Abstract
Keywords
Introduction
Significant atrioventricular valve (AVV) dysfunction occurs in 10% to 25% of functionally univentricular heart (FUVH) patients1,2 and is well-known to be an independent risk factor for death and transplant in these patients.1,3–9 Successful AVV repair significantly improves their long-term outcomes6,8,10–15 and may return them to the same risk profile as FUVH patients without AVV dysfunction.9,11,16 However, successful, durable AVV repair is difficult to achieve,1,6,9,10,13,14 and AVV repair itself may be a risk factor for death or transplantation.17,18 Thus, these patients face a dilemma regarding optimal treatment strategy.
An aggressive approach of early AVV repair in FUVH patients who are otherwise acceptable single ventricle palliation (SVP) candidates is advocated by many,1,4,5,18 while others recommend a selective approach with early consideration of heart transplantation.3,6,19 It is unclear which strategy is optimal, and it is unknown which factors should be considered for a selective approach. The aims of this study are to characterize the impact on outcomes of concomitant AVV intervention at each stage of SVP and to identify predictors of poor outcomes in patients undergoing concomitant AVV intervention at each stage of SVP.
Patients and Methods
Design and Data Source
The Society of Thoracic Surgeons Congenital Heart Surgery Database (STS-CHSD) was used to perform a series of independent retrospective cohort analyses at each stage of SVP. The STS-CHSD contains audit-validated, procedure-level data from over 90% of congenital heart surgery cases performed in the United States. 20 Analyses were conducted by the STS Research and Analytic Center. This study was determined to be exempt research by the Institutional Review Board (Advarra; Protocol #Mod01760092).
Patient Cohorts
The STS-CHSD was queried for patients with a FUVH diagnosis undergoing a procedure consistent with SVP from January 1, 2013, to December 31, 2022 (Supplemental Table 1). Patients undergoing 1.5 ventricle repair or a procedure corresponding to a previously completed SVP stage were excluded. Patients undergoing procedure combinations with ambiguous SVP stage were manually adjudicated to a single stage or excluded (Supplemental Table 2). Patients were then assigned to preliminary cohorts based on SVP stage. Cohort 1 included initial palliation procedures such as Norwood and hybrid stage 1 procedures, cohort 2 included superior cavopulmonary anastomosis procedures, and cohort 3 included Fontan procedures (Supplemental Table 1). Patients undergoing concomitant aortopulmonary amalgamation and superior cavopulmonary anastomosis were included in cohort 2 and assigned an operation value of “Comprehensive Stage 2” (Supplemental Table 2). Patients were excluded from cohort 1 if their age was missing or >1 year. Patients with pulmonary atresia with intact ventricular septum undergoing a conduit procedure were excluded from cohort 1. Patients whose age was missing, <30 days, or >1.5 years were excluded from cohort 2. Patients whose age was missing, <1.5 years, or >7 years were excluded from cohort 3 (Figure 1). These cohorts are independent of one another and unrelated except that they correspond to patients along with the same clinical pathway. Thus, each cohort was analyzed independently with no attempt made to compare across cohorts or to perform longitudinal analysis.
