Abstract
Background
Tricuspid atresia (TA) is the second most common form of functionally univentricular heart. For patients with TA and normally related great arteries (Type I), left ventricular outflow tract obstruction (LVOTO) is rare.
Methods
From the Congenital Heart Surgeons’ Society multi-institutional cohort of 445 patients with Type I TA enrolled from 1999 to 2024 from 42 sites, 14 infants (3%) had interventions for associated LVOTO, either at presentation or after their first TA-related intervention.
Results
Of seven infants initially undergoing Norwood/Damus-Kaye-Stansel (DKS), six survived to Stage II, of whom five survived with one developing pulmonary hypertension and four achieving Fontan. An additional seven infants who were first managed with pulmonary artery band placement subsequently had bidirectional superior cavopulmonary anastomosis (BCPA) and a DKS procedure; there were six survivors, all achieving Fontan. All ten survivors who underwent the Fontan procedure had normal left ventricular and mitral valve function at the latest follow-up. The overall Kaplan-Meier survival estimate at 20 years for these 14 patients was 79% (70% CI, 66%-88%), and the median follow-up was 8.3 years (0.24-21.5).
Conclusions
While infants with TA and transposition of the great arteries are more likely to have LVOTO, this can also occur in the setting of normally related great arteries. Infants with Type I TA and LVOTO can be managed in the neonatal period with the Norwood procedure ensuring complete arch relief with acceptable outcomes. If LVOTO becomes evident after initial pulmonary artery band placement, a subsequent DKS procedure facilitates satisfactory success to Fontan.
Keywords
Introduction
Tricuspid atresia (TA) is a cyanotic congenital heart defect and is considered the second most common form of functionally univentricular heart, requiring a multistage palliation approach.1–3 Tricuspid atresia with normally related great arteries (Type I) is the most common morphology, accounting for approximately 70% of cases, and left-sided obstruction is not usually a feature for these patients. 1 Conversely, left ventricular outflow tract obstruction (LVOTO) is common for patients with TA and transposition of the great arteries (TGA) (Type II), either evident at initial presentation or occurring secondarily following pulmonary artery banding (PAB) with the development of subaortic stenosis. Controlling unrestricted pulmonary blood flow with PAB may promote or exacerbate the development of subaortic stenosis, especially in the presence of aortic arch obstruction.4,5 The Damus-Kaye-Stansel (DKS) procedure at the time of Stage II has been performed safely in these patients with single ventricle physiology with or at risk of developing systemic outflow obstruction, alleviating or avoiding the negative hemodynamic effects of the obstruction.6-8
Left ventricular outflow tract obstruction is rare in patients with TA when the great arteries are normally related (Type I); thus, the literature describing this morphology is scarce. 1 We describe characteristics of 14 such patients who underwent LVOTO procedures and their subsequent outcomes through multistage palliation.
Patients and Methods
From the Congenital Heart Surgeons’ Society (CHSS) multi-institutional cohort of 445 patients with Type I TA enrolled from 1999 to 2024 from 42 sites, 14 patients (3%) had interventions for associated LVOTO, either at presentation or after their first TA-related intervention. This included seven infants who underwent Stage I palliation, meaning all those having initial palliative procedures that involved proximal aortopulmonary amalgamation and hybrid procedures (Group I), and seven infants who were first managed with PAB placement and subsequently underwent Stage II with a bidirectional superior cavopulmonary anastomosis (BCPA) combined with a DKS procedure (Group II). Patient demographics, anatomic features, surgical characteristics, subsequent interventions, and outcomes are described. Systemic obstruction was defined as reported in submitted echocardiography reports (recordings not available), either from qualitative appearance of narrowing or hypoplasia or quantitatively where data were available. For those cases, LVOTO was defined as Z-score values of less than −2.0 based on body surface area at the subaortic, aortic annulus, ascending aorta, transverse arch, or isthmus level. Characteristics of the ventricular septal defect (VSD)/bulboventricular foramen were derived from qualitative comments in the echocardiography reports; quantitative data were not reported.
The study was approved by the Research Ethics Board of the Hospital for Sick Children and that of all participating institutions. Families provided written consent as per local requirements.
Results
The first palliative interventions for the overall CHSS TA cohort of 445 infants was the Blalock-Taussig-Thomas (BTT) shunt for 222 patients, BTT shunt plus PAB placement for 10, BTT shunt plus main pulmonary trunk interruption for 29, PAB placement for 75, BCPA for 98, ductal stent placement for 1, and Stage I palliation (Norwood/DKS) for 7 (Group I).
