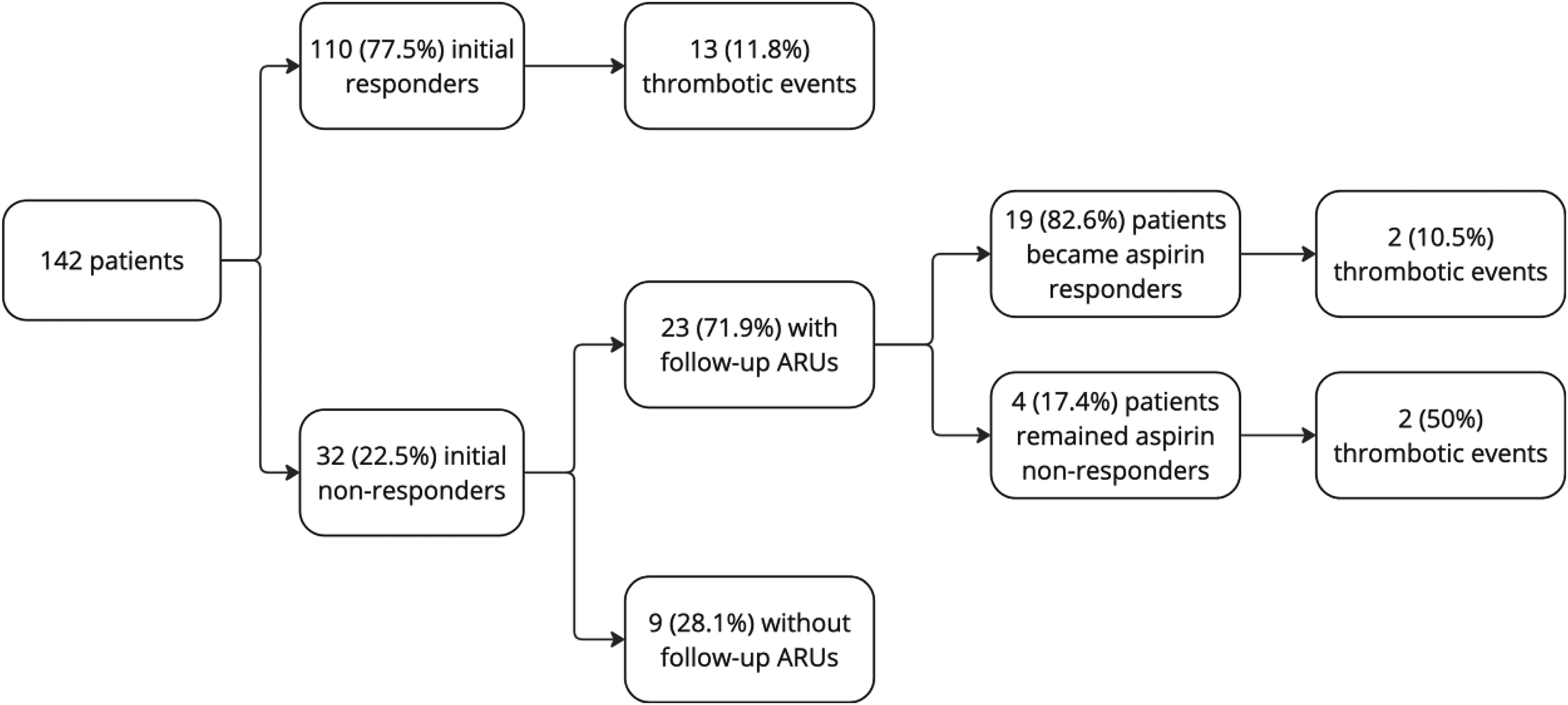
Flowchart of patient responsiveness and thrombotic events.
Of the 13 initial aspirin responders who experienced thrombotic events, 8 (61.5%) experienced the thrombotic event one to eight days after the most recent aspirin responsiveness assessment, 4 (30.8%) experienced the thrombotic event 12 to 15 days after the most recent aspirin responsiveness assessment, and 1 (7.7%) experienced the thrombotic event 30 days after aspirin responsiveness assessment. Of the two initial aspirin nonresponders who subsequently became responsive, 1 (50%) experienced the thrombotic event one day after the most recent aspirin responsiveness assessment and 1 (50%) experienced the thrombotic event 15 days after the most recent aspirin responsiveness assessment. Of the two initial aspirin nonresponders who remained nonresponsive, 1 (50%) experienced the thrombotic event 16 days after the most recent aspirin responsiveness assessment, and 1 (50%) experienced the thrombotic event 28 days after the most recent aspirin responsiveness assessment.
Comment
Previous studies have revealed significant variation in aspirin responsiveness between patients. This study was consistent in demonstrating this variability, with a nonresponsiveness rate of 22.5%, compared with previous reports of 10% to 26%.7,10 Importantly, the results from this institution demonstrated that the majority of nonresponders can be managed with either a dose escalation or simply repeat assessment after a longer treatment course. Nineteen of the 23 (82.6%) patients who were initial nonresponders, who had an increased dose of aspirin or longer duration of aspirin, became responsive, resulting in 138 (97.2%) of the original 142 patients achieving aspirin responsiveness. This is in line with previous data suggesting true aspirin nonresponsiveness occurs in only 2% of children. 17
Emani et al evaluated aspirin responsiveness specifically in postoperative CHD patients at high risk for thrombosis. 10 Their study found an association between aspirin nonresponsiveness with lower weight and an aspirin dose of 20.25 mg/kg when compared to higher doses of aspirin. While their study was not intended to evaluate aspirin dose management, they found two of their initially nonresponsive patients became responsive after dose escalation. Others have also questioned if aspirin nonresponsiveness was truly an intrinsic nonresponse or the result of underdosing. 10 In this study, we identified 19 of 23 (82.6%) patients who were initially nonresponders who became responsive with increased aspirin dose or duration, with seven becoming responsive with increased duration of aspirin alone, further demonstrating the variability of achieving aspirin responsiveness with current weight-based dosing guidelines. The other 12 patients received an increased dose of aspirin prior to becoming responsive. However, as these patients did not undergo a responsiveness assessment with greater aspirin duration prior to the dose escalation, we are unable to determine if any of these patients would not have required dose escalation and would have become responsive with greater duration alone.
