Abstract
We describe the case of a newborn male with a large fistula from the left main coronary artery to the right ventricle. This case illustrates a rare congenital coronary artery fistula and its successful surgical management in the neonatal period.
Introduction
Congenital coronary artery fistulas (CAFs) are anomalous coronary artery communications with a heart chamber or pulmonary vessel. There are no specific pediatric guidelines for CAF management. 1 We describe an asymptomatic newborn patient with a large and rare left main coronary artery (LMCA) to right ventricle (RV) fistula. The fistula was diagnosed with transthoracic echocardiogram (TTE) after a murmur was appreciated.
Case Report
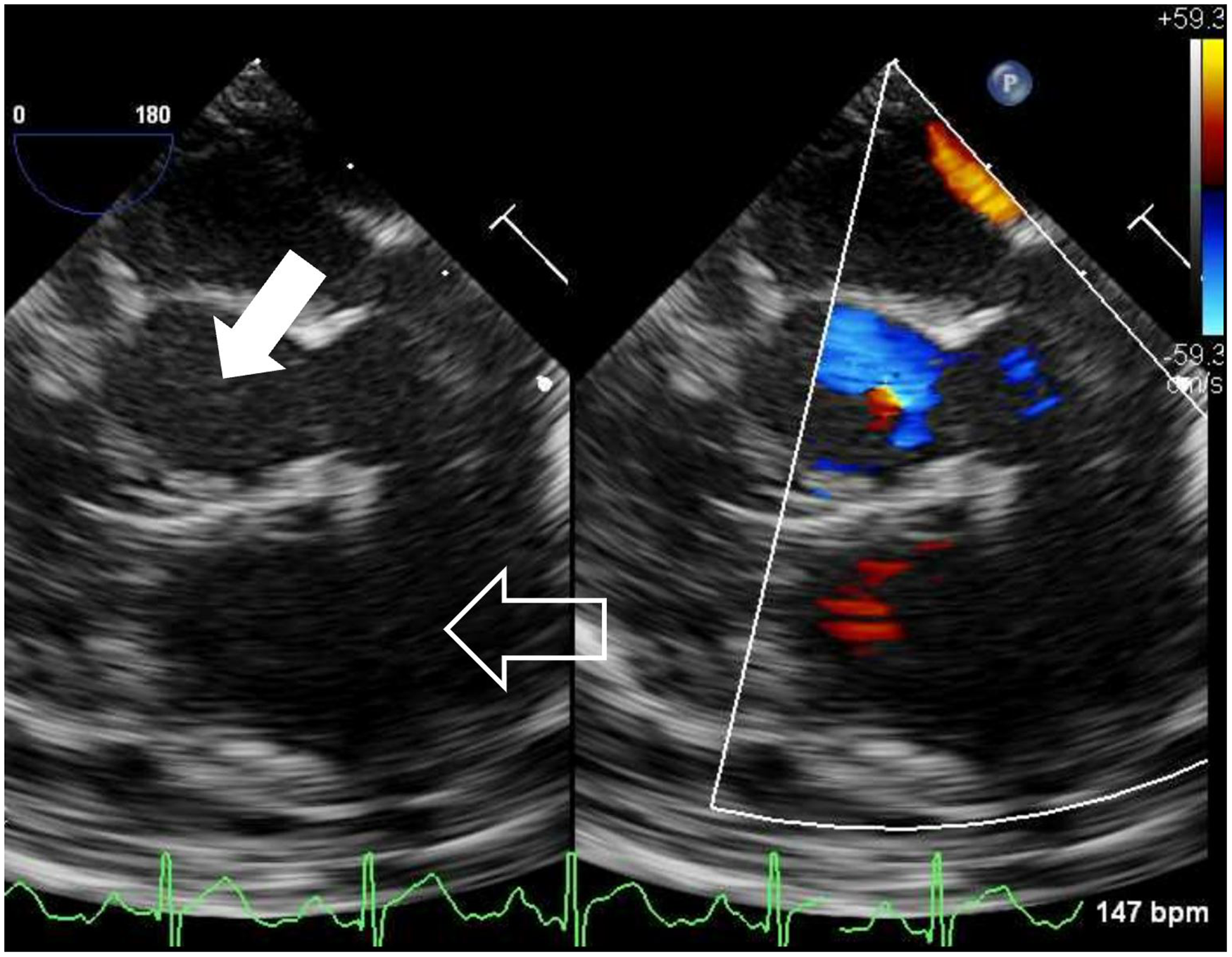
A four-day-old male, weighing 3.7 kg, was transferred from another hospital after a CAF was discovered by TTE. The patient was born at term following an uncomplicated pregnancy. The patient was on per-oral feeding with normal sinus rhythm, Apgar score 9/9, and a heart murmur. Transthoracic echocardiogram showed a large LMCA to RV fistula (Figure 1). The fistula coursed anteriorly and leftward, connecting with the RV inferiorly and leftwards of the pulmonary valve. The LMCA was dilated at 5 mm. The fistula was 8 mm at its widest, and 6 mm at