Abstract
Background
The arterial switch operation (ASO) is the preferred surgical procedure used to correct dextro-transposition of the great arteries. A known complication of the ASO is branch pulmonary arteries (PAs) stenosis, which may require reintervention. Our goal is to determine the frequency of reintervention after the ASO and any factors associated with reintervention.
Methods
This was a single center, retrospective study of infants who underwent the ASO from June 6, 2011 to February 21, 2021. The primary outcome was the prevalence of reintervention on the PAs following the ASO.
Results
Sixty-eight infants were analyzed; 9 (13%) patients had 10 reinterventions. The mean age at time of the ASO was 6.52 ± 6.63 days; weight was 3.34 ± 0.57 kg. Those with a reintervention had a longer bypass time (
Conclusion
The prevalence of branch PA reintervention following the ASO in our cohort was 13%. There is an association between longer cardiopulmonary bypass time and reintervention. After reintervention, there was an increase in PA diameter and a decrease in echo gradient.
Keywords
Introduction
The arterial switch operation (ASO) is the most common surgical procedure in the modern era used to correct the congenital heart defect dextro-transposition of the great arteries (d-TGA). 1 Dextro-transposition of the great arteries is one of the most prevalent causes of cyanotic congenital heart disease in newborns, and this condition affects approximately 1100 neonates each year in the United States. 2 The ASO is the primary corrective measure for d-TGA due to its low surgical mortality and good long-term prognosis. 3 However, there are known complications with this procedure. One noted complication is stenosis of the branch pulmonary arteries (PAs). This complication likely results from several factors including the anterior displacement of the branch PAs during the ASO referred to as the LeCompte maneuver. 4
Unfortunately, there is a paucity of data on the prevalence of repeat interventions on the branch PAs after the ASO, and the risk factors that may be associated with the need for PA reintervention. Our goal was to determine how frequently patients require an intervention on a branch PA after the ASO and what factors are associated with a higher risk for reintervention. We aimed to identify risk factors for PA reintervention in the early postoperative period.
Patients and Methods
The Institutional Review Board at the University of Louisville approved this project (# 21.0438, date of approval 06/16/2021), and the requirement for patient consent was waived. This was a single center, retrospective, chart review of all patients who had d-TGA physiology and underwent the ASO as a neonate from June 6, 2011, to February 21, 2021. Patients were excluded if they had other complex intracardiac conditions, but patients with ventricular septal defect (VSD) and atrial septal defect were included. The technique used at our center for the ASO was performed using the LeCompte maneuver for all patients. For the aortic and pulmonary transection, a longer ascending aorta is kept proximally which is then used to construct a long neopulmonary trunk. If deemed necessary, the aorta is shortened by the surgeon. Although this was done in the majority of cases, it was not done routinely. When performing the distal anastomosis with the PA bifurcation, the anastomosis is only extended by incisions to the left and/or right if needed to complete the surgery. The PA reconstruction is typically performed with clamp off, and untreated autologous pericardium is used as a pantaloon patch for this reconstruction. Throughout the ASO a running suture is used, and this suture is either 7-0 or 8-0 prolene. This technique was used throughout the study period. The only change in technique during the study period was switching from 7-0 to 8-0 as the primary suture size.
All data were obtained from available records within the electronic medical record at Norton Children's Hospital. The primary outcome of this study was need for intervention on a branch PA after the ASO. Demographic, preoperative, postoperative, operative, and interventional data were collected. Aortic root dilation was defined as Z-score >2 standard deviations from the norm.
Echo gradient data were retrieved from the first transthoracic echo following the procedure that examined the branch PAs. This study was always done prior to initial discharge after the ASO. Reintervention echo gradient datum was taken from the last echo prior to and the first echo after reintervention. Intracardiac hemodynamics were obtained from the catheterization report at the time of branch PA intervention. These are reported as peak to peak gradients from the branch PA to the main PA in mm Hg. This was measured using continuous wave doppler through the right or left branch PA, respectively. Diameters of the branch PAs were measured at the origin of each branch PA that was measured. Continuous variables were described using mean and SD or median and IQR; categorical variables were described using frequency and percentages. Between-group differences were assessed using Mann-Whitney
Results
Sixty-eight patients were analyzed in this study; 49 (72%) patients were male. In this cohort, the ASO was performed at an average age of 6.5 ± 6.6 days and at an average weight of 3.3 ± 0.6 kg (Table 1). Mean total follow-up was 6.17 ± 2.94 years. Mean follow-up for patients with reintervention was 7.57 ± 2.59, while mean follow-up for patients without reintervention was 5.94 ± 2.78 (
Baseline and Procedural Data.
