Abstract
Background
Some patients with pulmonary stenosis present with a complex multilevel disease that involves the valve, the leaflets, and the sinotubular junction (STJ) forming an hourglass appearance. We herein report the mid-term results of our experience with the reconstruction of the supravalvar narrowing using three pericardial patches.
Material and Methods
Retrospective analysis of patient charts and echocardiography studies of patients who underwent three-patch reconstruction of the pulmonary valve (PV) from 2013 to 2022. After PV transection distal to STJ, vertical incisions into the sinuses were performed, and leaflets were trimmed and thinned. The three sinuses were augmented using three pericardial patches.
Results
Nineteen patients underwent repair of hourglass supravalvar pulmonary stenosis. Mean weight at surgery was 9.4 kg (median 7.2, range 4.7-35); 16 patients underwent previous catheterization with unsuccessful balloon dilatation of the PV (13 pts.). Preoperative aortic/pulmonary annulus ratio was 1.02 (median 1, range 0.89-1.25). After surgery, gradients across the PV were significantly reduced (94 ± 26 vs 29 ± 9 mm Hg, P = .02). Postoperatively, 14 patients had mild or no pulmonary insufficiency (PI) and five had mild to moderate PI. At a mean follow-up of 71 months (median 78 months, range 8-137), gradients continued to decrease (29 ± 9 vs 15 ± 5 mm Hg, P < .001).
Conclusion
The three-patch technique for the repair of supravalvar pulmonary stenosis is simple, reproducible, and achieves excellent and long-standing relief of the right ventricular outflow tract gradient.
Introduction
Pulmonary stenosis is common accounting for approximately 6% of all congenital heart defects. When associated with other congenital cardiac lesions, it may occur in as many as 50% of all patients with congenital heart disease.1‐3
Morphologically, pulmonary stenosis was initially classified as involving the valve, the bifurcation, or the distal pulmonary arteries.4,5 In 1988, Dr Simha Milo described an additional type of pulmonary valve (PV) stenosis which he termed as “hour-glass deformity.” 5 In this anatomical subtype, there is a distinct narrowing and thickening of the sinotubular junction, the cusps are thickened and dysplastic, but the annulus is of acceptable size. At that time, the patients were repaired either by a valvectomy or a small transannular patch. Supravalvar pulmonic stenosis has been associated with a rare form of elastin arteriopathy, 6 supravalvar aortic stenosis, and Williams syndrome. 7 There is paucity of data regarding surgical repair of supravalvar pulmonary stenosis either by valvectomy, 5 Y-shaped patch repair, 8 autologous tissue repair, 9 and more recently the three-patch repair.6,10 In all publications, the number of patients treated was small, and there was no mid-term or long-term follow-up of the results. We have been using three-patch reconstruction of the supravalvar area for the last 10 years and we herein report our mid-term follow-up results.
Material and Methods
Retrospective analysis of patient charts and echocardiography studies of patients who underwent three-patch reconstruction of the PV from 2013 to 2022. Demographics, preoperative, operative, and postoperative variables were collected and echocardiographic indices of the patients last follow-up visits were obtained.
Operative Technique
After the induction of general anesthesia a median sternotomy incision was performed, the pericardium was opened longitudinally and a large patch of pericardium was harvested and treated with glutaraldehyde (0.6% glutaraldehyde, soaked for 10 min and doubly rinsed with saline solution). Heparin was administered, and aortic and bicaval cannulations were performed in the usual manner. Cold crystalloid cardioplegia was then administered through the aortic root and the left side of the heart was vented. The main pulmonary artery was transected by a horizontal incision, and the supravalvar area and the PV were then inspected. Usually, a tight supravalvar area was noted (Figure 1A), and three PV leaflets underneath the narrowing were found to be thickened and dysplastic (Figure 1A). In some of the patients in which balloon valvuloplasty had been performed previously, tears were noted in the PV leaflets.