Group I
All seven infants from Group I had unrestricted pulmonary blood flow through a large VSD without pulmonary stenosis (Type Ic), five of whom presented with congestive heart failure and two with cardiogenic shock (one with acute kidney injury). At hospital admission, six infants had normal left ventricle (LV) function and one had significant LV dysfunction; five had moderate LV dilation, three had mild, and one had moderate mitral valve regurgitation. Multilevel LVOTO was present in all patients (Figure 1), including transverse aortic arch hypoplasia in six infants, subaortic stenosis in four, isthmic narrowing in four, aortic valve stenosis in three, and hypoplastic ascending aorta in three and interrupted aortic arch (IAA) in one patient. Z-scores for systemic outflow structures were available for five patients and were in the range of −2.3 to −6.03. Age at Stage I ranged from 5 to 68 days and weight from 2.1 to 3.5 kg.
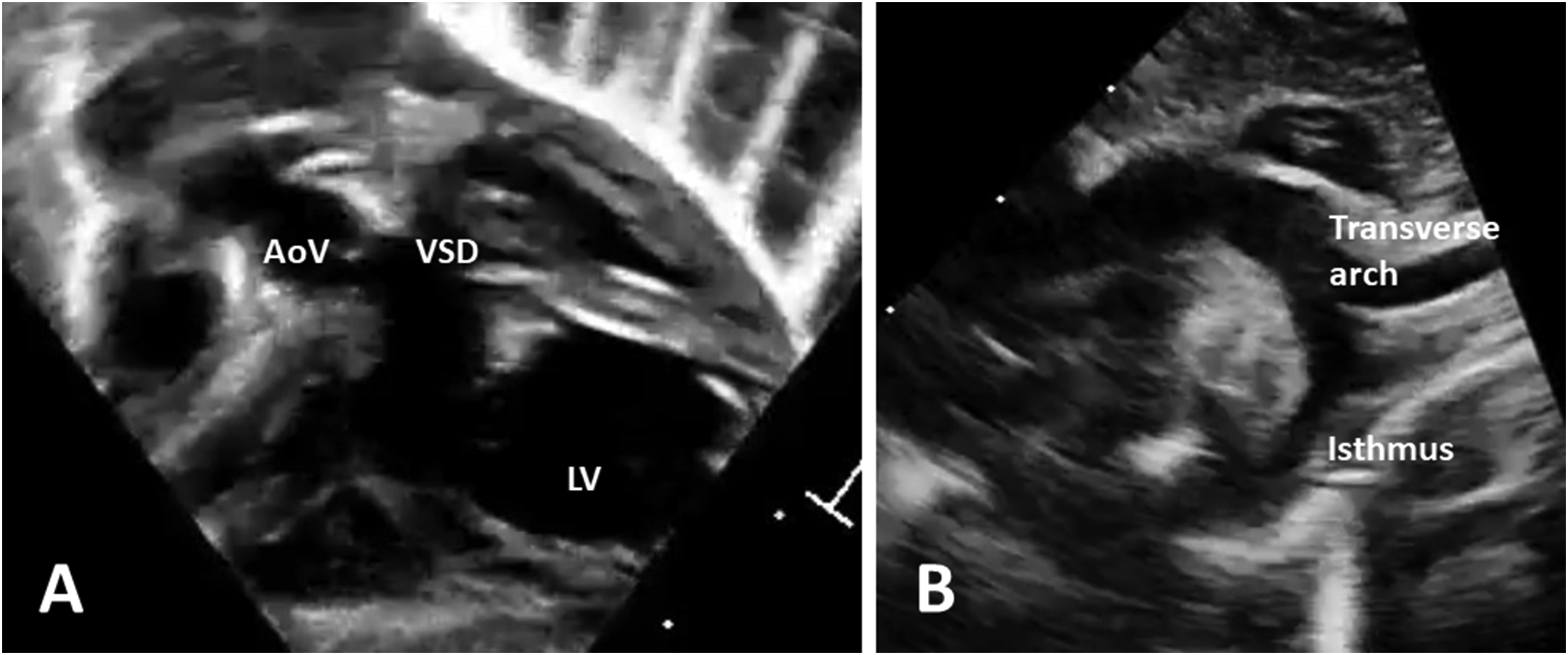
Baseline echocardiogram images of a patient with tricuspid atresia and left ventricular outflow tract obstruction who underwent a Norwood procedure. (A) The normally functioning but small aortic valve (5.6 mm, Z-score −1.6) and root (7.4 mm, Z-score −2.2). (B) The hypoplastic transverse aortic arch (4.4 mm, Z-score 3.6) and isthmus (2.3 mm, Z-score −5.2). AoV, aortic valve; LV, left ventricle; VSD, ventricular septal defect.
