Abstract
This consensus document for the performance of cardiovascular computed tomography (CCT) to guide intervention in the right ventricular outflow tract (RVOT) in patients with congenital heart disease (CHD) was developed collaboratively by pediatric and adult interventionalists, surgeons, and cardiac imagers with expertise specific to this patient subset. The document summarizes definitions of RVOT dysfunction as assessed by multimodality imaging techniques and reviews existing consensus statements and guideline documents pertaining to indications for intervention. In the context of this background information, recommendations for CCT scan acquisition and a standardized approach for reporting prior to surgical or transcatheter pulmonary valve replacement are proposed and presented. It is the first Imaging for Intervention collaboration for CHD patients and encompasses imaging and reporting recommendations prior to both surgical and percutaneous pulmonary valve replacement.
Introduction
Pulmonary valve dysfunction is common in patients with congenital heart disease (CHD) as an isolated lesion or in combination with other cardiac defects. With improved CHD survival,1,2 the sequelae of right ventricular volume or pressure overload that result from pulmonary valve dysfunction have come to light, including arrhythmias and right-sided heart failure.3,4 Patients with CHD and pulmonary valve dysfunction undergo one or more pulmonary valve replacement (PVR) procedures to avoid these complications.5–8 Historically, surgical PVR has been the standard of care and remains commonly performed today. 9 The first successful transcatheter valve was placed in the dysfunctional pulmonary conduit of a 12-year-old boy in 2000. 10 Transcatheter PVR (TPVR) is now performed in previously placed conduits and bioprosthetic valves of appropriate size.11–13 More recently, TPVR is used for the treatment of the native or patched right ventricular outflow tract (RVOT) as devices that accommodate a larger dimension have become available.14–18 Cardiovascular computed tomography (CCT) has emerged as an important modality in determining optimal candidates for surgical and TPVR and to guide intervention. 19 Cardiac advanced imagers in both pediatric and adult medicine may benefit from guidance to provide a reproducible assessment of RVOT anatomy prior to surgical and TPVR.
The consensus documents for transcatheter aortic valve replacement (TAVR) were instrumental in standardizing reporting and integrating CCT guidance into TAVR clinical practice.20,21 Similar multispecialty consensus recommendations to optimize CCT image acquisition, minimize diagnostic risk, and standardize reporting to guide PVR are not available. This document addresses this need and encompasses dysfunctional RVOT conduits and bioprosthetic valves, as well as native and patched RVOTs. This effort reflects the “heart team concept” and is written collaboratively by cardiac imagers, interventionalists, and surgeons with expertise specific to RVOT intervention in patients of all ages with CHD. Optimal care for these patients requires interventional and surgical collaboration throughout the age spectrum of the congenital cardiac patient. 22
This white paper includes the following, specific to patients with CHD:
A review of current definitions of pulmonary valve, bioprosthetic valve, and pulmonary conduit dysfunction as assessed by multimodality imaging techniques (with a comparison between imaging techniques and invasive catheter-derived hemodynamic measurements) A review of contemporary consensus statements and guideline documents pertaining to indications for intervention in the dysfunctional RVOT Presentation of recommendations for CCT scan acquisition, data processing, and image reconstruction for purposes of assessment of RVOT dysfunction Presentation of recommendations for a standardized approach for reporting on CCT of the dysfunctional RVOT for the guidance of surgical or TPVR Presentation of recommendations for a standardized approach for CCT assessment of coronary artery anatomy in patients with dysfunctional RVOT in preparation for surgical or TPVR Procedural overview for imagers
Multimodality Definitions of Pulmonary Valve, Bioprosthetic Valve, and Pulmonary Conduit Dysfunction
The degree of pulmonary valve, bioprosthetic valve, and conduit obstruction and regurgitation is determined noninvasively prior to recommendation for intervention (Table 1). Measures of pulmonary valve or conduit stenosis (PS) and regurgitation (PR) are most useful when trended over time and interpreted in relation to clinical symptoms and the effect on right heart size and function. Echocardiography is the modality most commonly used to serially assess RVOT dysfunction noninvasively after CHD intervention. Both echocardiography and cardiac magnetic resonance (CMR) imaging are used to assess PR, with CMR flow quantification considered the benchmark standard.23,24 Cross-sectional imaging is most reliable for the quantification of ventricular volumes and function in the tripartite RV. Cardiac magnetic resonance and CCT are both used to quantify ventricular volumes and function. 25 Cardiovascular computed tomography is performed when there are CMR contraindications, significant metallic artifacts, nonconditional EP devices, or need for coronary artery disease (CAD) assessment in preparation for RVOT intervention. Secondary effects of RVOT dysfunction on right atrial, inferior vena cava, or right ventricular size and the effect on ventricular septal position can be visualized in all modalities. Left ventricular (LV) size and function may be adversely affected by a severely enlarged right ventricle that inhibits LV diastolic filling. Evaluation of LV size and function is part of a comprehensive imaging evaluation in this patient subset. 26
Estimates of elevated RV pressure assume pulmonary resistance is normal, and the elevated pressure is due to RVOT obstruction.
Pulmonary regurgitation depends on RV compliance, may be minimal in severe RV failure or restrictive physiology and can decrease over time with progression of RV dysfunction. The categorization of regurgitant fraction into categories of significant, moderate to severe, and severe vary by publication.
Abbreviations: CFJW/RVOT, Doppler color flow jet width/right ventricular outflow tract diameter at the level of the pulmonary valve; CMR, cardiac magnetic resonance imaging; PHT, PR spectral Doppler pressure half time; PPG, catheterization peak to peak gradient; PR Index, duration of PR/duration of diastole on spectral Doppler;PR, pulmonary regurgitation; PSG, peak systolic gradient by spectral Doppler echocardiography; RVp, RV systolic pressure; TR, tricuspid regurgitation; V, velocity in m/s on spectral Doppler echocardiography or velocity encoded CM.
