Abstract
Objectives
Patients with dextro-transposition of the great arteries (d-TGA) frequently undergo balloon atrial septostomy (BAS) prior to the arterial switch operation (ASO) to promote atrial-level mixing. Balloon atrial septostomy has inherent risks as an invasive procedure and may not always be necessary. This study revisits the routine utilization of BAS prior to ASO.
Methods
Single-center, retrospective review of d-TGA patients undergoing the ASO from July 2018 to March 2023. Preoperative patient characteristics, pulse oximetry oxygen saturations (SpO2), cerebral/renal near-infrared spectroscopy (NIRS) readings along with prostaglandin status at the time of the ASO were analyzed with descriptive and univariate statistics.
Results
Thirty patients underwent the ASO. Of these, 7 (23%) were female, 25 (83%) were white, and median weight at ASO was 3.2 kg (range 0.8-4.2). Twenty-two (73%) patients underwent BAS. There were no demographic differences between BAS and no-BAS patients. Of those who underwent BAS, there was a significant increase in SpO2 (median 83% [range 54-92] to median 87% [range 72-95], P = .007); however, there was no change in NIRS from pre-to-post BAS. Six (27%) patients in the BAS group were prostaglandin-free at ASO. Balloon atrial septostomy patients underwent the ASO later compared with no-BAS patients (median 8 [range 3-32] vs 4 [range 2-10] days old, P = .016) and had a longer hospital length of stay (median 13 [range 7-43] vs 10 [range 8-131] days, P = .108).
Conclusions
While BAS is an accepted preoperative procedure in d-TGA patients to improve oxygen saturations, it is also an additional invasive procedure, does not guarantee prostaglandin-free status at the time of ASO, and may increase the interval to ASO. Birth to direct early ASO, with prostaglandin support, should be revisited as an alternative, potentially more expeditious strategy.
Keywords
Introduction
Balloon atrial septostomy (BAS), introduced by Rashkind and Miller, 1 is part of currently accepted standardized management in neonates with dextro-transposition of the great arteries (d-TGA). 2 Perceived benefits include stabilization of pulmonary vascular resistance and flexibility in scheduling the arterial switch operation (ASO). Recent trends suggest that a more selective strategy for BAS may be warranted. 3
While BAS may improve measured arterial oxygen saturations many patients are not successfully weaned from prostaglandin prior to anatomical correction.4,5 Additionally, BAS-related complications are not trivial and may place patients at a higher risk of subclinical brain injury or stroke. 6
Several recent studies have confirmed excellent outcomes with ASO as early as three days of age, in addition to significantly lowering associated economic burden. 7 Additionally, the reduced overall duration of preoperative hypoxia, with its attendant risk of periventricular leukomalacia 8 may confer significant long-term benefit. Finally, in an experience with the ASO for d-TGA spanning over 30 years, it has recently become our perception that inordinate delays between birth and definitive operation may be exacerbated by an interval BAS. Furthermore, with the advent of close preoperative monitoring of systemic oxygen delivery, it is our observation that in the current era, many if not most patients undergoing a successful BAS are not weaned from prostaglandin prior to surgery.
Given these considerations, we questioned the necessity of routine BAS prior to the ASO and sought to examine outcomes in neonates undergoing BAS prior to the ASO in comparison with those proceeding directly to operation.
Methods
Study Population
Approval was obtained from the Institutional Review Board of the University of Texas at Austin. This is a retrospective review of neonates who presented to Dell Children's Medical Center with d-TGA between July 2018 and March 2023.
Study Measures
A retrospective chart review of all neonates with d-TGA, with or without an intact ventricular septum (IVS), who underwent the ASO was conducted. Patients with Taussig-Bing anomaly were excluded from this study.
The aim of this study was to compare neonates who underwent initial BAS (BAS group) with neonates who went directly to ASO without intervening BAS (no-BAS group).