A Norwood procedure was performed for four of these seven patients (one with a Sano shunt). One patient with Type B IAA who presented with cardiogenic shock and acute kidney injury had hybrid palliation with bilateral PAB placement and ductal stent insertion. The remaining two patients had initial DKS with BTT shunt placement, but both required conversion to Norwood due to unresolved aortic arch obstruction noted when trying to wean from cardiopulmonary bypass (Figure 2). For these two patients, one had an aortic arch Z-score of −2.9, and the Z-score was not available for the other. For patients having the Norwood procedure, augmentation of the aortic arch was performed using homograft material in five patients, and a 3D CardioCel patch in one patient. Overall, two patients left the operating room on extracorporeal membrane oxygenation (ECMO). One patient was weaned successfully after three days, and the other patient underwent revision of a stenotic shunt and was able to be subsequently weaned from ECMO but died on postoperative day 56 from sepsis/multiorgan failure (MOF). The postoperative median length of stay for the six survivors was 53 days (11-68 days).
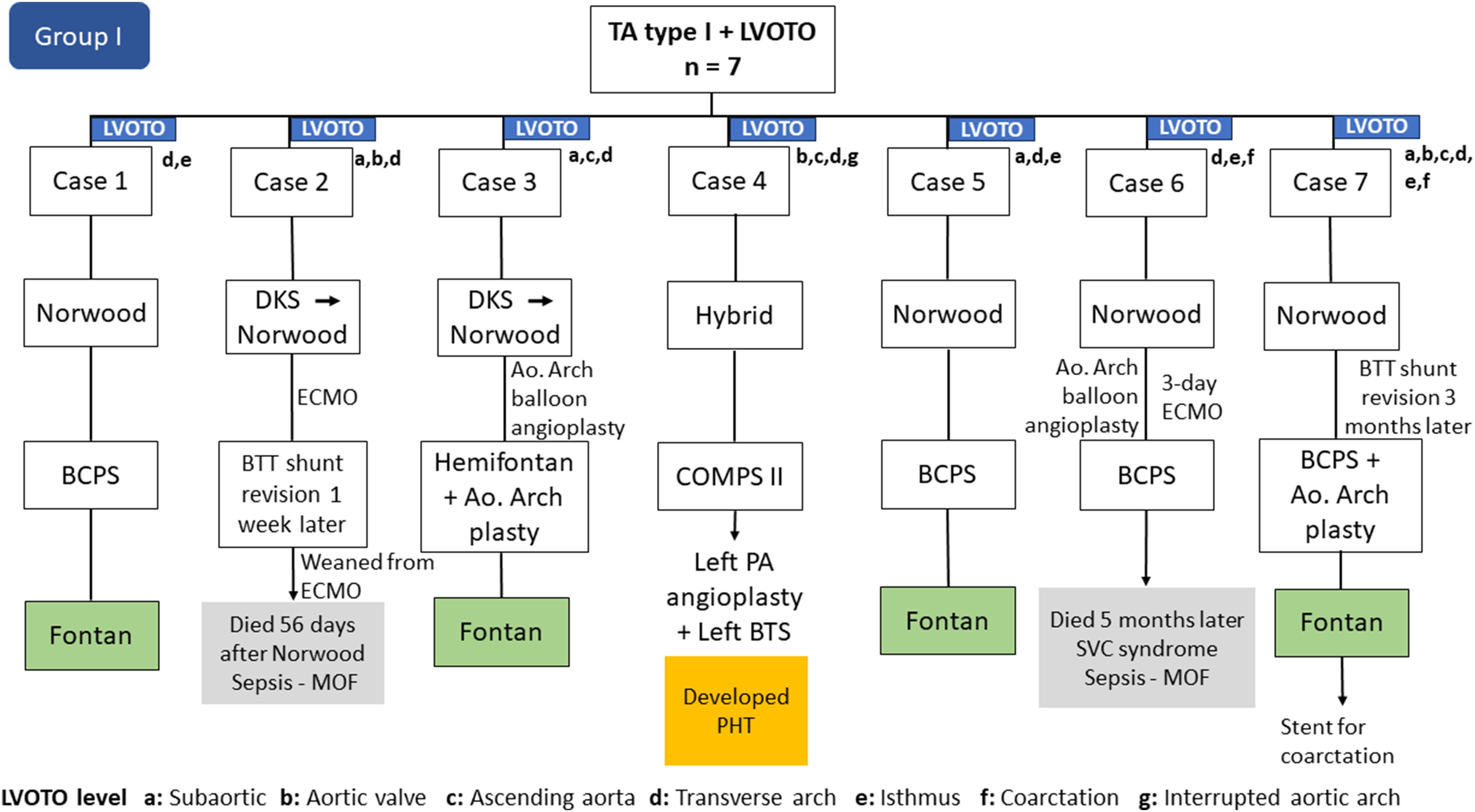
Flow diagram of pathways and interventions in Group I. Ao, aortic; BCPS, bidirectional superior cavopulmonary anastomosis; BTS, Blalock-Taussig-Thomas shunt; BTT, Blalock-Taussig-Thomas; DKS, Damus-Kaye-Stansel; ECMO, extracorporeal membrane oxygenation; LVOTO, left ventricular outflow tract obstruction; MOF, multiorgan failure; PA, pulmonary artery; PHT, pulmonary hypertension; SVC, superior vena cava; TA, tricuspid atresia; COMPS II, Comprehensive Stage II.