RV entry. Fistula blood flow was pressure restrictive, with a peak coronary-to-right-ventricle gradient of 50 mm Hg. While the aortic valve was normal, there was reverse diastolic blood flow throughout the aortic arch and proximal descending aorta suggesting a significant run-off lesion. The RV was normal in size and function, with no evidence of pulmonary hypertension. A small patent foramen ovale was present with left to right shunting.
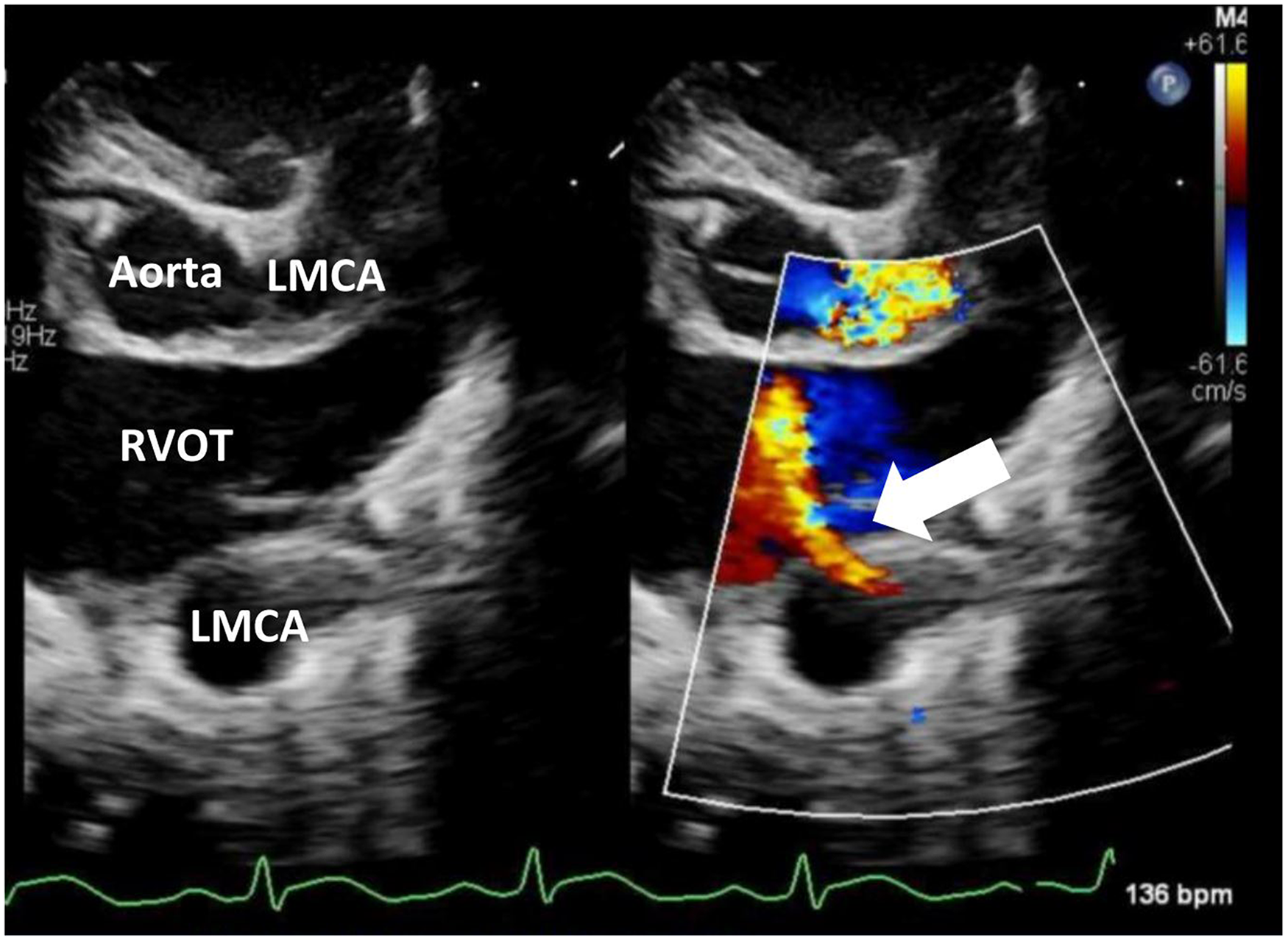
Transthoracic echocardiogram (TTE) showing the coronary artery fistula (CAF) draining into the right ventricular outflow tract (RVOT) (arrow). LMCA, left main coronary artery.
Given the risk of cardiac dysfunction due to coronary steal from the fistula, the baby underwent surgery on day of life 7. After a median sternotomy, a portion of anterior pericardium was resected and treated with glutaraldehyde to serve as patch material. Surface inspection of the heart showed no sequela of a fistula. A thrill was palpable over the right ventricular outflow tract (RVOT). Following heparin administration, the aorta and right atrium were cannulated, and cardiopulmonary bypass was initiated. An additional cannula was placed in the inferior vena cava. The patient was cooled to 28 °C, and a left atrium vent was placed through the superior right pulmonary vein. Both branch pulmonary arteries were snared, aortic cross clamp applied, and cardioplegia administered in an antegrade fashion. Prompt arrest of the heart and visualization of full coronaries during cardioplegia administration suggested sufficient cardioplegia delivery. To further ensure myocardial protection, an additional dose of cardioplegia was administered along with topical ice. The RVOT area was opened and exposed with stay sutures. The fistula was identified near the pulmonary valve (Figure 2). The fistula opening was closed with an autologous pericardial patch utilizing running sutures (Figure 3). Antegrade cardioplegia was administered to verify the complete closure of the fistula opening and provide additional myocardial protection. The RVOT area was then closed with another autologous pericardial patch (Figure 4). The patient was warmed and weaned off bypass. We did not transect the main pulmonary artery to patch the LMCA given the potentially increased thrombus risk associated with LMCA patching. Aortic cross clamp time was 45 min, and bypass time was 70 min. Postoperative transesophageal echocardiography showed good cardiac function, complete fistula closure, and no reverse diastolic aortic blood flow (Figure 5).