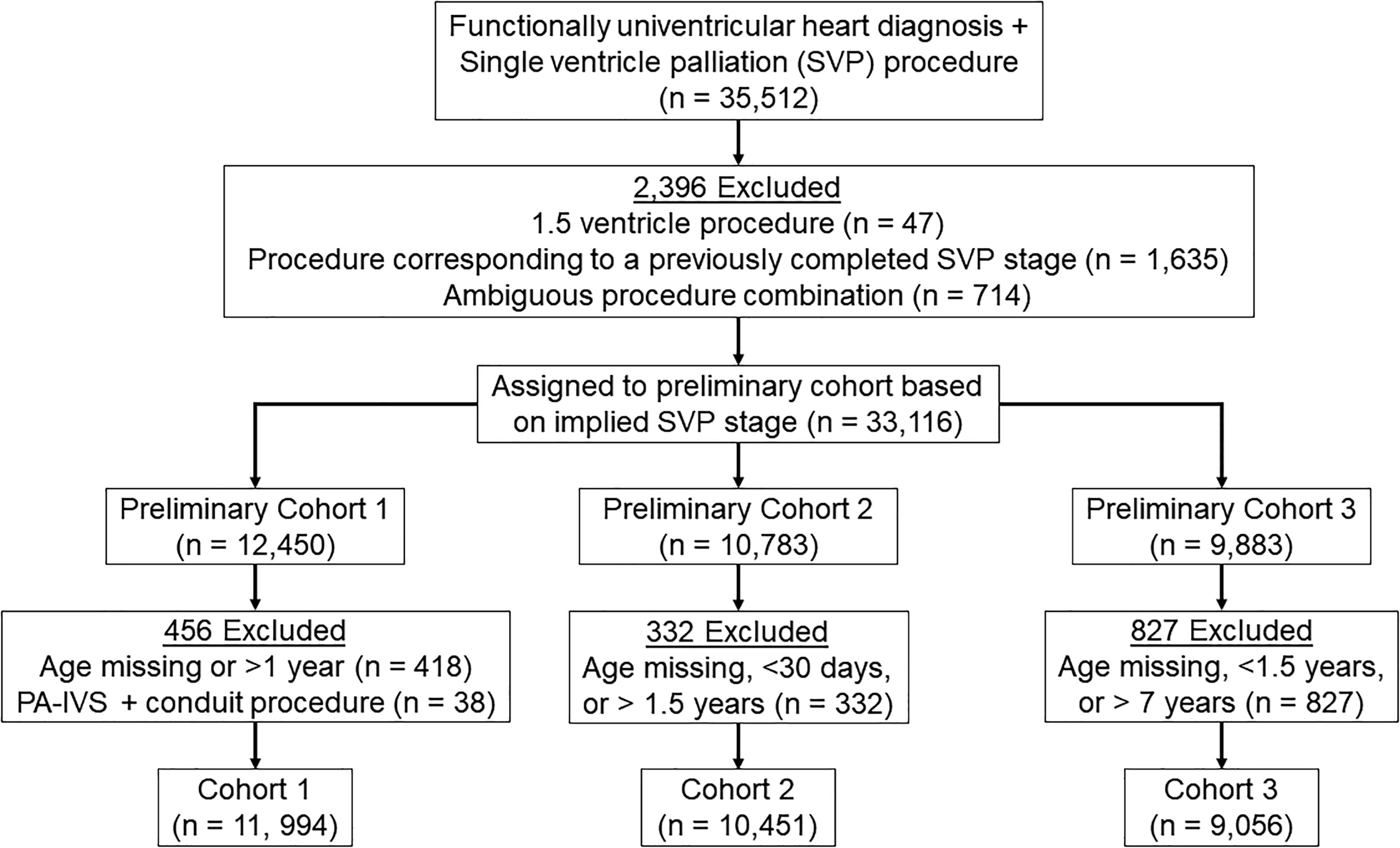
Cohort creation diagram. PA-IVS, pulmonary atresia with intact ventricular septum; SVP, single ventricle palliation; PA-IVS, pulmonary atresia with intact ventricular septum.
Outcomes
A composite end point of operative mortality and/or major morbidity was the primary outcome analyzed. Operative mortality was defined as death within 30 days of procedure or prior to hospital discharge. Major morbidity was defined as the occurrence of any of the following within 30 days of procedure: renal failure requiring dialysis, neurologic deficit persisting at discharge, arrhythmia requiring permanent pacemaker, mechanical circulatory support, phrenic nerve injury, or unplanned reoperation. 21 The individual components of the primary outcome were analyzed as secondary outcomes.
Analysis
Within each cohort, patients were stratified by the occurrence of concomitant AVV intervention (Supplemental Table 3). Bivariate analysis of demographics, diagnosis, comorbidities, preoperative risk factors, operative characteristics, and outcomes with and without concomitant AVV intervention was conducted within each cohort using an appropriate test of statistical inference for each variable (χ2, Fisher exact, Wilcoxon rank sum). Multivariable analysis of the concomitant AVV intervention subgroup of each cohort was conducted to find preoperative predictors of composite operative mortality or major morbidity by multiple logistic regression. Models were created by backward stepwise regression starting with a fully saturated model. Age group, weight, ventricular dominance, and operation were included in the minimum model. Statistical significance was assigned to P-values <.05.
Results
Cohort 1
Complete bivariate analysis of demographics, diagnoses, comorbidities, preoperative risk factors, and operative characteristics of those with and without concomitant AVV intervention for cohort 1 is reported in Supplemental Table 6. Concomitant AVV intervention was more prevalent among cohort 1 patients who were in age group 1 to 12 months (3.7% vs 2.0% [age group 0-1 month], P < .001), right ventricle (RV) (2.8%) or ambiguous ventricle (2.4%) dominant (vs 1.0%; left ventricle [LV] dominant, P < .001), and undergoing Norwood procedure (2.8% vs 1.3% [Shunt] vs 1.9% [pulmonary artery band] vs 0.6% [other], P < .001). Cohort 1 patients undergoing concomitant AVV intervention were heavier (3.33 kg vs 3.15 kg, P < .001) and had a higher prevalence of prior AVV intervention (1.1% vs 0.1%, P = .009), preoperative mechanical ventilation (50% vs 36%, P < .001), and reoperative surgery (18% vs 7.8%, P < .001). Total operative time (355 min vs 263 min, P < .001), cardiopulmonary bypass time (182 vs 121 min, P < .001), and cross clamp time (78 vs 42 min, P < .001) were longer for patients undergoing concomitant AVV intervention.