Right ventricular outflow tract obstruction can be dynamic, and gradients may vary with sedation, anesthesia, or loading conditions. Patient-prosthesis mismatch may occur with somatic growth in the presence of a conduit or bioprosthetic valve placed in childhood. When possible, both invasive and noninvasive studies should be performed with the least sedation possible to reflect the baseline hemodynamic status of the patient most reliably. In a patient with significant right-sided heart failure, the RVOT gradient may not accurately reflect the severity of obstruction, which is similar physiology to low-flow low-gradient aortic stenosis. In a patient with significant pulmonary hypertension, the RVOT gradient may also underestimate the degree of stenosis.
PR is affected by RV end-diastolic pressure, which is often elevated in patients with right heart disease. The degree of PR may change over time and can decrease with the progression of RV restrictive or constrictive physiology, diastolic dysfunction, and RV failure. In severe RV failure, PR may be minimal in the presence of an incompetent pulmonary valve.
Multimodality Evaluation of Pulmonary Valve, Conduit, and Bioprosthetic Valve Stenosis
Echocardiography: Native pulmonary valve, RVOT conduit, and bioprosthetic valve stenosis are routinely and serially evaluated with color and spectral Doppler echocardiography. The severity of RVOT obstruction is evaluated by Doppler peak velocity, and peak and mean gradients are obtained using the Bernoulli equation and categorized as mild, moderate, and severe (see Table 1).27,28 Classic teaching is that the peak systolic Doppler gradient will correspond to the catheter systolic peak-to-peak gradient (PPG) in valvular pulmonary stenosis. However, some studies show catheterization PPG correlates more closely to the echo mean Doppler gradient, and, therefore, some clinicians use the echo mean gradient to determine whether intervention is indicated. 29 Similarly, peak Doppler gradients obtained in patients with multiple levels of RVOT obstruction or with long segment narrowing tend to be overestimated, and mean gradients are often used. Due to the variability of echo Doppler gradient correlation to catheter PPG, tricuspid regurgitation (TR) jets are used to evaluate right ventricular pressure and to assess RVOT obstruction indirectly, assuming there is normal pulmonary artery pressure.29,30 The TR jet peak velocity and the ratio of estimated RV pressure to systemic pressure can be quantified, if systolic blood pressure is recorded.
Intracardiac echo: Intracardiac echo is sometimes used to determine the etiology of new-onset valvular dysfunction after TPVR, when transthoracic echo is nondiagnostic, and the limitations of Doppler echocardiography described above would be relevant. 31
Cardiac magnetic resonance: Velocity encoded cine flow analysis can determine the peak velocity of measured flow in the RVOT but is typically confirmatory and not the method by which recommendations for intervention are made in cases of RVOT obstruction. 32
Cardiovascular computed tomography: Cardiovascular computed tomography cannot provide hemodynamic information regarding the severity of RVOT obstruction. Effective valve orifice has not been correlated with degree of stenosis. Valve thickening, hypoattenuated leaflet thickening (HALT), 33 vegetation, thrombus, and calcification can be visualized and may help to determine the etiology of valve dysfunction and help guide appropriate treatment. 34 (Figure 1).
Multimodality Definitions of Pulmonary Valve, Conduit, and Bioprosthetic Valve Regurgitation
Echocardiography: Echocardiographic PR descriptions are qualitative and dependent both on acquisition techniques and patient physiology. Doppler Nyquist limits affect the visualization of low-velocity PR and should be optimized. In patients with CHD and multiple prior sternotomies, acoustic windows are challenging, and optimal 2D, color, and spectral Doppler imaging may not be possible.
Studies comparing echocardiographic parameters of PR to MRI flow quantification have shown echo may help differentiate between mild to moderate and severe PR, if multiple parameters are used.35,36 Color flow Doppler parameters of PR include proximal diastolic color flow jet width (CFJW) and ratio of the CFJW to the RVOT diameter. A CFJW/annular ratio > 0.5 is predictive of an regurgitant fraction (RF) > 20% and a ratio > 0.7 is predictive of severe PR on retrospective studies in small patient subsets.37–39 The dynamism and variability of pulmonary annular dimension measurement in palliated CHD is significant. By convention, the annulus is measured in diastole and may underestimate the annular size in a patient with patched RVOT. 40
Color and pulse Doppler interrogation allows detection of diastolic flow reversal in the main and/or branch pulmonary arteries and anterograde flow in late diastole. Rapid equalization of the regurgitant jet, flow reversal in the branch pulmonary arteries, and end-diastolic anterograde flow in the pulmonary artery may be markers of severe PR.28,39 Spectral Doppler quantifies PR jet deceleration time, pressure halt time, and duration of the PR jet compared to the diastolic duration (PR index).35,41,42
Cardiac magnetic resonance: Cardiac magnetic resonance imaging is the current standard for noninvasive quantitative assessment of PR.39,28 Cardiac magnetic resonance quantifies PR by velocity-encoded cine imaging of the RVOT measuring anterograde versus retrograde flow. PR estimated by stroke volume differences between the right and left ventricle correlates well with flow analysis in the absence of other regurgitant lesions or cardiac shunts.43,44 Phase contrast acquisitions can be acquired in 2D planes perpendicular to the direction of flow or using 4D flow which allows for off-line analysis in multiple different planes.43,45 There is variability in categorization of severity of PR based on quantified RF. Most commonly, PR is graded as mild if the RF is <20%, moderate if between 20% and 40%, and severe if >40%.32,46 Other classifications categorize RF > 25% as significant 47 or severe as >30% to 40% RF. 30 Transcatheter pulmonary valve replacement recommendations classify PR > 30% as severe. 48
Cardiovascular computed tomography: Cardiovascular computed tomography is considered equivalent to CMR for estimation of ventricular volumes and function if scanners with the appropriate temporal resolution are used and the RV is well opacified with contrast.49–51 The short-axis stack should be reconstructed at 5% intervals to allow 20 frames/cardiac cycle and evaluated in a similar slice thickness to CMR (6-8 mm). Like CMR, stroke volume differences from a short axis orientation from an ECG-gated functional CT scan can be used to estimate PR by stroke volume differences between the right and left ventricle, if there is no cardiac shunt or other valvular regurgitation. When multiple shunts or regurgitant lesions are present, only the total stroke volume difference can be reported, with other modalities required to determine the significance of each.