All patients were initiated on prostaglandin infusion immediately after diagnosis. The decision to perform BAS was based on the initial clinical assessment and incorporated a composite of clinical cardiopulmonary status, echocardiographic evaluation of interatrial gradient, and initial arterial oxygen saturation by pulse oximeter. It was also influenced by the individual assessment of the admitting physician in the setting of a typically liberal BAS strategy. Patients who were stably maintained on prostaglandin infusion did not undergo BAS.
Demographic data, birth weight, weight, and age at the ASO, mean preductal systemic arterial oxygen saturations by pulse oximeter (SpO2) before and after BAS, markers of oxygen delivery measured by cerebral and renal near-infrared spectroscopy (NIRS) before and after BAS, BAS-related complications, freedom from prostaglandin infusion at the time of the ASO, timing of the ASO, evidence for low cardiac output syndrome (LCOS) within 48 h post-ASO, in-hospital mortality, and hospital length of stay (LOS) were compared.
Based on hourly nurse charting, the lowest documented SpO2 and cerebral/renal NIRS saturations were collected for each of the time intervals specified as pre-BAS, post-BAS, and pre-ASO; the mean SpO2 and NIRS readings in the pre-BAS, post-BAS, and pre-ASO time frames were calculated by obtaining the total of the hourly documented readings (for a maximum of 12 h) and dividing the total by the number of hours within each time interval; the mean value thus calculated for each time interval was used for subsequent analysis. Low cardiac output syndrome was defined as the presence of oliguria (urine output <0.5 cc/kg/h), escalation of inotropic support (for echocardiogram-assessed poor ventricular function), elevated left atrial pressures (>10 mm Hg) as measured by a left atrial catheter placed intraoperatively, or elevated serum lactate levels >3 mmol/L within the first 48 h following ASO.
Statistical Analysis
The demographic and patient characteristics were described using frequencies and percentages for categorical data, while medians, minimum-maximum range, and interquartile ranges (IQRs) were used for continuous data. Fisher exact test for categorical variables and the Wilcoxon-Mann-Whitney test for continuous variables were used to compare the BAS group with the no-BAS group. Wilcoxon signed-rank tests were employed to evaluate the alterations in continuous data from pre- to post-BAS. A two-tailed alpha level of .05 was used to determine statistical significance. All statistical analyses were performed using SAS (Statistical Analysis Software 9.4, SAS Institute Inc).
Results
Thirty patients were identified who underwent the ASO during the study period; 7 (23%) were female and 25 (83%) were white. Postnatal 2-D transthoracic echocardiogram evaluation diagnosed 15 (50%) patients with intact ventricular septum and 15 (50%) patients with ventricular septal defect (VSD). Two (6.7%) patients had associated aortic arch hypoplasia. There were 4 (13.3%) babies born at gestational age <36 weeks, and 4 (13.3%) babies had associated chromosomal anomalies. The median gestational age was 39 weeks [IQR 38-39] (range 27-41), median birth weight was 3.2 kg [IQR 2.9-3.6 kg] (range 0.8-4.2 kg), and weight at the ASO was 3.2 kg [IQR 3.0-3.5 kg] (range 0.8-4.2 kg) (Table 1). Balloon atrial septostomy was performed in 22/30 (73.3%) patients (BAS group), and 15 of 22 (68.1%) had BAS performed within 24 h of birth; 8 (26.7%) patients went directly to the ASO without intervening BAS (no-BAS group).
Patient Characteristics of Patients Who Underwent the ASO Stratified by BAS Status.