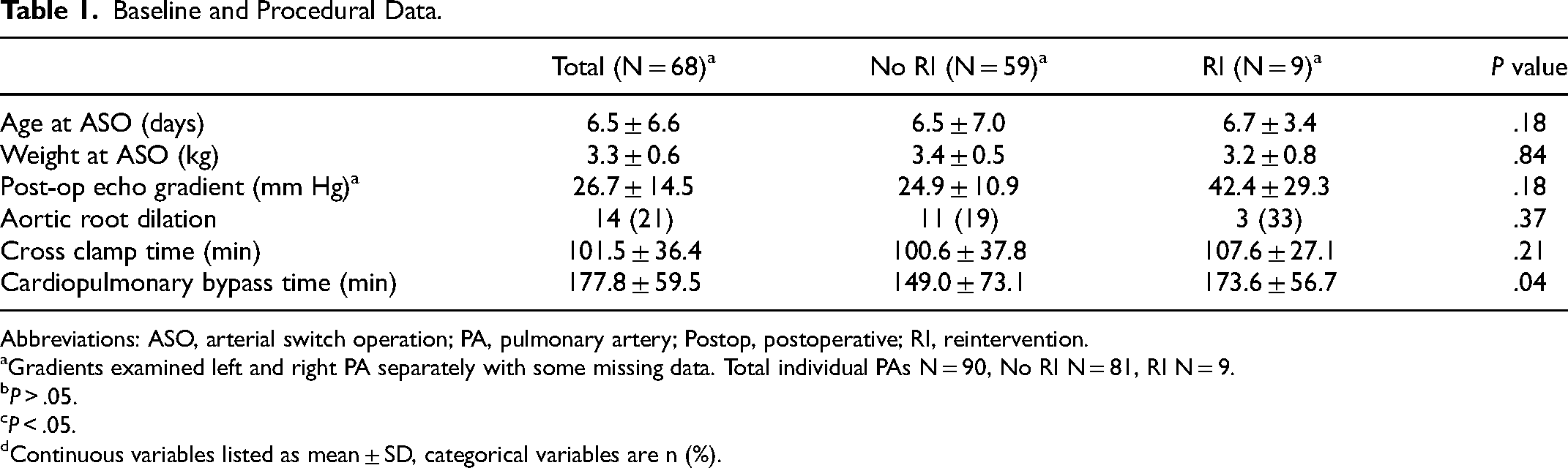
Abbreviations: ASO, arterial switch operation; PA, pulmonary artery; Postop, postoperative; RI, reintervention.
Gradients examined left and right PA separately with some missing data. Total individual PAs N = 90, No RI N = 81, RI N = 9.
Continuous variables listed as mean ± SD, categorical variables are n (%).
Nine (13%) of these patients had 10 reinterventions on 13 branch PAs. The distribution of the reinterventions included four balloon angioplasties, two stent placements, and six surgical procedures (one surgical procedure included angioplasty of two branch