Summary of Existing Scientific Statements and Guideline Documents Addressing Intervention in the Dysfunctional Right Ventricular Outflow Tract in Patients With Congenital Heart Disease
The goal of lifetime management of the RVOT is to maximize functional capacity and preserve right heart function by carefully balancing the risks of multiple surgical and/or transcatheter interventions with the benefit of restoring pulmonary valve function at all stages of patients’ lives. Despite strong interest and much study regarding the timing of restoring pulmonary valve function, the optimal timing is still unclear, and the risk–benefit ratio may be different for every patient.53–55 There are no published consensus recommendations for timing of PVR that are specific to the pediatric age range. For this reason, a commonly referenced expert opinion paper by Geva from 2013 has been included, as it offers a list of proposed indications for PVR in patients of all ages with tetralogy of Fallot (TOF) based on the author's interpretation of the current literature at the time. 47 Adult CHD (ACHD) guidelines have been proposed and published by American, European, and Canadian cardiovascular societies. These are based on a level of evidence that does not include a randomized trial, and much of the guidance is based on consensus of expert opinion. Recommendations are based on a combination of clinical symptoms, the degree of RVOT dysfunction, and the effects on RV size and systolic function. Recent prospective studies in adults who have undergone PVR after repair of TOF suggest that current criteria for intervention are predictive of RV remodeling but have limited value for predicting important adverse clinical events, such as progression of arrhythmia or heart failure and sudden death.56–58 Decision-making in patients with mixed valve disease is particularly difficult since they may not reach the criteria for pulmonary stenosis or pulmonary regurgitation alone but still have hemodynamically significant RVOT dysfunction. The current recommendations for intervention are likely to change, as new information is incorporated into clinical practice. Table 2 (A, B, C) is a summary of existing scientific statements and guideline documents addressing intervention in the dysfunctional RVOT in patients with CHD.
Indication for Intervention in Pulmonary Stenosis (PS)/Right Ventricular Outflow Tract Obstruction (RVOTO)
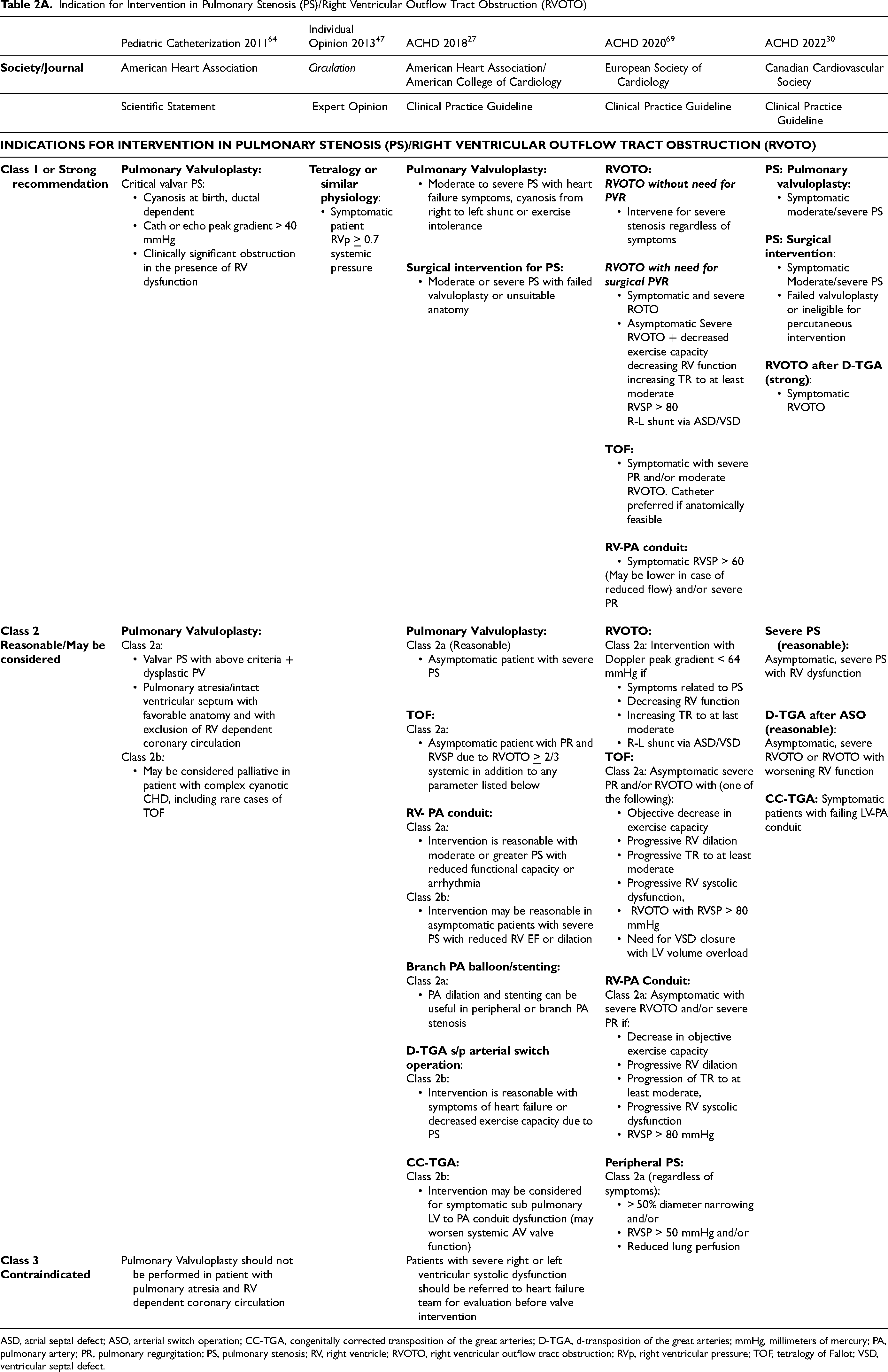
ASD, atrial septal defect; ASO, arterial switch operation; CC-TGA, congenitally corrected transposition of the great arteries; D-TGA, d-transposition of the great arteries; mmHg, millimeters of mercury; PA, pulmonary artery; PR, pulmonary regurgitation; PS, pulmonary stenosis; RV, right ventricle; RVOTO, right ventricular outflow tract obstruction; RVp, right ventricular pressure; TOF, tetralogy of Fallot; VSD, ventricular septal defect.