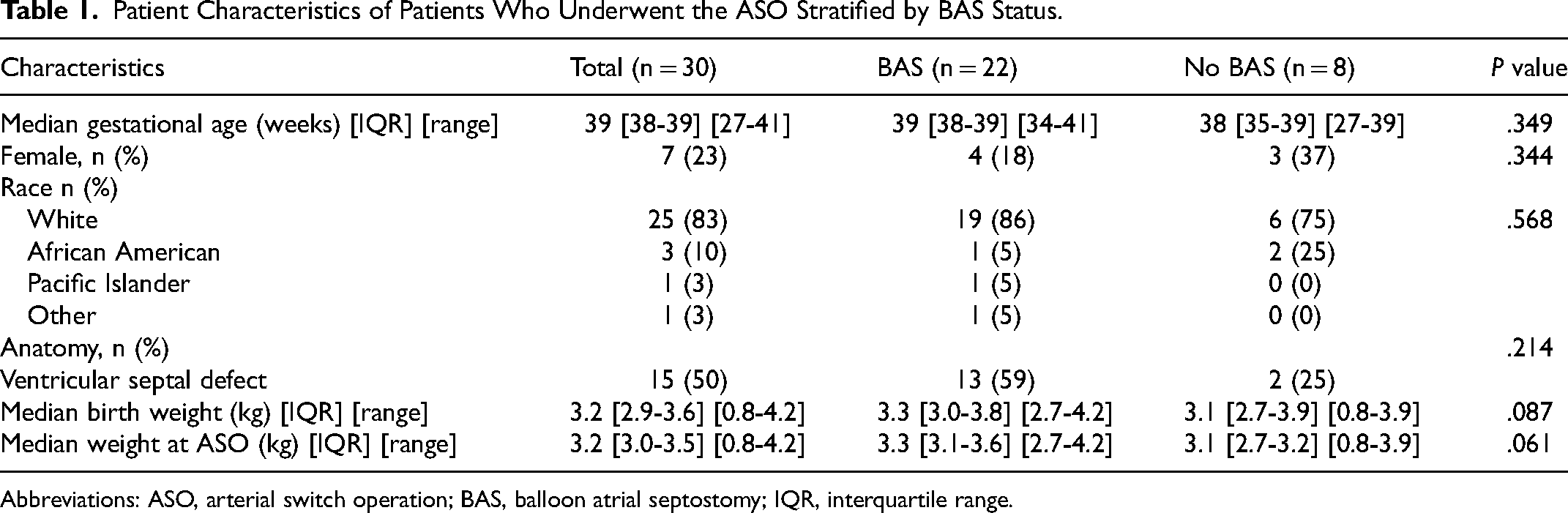
Abbreviations: ASO, arterial switch operation; BAS, balloon atrial septostomy; IQR, interquartile range.
Figure 1 illustrates the SpO2 and cerebral/renal NIRS readings before and after BAS. There was a significant improvement in SpO2 post-BAS (median 83% [IQR 75%-88%] (range 54%-92%] to median 87% [IQR 85%-90%] [range 72%-95%]), P = .007. There were no significant changes in the cerebral/renal NIRS readings post-BAS.
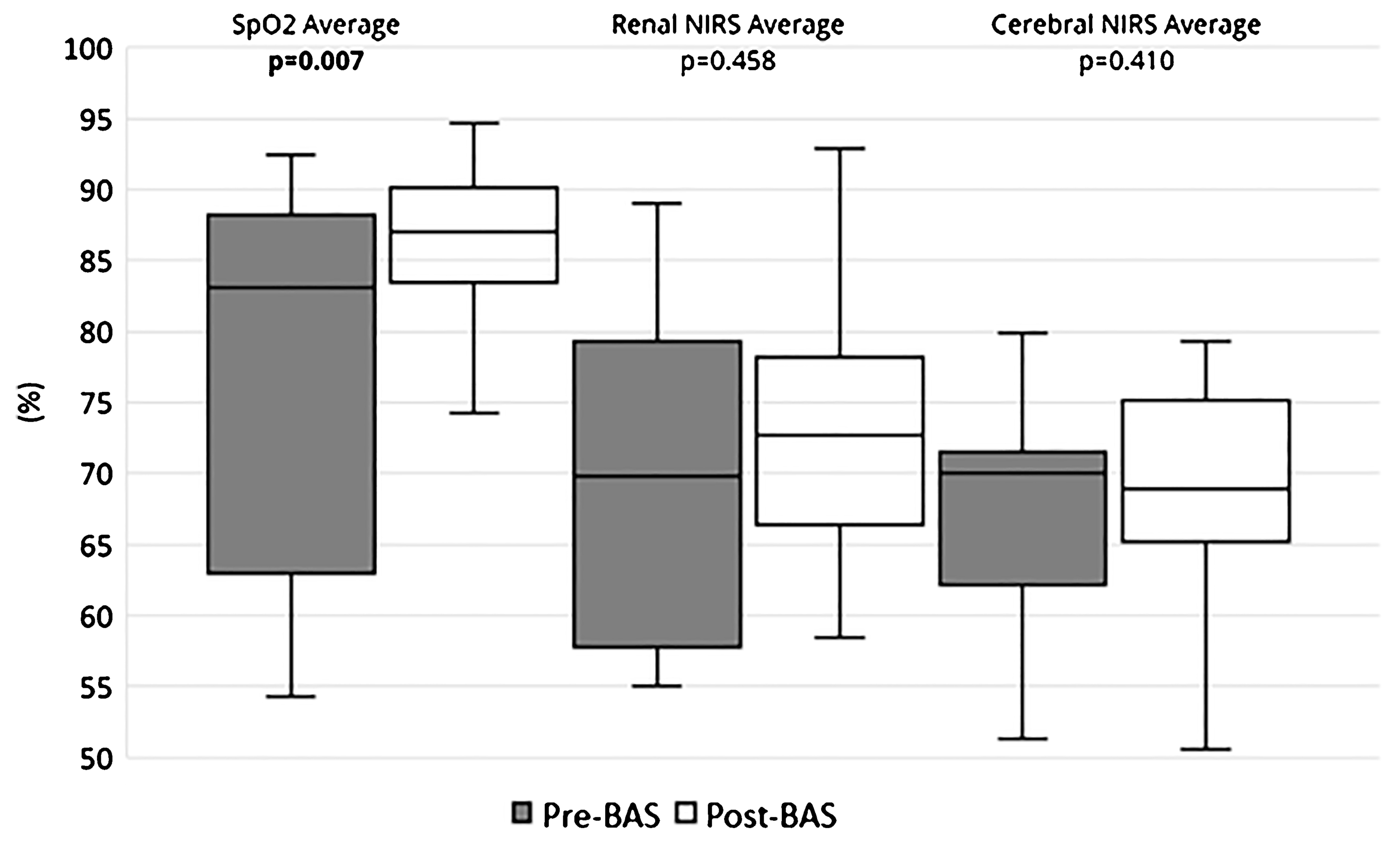
Oxygen saturations and cerebral/renal NIRS pre- and post-BAS.
Of the 22 patients in the BAS group, six (27%) were never able to be weaned from prostaglandin. In the remaining 16 patients of the BAS group, the attempted wean from prostaglandin was successful in 6 (38%) patients and reinitiated in 10 (62%) patients. Figure 2 shows a scatter-plot distribution of prostaglandin reinitiation timing in the BAS group; most patients (n = 7/10 (70%)) were reinitiated within 24 h of BAS. In the no-BAS group of eight patients, there was one (13%) patient with a VSD, who was weaned off prostaglandin, P = .638 (Table 2). There was no significant difference in prostaglandin-free status between patients with or without a VSD (VSD: n = 5/7 (71%); IVS: n = 2/7 (29%), P = .195) (Table 3).