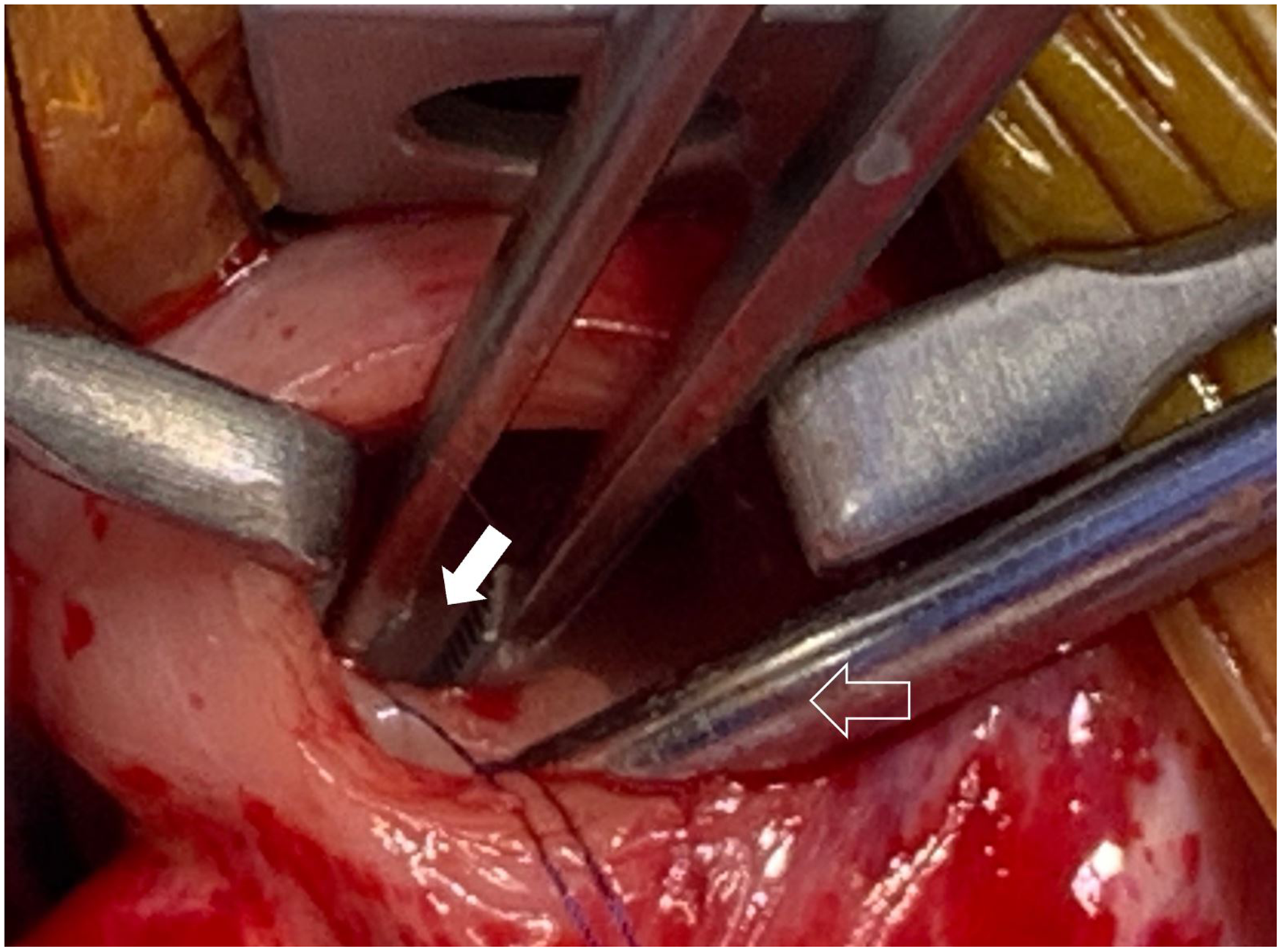
Intraoperative photograph showing the fistula opening (full arrow) and sucker in the pulmonary valve (outlined arrow).