Following an initial LVOTO procedure, one patient had BTT shunt revision three months after Norwood. Aortic arch reintervention was performed in three infants before or at Stage II; one of these patients had initial DKS and intraoperative conversion to Norwood, but the final repair did not extend to the descending aorta. This patient required balloon angioplasty before Stage II and aortic arch repair at Stage II. All six survivors proceeded to subsequent Stage II palliation. Afterward, one patient required angioplasty and stenting for superior vena cava syndrome associated with anastomotic obstruction and died five months later from sepsis/MOF. The patient with IAA had a comprehensive Stage II procedure and subsequently underwent angioplasty and an additional left BTT shunt placement for left pulmonary artery thrombosis. Weeks later the patient developed pulmonary hypertension and was not considered a candidate for Fontan. The remaining four patients underwent the Fontan procedure; two had a lateral tunnel connection of 18 mm and 20 mm and the other two had an extracardiac connection with a polytetrafluoroethylene (PTFE) conduit of 20 mm. All four patients had normal LV function and less than mild mitral valve regurgitation at the latest follow-up after Fontan (0.1, 0.2, 7.2, 19.6 years).
Group II
In Group II (LVOTO procedure at Stage II), all seven infants initially had unrestricted pulmonary blood flow through a large VSD and received PAB placement as initial intervention (Figure 3); these patients represent 9% of the 75 patients from the total cohort who had PAB placement. Four of them presented with tachypnea, two with congestive heart failure, and one had both. All seven patients had oxygen saturation levels above 95%. Age at PAB placement ranged from 4 to 67 days with patient weight ranging from 2.7 to 3.7 kg. All seven infants had normal left ventricular function. Mild mitral valve regurgitation was noted in three infants and moderate or greater in two. The median length of hospital stay was five days (3-29 days).