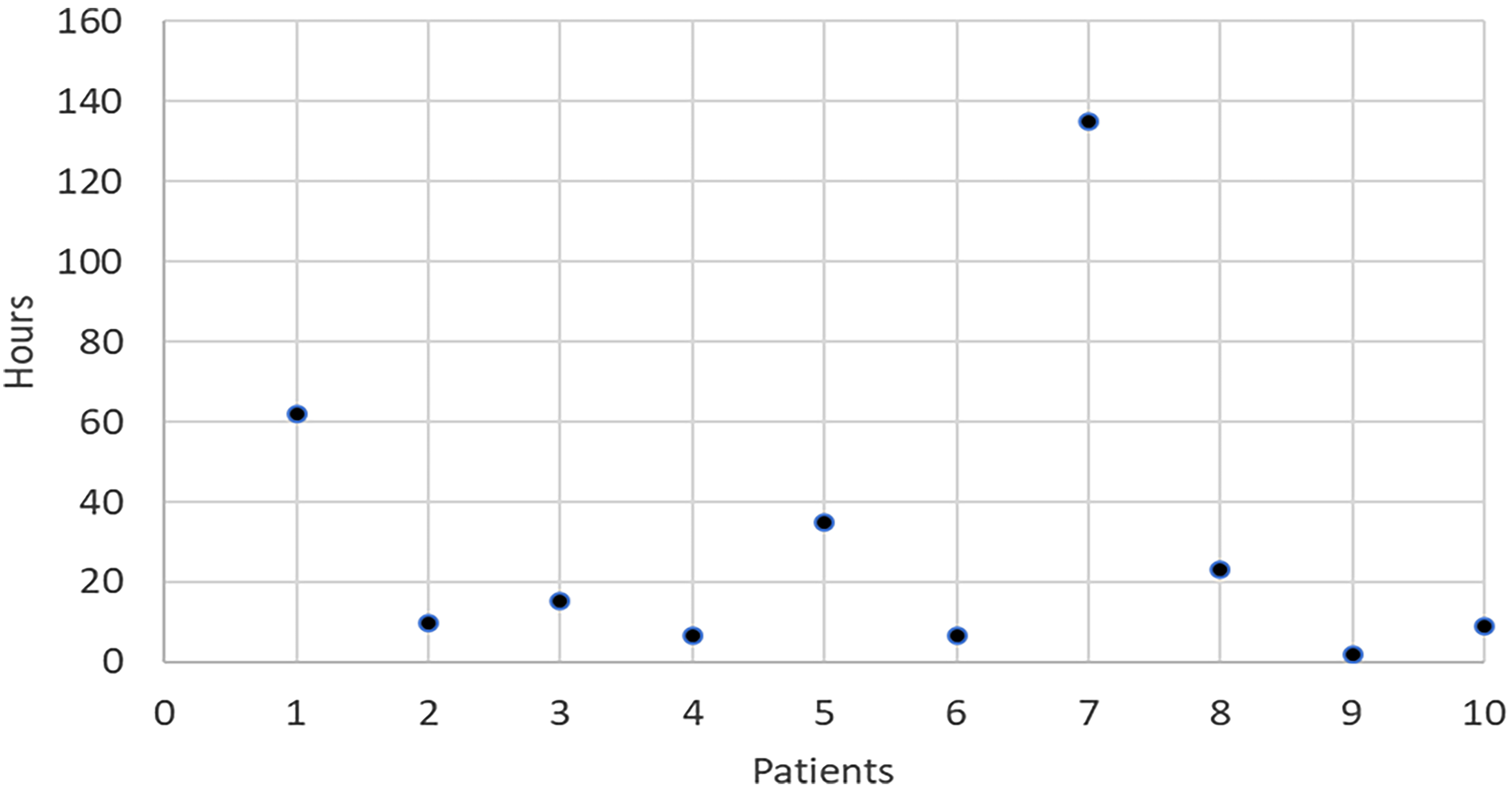
Scatter-plot frequency of PGE reinitiation after balloon atrial septostomy (BAS) (n = 10).
Outcomes of Patients Who Underwent the ASO Stratified by BAS Status.
