Abstract
Double-chambered left ventricle (DCLV) is a rare congenital cardiac lesion. It is usually an incidental finding in children and young adults. Diagnosis during fetal life is exceedingly rare. We report a case of DCLV diagnosed in the prenatal period associated with a ventricular septal defect (VSD). Transthoracic echocardiographic and magnetic resonance imaging confirmed the prenatal findings. At seven-month follow-up, the baby was asymptomatic. Transthoracic echocardiographic check-up revealed spontaneous closure of the VSD and stable aspect of the main left ventricle and the accessory chamber.
Introduction
Double-chambered left ventricle (DCLV) is an extremely rare congenital cardiac lesion. 1 Diagnostic criteria are not well-established. Double-chambered left ventricle is usually an incidental finding in children and young adults. Diagnosis during fetal life is exceedingly rare. Clear data about prognosis, potential complications, and management are lacking. We report a case of DCLV diagnosed in the prenatal period and followed up postnatally.
Case Presentation
A 29-year-old primigest woman was referred to our department at 25 weeks gestation for “extra cardiac mass” in a female fetus. There was no consanguinity. The medical history was unremarkable. The pregnancy was reported to be uncomplicated. The fetal echocardiogram (Figure 1) revealed the presence of a morphologically left ventricle (LV) partitioned by a muscular shelf into two side-by-side chambers. Atrioventricular and ventriculo-arterial connections were normal. The main LV chamber was connected directly with the mitral and aortic valves. The accessory chamber was large, had reached the apex, measured 26 by 16 mm, and connected to the lateral wall of the main chamber through a large opening of 9 mm. The lateral wall of the accessory chamber was confined within the external contours of the LV. It had a similar appearance and thickness as the rest of the LV, and it contracted during systole and expanded during diastole synchronously with the rest of the ventricles characterizing a DCLV. An apical ventricular septal defect (VSD) of 5 mm was also visualized. Flow across the atrioventricular and semilunar valves was normal with no regurgitation. There were no signs of fetal heart failure or cardiac effusion. The fetal cardiac rhythm was normal. At follow-up, the pregnancy remained uncomplicated and it was carried to term.