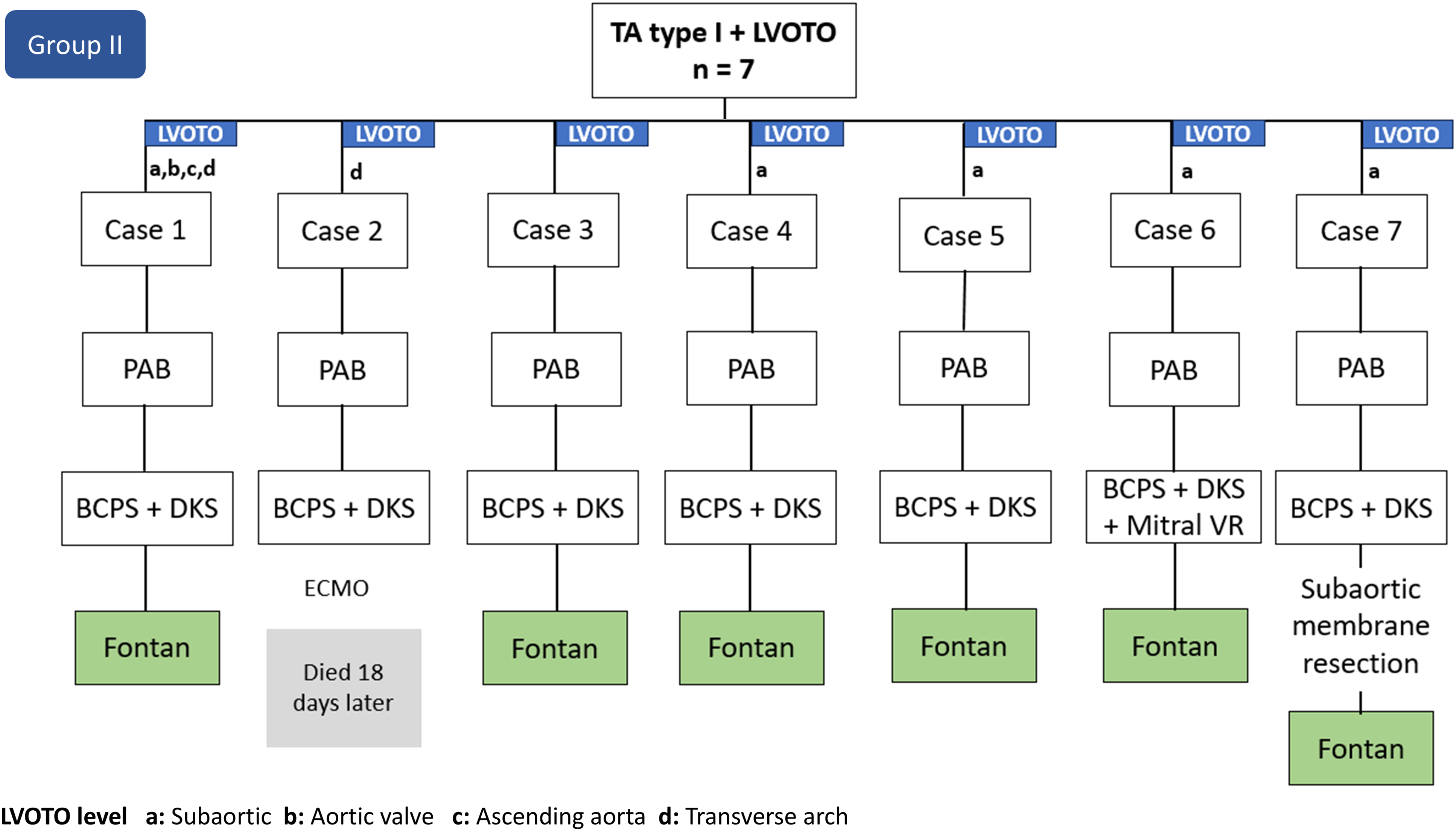
Flow diagram of pathways and interventions in Group II. BCPS, bidirectional superior cavopulmonary anastomosis; DKS, Damus-Kaye-Stansel; ECMO, extracorporeal membrane oxygenation; LVOTO, left ventricular outflow tract obstruction; mitral VR, mitral valve replacement; PAB, pulmonary artery band; TA, tricuspid atresia.
At Stage II, these patients underwent BCPA and DKS procedures; one patient had concomitant mitral valve repair of an anterior leaflet cleft. The recorded rationale for performing a DKS procedure was the presence of subaortic narrowing for five infants and aortic arch hypoplasia for two (aortic arch Z-scores were −4.11 and −3.12). Five patients had qualitative evidence of mild LVOTO. Age at Stage II ranged from 4.1 to 8.1 months and weight from 5.2 to 7 kg with a median length of hospital stay of 14 days (7-26). Two days after Stage II, one patient had cardiac arrest of unknown etiology, required ECMO, and died 18 days after support was withdrawn. One infant required resection of a subaortic membrane in the interstage period, before the Fontan operation. The six surviving patients who underwent Fontan had an extracardiac connection with a PTFE conduit of 16 mm in one, 18 mm in three, and 20 mm in two patients. At the latest follow-up after Fontan (3.3, 8.0, 11.6, 12.5, 14.1, and 20.2 years), all have good ventricular function with less than mild mitral valve regurgitation and excellent self-reported functional health status.
The Kaplan-Meier (KM) survival estimate at 20 years after initial hospital admission was 71% (5/7) (70% confidence interval: 52%-85%) for Group I and 86% (6/7) for Group II (70% confidence interval: 67%-95%; P = .53). The overall KM survival estimate at 20 years for the 14 patients who had LVOTO procedures (Group I and II combined) was 79% (11/14) (70% confidence interval: 66%-88%). The survival of this combined group was not significantly different from the remaining larger TA cohort without LVOTO (n = 431; 87% survival at 20 years from first admission [70% confidence interval: 85%-89%]; P = .18; Figure 4).