Indications for Intervention in Pulmonary Regurgitation (PR)
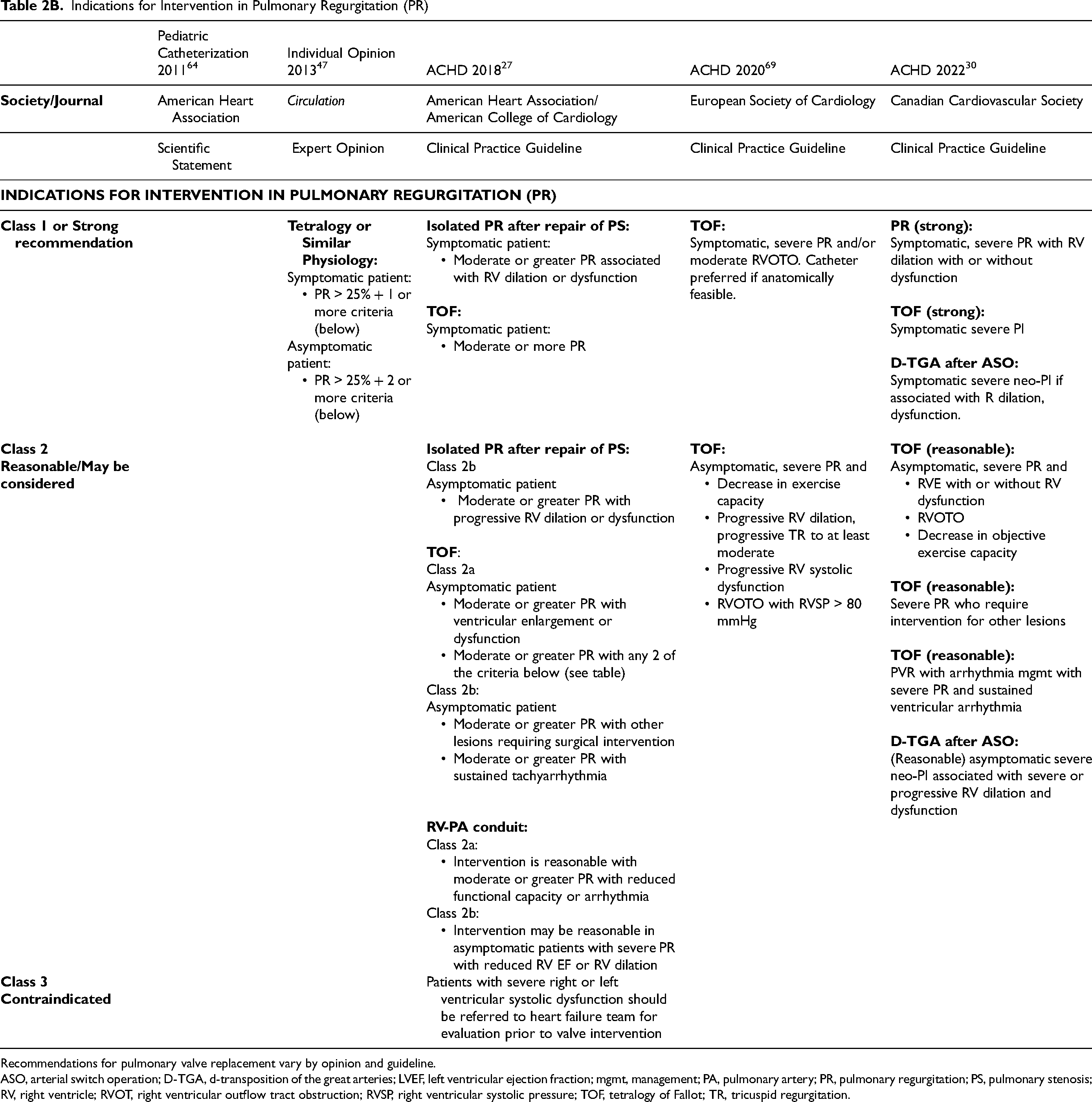
Recommendations for pulmonary valve replacement vary by opinion and guideline.
ASO, arterial switch operation; D-TGA, d-transposition of the great arteries; LVEF, left ventricular ejection fraction; mgmt, management; PA, pulmonary artery; PR, pulmonary regurgitation; PS, pulmonary stenosis; RV, right ventricle; RVOT, right ventricular outflow tract obstruction; RVSP, right ventricular systolic pressure; TOF, tetralogy of Fallot; TR, tricuspid regurgitation.