Bivariate analysis of outcomes with and without concomitant AVV intervention for cohort 1 is reported in Table 1. Concomitant AVV intervention was associated with a higher occurrence of composite operative mortality or major morbidity (62% vs 46%, P < .001) as well as all components of the composite outcome except neurologic deficit (3.0% vs 2.0%, P = .2) and paralyzed diaphragm (4.1% vs 3.1%, P = .4).
Cohort 1: Bivariate Analysis of Outcomes Versus Concomitant AVV Intervention.
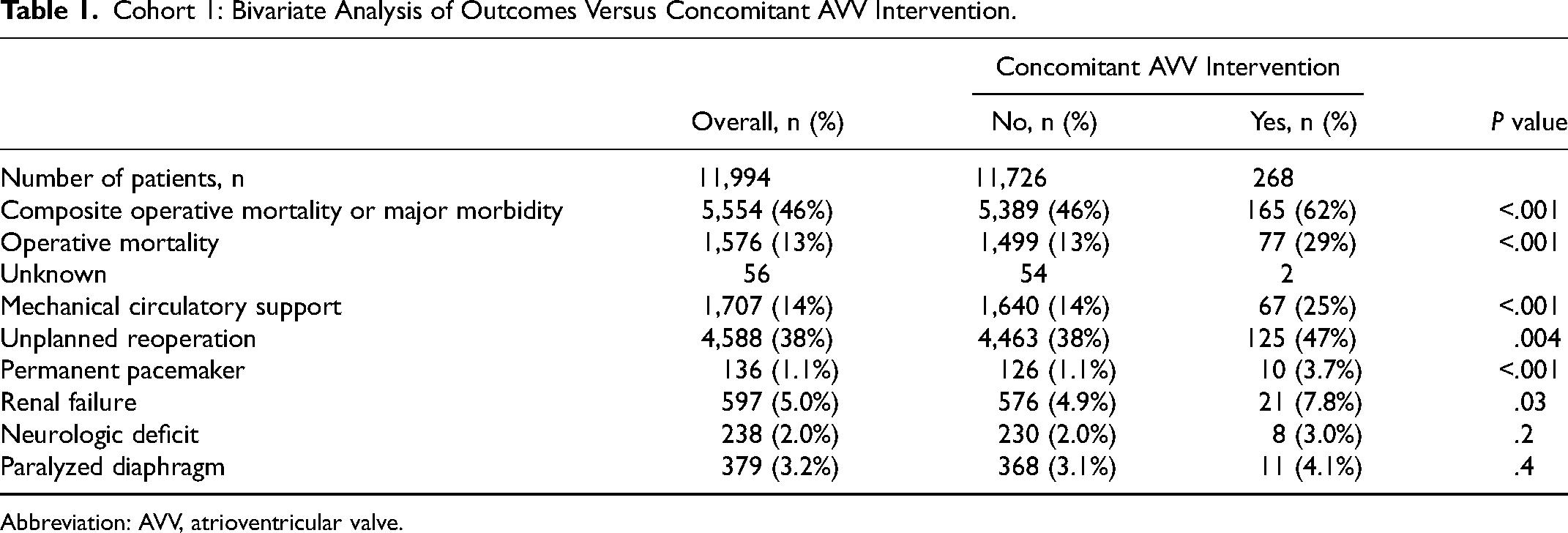
Abbreviation: AVV, atrioventricular valve.
Multivariable analysis of predictors of composite operative mortality or major morbidity for cohort 1 patients undergoing concomitant AVV intervention is reported in Table 2. Predictors independently associated with a higher occurrence of composite mortality or major morbidity are black race (reference white, odds ratio [OR] = 3.151, 95% confidence interval [CI] = 1.181-9.649, P = .03) and ambiguous ventricular dominance (reference LV, OR = 3.355, 95% CI = 1.242-9.408, P = .018). Each marginal increase of 1 kg of weight is independently associated with a decreased occurrence of the composite outcome (OR = 0.627, 95% CI = 0.457-0.843, P = .003).