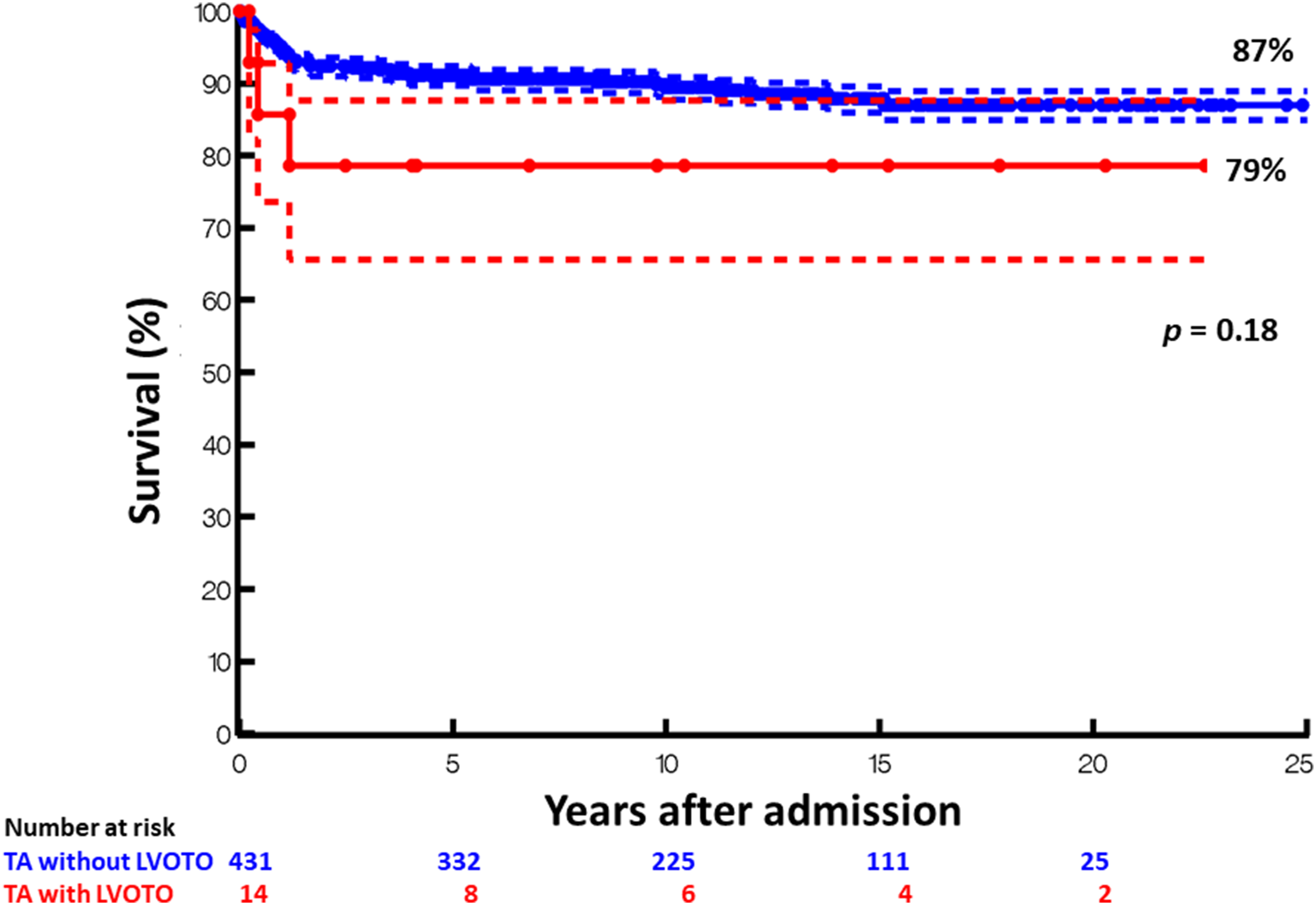
Kaplan-Meier curves of tricuspid atresia patients with normally related great arteries who did (red line, Group I and II combined) versus did not (blue line) undergo procedures for left ventricular outflow tract obstruction. Dashed lines represent 70% confidence interval. LVOTO, left ventricular outflow tract; TA, tricuspid atresia.
