Abstract
Nickel is a component of nitinol, an alloy used in several medical devices. Allergy to nickel may place patients at a high risk for severe hypersensitivity reactions. We report a rare case of a patient who developed severe ulcerative colitis ten years following closure of an atrial septal defect with the Amplatzer Septal Occluder device.
Keywords
Introduction
Recent epidemiologic data suggest that atrial septal defects (ASDs) occur in 1.6 per 1,000 live births. 1 Over the last two decades, the increased utilization of percutaneous ASD closure has been driven by the advantages of an earlier recovery to normal activity and the avoidance of sternotomy and cardiopulmonary bypass. 2 However, percutaneous device closure is not entirely without risk. The Amplatzer (Abbott) Atrial Septal Occluder (ASO) is a double-disc device comprised of polyester material and a wire mesh made of Nitinol (Nickel Titanium Naval Ordnance Laboratory), which is a shape-memory alloy of 55% nickel, and titanium. 3 Allergy to nickel can have a variable presentation; we describe the clinical course of a patient who developed a severe autoimmune reaction manifesting as ulcerative colitis (UC), following ASO device closure.
Case Report
A 27-year-old female presented to her primary care physician with complaints of eczema, hives, cystic acne, bleeding gums, and migraines. Her past medical history was significant for a moderate-sized ASD which was closed using the ASO device when she was 12 years old. Within 4 to 6 months of implant, she experienced hives and migraines; these symptoms flared episodically over the course of nine years. At around 22 years of age, she developed symptoms of chronic abdominal pain and fatigue and was diagnosed with irritable bowel syndrome. Two years later, her abdominal symptoms had deteriorated significantly. She underwent colonoscopy and was diagnosed with biopsy-proven UC. Over the next several years, she was tried on successive medical management regimens (Humira, Prednisone, Inflectra, Entyvio, and Remicaide), and her UC proved refractory. She was ravaged by the frequent and episodic intensity of her abdominal symptoms and developed significant weight and energy loss. Surgical evaluation for a total colectomy and end-ileostomy was initiated. As part of the work-up for UC, skin patch allergy testing was performed. Strong allergic reactions to nickel, palladium, cobalt, and vanadium were noted. The nickel content in the ASO device led to the hypothesis that protracted nickel exposure may be the causative factor.
The patient was evaluated by echocardiogram and subsequently underwent surgical device explanation via sternotomy and cardiopulmonary bypass. The resultant ASD was closed with autologous pericardial patch. The ASO device was noted to be very adherent to the underlying tissues necessitating careful piecemeal extractions for complete removal, and with great caution exercised to prevent damage to the adjacent valve tissue, valve apparatus, and conduction system. Concern for potential reaction to metallic sternal wires led to use of nonabsorbable sutures for sternal closure. The patient had an uneventful postoperative course and was discharged home on the third postoperative day. Pathologic examination showed paucicellular fibroelastic tissue with mild myxoid change, and no areas of acute inflammation, giant cell change, or dystrophic calcification (Figure 1).
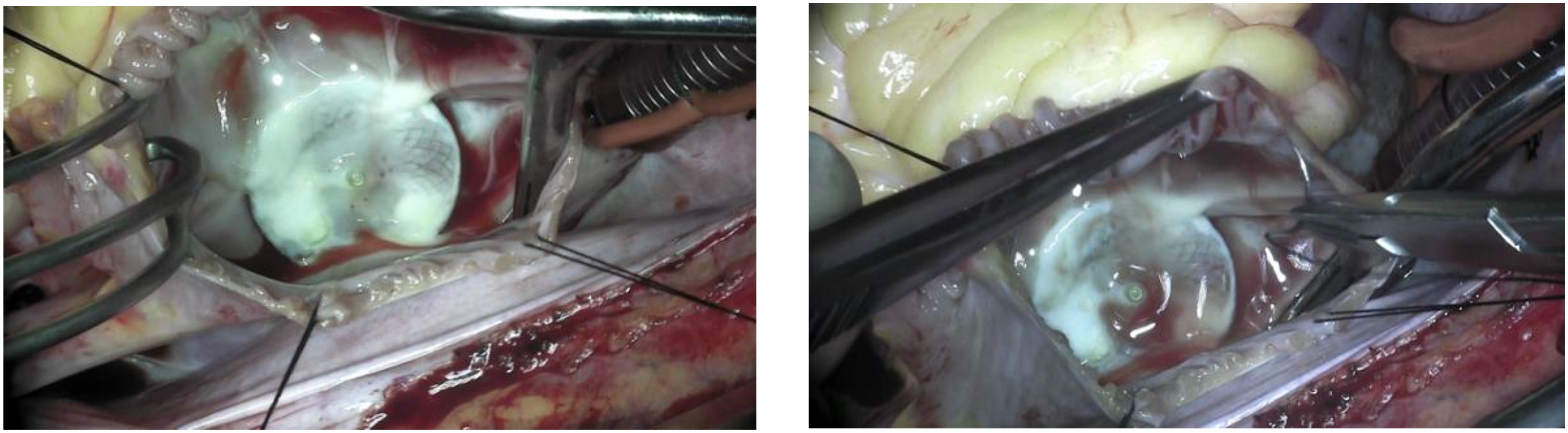
Intraoperative view of the Amplatzer Septal Occluder device. The device was well-adherent with a prominent fibrotic reaction.