Cohort 1: Multivariable Analysis of Predictors of Composite Operative Mortality or Major Morbidity for Patients Undergoing Concurrent AVV Intervention.
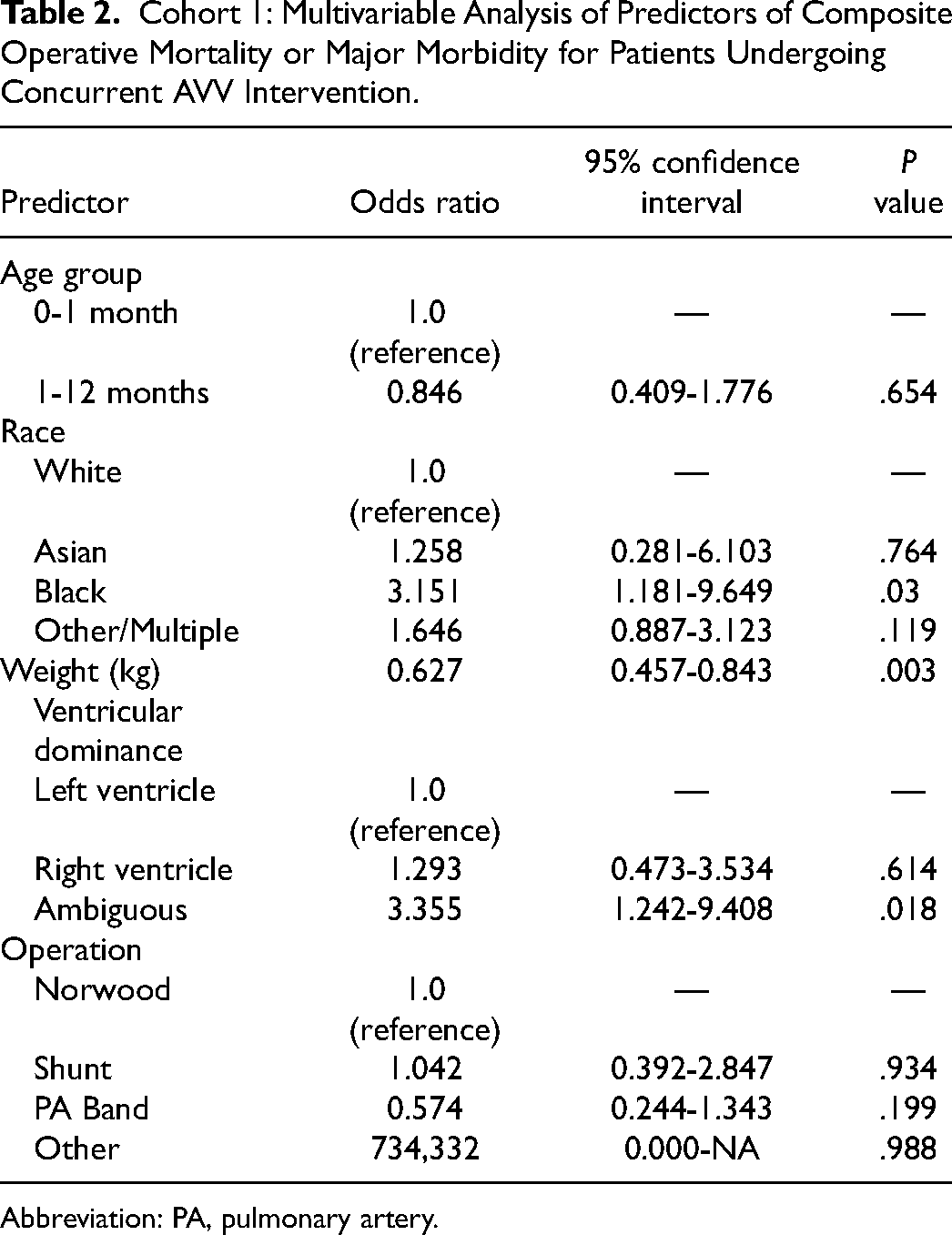
Abbreviation: PA, pulmonary artery.