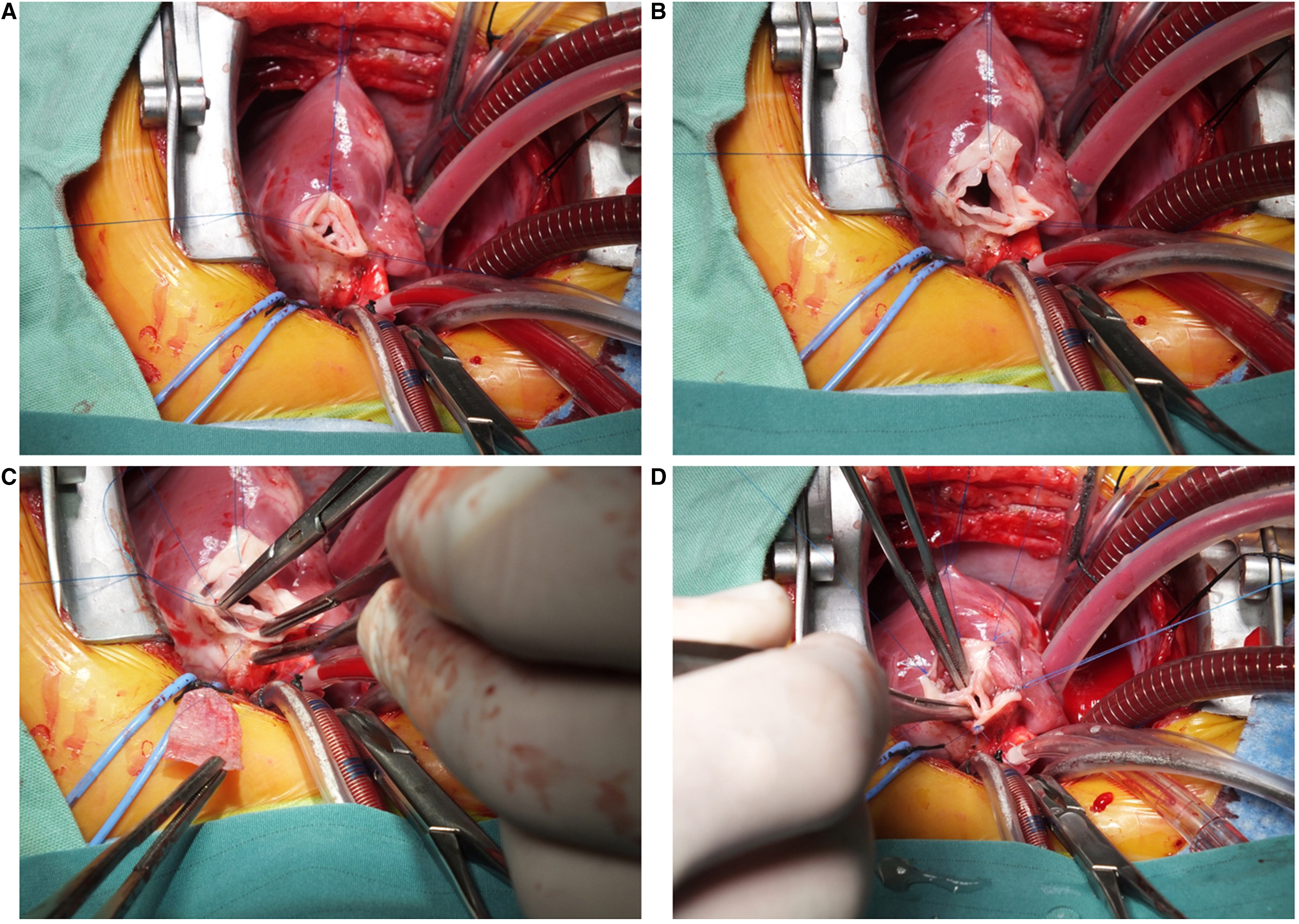
Operative technique for supravalvar pulmonary artery repair. Transection of the main pulmonary artery at the level of the sinotubular junction. Usually, a tight supravalvar area was noted (A). Incisions are made deep into the pulmonary valve sinuses (B). Subsequently, three pericardial patches are used to reconstruct the incised sinuses (C). and complete reconstruction (D). Color version is available online.