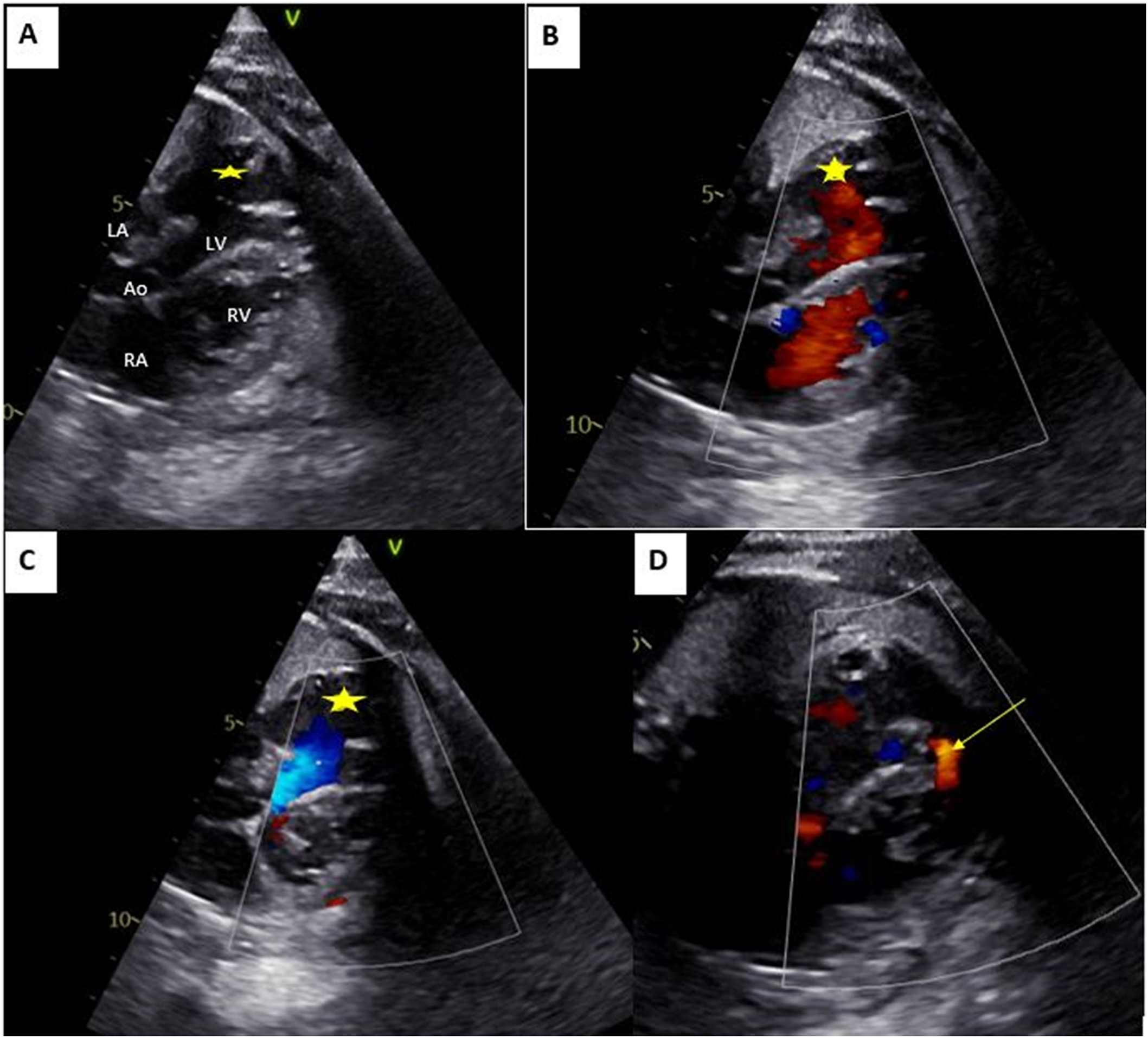
Fetal echocardiography findings. Large accessory chamber (star) having walls that are similar in appearance and thickness to the rest of the LV, connecting to the lateral wall of the LV with a large collar (A). Color Doppler visualizing flow during diastole (open mitral valve and closed aortic valve) from the LV to the accessory chamber (B), and the flow during systole from the accessory chamber to the LV (C). Flow across the apical ventricular septal defect is demonstrated by the yellow arrow (D). Ao: aorta; LA: Left atrium; LV: Left ventricle; RA: right atrium; RV: right ventricle.
At birth, the baby was asymptomatic. A systolic heart murmur was identified on cardiac auscultation. Electrocardiogram showed a regular sinus rhythm. Chest x-ray showed cardiomegaly. Transthoracic echocardiogram (TTE) confirmed the prenatal findings. The apical VSD was restrictive. The LV was mildly dilated (+3,4 z-score) with an ejection fraction of 55% using the Teichholz method, and of 48% using the biplane Simpson method. The right ventricle had normal size and function. The accessory chamber remained large connecting to the lateral wall of the main chamber through a large opening. The wall of the accessory chamber was confined within the lateral wall of the LV. It was thick and the myocardium was well visualized with synchronous contractility with the rest of the ventricles. No thrombus was visualized. Coronary arteries were of normal origin. Color Doppler showed bidirectional flow across the two LV chambers, a mosaic flow across the VSD, and mild mitral regurgitation. Figure 2 illustrates the TTE findings. In order to better characterize this additional chamber, cardiac magnetic resonance imaging (MRI) was performed confirming the three-layered wall with synchronized contraction and the large connection to the main chamber. LV ejection fraction was 40%. The indexed LV and accessory chamber volumes on cardiac MRI were 66 mL/m² SC and 30 mL/m² SC, respectively. Late gadolinium enhancement was not studied (Figure 3).