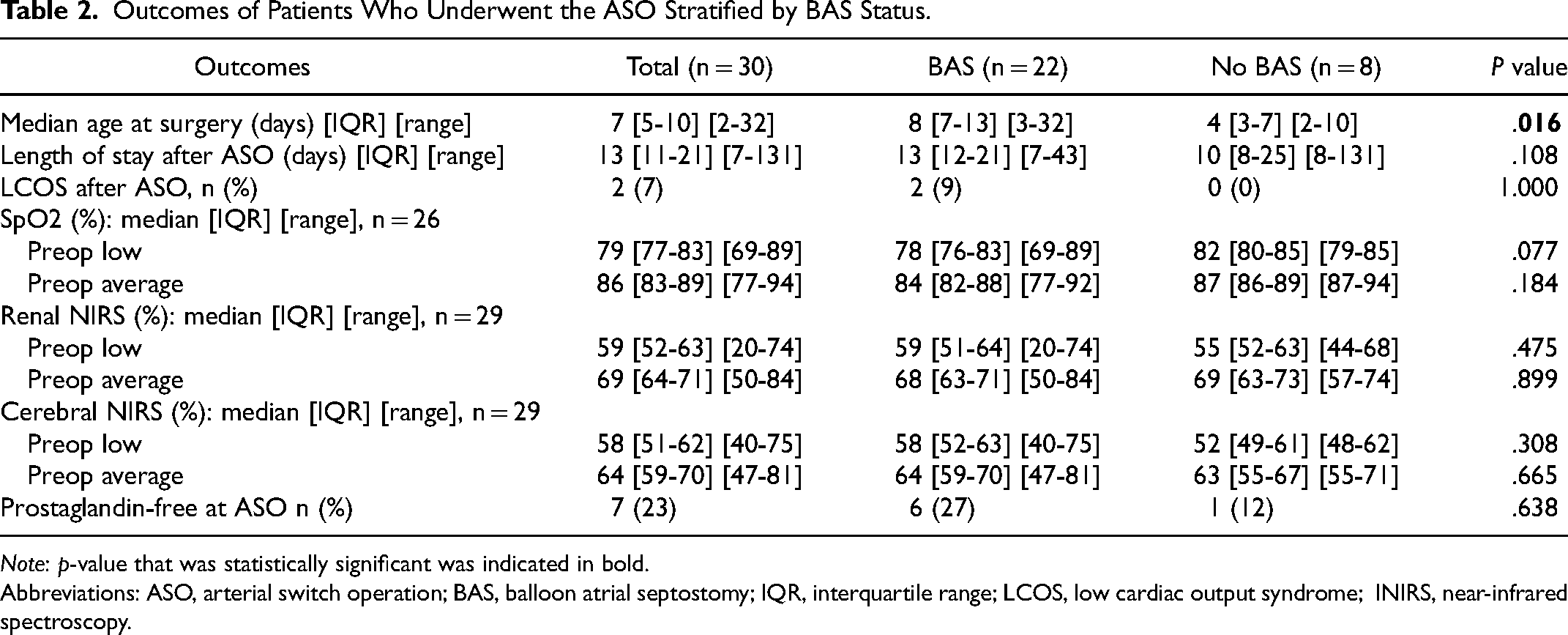
Note: p-value that was statistically significant was indicated in bold.
Abbreviations: ASO, arterial switch operation; BAS, balloon atrial septostomy; IQR, interquartile range; LCOS, low cardiac output syndrome; INIRS, near-infrared spectroscopy.
Cross-Tabulation of Anatomy and Prostaglandin Status at the Time of the ASO.

Abbreviations: ASO, arterial switch operation; d-TGA, dextro-transposition of great arteries; VSD, ventricular septal defect.
Table 2 shows the outcomes of patients who underwent the ASO by their BAS status. The median age at the ASO was eight days [IQR 7-13] [range 3-32] in the BAS group, and four days [IQR 3-7] (range 2-10) in the no-BAS group, P = .016. The median hospital LOS was 13 days [IQR 12-21] (range 7-43) in the BAS group, and 10 days [IQR 8-25] (range 8-131) in the no-BAS group, P = .108. There were no (0%) patients with LCOS within the first 48 h in the no-BAS group, whereas there were 2 (9%) patients with LCOS in the BAS group (P = 1.000), which had resolved between 48- and 72-h post-ASO. There were no BAS-related complications or in-hospital deaths in either group. There were no complications related to postoperative pulmonary hypertensive crises in the no-BAS group, based on clinical course and left atrial catheter readings consistently <10 mm Hg.