Our study did not identify any differences in demographics, clinical characteristics, cardiopulmonary bypass time or testing practices between aspirin responders and nonresponders. This contrasts with a study by Newland et al which found that patients of male sex were more likely to be aspirin responsive compared with patients of female sex. 11 Further work should attempt to identify other factors (eg, biologic or genetic) which may identify those at risk for being nonresponders to aspirin therapy.
In this investigation, 12% (17/142) of patients had a thrombotic event within 30 days of their initial aspirin responsiveness assessment. Thirteen (76.5%) of these 17 patients were aspirin responders on their initial assessment. Thus, we did not observe an association between initial aspirin nonresponsiveness and thrombotic events within 30 days. This compares with the aforementioned Emani study in which 10 (10.5%) patients were initially aspirin nonresponsive and six of those ten had a thrombotic event. This discrepancy may represent the result of aspirin responsiveness being utilized in clinical decision-making surrounding antiplatelet and anticoagulation therapy in this patient population. That said, of the 13 patients who had thrombotic complications, eight developed the thrombotic event one to eight days after aspirin initiation. Despite having a therapeutic aspirin responsiveness assessment at the time of thrombosis, it could be postulated that the thrombotic event started prior to the initiation of aspirin.
Our study identified that patients with cyanotic heart disease, MBTTS, those with a longer duration between initial aspirin responsiveness assessment and cardiopulmonary bypass, and younger and smaller patients at initial ARU experienced more thrombotic events. Furthermore, a univariate logistic model revealed that patients with cyanotic heart disease were 12 times more likely to experience a thrombotic event. There were no significant differences in gender, race, platelet count, administration of heparin at time of initial aspirin responsiveness assessment, duration of aspirin prior to aspirin responsiveness assessment, initial aspirin dose (mg/d or mg/kg/d), final aspirin dose (mg/d or mg/kg/d), and initial or final aspirin responsiveness assessment between patients with and without a thrombotic event.
Our study did not demonstrate a significant difference in initial and final ARUs (513 [IQR: 445-541] vs 521 [IQR: 473-543] initial; 506.5 [IQR: 469.5-544] vs [512 [IQR: 462.5-529] final), and therefore aspirin responsiveness, when comparing patients with and without thrombotic events. Additionally, 11.8% (13/110) of patients experiencing a thrombotic event were responsive to aspirin prior to thrombosis. While our analysis did not demonstrate an association between thrombosis and aspirin responsiveness utilizing the VerifyNow Aspirin assay definition of greater than 550 ARU, previous research by Newland et al has demonstrated this association. 11 This suggests that further evaluation of a clinically meaningful threshold for the VerifyNow Aspirin assay is needed in the pediatric population. Given these findings, utilizing an aspirin responsiveness definition of greater than 550 ARU as a means to detect platelet dysfunction may need reconsideration in the pediatric population. Furthermore, while previous investigations have suggested an association between aspirin nonresponse and thrombosis, this does not mean that achieving aspirin responsiveness will reduce their risk for thrombosis. Further studies with larger sample sizes are needed to investigate the risk profile for thrombosis in CHD patients with initial nonresponsiveness who then become responsive.
The pediatric CHD population is at significant risk for thrombotic events. When these events occur, they have the potential to cause significant long-term morbidity, including neurological deficits and death. 7 Other groups have demonstrated in the cardiac patient population that when assessed, those who showed adequate aspirin response were at lower risk for thrombotic events. If true, this accentuates the potential importance of achieving aspirin responsiveness. Given that many initially aspirin nonresponsive patients become responsive with an increased duration of time, reassessing aspirin responsiveness 48 to 72 h after a nonresponsive result is reasonable. However, for patients with concern for a high risk of thrombosis, increasing the aspirin dose at the initial nonresponsive result may be considered given a relatively low risk of bleeding complications. Transitioning patients from aspirin therapy to an anticoagulative therapy with Lovenox may be considered in patients that are at high risk of thrombosis or persistently nonresponsive on repeat aspirin assays.
Limitations
This is a retrospective review from a single institution. As such, there is the possibility of bias in patients selected for aspirin responsiveness assessment. Moreover, our data reflect institutional practices and policies outside of aspirin responsiveness testing which may impact the results of the study. Additionally, we did not measure blood thromboxane B2 or urine 11-dydrothromboxane B2 levels to confirm elevated levels in patients nonresponsive to aspirin. This study involves a relatively small sample of patients and events, all with significant clinical heterogeneity, which may result in insufficient power to determine differences.
Conclusions
Aspirin nonresponsiveness is not uncommon in the pediatric CHD population, and responsiveness should be monitored in all patients started on aspirin. Patients who are initially nonresponsive should undergo an increased duration and/or dose escalation of aspirin, and then be reassessed. Further research is needed to determine the effect of responsiveness-driven aspirin management on thrombotic events.
Footnotes
Abbreviations
Acknowledgments
C.S. and A.W. wrote the main manuscript text and C.S. prepared Tables 1-5 and Figure 1. All authors reviewed the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