Cohort 2
Complete bivariate analysis of demographics, diagnoses, comorbidities, preoperative risk factors, and operative characteristics of those with and without concomitant AVV intervention for cohort 2 is reported in Supplemental Table 7. Concomitant AVV intervention was more prevalent among cohort 2 patients who were RV (8.1%) or ambiguous ventricle (8.1%) dominant (vs 3.1% [LV dominant], P < .001) and undergoing urgent surgery (9.4% vs 5.9% [elective] vs 7.0% [salvage/emergent], P < .001). Cohort 2 patients undergoing concomitant AVV intervention were younger (157 vs 162 days, P = .014), lighter (6.06 vs 6.28 kg, P = .001), and had a higher prevalence of prior AVV intervention (7.8% vs 1.2%, P < .001), preoperative mechanical ventilation (20% vs 10%, P < .001), and other preoperative risk factors (Supplemental Table 5) (54% vs 46%, P < .001). Total operative time (317 vs 239 min, P < .001), cardiopulmonary bypass time (143 vs 95 min, P < .001), and cross clamp time (45 vs 0 min, P < .001) were longer for patients undergoing concomitant AVV intervention.
Bivariate analysis of outcomes with and without concomitant AVV intervention for cohort 2 is reported in Table 3. Concomitant AVV intervention was associated with a higher occurrence of composite operative mortality or major morbidity (37% vs 19%, P < .001) as well as all components of the composite outcome except neurologic deficit (1.0% vs 0.8%, P = .4) and paralyzed diaphragm (4.2% vs 3.1%, P = .12).
Cohort 2: Bivariate Analysis of Outcomes Versus Concomitant AVV Intervention.

Abbreviation: AVV, atrioventricular valve.
Multivariable analysis of predictors of composite operative mortality or major morbidity for cohort 2 patients undergoing concomitant AVV intervention is reported in Table 4. Predictors independently associated with a higher occurrence of composite mortality or major morbidity are preterm birth (OR = 1.776, 95% CI = 1.049-3.005, P = .032), presence of other preoperative risk factors (Supplemental Table 5) (OR = 1.445, 95% CI = 1.033-2.028, P = .032), mechanical ventilation prior to surgery (OR = 2.463, 95% CI = 1.613-3.786, P < .001), and comprehensive stage 2 procedure (reference superior cavopulmonary anastomosis, OR = 2.476, 95% CI = 1.143-5.494, P = .022).
Cohort 2: Multivariable Analysis of Predictors of Composite Operative Mortality or Major Morbidity for Patients Undergoing Concurrent AVV Intervention.