PAs). The average age at reintervention was 0.8 ± 0.7 years, the mean time from the ASO to reintervention was 0.8 ± 0.7 years, and the average weight at reintervention was 8.0 ± 4.0 kg. Forty-five percent of reinterventions were on the right PA only, while 11% of reinterventions were on the left PA only, and 44% of the reinterventions included both (Figure 2). There was no increased risk for reintervention on the right versus left PA (
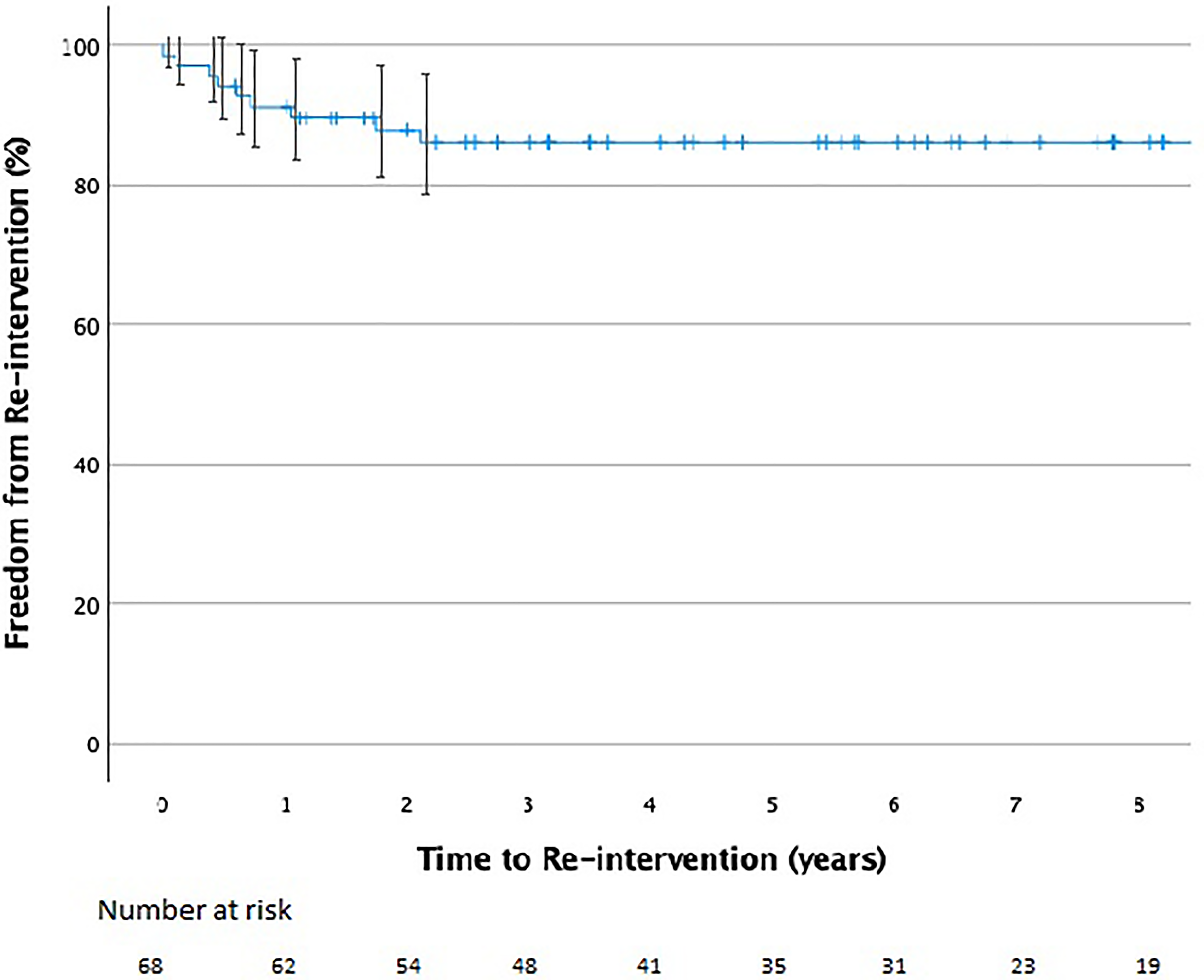
Kaplan-Meier curve showing freedom from reintervention on the branch pulmonary arteries over time after the arterial switch operation.