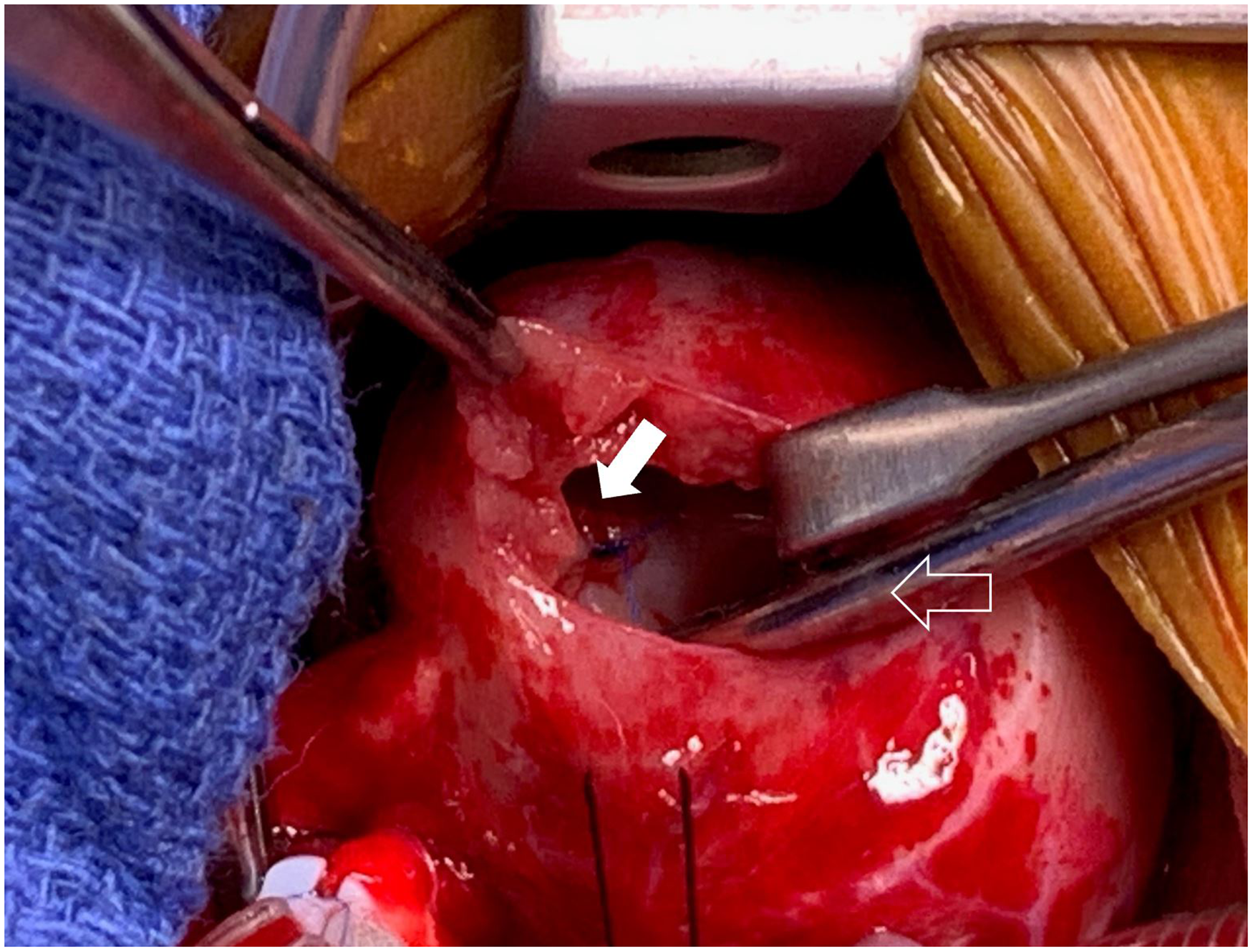
Pericardial patch closure of the fistula opening (full arrow), sucker across the pulmonary valve (outlined arrow).