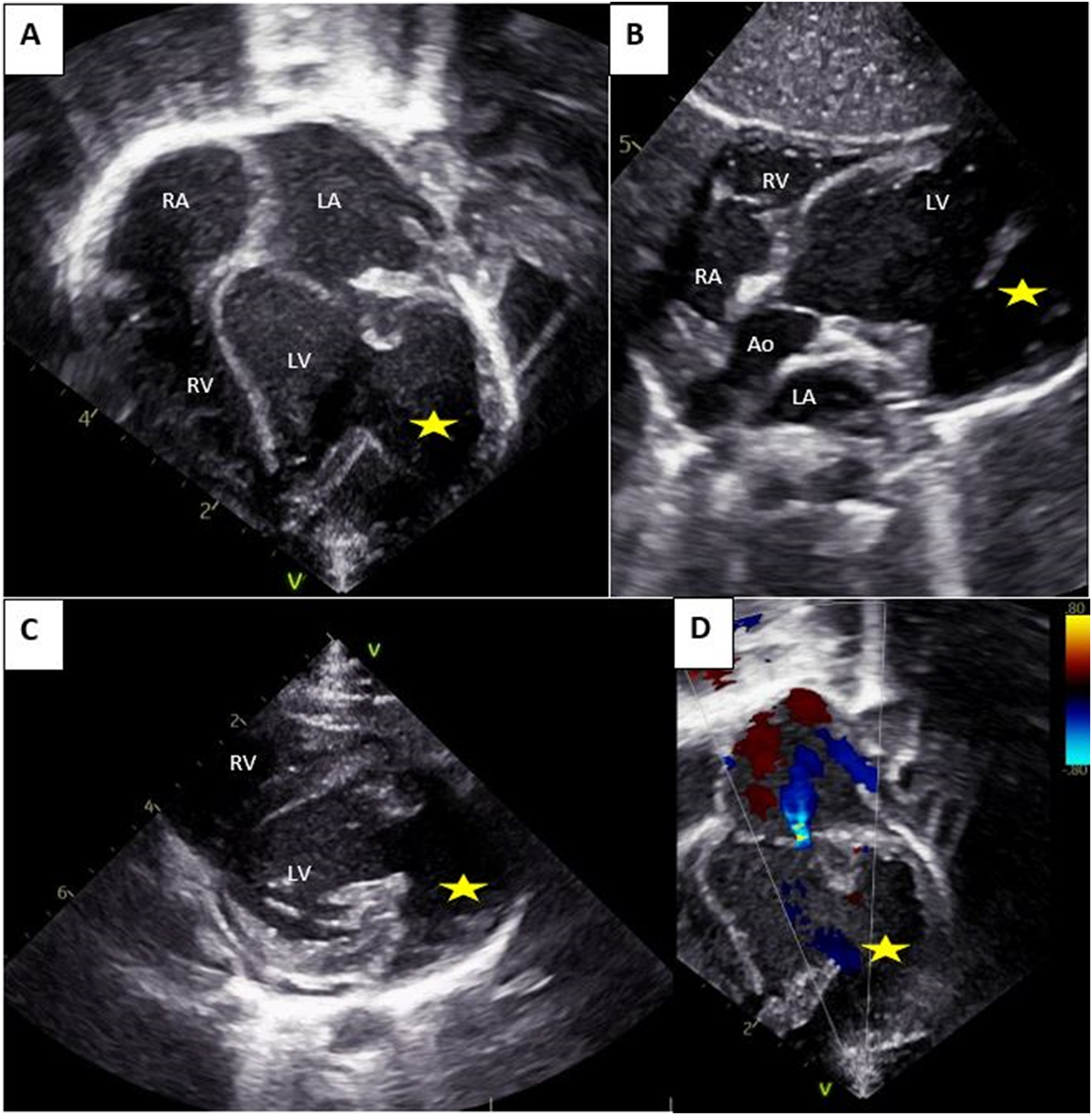
Transthoracic echocardiographic findings. Illustrating the large accessory chamber (star) with three cardiac layers connecting to the lateral wall of the LV via a large collar in the apical 4 chamber view (A), subcostal view (B) and parasternal short axis view (C). Color Doppler showing mild mitral regurgitation. Ao: aorta; LA: Left atrium; LV: Left ventricular; RA: lustrating the large accessory chamber ( ) with three cardiac layers connecting to the lateral wall of the LV via a large collar in the apical 4 chamber view (A), subcostal view (B) and parasternal short axis view (C). Color Doppler showing mild mitral regurgitation. Ao: aorta; LA: Left atrium; LV: Left ventricle; RA: right atrium; RV: right ventricle.