Parameters used for Quantification of Hemodynamic Abnormalities in RVOT Dysfunction (PI/PS)
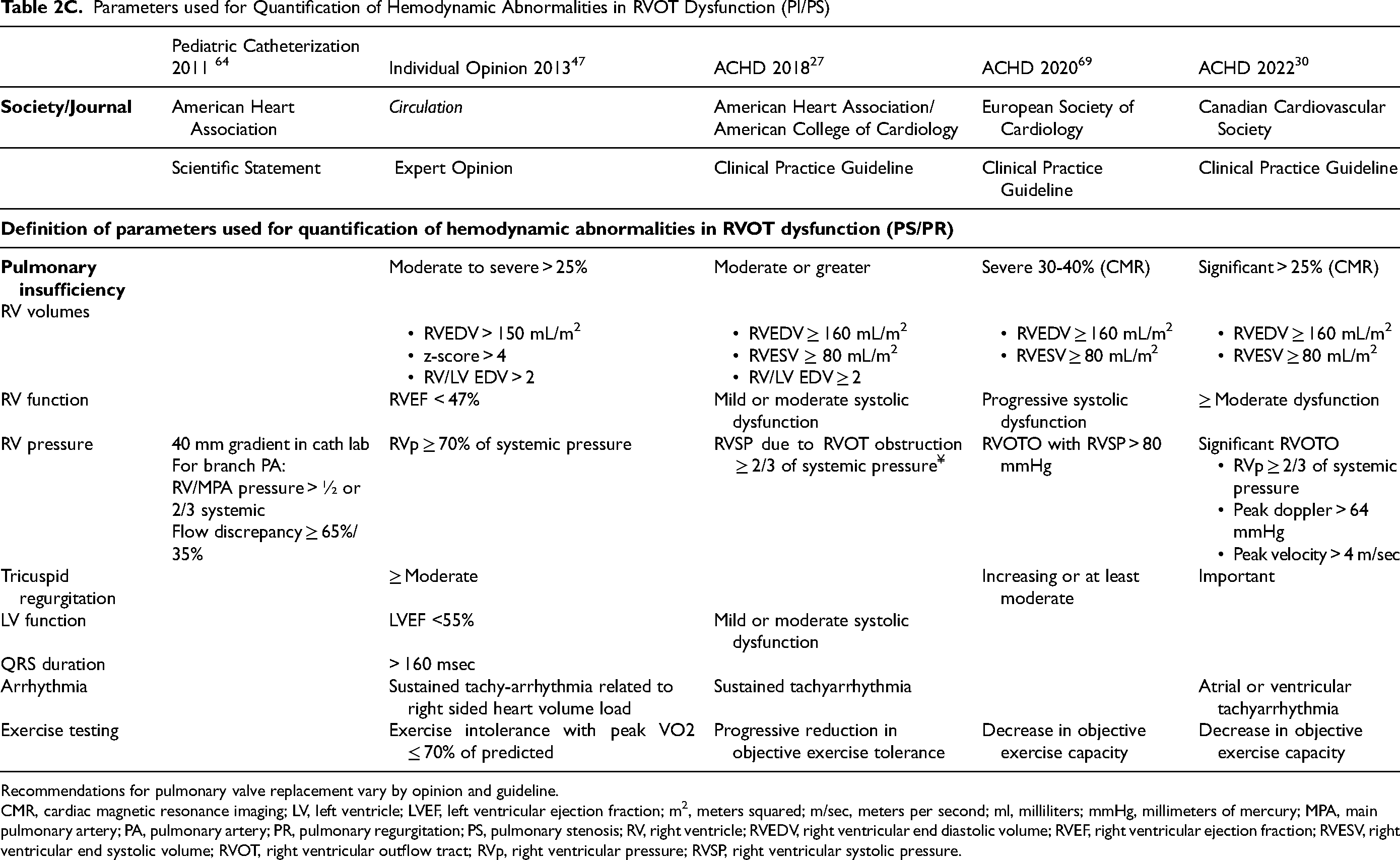
Recommendations for pulmonary valve replacement vary by opinion and guideline.
CMR, cardiac magnetic resonance imaging; LV, left ventricle; LVEF, left ventricular ejection fraction; m2, meters squared; m/sec, meters per second; ml, milliliters; mmHg, millimeters of mercury; MPA, main pulmonary artery; PA, pulmonary artery; PR, pulmonary regurgitation; PS, pulmonary stenosis; RV, right ventricle; RVEDV, right ventricular end diastolic volume; RVEF, right ventricular ejection fraction; RVESV, right ventricular end systolic volume; RVOT, right ventricular outflow tract; RVp, right ventricular pressure; RVSP, right ventricular systolic pressure.
Adults With Native Valvar Pulmonary Stenosis
Intervention for moderate or severe native pulmonary valve stenosis is typically balloon valvuloplasty. Patients with marked dysplasia of the valve, annular hypoplasia, or supravalvular pulmonary stenosis may not respond to valvuloplasty and may require PVR, however. There are no data to guide the timing of PVR in patients with PR after treatment for isolated valvar pulmonary stenosis. 27 All consensus documents recommend intervention in symptomatic patients and consideration of intervention in asymptomatic patients with significant or progressive RV dilation, dysfunction, and objective decrease in exercise capacity.
Adults With Repaired Tetralogy of Fallot
Timing for PVR in adults with repaired TOF also remains uncertain but is better investigated. Studies have determined preoperative RV volumes that will normalize after PVR. Based on these reports, the major cardiovascular professional societies in the U.S., Canada, and Europe have published recommendations for the timing of PVR based on clinical symptoms and the degree of RV dilation and dysfunction.27,47,59–61 All current guidelines recommend PVR for symptomatic adults with moderate or greater PR. 27 Pulmonary valve replacement is considered reasonable in asymptomatic adults with moderate to severe PR and ventricular enlargement, ventricular dysfunction, or progressive arrhythmia. Surgical PVR may be reasonable in adults with moderate or greater PR who are undergoing surgery for other cardiac intervention. 27
While most studies on timing of PVR have focused on returning the RV to a normal size, few have focused on decreasing sustained ventricular tachycardia and death. 7 The International Multicenter TOF Registry (INDICATOR) cohort of patients with comprehensive preoperative evaluation prior to PVR showed that an older age (≥28 years) at PVR and pre-PVR RV hypertrophy (RV mass-to-volume ratio ≥0.45 g/mL) and dysfunction (RVEF <40%) were predictive of a shorter time to postoperative death and sustained ventricular tachycardia. 57 A subgroup analysis revealed that RV systolic pressure ≥40 mm Hg, decreased RV systolic function, and RV hypertrophy were associated with the primary outcome of sustained ventricular tachycardia, aborted sudden cardiac death, or death. Interestingly, pre-PVR RV end-diastolic volume, pulmonary RF, and LV ejection fraction were not associated with the primary outcome.
Adults With Right Ventricular Outflow Tract Conduits
Current guidelines consider intervention on an RVOT conduit reasonable for adults with moderate or greater PR or moderate or greater stenosis with reduced functional capacity or arrhythmia. In the absence of symptoms, intervention may be reasonable (Class IIb) in the presence of severe stenosis or severe regurgitation with reduced RV ejection fraction or RV dilation. 27 The most common underlying CHD diagnoses and surgeries with RVOT conduits include TOF, post-Ross operation, post-Rastelli operation, and post-repair of truncus arteriosus.