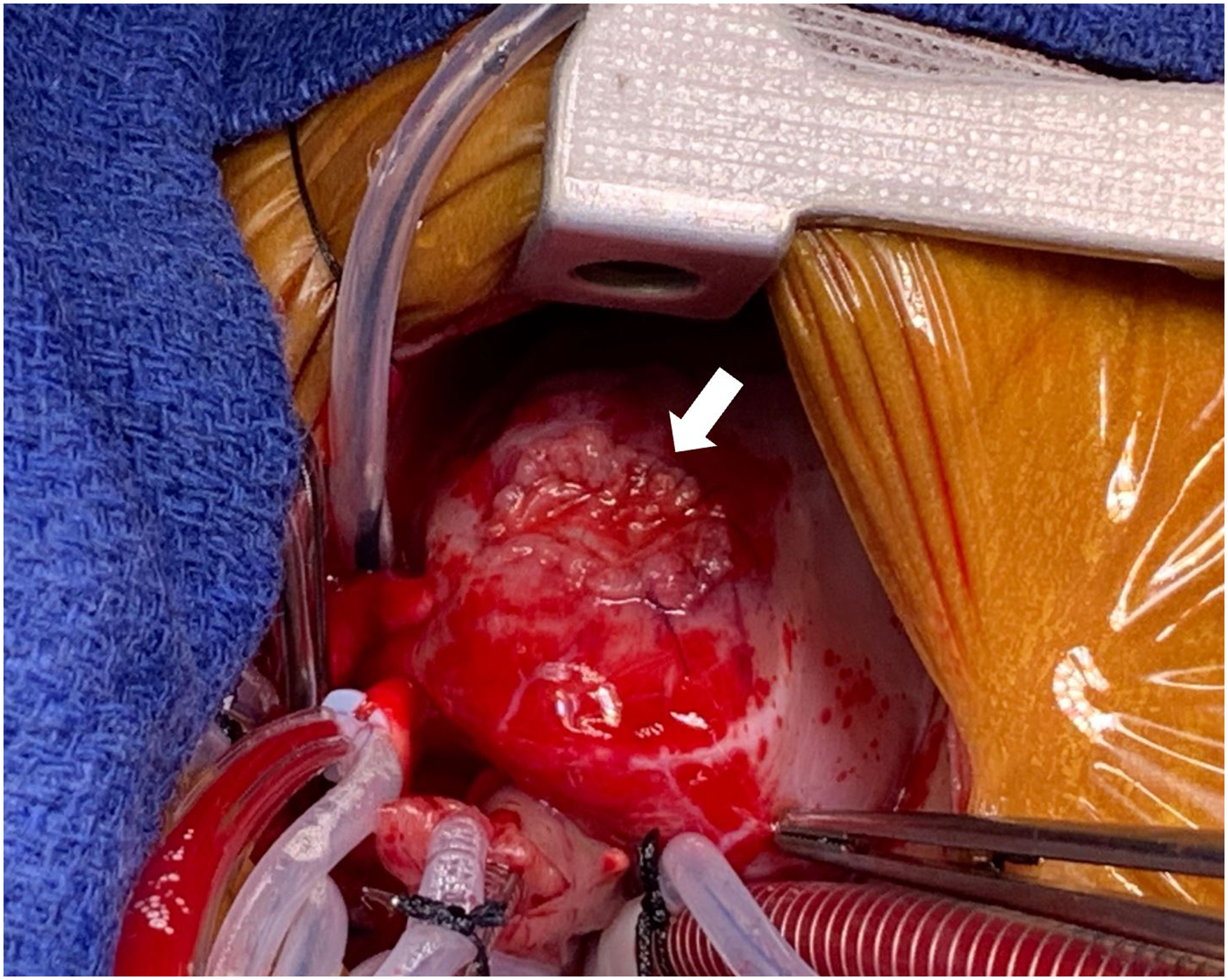
Autologous patch closure of the right ventricular outflow tract (RVOT) (arrow).