Comment
Classification
There are a variety of morphological types of TA. The most common subtype (Type I) is associated with ventriculoarterial concordance. Type II TA is associated with TGA, 1 where the aorta arises from the hypoplastic right ventricle, creating a set-up for systemic outflow obstruction in fetal and neonatal life. Type III TA includes malposition defects other than TGA.
Prevalence and Survival
For patients with TA, the presence of systemic outflow obstruction has been reported to be ∼20%. 1 Alsoufi et al reported 105 patients with TA, 23 of whom had LVOTO or the potential to develop it. 1 Of these, 19 had ventriculoarterial discordance (Type II and III), and four were Type I with unrestricted pulmonary blood flow. The Norwood procedure was performed for 18 patients, with an eight-year survival of 78%. Most often, LVOTO in TA occurs in the setting of TGA, as described by Tandon and Edwards. 9 Lan et al reported a series from 1983 to 2002 of 105 patients with double inlet LV (DILV) and 35 patients with TA and TGA, with an overall mortality of 49% (17/35 deaths) in the TGA group. 10 The majority of the perioperative deaths occurred in neonates who underwent DKS (9/41 deaths, 22%).
More recently, other series of patients undergoing the Norwood procedure for hypoplastic left heart syndrome (HLHS) and non-HLHS anomalies have reported 62% to 68% survival at five years.11-13 In a large cohort of infants with HLHS, Mascio et al reported that the overall hospital survival after the Norwood procedure was 74% 14 ; this reported intermediate-term survival is lower than the 79% 20-year survival noted within our study, which is likely related to the fact that the systemic ventricle for infants with TA is morphologically a LV. Nonetheless, infants with TA that require a Norwood/DKS procedure have some of the increased risk associated with those procedures.
Reinterventions
It has been shown that after Stage I palliation, unplanned reoperations, such as shunt revisions and percutaneous reinterventions, are associated with increased morbidity and mortality, as we also noted. 11 In our series, the two patients who had an initial DKS procedure did not have aortic coarctation, and one may think that this is why the aortic arch repair was initially limited. However, based on echocardiogram review, these patients did have aortic arch hypoplasia (Z-score of −2.9). After trying to wean from cardiopulmonary bypass, a gradient was more evident in the arch. Hence, for these patients, a more extensive initial repair of the aortic arch reaching the descending aorta may have been indicated.
Although recurrent aortic arch obstruction is common after the Norwood procedure, with a reported incidence ranging from 2% to 40%, it has been shown that surgical or percutaneous reinterventions are not associated with a higher risk of mortality. 15 The risk is more significant early after the first palliation. Intervention for recoarctation is usually performed at the pre-Stage II catheterization or at Stage II in ∼20% of patients after single ventricle reconstruction. 15 In our series, three out of five surviving patients after Norwood/DKS underwent aortic arch reinterventions before or at Stage II. One possible factor that may contribute to the development of recurrent arch obstruction is that these neonates with small ascending aorta and aortic arch usually have less native tissue for reconstruction, requiring larger homograft patches for aortic arch repair as described by Walters et al. 16 In addition, the lack of an adequate interdigitating technique for arch reconstruction may predispose patients for recoarctation and need for arch revision.17-19
For patients with Type I TA and systemic outflow obstruction, early management with the Norwood procedure should be intended to completely relieve any stenoses, protecting the pulmonary circulation and preventing deleterious effects of chronic pressure or volume overload on the ventricle. This helps to preserve LV and mitral valve function, 20 as was observed in our case series. Effectively reducing the afterload avoids a sudden increase in systemic vascular resistance and a burden for the single ventricle early in the postoperative period; it also has a positive effect on inflammatory response and reduction in early mortality.13,21
The bulk of the literature addresses patients with TA, LVOTO, and discordant ventriculoarterial connection. After PAB placement for this subtype, myocardial hypertrophy may quickly progress and further cause or exacerbate LVOTO at the level of the VSD. In some infants, PAB placement unmasks a restrictive VSD and, thus, subaortic narrowing. 4 In our series, all patients had large unrestrictive VSDs without pulmonary stenosis, which may be why the LVOTO in Group II was mild.