Comment
Standardized protocols for d-TGA management immediately after birth often include a liberal approach to performance of BAS prior to the ASO. 2 This may allow for flexibility in operative scheduling which could be an advantage when resource optimization is a consideration. Furthermore, the perception has been that BAS allows promotion of patients to prostaglandin weaning prior to definitive surgery.
Utilization of BAS prior to ASO is variable and largely determined by institutional practices. The European task force guidelines suggest that BAS may improve mixing in patients with refractory hypoxemia or with LCOS. 3 The role for non-urgent BAS, however, is less clear.
It has been our concern that routine BAS may have the unintended consequence of unnecessarily increasing the interval between birth and the ASO. Increased arterial oxygen saturations post-BAS may lend support to the opinion that later surgery is acceptable. We have noted that the initial improvement in arterial oxygen saturations achieved by BAS may not be sustained, leading to the re-initiation of prostaglandins. The absence of uniform improvement in oxygen delivery, and the increased interval from birth to the ASO has led us to revisit our practice paradigm. In our current thinking, a practical alternate strategy would be to commence prostaglandin infusion soon after birth in patients with prenatally diagnosed d-TGA, and, after standard perinatal assessments are completed, proceed directly for operative repair, ideally within 48 h after birth. Although institutional biases and standardized protocols established at centers over the last several years may pose a barrier, it is our belief that birth to direct repair with prostaglandin support may be a potentially superior strategy.
As early as 1992, Baylen et al demonstrated that patients who underwent the ASO without a preceding BAS had a clinical course similar to those that did undergo BAS. Of 23 neonates with d-TGA, 15 patients, on prostaglandin infusion, did not undergo BAS based on clinical cardiopulmonary status, echocardiography evaluation, p02 >30 mm Hg, pulse oximeter arterial oxygen saturations >70%, and pH within physiologic range. In eight patients, a restrictive PFO with limited inter-atrial shunt led to BAS, usually within 48 h of birth, and was associated with a significant increase in arterial pO2. The mean age at which the BAS group underwent the ASO was significantly greater; apart from this, there were no differences between the two groups in their postoperative or clinical course. 9
Other recent reports have noted a declining trend in non-urgent BAS.4,10 While the primary aim of BAS is arterial oxygen saturation improvement and stability, this is not achieved in up to 20% of patients and it is not always sustained. 11 Additionally, there appears to be a poor correlation between ASD size and oxygen delivery. 12 Among other concerns, it is also well-recognized that BAS, as an invasive procedure, carries the specific risk profile of stroke, thrombus, femoral/iliac vein thrombosis, and perforation.6,13,14
In our cohort of patients, the BAS group did achieve a numerically significant immediate improvement in arterial oxygen saturations; however, despite this initial improvement, less than a third of them were able to remain off prostaglandin. This finding is also consistent with Zaleski et al who found that although there was improvement in arterial oxygen saturations, the performance of BAS did not eliminate the need for prostaglandin at the time of surgery. 4 Several studies have noted that despite the improvement in arterial oxygen saturations with BAS, the rate of prostaglandin discontinuation was only 38% to 56%.5,15,16 When the original protocol for BAS prior to the ASO was first derived, advanced monitoring devices such as NIRS were often not routinely available, and prostaglandin weaning decisions tended to be based on bedside assessment and clinical judgment. While the advent of these sophisticated monitoring devices for tissue perfusion in the modern-day ICU has enabled improved assessment of oxygen delivery, ironically it has also made it more challenging to determine the timing of prostaglandin withdrawal. Many practitioners agree that continuation of prostaglandin infusion serves as a pulmonary vasodilator and that it also protects from rebound hypoxemia. 12 Continuing the prostaglandin infusion through surgery could therefore be beneficial; it may also often be sufficient without the need for adjunct BAS.