Three deep vertical incisions were then performed into each of the three sinuses, and the valve was suspended using stay stitches (Figure 1B). At this point, attention was drawn to the valve itself; commissurotomy was performed if needed, leaflet tears were repaired, and valve leaflets were thinned. Subsequently, three pericardial patches were used to reconstruct the incised sinuses (Figure 1C). Once the reconstruction was completed (Figure 1D), the main pulmonary artery was reanastomosed distally to the pulmonary artery bifurcation occasionally needing pericardial patch augmentation of the pulmonary artery bifurcation. The aortic cross clamp was then removed and the patient was gradually weaned from cardiopulmonary bypass. Intraoperative transesophageal echocardiograpy was performed in all cases, and if there was any doubt regarding postoperative right ventricular pressure, a direct right ventricular pressure measurement was made (Video 1).
Results
Nineteen patients underwent repair of hourglass supravalvar pulmonary stenosis. There were no operative deaths. Mean weight at surgery was 9.4 kg (median 7.2, range 4.7-35); 16 patients underwent previous catheterization of whom 13 underwent unsuccessful balloon dilatation of the PV. Preoperative aortic/pulmonary annulus ratio was 1.02 (median 1, range 0.89-1.25). After surgery, gradients across the PV were significantly reduced (94 ± 26 vs 29 ± 9 mm Hg, P = .02). Postoperatively, 14 patients had mild pulmonary insufficiency (PI) and five had mild to moderate PI. At a mean follow-up of 71 months (median 78 months, range 8-137), gradients continued to decrease (29 ± 9 vs 15 ± 5 mm Hg, P < .001) (Figure 2). Twelve patients had none to mild PI, and seven had mild to moderate PI.
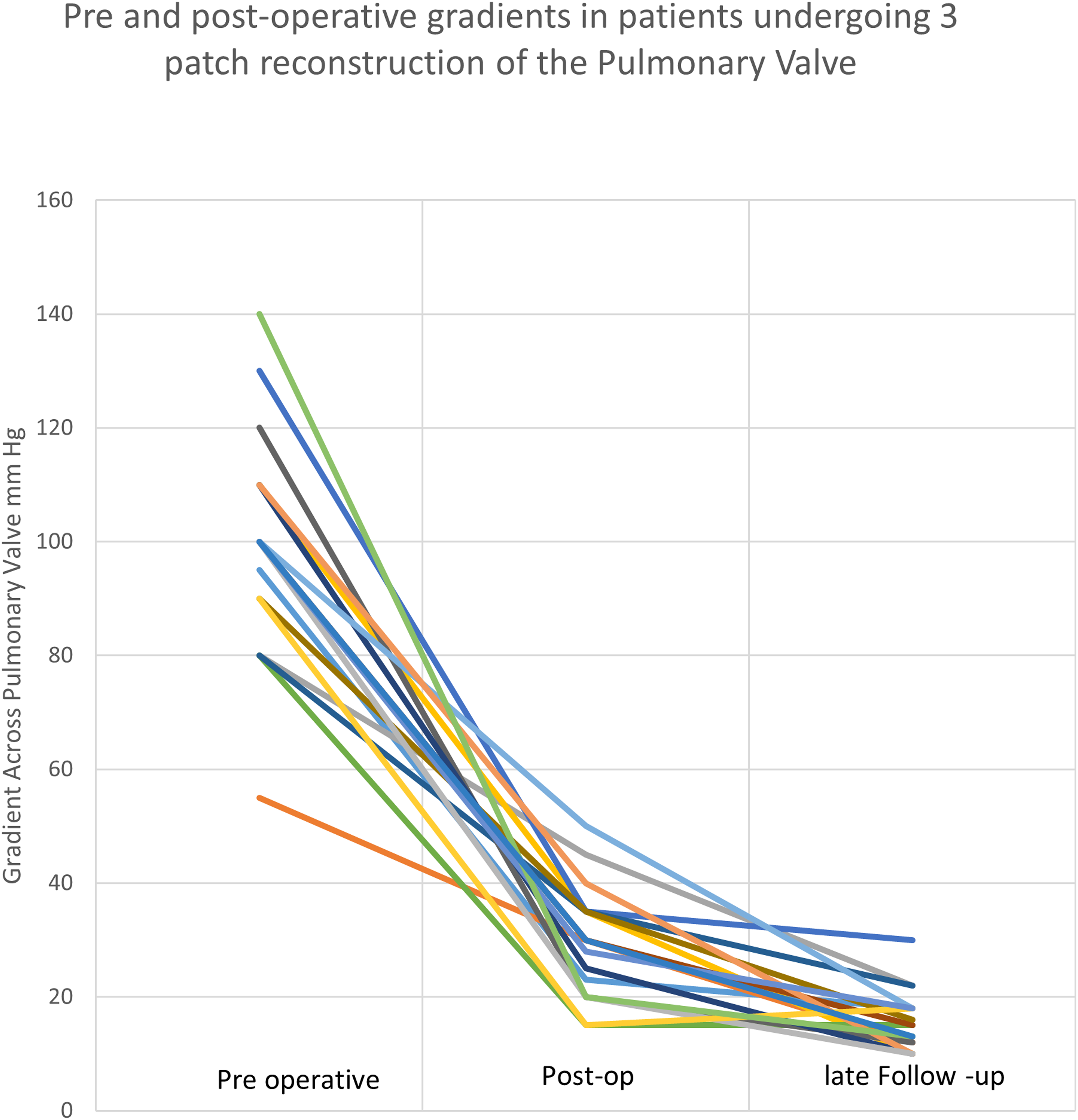
Preoperative and postoperative gradients in patients who underwent three-patch reconstruction of the pulmonary valve. Color version is available online.
Statistical Analysis