Postoperative transesophageal echocardiography (TEE) showing no evidence of residual fistula. Aortic valve (full arrow) and right ventricular outflow tract (RVOT) (outlined arrow).
The patient was extubated on postoperative day 2 and maintained on prophylactic heparin (15 U/kg/h) until postoperative day 4, when he was transitioned to aspirin. The patient was discharged home on postoperative day 6. Predischarge TTE showed a small thrombus in the cul-de-sac (between the coronary artery and the patch closure). At one-month follow-up, the patient was doing well with no reported complications and reduced thrombus size (Figure 6). At three-month follow-up, there was no evidence of residual thrombus.
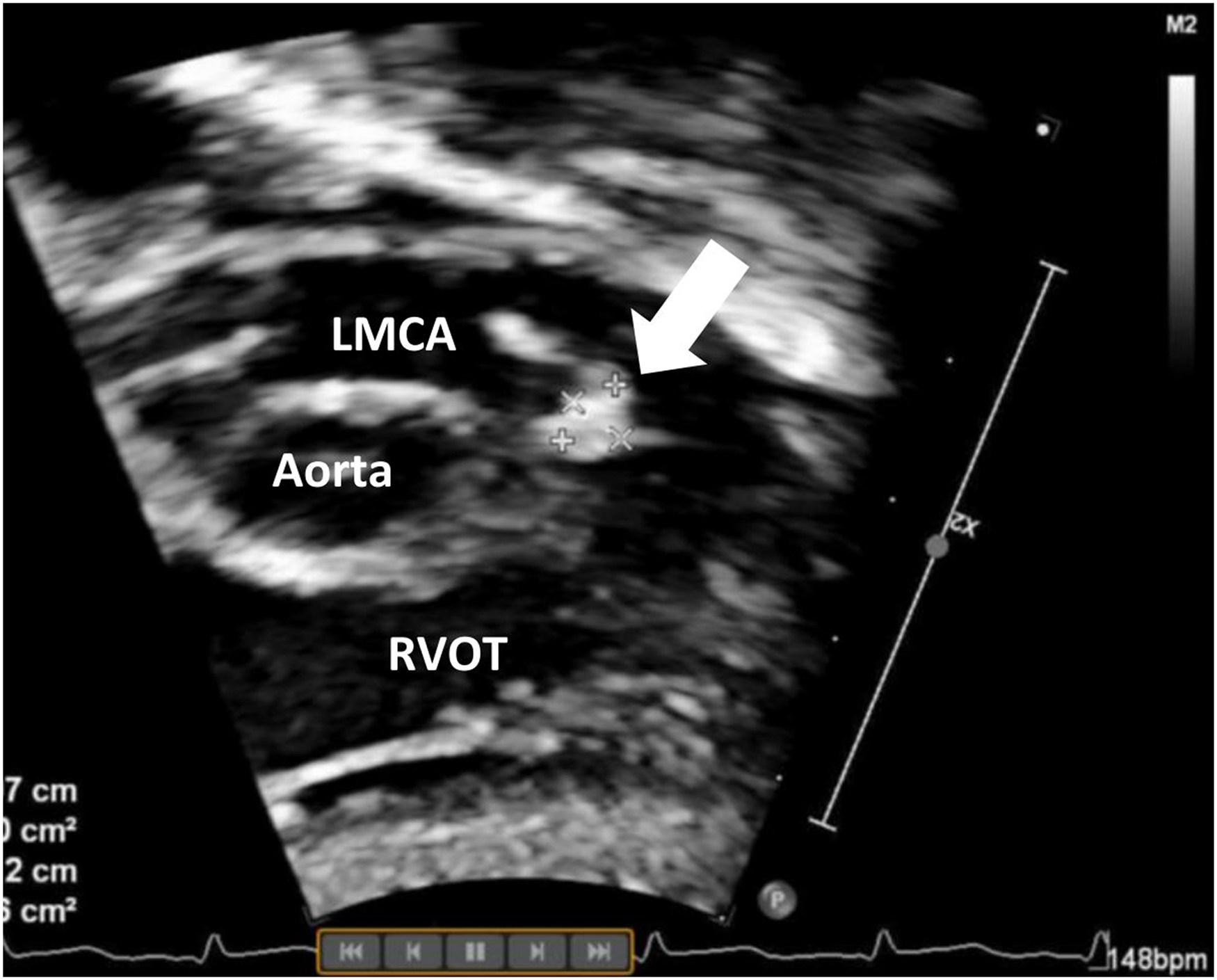
One-month follow-up transthoracic echocardiogram (TTE) showing a 4- by 5-mm cul-de-sac thrombus (arrow). LMCA, left main coronary artery; RVOT, right ventricular outflow tract.