Adults After Arterial Switch Operation for d-Transposition of the Great Arteries and Subpulmonary Outflow Tract Obstruction With Congenitally Corrected Transposition of the Great Arteries
Intervention is recommended for symptomatic adult patients with severe RVOT obstruction after arterial switch operation (ASO) and may be considered for severe RVOT obstruction or RVOT obstruction with worsening RV function. 30 Patients after LeCompte maneuver may have neopulmonary valve stenosis, supravalvular stenosis, and/or branch pulmonary artery stenosis. The reimplanted coronary artery relationship to the neopulmonary root may complicate TPVR, and this patient population is considered at high risk for coronary compromise (see coronary section). A subpulmonary LV in congenitally corrected transposition of the great arteries (CC-TGA) may also be affected by outflow tract dysfunction secondary to congenital valve abnormality, after conventional repair of pulmonary valve dysfunction, acquired dysfunction of previously placed conduit, or from ventricular septal shift from an enlarged systemic RV. Etiology must be determined prior to intervention. In a patient with CC-TGA after a physiologic repair including both atrial baffle and ASO (“double switch”), the venous pathway to the RV should be detailed to determine catheter course for TPVR. The Canadian guidelines classify intervention as reasonable in a symptomatic CC-TGA patient with failing LV-PA conduit, but relief of LV-PA conduit obstruction with TPVR has been shown to worsen systemic RV dysfunction or TR in the majority of patients. Worsening RV dysfunction and TR are associated with lower residual LV-PA gradients and greater interventricular septal shift. 62
Children With Repaired Tetralogy of Fallot
There is no published consensus on the timing of PVR in children and adolescents. Adult guidelines are often extrapolated to children. Indexing RV end-diastolic volumes to body surface area (BSA) in children often leads to larger numbers than in adults and may lead to early PVR when risk/benefit is not clearly understood. As children grow, the indexed volumes may normalize as BSA increases. In a single center study of patients with repaired TOF, those who had PVR before age 16 years had better preservation of RV morphology and function and shorter duration of QRS at the latest follow-up (mean 24 years after TOF repair) in comparison to those who had later PVR. However, it did not translate to better event-free survival over this time interval. 63 More data are needed to determine the optimal timing of PVR in children and adolescents with TOF.
Children and Adults With Pulmonary Atresia/Intact Ventricular Septum
Patients with pulmonary atresia/intact ventricular septum often do not have a normal tripartite ventricle and have inlet, apical trabecular, or outflow tract hypoplasia. Initial RVOT palliation may include perforation of membranous pulmonary valve atresia with balloon valvuloplasty, an RVOT patch, or a pulmonary conduit in the absence of RV-dependent coronary circulation. Patients with RVOT dysfunction in the setting of a hypoplastic trabecular RV may not meet RV volume criteria for PVR but may have progressive TR and right atrial dilatation due to diastolic dysfunction or restrictive physiology. These factors should be reported, and the right-to-left ventricular ratio is particularly relevant in this patient subset.
Children With Right Ventricular Outflow Tract Conduits
The most recent recommendations for pediatric transcatheter interventions were published in 2011, just one year after the Melody TPV (Medtronic, Inc.) received U.S. Food and Drug Administration (FDA) approval for Humanitarian Device Exemption. Like the 2018 AHA/ACC ACHD guidelines, TPVR was given a Class IIa recommendation as reasonable to consider for treatment of moderate to severe PR or stenosis of the RVOT conduit, provided the patient meets criteria for the valve used. 64 Like young patients with TOF with native RVOT dysfunction, the timing of intervention in pediatric patients with pulmonary conduit and bioprosthetic valve dysfunction is not clear.
Indications for Approved Transcatheter Pulmonary Valves
Both the Melody TPV and the SAPIEN 3 Transcatheter Heart Valve (THV) System (Edwards Lifesciences Corporation) are indicated to treat pediatric and adult patients who have a clinical indication for intervention on a dysfunctional RVOT conduit or surgical bioprosthetic pulmonary valve that has ≥ moderate regurgitation and/or a mean RVOT gradient by echocardiogram of ≥35 mm Hg.65,66 The Harmony TPV System (Medtronic, Inc.) and the SAPIEN 3 TPV System with Alterra Adaptive Pre-stent (Edwards Lifesciences Corporation) are both indicated for use in the management of pediatric and adult patients with severe PR by echocardiography who have a native or surgically repaired RVOT and have clinical indications for PVR.48,67 The Harmony valve indications include patients with indication for “surgical PVR, with severe PR determined by echocardiography or RF ≥ 30% by CMR.” 48
Mixed Valve Dysfunction
Recommendations for intervention in the dysfunctional RVOT in patents with CHD are primarily based on the effects of severe PS or severe PR. Many patients have mixed RVOT dysfunction and may not meet criteria for isolated regurgitation or stenosis. In these patients, symptoms and changes in parameters of RV size and function are vitally important to clinical decision-making.
Indexed RV Volumes
Most indications for PVR include RV end diastolic and end systolic volumes indexed to BSA. In patients with large body mass index (BMI), volumes should be indexed to height, since volumes indexed to BSA may underestimate the degree of RV enlargement. Evaluating total RV volume or volumes indexed to BMI, height, or BSA calculated with ideal body weight should be considered in these patients. 68 In pediatric patients with low BMI, as noted above, volumes may be overestimated and may normalize with growth. Volume criteria are not applicable to patients who have undergone “1 ½” ventricle repair, which includes a superior cavopulmonary artery connection with patent RVOT. In these patients, the RV volumes and pulmonary artery flow reflect inferior vena cava flow only.
Cardiovascular Computed Tomography Scan Acquisition Recommendations for Assessment of Right Ventricular Outflow Tract Dysfunction in Patients With Congenital Heart Disease
There are many comprehensive documents regarding CCT image acquisition optimization in pediatric and adult patients and recommendations specific to CHD.70–72 Brief recommendations on patient preparation and scan acquisition are as follows:
Patient Preparation
Patient history: Detailed cardiac history should be obtained, including prior interventions, vascular access complications, operative details of RVOT intervention (transannular or limited RVOT patch, valvotomy, size of bioprosthetic valve or RVOT conduit, prior aortopulmonary shunt, collateral unifocalization or branch pulmonary artery intervention).