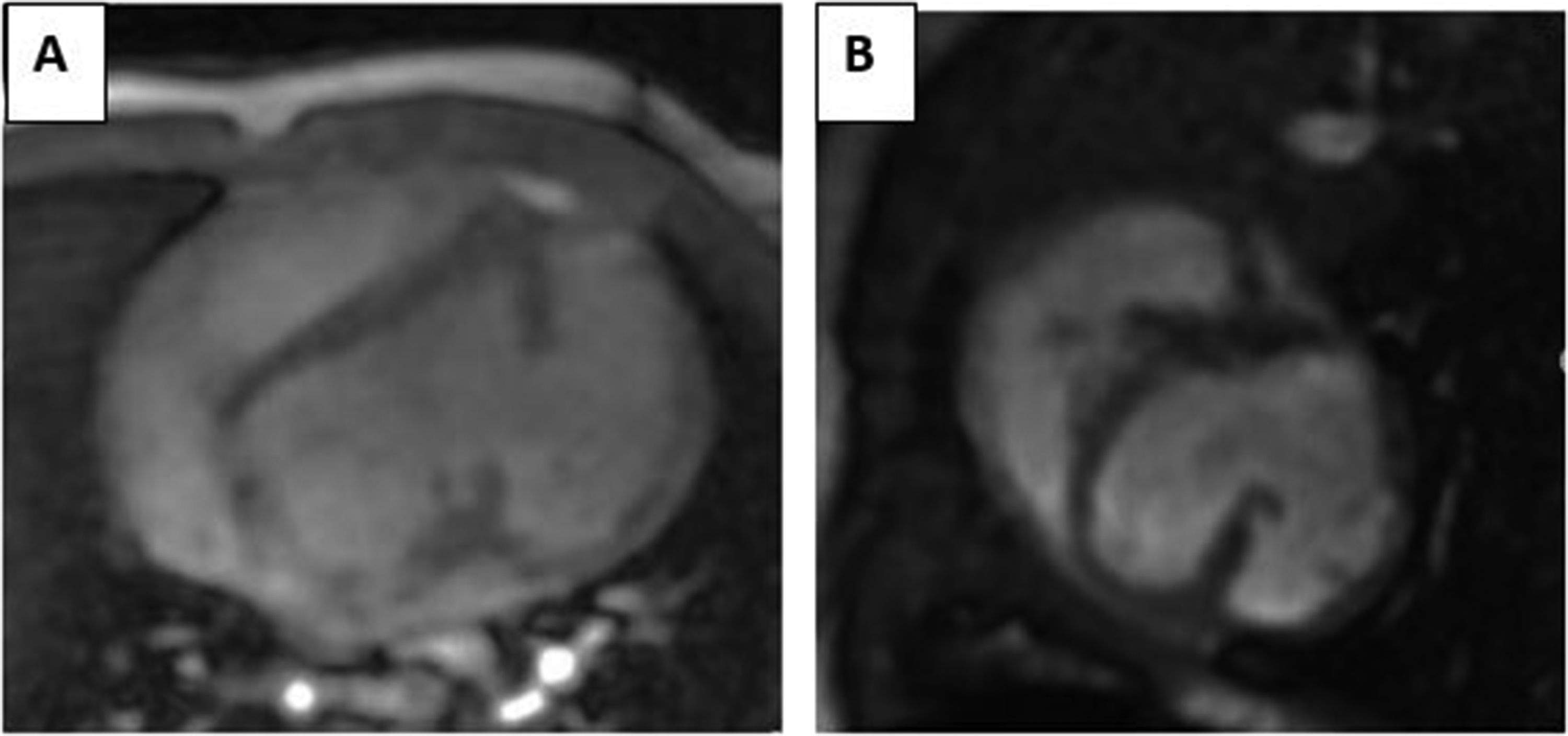
Cardiac magnetic resonance imaging. MRI showing the accessory chamber in four chamber view (A) and short-axis view (B).