Comment
This case describes a rare CAF between the LMCA and RVOT. In a study of 46 patients with congenital CAFs, none had an LMCA to RV fistula. 2 The first clinical sign of the fistula was a murmur, a clinical finding reported in >95% of cases.1,3 In our case, color Doppler TTE showed a clear flow pattern and was sufficient to diagnose the CAF and plan the operation. However, cross-sectional imaging, including computed tomography (CT) angiogram and cardiac catheterization, should be liberally utilized if the anatomy remains unclear, including if multiple fistula openings are suspected. Kuwahara et al describe a newborn with a right CAF with multiple openings that was surgically treated. 4 Given the high quality of our TTE imaging, it was unnecessary to expose the patient to invasive catheterization or CT radiation. A study of 63 patients with CAFs found TTE to be as accurate as coronary arteriography in diagnosing and describing CAFs. 5 A neonatal CAF imaging case study found echocardiogram to be as accurate as CT scans. 6
The optimal timing of asymptomatic CAF repair is debated.1,3,7 In this asymptomatic case, diastolic flow reversal in the aorta suggested significant fistula run-off. We hypothesized that if left untreated, the fistula flow into the RV would increase as the patient's pulmonary vascular resistance dropped during early life. The increased fistula flow could result in myocardial dysfunction from coronary steal similar to the pathophysiology seen in patients with unrepaired anomalous left coronary artery from the pulmonary artery. Given this risk, we repaired the CAF in the neonatal period. In a study of patients prenatally diagnosed with CAF, two of three patients with to-and-fro aortic flow developed heart failure (HF) as neonates. 7 Another study (N = 48) found an association between HF and large fistula size. 3
Coronary artery fistulas can be treated through transcatheter or surgical closure. A CAF closure study (63 transcatheter, median age four years; 58 surgical, median age five years) found significantly higher rates of myocardial ischemia (10% vs 0%), residual shunts (17% vs 4%), and new moderate-to-severe valve regurgitations (12% vs 0%) among the transcatheter cohort. 1 Another study (N = 48, median age 58-days) found no differences between transcatheter and surgical outcomes. 3 In our case, surgery was chosen as the fistula's large size prevented transcatheter closure.
Expecting a thrombus to form in the cul-de-sac, we prophylactically administered heparin postoperatively and transitioned to aspirin on postoperative day 4. Coronary thrombosis and myocardial infarction have been reported following CAF closure, though more commonly in older patients.2,3 We recommend utilizing prophylactic postoperative heparin when there is a known thrombus risk, as postoperative heparin may improve remodeling. 3 In our case, at three-month follow-up, there was no evidence of thrombus. We will continue the aspirin for another three months with serial echocardiograms. Thrombus progression would warrant anticoagulation with warfarin for ∼3 to 6 months (target international normalized ratio of 2-3) with monthly TTE to reassess the treatment plan.
We describe a rare LMCA to RV fistula that was successfully treated with surgery in an asymptomatic neonate. Additional studies are required to develop guidelines for asymptomatic neonates with CAFs.
Footnotes
Authors’ Statement
Parental consent was provided for the publication of this case report.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