The patient noticed immediate and complete relief from her symptoms of eczema, hives, and migraines after device removal. Follow-up with the gastroenterology service at six months revealed a steady resolution of her abdominal symptoms with colonoscopy evidence of only mild residual colitis. Over the next several months, she came off her steroids, and the multiple medications regimen was tapered down to a single biologic agent. At two years from surgery, she has remained in complete clinical remission with no further UC flares and has maintained steady and stable weight gain with normal energy levels. In her own words, the patient describes her remarkable clinical improvement following device removal as “the difference between night and day.” Repeat colonoscopy at two years following surgery shows only mildly disorganized glandular architecture with no evidence of active colitis in biopsy specimens from the terminal ileum, colon, and rectum. Given the patient’s previous prolonged duration of abdominal symptoms, the prevailing opinion of her gastroenterologist is to discontinue the single biologic agent she is currently taking and wean her very conservately over a period of several months.
Discussion
Percutaneous device closure of ASD is an attractive option since it allows for a scarless and rapid recovery. Although the procedural ease of ASO has driven its utilization, 2 these devices are not without risk.
The prevalence of nickel allergy is reported as 8% to 28% in females of Scandinavian descent. 3 Chronic internal exposure to nickel is known to elicit cutaneous and systemic allergic reactions. 4 Symptomatology is diverse and includes urticaria and hives, migraines and vertigo, and chest pain, palpitations, and fatigue. 5 In a large series of ASO device explant patients, the temporal relationship from device implantation to symptom onset ranged from one day to one year; however, the patients were managed medically for a median of eight years (range: 6 months-18 years) before being assessed for device removal. 5 The long duration between implantation and removal was explained by the diverse spectrum of debilitating yet nonspecific symptoms.
We report a possibly unique manifestation of nickel allergy: a patient with refractory UC which occurred nearly ten years following ASO implantation. A significantly higher prevalence of hypersensitivity to nickel and palladium in patients with UC has been shown, suggesting that aberrant immune activation may lead to mucosal inflammation. 6 The immunologic response may be due to both innate and adaptive type-IV delayed reactions.
Protracted or lifelong immunotherapy and the permanent alteration in bowel configuration with end-ileostomy are consequences of UC that significantly impacts quality of life. The consideration of nickel allergy as the possible trigger for UC in our patient led to the surgical explantation of the ASO device, which induced a complete clinical remission and averted a total colectomy.
Although routine preimplant patch testing is not currently recommended by the American Contact Dermatitis Society, 4 and there are no definitive data to suggest that preimplant patch testing correlates with the development of subsequent adverse or allergic reactions, 7 it may be prudent to screen for nickel allergy prior to implanting a nickel-containing device, given the relatively high incidence of nickel allergy in young white females. Serum levels of nickel have been shown to peak at one month and return to baseline values by one year after implantation yet there does not appear to be a correlation between serum nickel levels and allergic reactions to nickel. 8 The utility of testing nickel levels in late-presenting patients is unclear, and more sophisticated tests may need to be developed. It is entirely probable that implantation itself may cause subsequent sensitization 7 ; postimplant patch testing, especially when new or refractory symptoms develop after device implantation, is recommended. 4 Patient education and physician awareness are necessary for early postimplant patch testing. This can lead to an earlier diagnosis and a quicker definitive intervention thereby optimizing the clinical course and outcomes.
Footnotes
Authors' Statement
The author confirms that permission was granted by the subject patient to publish this case report. The author also confirms that I obtained the patient's consent directly and cross-checked all details pertaining to history and clinical course.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