Statistical analysis was performed using Graphpad In-stat (GraphPad Prism version 3.0.1 for Windows, GraphPad Software, www.graphpad.com). Patient characteristics, postoperative, and long-term follow-up were displayed using descriptive statistics. The unpaired t test was used to compare continuous variables with normal distribution, and the Mann-Whitney U test was used to compare nonparametric variables. Categorical variables were compared using the χ2 test. Statistical significance was defined as P value <.05.
Discussion
Three-patch reconstruction of a semilunar valve for the treatment of supravalvar aortic stenosis was first described by Brom and has proven to be effective and safe with good long-term results. 11 Surprisingly, its use in the pulmonary circulation has been rarely reported.6,8‐10 Sojak et al reported reconstruction of the pulmonary supravalvular area using three pericardial patches in an eight-month-old baby, 10 Peng et al reported good short-term results in four patients. 6 To our knowledge there are no data regarding the mid-term and long-term results of this reconstructive technique.
For the last ten years, we have been performing three patch reconstruction of the PV in selected cases with a distinct supravalvar narrowing. Supravalvar pulmonary stenosis has a typical angiographic feature of an hourglass deformity (Figure 3) which has been linked to elastin arteriopathy and to Williams syndrome although isolated cases of hourglass pulmonary stenosis have been reported.6,8,9 In our experience, we have found complex pathologies extending below the supravalvar region involving the commissures (fusion) and the valve leaflets (thick and dysplastic leaflets and short free edges). Although the mainstay treatment of isolated PV stenosis has been balloon dilatation of the PV with excellent results, 12 our report demonstrates that transcatheter balloon dilatation of supravalvar pulmonary stenosis has been mostly unsuccessful and in many cases caused PV leaflet tears. Out of our 19 patients, 16 underwent cardiac catheterization and 13 of them had failed balloon dilatation of the supravalvar area and PV. Intraoperative inspection revealed an unaffected supravalvar area and leaflet tears that were subsequently repaired. The supravalvar area was found to be thick and fibrotic and it is not surprising that it was not amenable to balloon dilatation.