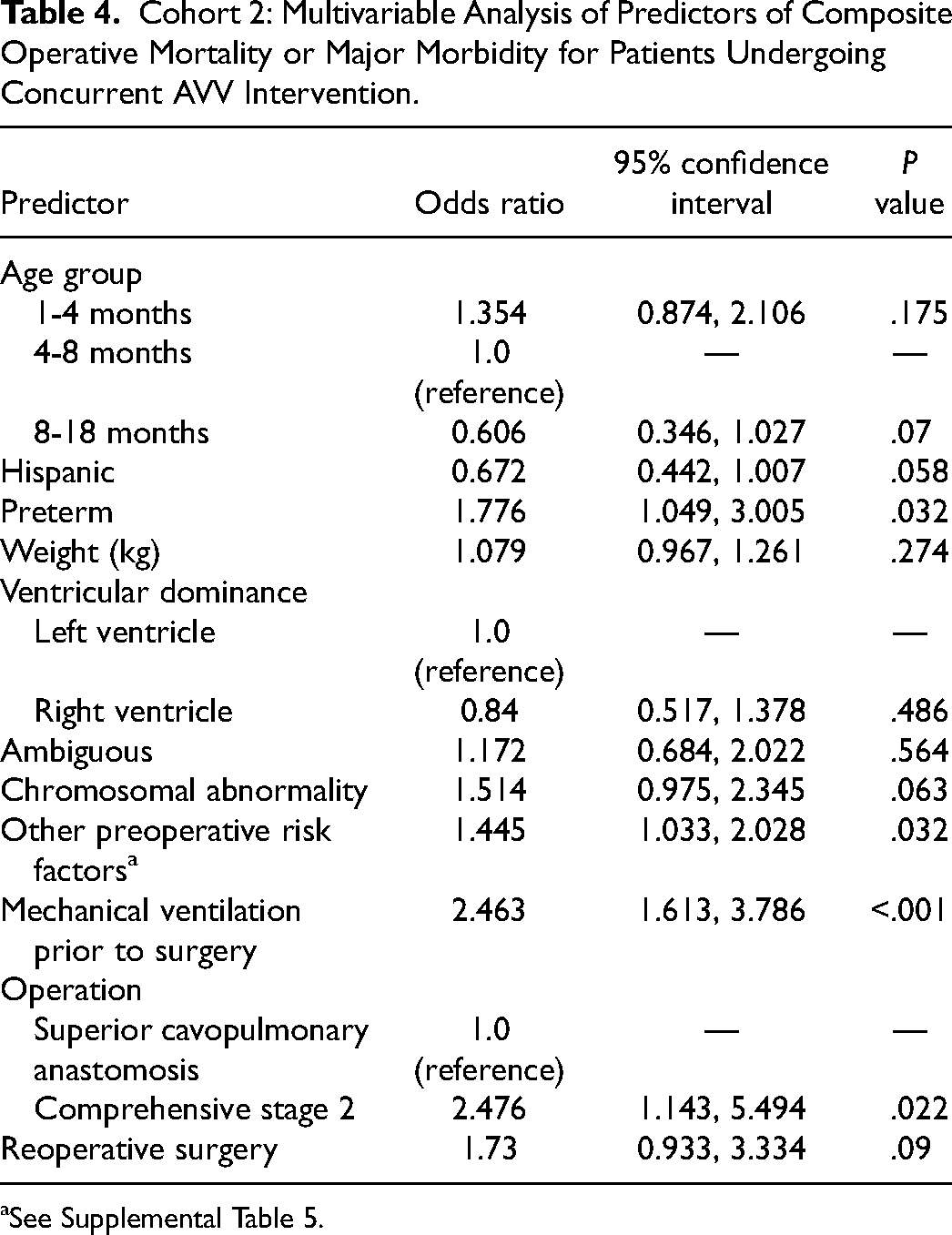
See Supplemental Table 5.
Cohort 3
Complete bivariate analysis of demographics, diagnoses, comorbidities, preoperative risk factors, and operative characteristics of those with and without concomitant AVV intervention for cohort 3 is reported in Supplemental Table 8. Concomitant AVV intervention was more prevalent among cohort 3 patients who were RV (10.4%) or ambiguous ventricle (10.8%) dominant (vs 2.8% [LV dominant], P < .001). Cohort 3 patients undergoing concomitant AVV intervention were lighter (14.2 vs 14.6 kg, P = .005) and had a higher prevalence of prior AVV intervention (7.8% vs 1.2%, P < .001), major noncardiac abnormality (22% vs 15%, P < .001), and shock at time of surgery (0.4% vs <0.1%, P = .004). Total operative time (335 vs 258 min, P < .001), cardiopulmonary bypass time (136 vs 89 min, P < .001), and cross clamp time (58 vs 0 min, P < .001) were longer for patients undergoing concomitant AVV intervention.
Bivariate analysis of outcomes with and without concomitant AVV intervention for cohort 3 is reported in Table 5. Concomitant AVV intervention was associated with a higher occurrence of composite operative mortality or major morbidity (22% vs 14%, P < .001) as well as all components of the composite outcome except neurologic deficit (1.8% vs 1.3%, P = .3) and paralyzed diaphragm (1.9% vs 1.7%, P = .7). Notably, concomitant AVV intervention was associated with a much higher occurrence of permanent pacemaker placement (5.2% vs 1.2%, P < .001).
Cohort 3: Bivariate Analysis of Outcomes Versus Concomitant AVV Intervention.

Abbreviation: AVV, atrioventricular valve.
Multivariable analysis of predictors of composite operative mortality or major morbidity for cohort 3 patients undergoing concomitant AVV intervention is reported in Table 6. Predictors independently associated with a higher occurrence of composite mortality or major morbidity are ambiguous ventricular dominance (reference LV, OR = 1.939, 95% CI = 1.036-3.806, P = .045) and presence of other preoperative risk factor (Supplemental Table 5) (OR = 1.608, 95% CI = 1.110-2.335, P = .012).
Cohort 3: Multivariable Analysis of Predictors of Composite Operative Mortality or Major Morbidity for Patients Undergoing Concurrent AVV Intervention.