Franken et al reported that for patients with TA with TGA or DILV, the presence of systemic outflow tract obstruction at birth was the only factor in a multivariable analysis that was associated with a higher risk of death (hazard ratio: 3.7). 22 They recommended performing a DKS at Stage II, given the high proportion of patients with TA and TGA who ultimately develop systemic outflow tract obstruction (27%). This is in contrast to our overall Type I TA cohort, where only 9% of patients who had PAB placement developed LVOTO. Additionally, various studies have demonstrated that the presence of aortic arch obstruction is recognized as the most influential risk factor for systemic outflow tract obstruction after PAB placement.4,5,23,24
In the series by Alsoufi et al, TA patients with subaortic obstruction, a small aortic valve annulus, and/or small ascending aorta/proximal arch were more likely to undergo a Norwood operation, while patients with aortic coarctation or distal arch hypoplasia without significant subaortic obstruction were considered for arch repair plus PAB placement. 11 Of note, subaortic stenosis usually develops in patients with associated aortic coarctation, and a subvalvar gradient may be hard to accurately appreciate in the setting of ductus-dependent systemic circulation to the lower body. 24
The hybrid procedure with bilateral PAB and ductal stent placement has been previously used as a management strategy for patients with HLHS and IAA.24,25 This approach may be the best initial palliative option for high-risk neonates, such as our individual patient who presented in shock with an acute kidney injury. The aortic arch repair for this patient was deferred for second-stage palliation.
As the infants with TA in our series had normally related great arteries, the risk for systemic outflow tract obstruction would be expected to be less. We presume the surgical teams who managed infants in Group II did not consider the systemic outflow tract obstruction to be significant at the time of initial PAB placement and may be why the aortic arch hypoplasia may not have been addressed until Stage II.
It has been previously shown that survival after the Norwood procedure may be related to surgeon and center-specific factors/experience, and some may want to avoid the risks of this strategy. 23 We believe that patients with Type I TA who undergo PAB placement and those who present with subtle signs of LVOTO should undergo an early BCPS with a concomitant DKS procedure either anticipating or treating the systemic outflow tract obstruction. Patients with Type I TA and aortic coarctation without aortic arch hypoplasia or subaortic stenosis might benefit from coarctation repair and PAB placement. Furthermore, these patients should be flagged as a group who may be more at risk of developing subaortic stenosis and may need a DKS procedure at the time of Stage II.
While infants with TA and TGA are much more likely to have LVOTO, this study has shown that there is a rare, but important, occurrence of LVOTO in neonates with TA and normally related great arteries. Infants with type I TA accompanied by LVOTO can be managed successfully with Stage I palliation, with the recommendation to pursue complete arch augmentation in all patients. As the experience and survival after the Norwood procedure have dramatically improved over the years, addressing the systemic outflow tract obstruction in patients with TA and concordant ventriculoarterial connection should be performed early to avoid development of subaortic stenosis and ventricular dysfunction.
Limitations
Although this is one of the largest reported experiences and from a multi-institutional cohort, one of the limitations of this study remains a small sample size that prohibits comparative analysis and robust recommendations. Additionally, due to the retrospective nature of the study, clinical decision-making is unknown, and this might have impacted management including surgical choices. Also, this study enrolled patients over a 25-year period, potentially leading to era effects. Moreover, the literature on patients with TA and normally related great arteries is scarce, making it challenging to accurately compare our results with similar studies.
Summary
The CHSS cohort experience suggests that, despite there being a normal ventriculoarterial relationship between the LV and aorta, a DKS anastomosis and complete alleviation of aortic arch obstruction is a reliable way of ensuring unobstructed systemic outflow pathway in patients with type I TA and LVOTO. Initial palliation with PAB placement may be considered for patients with borderline arch dimensions, allowing infant growth, and greater stability, or in patients where the risk of performing a Norwood is higher. After PAB placement, these patients should be closely followed for signs of either unmasked or evolving LVOTO. The DKS procedure can be performed successfully at the time of Stage II, and it is essential in circumventing the development of subaortic obstruction while diminishing the need for later aortic arch reintervention. However, left ventricular and mitral valve functions remain well preserved, extending to the post-Fontan period. In this context, the risk of adverse outcomes is higher compared with uncomplicated Type I TA. Clinicians should be aware of this rare but important morphological subtype of patients with TA and normally related great arteries.
Footnotes
Abbreviations
Acknowledgments
The authors thank Sally Cai, Maria Micalowska, Apoorva Anand, and Tharini Paramanathan from the CHSS-CRQ staff and acknowledge the invaluable contributions of participants in the tricuspid atresia cohort and research coordinators at CHSS institutions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