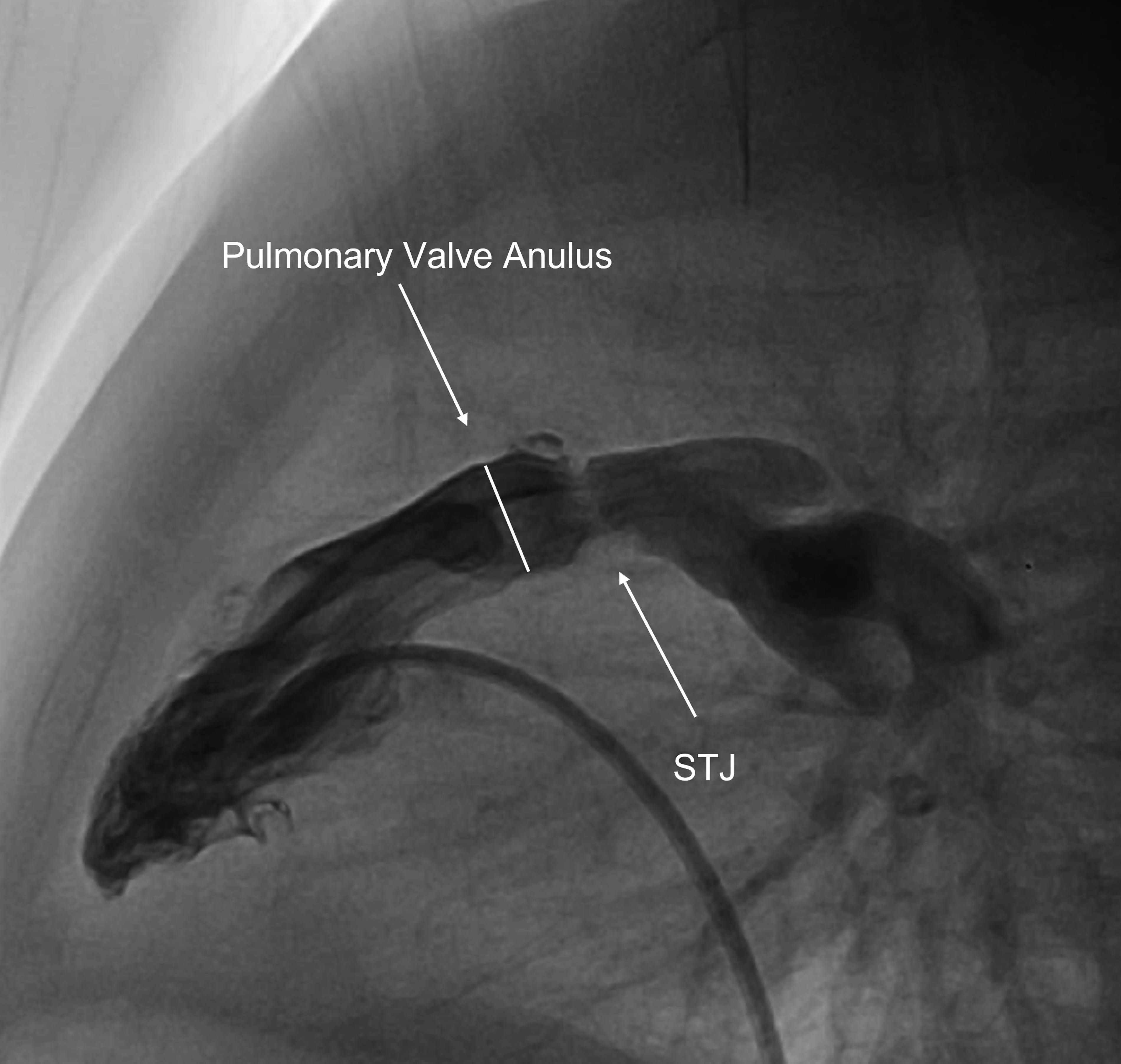
Angiographic appearance of supravalvar hourglass pulmonary stenosis. STJ, sinotubular junction.
Surgically, the limiting factor determining the success of this type of repair is the length of the free leaflet edges of the PV. If these edges are short, they limit the amount of sinus augmentation, and if the augmentation is too big for the leaflet, the free leaflet edge assumes a sail-like appearance causing significant PV regurgitation.
We have been using glutaraldehyde-treated pericardium for supravalvar reconstruction; however, several recent reports have been published in which fresh autologous pericardium has been used for the repair of congenital cardiac defects. Fresh autologous pericardium was found to be durable without evidence of late patch calcification, stiffness, or aneurysmal degeneration and can be an excellent alternative patch material for repair.13,14 Mid- and long-term follow-up have been encouraging as gradients remained low and no reinterventions were needed.
Conclusions
The three-patch technique for the repair of supravalvar pulmonary stenosis is simple, reproducible, and achieves excellent immediate and long-standing relief of the right ventricular outflow tract gradient.
Footnotes
Author note
Institutional Review Board approval was granted 0906-20 RMC.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