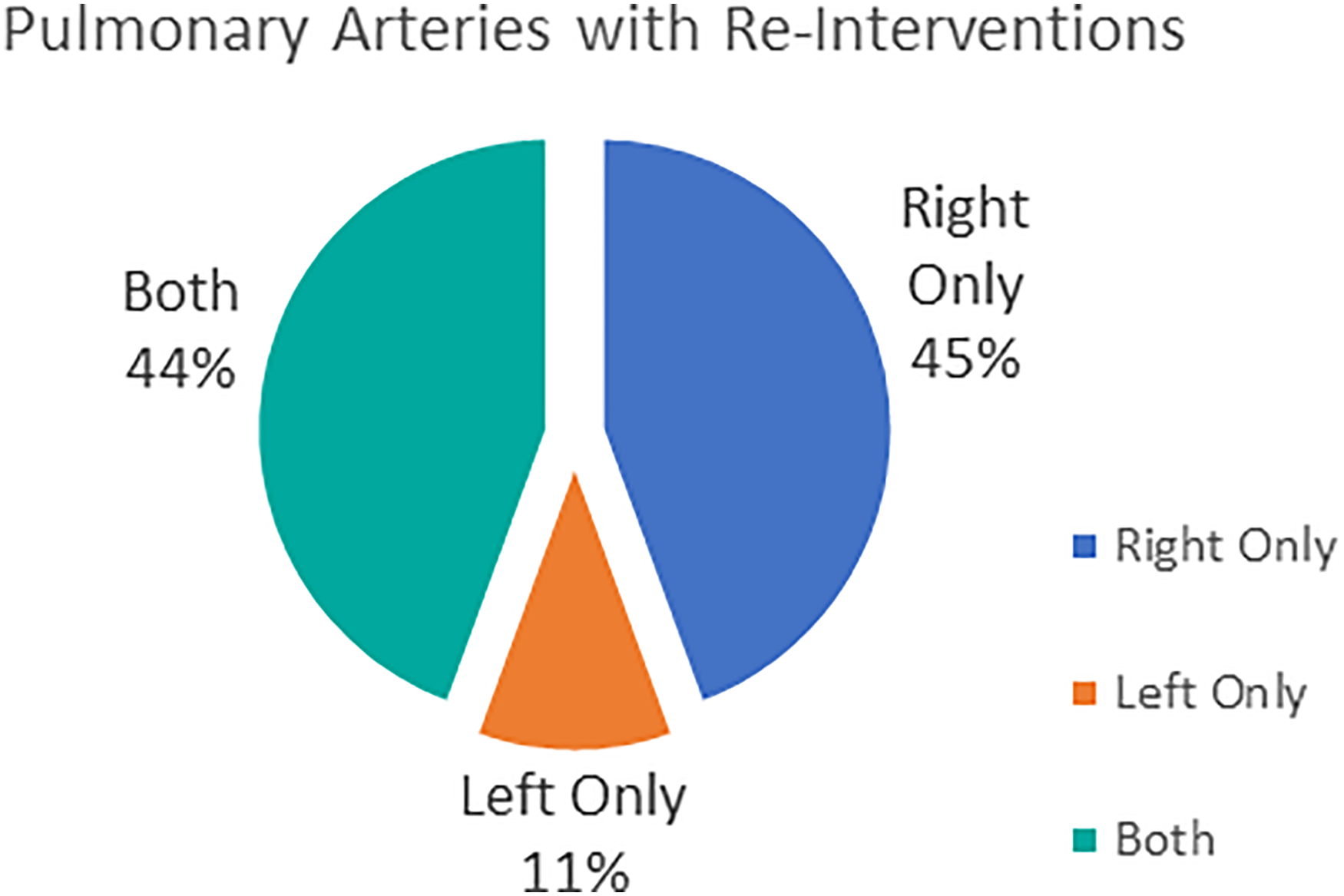
Analysis of which pulmonary arteries required reintervention. There was no statistically significant difference between which pulmonary artery required reintervention.
There was no difference in mean postoperative echo gradient for reintervention compared with those without reintervention (42.4 ± 29.3 mm Hg vs 24.9 ± 10.9 mm Hg,
After reintervention, there was an improvement in the minimal PA diameter with a mean diameter of 3.0 ± 1.3 mm before reintervention compared with 5.1 ± 2.3 mm after reintervention (
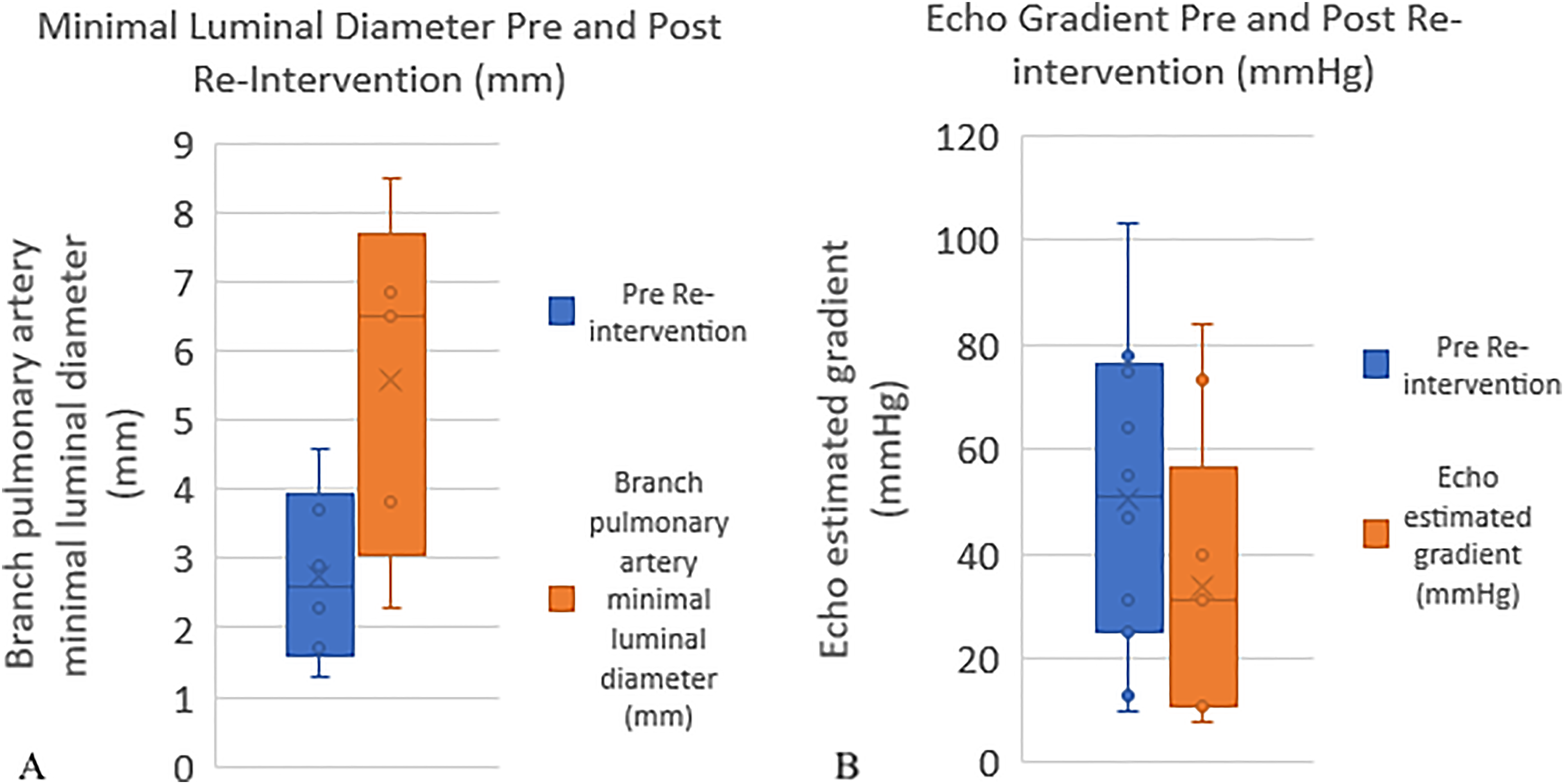
(A) Bar and whisker plot showing improvement in minimal branch pulmonary artery diameter after reintervention. The plot on the left shows minimal branch pulmonary artery diameter prior to intervention, the plot on the right is after intervention. (B) Bar and whisker plot showing improvement in echo gradient across the branch pulmonary artery after reintervention. The plot on the left shows the estimated gradient by echo prior to intervention and the plot on the right is after intervention.
Comment
Reintervention on the branch PAs is common after the ASO with a prevalence of 13% in this cohort after an average follow-up of approximately six years (6.17 ± 2.94 years). Most of these reinterventions occurred in the first year after the ASO. This falls in range with previously published reports of reoperation after the ASO. A large single center study by Luo et al found a 10.7% incidence of early reintervention (during the intensive care stay), with a 9.4% late intervention; nearly one-third of patients who required early intervention needed a subsequent intervention. 5 A review of surgical outcomes showed freedom from reoperation ranged from 79% to 97% ten years after the ASO. 6 This included all reoperations and was not limited to branch PAs, but only included surgical reoperation. Our study examined reintervention rates including surgical and transcatheter interventions.