In considering BAS prior to the ASO, a factor that is often overlooked is the risk associated with multiple exposures to volatile anesthetic agents. An association between volatile anesthetic exposure, brain injury detected by magnetic resonance imaging, ICU LOS, and lower neurodevelopmental outcome scores at 12 months of age was shown in neonates undergoing complex congenital heart surgery. 15 Reducing the frequency of exposure to these agents by reducing the number of procedures may be beneficial for patients requiring the ASO.
A further argument for early ASO without intervening BAS can be made from a neuroprotective standpoint. Multiple studies have shown the adverse relationship between prolonged hypoxia and neurodevelopment outcomes. Low arterial oxygen level was an independent risk factor for periventricular leukomalacia and longer time to surgery may be an additive risk factor for its development.8,17 While there are several mitigating factors that increase the risk of perinatal hypoxemia, it is true that neonatal brains are more resilient and mature rapidly especially as myelination progresses after birth; reduced duration of hypoxemia and decreased fluctuations in arterial oxygen saturations prior to the ASO may be beneficial.
Although there may be a concern for postoperative pulmonary hypertensive crises with early ASO, none of the patients in our cohort demonstrated a left atrial pressure exceeding 10 mm Hg in the postoperative period. It has been reported by Nevvazhay et al that the ASO within the first 24 h of birth can be safely accomplished without additional morbidity or mortality. 18 Chasovsky et al, in evaluating the use of autologous umbilical cord blood transfusion (AUCBT), randomized 21 neonates to undergo the ASO in the first few hours of life. Although their primary objective was the use of AUCBT, it was remarkable that the early ASO group showed no increase in major morbidity or mortality. 19 These studies show that early direct ASO can be safely accomplished; minimizing the overall duration of preoperative hypoxemia that could potentially confer a long-term neuroprotective benefit.
A final consideration is cost. The multiple interventions and the accumulation of hospital days, as the interval from birth to the ASO increases, impose a significant economic burden. In a study of 140 infants operated at a single center between 2003 and 2012, 80% of neonates had BAS, and the usual age at the ASO was five days (range 1-12 days). The authors showed that for babies between the ages of 1 to 3 days, for every day later that surgery was performed, the odds of a major morbidity decreased by 46% (range 3%-70%), and for ages older than three days, for every day later that surgery was performed, the odds of a major morbidity increased by 47% (range 23%-66%). Also, for every day later that surgery was performed, reimbursements increased by 8% across all ages (P < .001). They inferred that the ASO is ideally performed on DOL 3. 7 In our study, the median time to the ASO in the BAS group was eight days, whereas it was four days in the no-BAS group. While there were no differences in overall outcomes between the two groups, the LOS in the no-BAS group was shorter (median 10 vs 13 days). Although our cohort is small, these results suggest that there could be a resource utilization benefit to earlier and direct ASO without intervening BAS.
Study Limitations
The small number of patients in each group confers limited power to the study. Balloon atrial septostomy was mostly driven by the practice of a standardized management strategy developed in 1995. 2
One of the less-clearly-defined issues of selection for BAS is the personal bias of the physician making the initial clinical assessment. Although no specific criteria were utilized to select for no-BAS, these patients were stably maintained on prostaglandin infusion, thereby enabling direct repair. While our results reflect retrospective observations and are derived solely from existing practice trends, an unintentional selection bias may be a factor appearing to favor the no-BAS strategy. ASD size or pressure gradient across the atrial septum pre-BAS and post-BAS were not routinely measured or recorded in our cohort. Decisions regarding BAS, PGE reinitiation and timing of definitive repair were made according to the care team's clinical judgment. The subgroup analysis between IVS and VSD was limited due to small patient numbers.
Conclusion
Balloon atrial septostomy may be over-utilized prior to the ASO and is probably not always necessary. Birth to direct early ASO, with prostaglandin support, may be a potentially superior alternative: the postoperative outcomes are similar to those undergoing BAS and later surgery, and there may be the additional advantage of optimal resource utilization. Larger multicenter studies may be required to assess the relative risks and benefits of these different preoperative management strategies.
Footnotes
Authors’ Note
IRB approval was obtained from the Institutional Review Board of the University of Texas at Austin.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