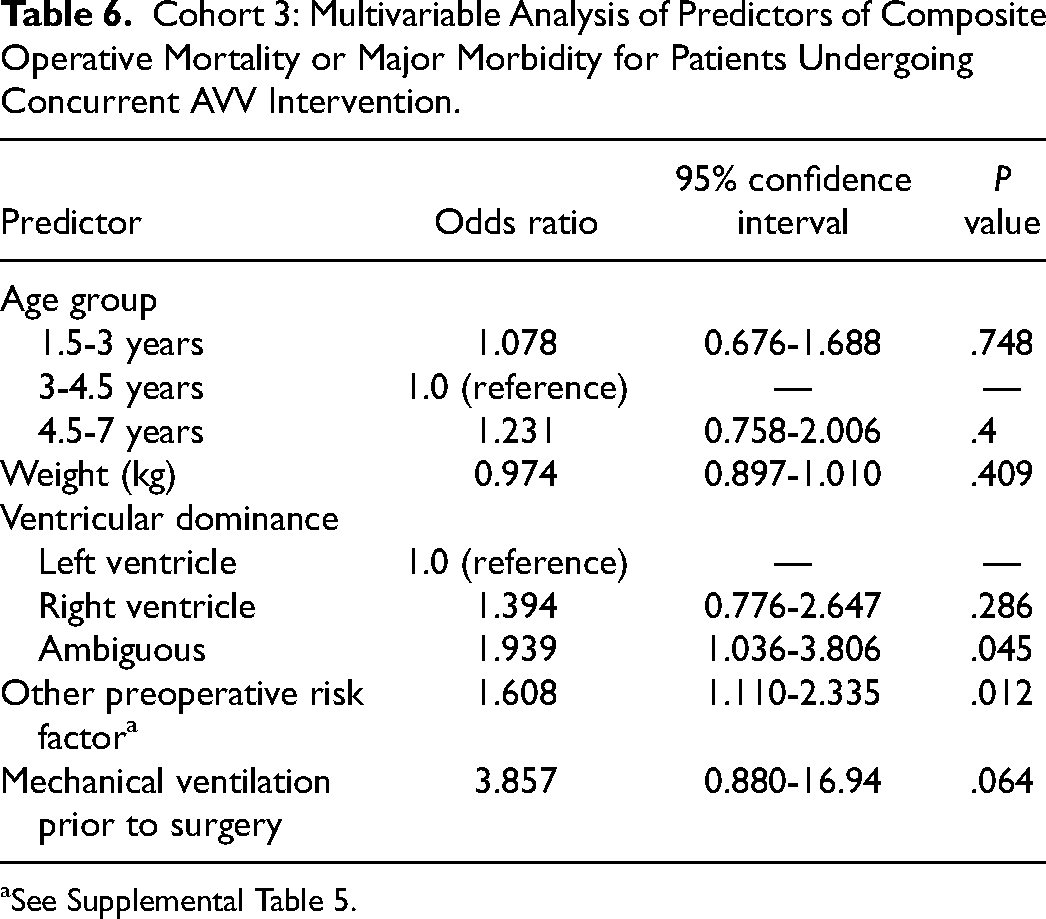
See Supplemental Table 5.
Comment
Concomitant AVV intervention is associated with an increased risk of operative mortality and major morbidity, including requirement of mechanical circulatory support, unplanned reoperation, permanent pacemaker, and renal failure at all stages of SVP. This is consistent with prior studies showing that AVV dysfunction and concomitant AVV intervention are risk factors for morbidity and mortality in these patients.1,3–9,17,18 This study adds to the body of knowledge on the subject by confirming that these findings hold true for short-term outcomes across all FUVH patients undergoing SVP.
Increased weight was found to be associated with a decreased occurrence of composite operative mortality or major morbidity in cohort 1 but not cohorts 2 and 3. This is consistent with the notion that weight is a more important factor when operating on neonates than older infants and children. The finding that a comprehensive stage 2 procedure was independently associated with an increased occurrence of composite operative mortality or major morbidity was expected. A comprehensive stage 2 operation is longer and more technically challenging than a superior cavopulmonary anastomosis and is reserved by some centers for high-risk patients. Therefore, comprehensive stage 2 would be expected to have worse outcomes when compared directly with superior cavopulmonary anastomosis. This study did not examine longitudinal outcomes, so it is impossible to compare the entire pathways leading up to and including these procedures. Thus, we cannot make any conclusion comparing these approaches with SVP. The presence of one or more preoperative noncardiac risk factors (Supplemental Table 5) was identified as independently associated with composite operative mortality or major morbidity in cohorts 2 and 3. It is, however, not possible to objectively speculate as to why any among this diverse list of noncardiac preoperative factors was associated with composite operative mortality or major morbidity.
Black race was identified as being independently associated with composite operative mortality or major morbidity for cohort 1. This is consistent with prior studies showing black race to be a risk factor for poor surgical outcomes. 22 This finding highlights the need to ensure our databases are well-equipped to study healthcare disparities. Ongoing investigation into the reasons for these differences is critical to improve outcomes.