Our data demonstrate that an increased cardiopulmonary bypass time during the ASO is associated with more frequent PA reintervention. This may suggest that a patient with a lengthier and likely more complex ASO procedure is at an increased risk of needing one or more reinterventions on the branch PAs in the future. There was not a significant difference in reintervention rate if VSD closure was also needed, so the increased length for septal defect closure alone did not account for longer bypass times. Regardless, it is documented that prolonged CPB time (>180 min) is significantly associated with increased mortality as well as higher risks of complications. 7 Along with prolonged CPB time, it has been shown that more complex anatomy—including Taussig-Bing anomaly—is a risk factor for any reoperation. 8
Studies show that other preoperative characteristics such as timing of the ASO may affect the outcomes and mortality of patients following the ASO. 9 This led us to hypothesize that age at the ASO, weight at the ASO, or other preoperative factors may be associated with reintervention, but the data did not find any associations with these preoperative factors. We also hypothesized that aortic root dilation may be associated with reintervention due to the proximity of branch PAs to the aortic root. An enlarged aortic root may impinge on the PAs and cause stenosis, leading to the need for reintervention on these arteries. Aortic root dilation has been well-demonstrated as a risk factor for PA stenosis in patients with tetralogy of Fallot (TOF) as well as hypoplastic left heart syndrome. 10 In fact, aortic dilation is highly correlated with the severity of right ventricular outflow tract stenosis in TOF patients. 11 However, in our cohort, there was no statistical association between aortic root dilation or any other preoperative and operative factors with the need for reintervention. This might be a result of small sample size and lack of power of this study to determine the association. This may also be related to the timing of this study. Follow-up was only six years (6.17 ± 2.94 years), and significant aortic root dilation may not have had enough time to develop.
We did not demonstrate an association between higher postoperative echo gradient following the ASO and increased risk for reintervention. This indicates that either echo gradient may not always be predictive of the need for reintervention or the stenosis develops later after the initial postoperative period. Echocardiography is limited in its uses and additional imaging via computed tomography (CT) and magnetic resonance imaging (MRI) may be needed to properly evaluate the PAs. 12 There are instances in which MRI has diagnosed pulmonary stenosis which was not detected during echocardiography. 13 Magnetic resonance imaging is useful in diagnosing pulmonary stenosis due to the multiplanar analysis of vessels as well as hemodynamics of pulmonary blood flow that MRI allows, and MRI does not require contrast agents or exposure to radiation. 13 Both MRI and CT are often used in addition to echo for evaluation of branch PA stenosis following the ASO. Computed tomography can also be useful in monitoring the morphology of the PAs after stent procedures. 14 However, neither of these advanced imaging methods were measured frequently in this study, preventing proper evaluation and analysis of these tools in our cohort. The determination to refer for advanced imaging is physician-specific and may have been restricted or biased based on primary physician preference for these patients. The previously mentioned study by Luo et al looked at the effect of preoperative anatomic factors on reintervention. 5 They found that smaller preoperative branch PAs predicted late branch PA reintervention. These data can be collected using transesophageal echocardiogram as well as MRI and CT. Thus, as wider availability and quicker scanning times are now present, preoperative evaluation with advanced imaging modalities may be able to determine morphologies that are risk factors for future reoperation.
It is important to note that our research was limited by the retrospective nature of this study as well as the study being limited to a single institution. Specific details about surgical technique were limited due to the retrospective nature of this study, surgeon-specific preferences, and varying surgeons performing the ASO over time. Single institution also biases the interventions to be performed based on our institution criteria which may not be universal. The data available in the institutional electronic medical record were, at times, incomplete thus leading to missing variables. We also have a limited follow-up time, averaging six years. While most interventions in this cohort were performed within the first year, there may be a bimodal distribution of reintervention that was beyond the follow-up and took place at a later point than the range of our studied data. It is documented that the first PA reintervention may occur as late as 25 years after the ASO, with a patient's first reintervention occurring at various intervals ranging from 5, 10, or 15 years after the ASO. 6 We may be missing patients who will undergo intervention as part of this later group of reinterventions.
In conclusion, there was a 13% prevalence of reintervention on branch PAs in our cohort with mean follow-up of a little over six years. Our data also show that the reinterventions are indeed successful in lessening the stenosis of PAs; this is indicated by both a statistically significant increase in minimal PA diameter and a decrease in echo gradient following the reintervention. Despite this, future research is needed using information from a larger database to examine if similar prevalence holds with a larger patient group and to examine if there are associated risk factors within a larger patient population. Advanced imaging may be able to determine morphologies at increased risk for reintervention. Lastly, a longer follow-up period will also be beneficial to determine if intervention is needed into adulthood.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