Iodinated contrast allergy screening, pregnancy, renal, and thyroid function testing: Screening should follow standard institutional protocols. Most centers ask about pregnancy or require pregnancy testing for women starting in adolescence, but this is not required by the American College of Radiology for CCT. 73 Renal function testing for adults prior to CT imaging is also required by most centers. When renal dysfunction is present, prehydration and/or low-dose contrast protocols should be utilized. Iodinated contrast allergy is more common in adults than children, and standardized protocols for pretreatment of contrast allergy and hemodynamic monitoring with contrast administration should be followed. 70 Thyroid testing is recently recommended for neonates undergoing testing utilizing iodinated contrast, but this is not applicable for older children, adolescent and adult patients being screened for TPVR. 74
Premedication: Beta blockade and coronary vasodilators can be used per institutional protocol if detailed coronary artery imaging is indicated to exclude CAD. This is commonly recommended in adult or pediatric patients with symptoms suggestive of ischemia, men over the age of 40 years, and postmenopausal women. 27 Beta-blockers may be relatively contraindicated in patients with pulmonary hypertension and moderate to severe LV outflow obstruction, including aortic stenosis. For assessment of the coronary relationship to the sternum and RVOT, premedication with beta-blockers for those with high heart rates is not required. Although cardiac motion artifact may degrade detailed imaging of the distal coronary arteries, this would not affect PVR planning, which requires evaluation of the proximal coronaries.
Intravenous (IV) access site: Peripheral IV access should utilize the best access site, unless there are known venous occlusions. The right antecubital vein may be preferred (assuming normal right-sided superior vena cava) to avoid contrast in the innominate vein that has potential for scatter artifact. If scatter artifact is present in the innominate vein or superior vena cava, it typically will not affect image quality for assessment of the main and branch pulmonary arteries, however.
Contrast injection: A contrast injection that opacifies both the right and left ventricle in the same image acquisition should be used. A typical contrast injection for LV opacification is 15 to 17 s of contrast followed by saline; the total contrast injection time for biventricular opacification should be at least 20 s. Timing will depend on patency of the systemic veins used for injection, degree of RV dysfunction, and tricuspid or pulmonary regurgitation. If contrast is still being injected at the time of image acquisition, scatter artifact will be present but can be minimized by diluting the contrast or decreasing the contrast injection rate for the later portion of the contrast injection. A two-phase contrast injection followed by saline is often called a triphasic injection protocol.
Biventricular contrast opacification can be accomplished in three ways:
Extend the contrast bolus for several seconds longer than normal so that contrast remains in the RV and PA at the time of image acquisition. Give a normal contrast bolus followed by an injection of 50% contrast/50% saline at the same rate. Give a normal contrast bolus followed by an injection of contrast at a lower rate (usually 50%).
Contrast injection rate by patient size and IV gauge is well outlined in other documents.71,72,75 Some TPVR screening protocols trigger image acquisition to optimize opacification in the pulmonary artery. This is not an optimal approach to allow visualization of the coronary arteries to predict coronary compromise with RVOT stenting but allows ease in creating 3D modeling of the isolated RVOT.
Sedation and anesthesia
A breath-hold sequence is optimal to avoid respiratory motion artifact, and developmentally normal patients older than seven years can usually cooperate. For an anatomic scan of anatomy without need for functional analysis, breath hold may not be required if the patient is able to hold still in the scanner during image acquisition. For younger patients, sedation and anesthesia may be needed for breath-holding if a functional scan is required. Institutional protocols should be used.
Cardiovascular Computed Tomography Scan Acquisition Parameters for Optimization of the Right Ventricular Outflow Tract
Scan range
The optimal scan range for CT evaluation of the RVOT should include the cardiac silhouette and extend cranially to include the main and branch pulmonary arteries. In patients with a history of TOF with pulmonary artery atresia and major aortopulmonary collaterals, the entire lung field should be included to assess for distal pulmonary artery stenoses that may require intervention. In patients with a history of aortic arch intervention or aortic dilatation, scan range should include the aorta. In patients with a history of a Ross procedure or with d-TGA after ASO, the scan range should extend past the area of the neoaortic and neopulmonary anastomoses and for truncus arteriosus should include the ascending aorta distal to the truncal root, which is often dilated. After the Rastelli or Nikaidoh operations, the scan range should include pertinent anatomy based on echocardiographic findings, such as the distal pulmonary arteries and proximal ascending aorta. If a prior MRI or catheterization has been done and the CT is specifically for PVR planning, coverage should prioritize visualization of the superior cardiac silhouette, RVOT, and branch pulmonary arteries. Assessment of venous vessel size for sheath sizing is not standard on current protocols but can be performed on an individual basis. Contrast injection would need to be modified in addition to extending the scan range to include the central venous vessels.
Volumetric scanners with a 16-cm scan range will cover the thorax in a child or the superior cardiac silhouette and branch pulmonary arteries in an adult in one acquisition. If the entire cardiac silhouette and branch pulmonary arteries or aorta are needed in a large adult, two volumetric scans may be needed for anatomic coverage. Due to higher radiation dose with ECG-gated scans, a limited scan range acquisition can be followed by a nongated helical scan with longer scan range in large adults needing full thoracic coverage. Dual source scanners have a smaller coverage for prospectively ECG-triggered axial scan mode (5 cm) and need several heart beats for anatomic coverage in a prospectively ECG-gated scan. The highest pitch scan mode on dual source scanners can be adjusted to the anatomy needed, acquiring the data in a single heartbeat and usually starting the acquisition in early diastole.
Scan sequence
For native RVOT and patched RVOTs, an ECG-gated functional cardiac scan should be performed with pulse modulation, indicating there is a narrow fully radiated acquisition window, and the remainder of the cardiac cycle has reduced radiation exposure (typically 20%). The fully radiated acquisition window should be kept minimal (10% of cardiac cycle) and adjusted to heart rate, typically systole for heart rates over 80 bpm and diastole for heart rates below 65 bpm. For heart rates between 65 and 80 bpm, an acquisition window including end-systole and early diastole can be used if detailed coronary imaging is needed to exclude coronary disease. For most pediatric and young adult patients, coronary origin and course are needed but not detailed definition of distal coronary vessels. Current screening protocols have systolic acquisition window varying between 30% and 45%, and diastolic acquisition windows vary between 70% and 90%. The minimum acquisition window should be utilized and can be narrowed to a single phase for the fully radiated dataset for both functional and anatomic imaging. For conduits, functional imaging may not be required but can be helpful in predicting coronary compression when anomalies are present and should be individualized based on patient-specific history and anatomy.