Preterm birth was identified as being independently associated with composite operative mortality or major morbidity for cohort 2. Preterm patients are known to have parenchymal lung disease that may predispose them to having increased pulmonary vascular resistance (PVR) throughout life. Cohort 1 patients have shunted physiology in the postoperative period, so elevated PVR is not expected to be a large concern for them and may even be helpful depending on their hemodynamic balance. Cohort 2 patients rely on passive blood flow through the pulmonary vasculature and would be expected to be more sensitive to elevated PVR. It has been shown that elevated PA pressure prior to stage 2 is a risk factor for Fontan completion failure, 5 which is consistent with this finding. Patients included in cohort 3 have already demonstrated that their PVR is low enough for passive pulmonary blood flow, and thus, preterm birth would no longer be expected to be a risk factor for them. Similarly, preoperative mechanical ventilation was found to be independently associated with composite operative mortality or major morbidity for cohort 2. This is consistent with the concept that the condition of the lungs and pulmonary vasculature is a strong driver of stage 2 outcomes. Careful consideration should be given to patients who are preterm or require mechanical ventilation prior to undergoing concomitant AVV intervention during stage 2 SVP.
Ambiguous ventricular dominance was found to be independently associated with composite operative mortality or major morbidity in cohorts 1 and 3. Ambiguous ventricular dominance included patients with unbalanced atrioventricular canal, heterotaxy syndrome, or other as their FUVH diagnosis. All three of these presumably place patients at risk for reasons other than their dominant ventricle morphology and have previously been shown to be risk factors for poor outcomes.1,19 Interestingly, RV dominance was not found to be associated with composite operative mortality or major morbidity by multivariable analysis. This is contrary to several prior studies.1,10,23–26 However, all of these studies looked at long-term outcomes, whereas this study looked at short-term outcomes. While RV dominance may be an important risk factor for poor long-term outcomes, it does not appear to influence short-term outcomes. Careful consideration should be given to patients with unbalanced atrioventricular canal, heterotaxy syndrome, or other FUVH diagnoses prior to concomitant AVV intervention during stages 1 and 3 SVP.
This study has several limitations. First, its retrospective design precludes conclusions of causality. Next, SVP is a treatment strategy, not a diagnosis or procedure. Thus, it is not directly captured in the STS-CHSD. Single ventricle palliation strategy and stage were inferred from combinations of diagnosis, procedure, and age. It is possible that some patients were improperly included/excluded from the study, particularly the patients excluded for ambiguous SVP stage. Next, the STS-CHSD lacks the data necessary for longitudinal analysis; thus, we were unable to analyze this patient population through the entire SVP pathway. This limitation included the inability to link, with confidence, sequential hospital encounters that may correspond to a given patient, especially across multiple participant institutions. Next, these cohorts are very heterogeneous, and extrapolating these findings to any given patient is challenging. Next, we limited our time frame to the most recent ten years with complete data with the intent of limiting “era” effects. This may have been too broad of a time frame to completely eliminate this effect. Finally, as is the case with all retrospective database analyses, there are several relevant data points that are not captured, not detailed, or incomplete. These include, but are not limited to, ventricular dominance, ventricular function, pre- and postoperative AVV function, hemodynamics, rhythm, AVV repair technique, and long-term outcomes. Moderate to severe AVV regurgitation is captured by the STS-CHSD, but it was missing or unknown for 46% of patients in this study. We therefore reported the variable in our bivariate analysis for completeness but chose to omit it from further analysis.
Concomitant AVV intervention is a risk factor for operative mortality or major morbidity in FUVH patients at each stage of SVP. Several risk factors were identified as being independently associated with these poor outcomes and may be useful in guiding decision-making for these patients. Further investigation is necessary to define the optimal treatment strategy for this challenging patient population.
Supplemental Material
sj-xlsx-1-pch-10.1177_21501351241269924 - Supplemental material for Impact of Atrioventricular Valve Intervention at Each Stage of Single Ventricle Palliation
Supplemental material, sj-xlsx-1-pch-10.1177_21501351241269924 for Impact of Atrioventricular Valve Intervention at Each Stage of Single Ventricle Palliation by John D. Vossler, Aaron W. Eckhauser, Eric R. Griffiths, Reilly D. Hobbs, Linda M. Lambert, Lloyd Y. Tani, Niharika Parsons, Robert H. Habib, Jeffrey P. Jacobs, Marshall L. Jacobs and S. Adil Husain in World Journal for Pediatric and Congenital Heart Surgery
Footnotes
Abbreviations
Acknowledgments
The data for this research was provided by The STS National Database Access and Publications Research Program. Analysis was performed by the STS Research and Analytic Center.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