At seven-month follow-up, the patient was asymptomatic. The physical examination was normal. A 24 Holter monitor revealed permanent sinus rhythm with no abnormalities. The TTE check-up revealed the spontaneous closure of the VSD. The LV was dilated (+3,5 z-score) with an ejection fraction of 45% (biplane Simpson method). The size of the accessory chamber remained stable and contracted synchronously. The mitral regurgitation remained mild. After consulting the heart team, the diagnosis of DCLV was confirmed, and the decision to regular follow-up was made taking into consideration the parents’ opinions.
Discussion
Double-chambered left ventricle is an extremely rare congenital cardiac lesion. 1 It is generally defined by the presence of a muscle bundle or septum dividing the LV into a main chamber connecting with the mitral and the aortic valves and an accessory chamber. Double-chambered left ventricle may be misdiagnosed as a lateral wall LV aneurysm. The presence of normal external contours of the LV with a thick three-layered wall and synchronized wall motion are the main features distinguishing the DCLV. According to the arrangement of the two parallel chambers, DCLV was categorized into two types: Type A with superior–inferior arrangement which may be associated with obstruction in the middle of the LV. This type was more frequently reported in the literature. 1 – 3 Type B consists of a left-right arrangement with the accessory chamber connecting to the LV at the lateral wall via a large opening, as it is the case in our patient. In a retrospective clinicopathological study, only two of nine cases with DCLV had a large communication between the LV chambers. 4 Associated cardiac lesions were absent in the Zhang et al series 1 and present in all cases with DCLV in the Kumar et al study. 4 A VSD with spontaneous closure was described in our patient.
The clinical manifestations of the DCLV were not specific. The diagnosis in the fetus, like the case of our patient, was very exceptional. Given the extreme rarity of DCLV, making the diagnosis prenatally requires a high index of suspicion. The detection of an additional cardiac chamber on fetal echocardiography should prompt the examiner to study the wall thickness of this chamber and its contraction. Having walls that are similar in appearance and thickness to the rest of the LV is the first distinctive feature. Synchronous contraction is the second main feature. Synchronous contraction of the accessory chamber during fetal life is easy to study using color Doppler: in systole, the accessory chamber contracts with the rest of the ventricle directing flow to the main LV, while it expands during diastole directing flow to the accessory chamber. To our knowledge, only two cases of DCLV diagnosed in the fetus and followed up after birth were reported. 5 , 6 The first case was a type A DCLV with no complications followed-up for one and a half years after birth. 5 The second case was a type B DCLV which was complicated in the first months with LV dilation and dysfunction. 6 Our patient presented a mild LV dilation with a stable aspect and function during the first seven months.
Transthoracic echocardiography is the first imaging modality for the diagnosis. The echocardiographic findings were consistent with the diagnosis of DCLV in our patient. Bao et al illustrated its efficiency in establishing a precise diagnosis. 3 Magnetic resonance imaging allows a better characterization of the myocardial tissue. The absence of late gadolinium enhancement is in favor of the DCLV. Nonetheless, histopathology is sometimes required in making the correct diagnosis.
The extreme rarity of DCLV explains the lack of prognostic and management data. Complications such as arrhythmia, embolism, and left ventricular dysfunction were reported. 6 , 7
Surgical treatment may be indicated for the associated lesions or for midventricular obstruction in type A DCLV. 3 Given the wide communication to the main LV in type B DCLV, surgical correction is indicated for severe heart failure, which consists of heart transplantation. In the case of good tolerance, as in our patient's condition, regular follow-up is indicated. 2
Conclusions
Double-chambered left ventricle is an extremely rare congenital cardiac lesion. Making the exact diagnosis is challenging. This case highlights the possibility of antenatal diagnosis and the necessity of a careful study to establish the diagnosis. Therapeutic management is likewise challenging in the absence of well-established guidelines.
Footnotes
Acknowledgments
The authors would like to acknowledge the pediatric and congenital cardiology department of Necker for their help and contribution in the diagnosis and the management of the patient.
Authors’ Statement
Written consent from the patient's parents was obtained for the publication of this case report.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