Scanner Output and Dose optimization
Young patients with CHD often have a high cumulative radiation dose from multiple diagnostic tests and interventional procedures, and some studies have shown an association between cumulative radiation dose and the future development of malignancy.76,77 Historically, radiation exposure was primarily from diagnostic and interventional catheterization. Patients with chromosomal and congenital anomalies including CHD, however, have a significantly increased rate of malignancy, even for defects unlikely to require radiation-based diagnostics, such as an isolated ventricular septal defect. 78 Nonetheless, it is prudent to minimize radiation dose as much as possible. Scanner output (kilo-volt peak: KVp and milliampere: mA) can be significantly reduced if detailed coronary imaging is not required, and a higher level of noise can be tolerated. Modern scanners have been shown to have far lower radiation exposure than historical standards when used in centers with expertise imaging CHD patients.79,80 Two studies documented 85% to 90% dose reduction in the CHD patient population with adoption of dose optimization and newer scanner platforms.81,82 A recent multi-institutional study of CCT performed in patients of all ages with CHD showed low radiation dose across the age range even for functional CT imaging. 79 The average radiation dose estimate for an anatomic scan was < 1 millisievert (mSv) in 1,516 CCT scans in CHD patients from neonates to adult patients and the average radiation dose for a functional CCT scan in 226 studies across the age range was < 2 mSv. For reference, a commonly cited paper estimating cancer from CT in pediatric patients reported a radiation dose up to 69 mSV per scan, a dose over 20 mSv in approximately 8% of chest CT and the dose associated cancer risk was between 30 and 50 mSv. 77
Standardized Approach for Assessing and Reporting the Dysfunctional Right Ventricular Outflow Tract for Pulmonary Valve Replacement Sizing and Landing Zone
Definition of the Transcatheter Pulmonary Valve Replacement Landing Zone
The landing zone for a TPVR can be in a native RVOT, a patched RVOT, the MPA, the neo-PA after ASO, a surgical RVOT conduit, or a surgical or percutaneously placed bioprosthetic valve. In the native RVOT, the landing zone may be the valve annulus or the main pulmonary artery. The TPV may extend below the annulus, depending on the size and proposed valve. A clear understanding of the underlying anatomy will determine whether a patient is a candidate for a percutaneous or surgical pulmonary valve, the potential landing zone, and the best device for the patient's RVOT anatomy.
Native and Patched Right Ventricular Outflow Tracts
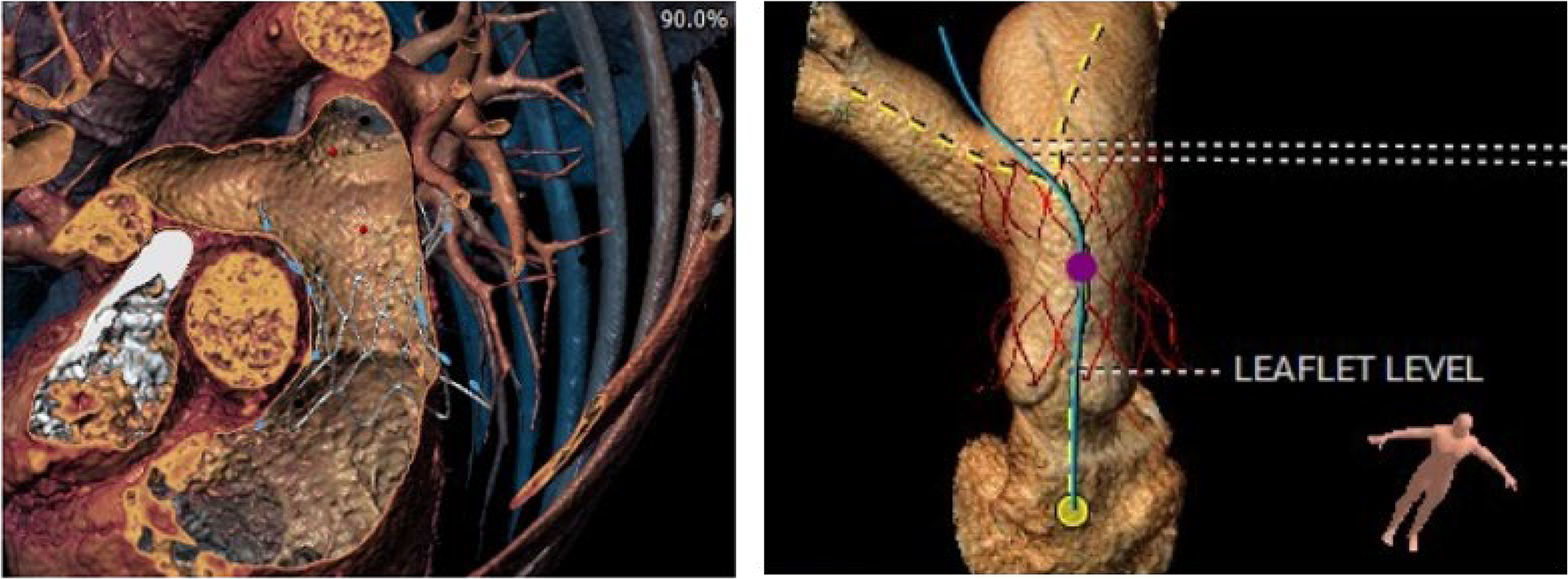
The anatomy of the native and patched RVOT is complex, and there can be significant dynamism throughout the cardiac cycle (Figures 2 and 3). The maximum RVOT diameter and length may be in systole or diastole depending on underlying anatomy and prior intervention. The RVOT below the annulus is comprised of muscular tissue, contracting in systole, and relaxing in diastole, resulting in larger RVOT cross-sectional dimensions in diastole compared to systole for the native RVOT. This has been quantified in normal healthy volunteers by cine cardiac MRI with recommendations to assess pulmonary annular dimensions in diastole. 50 3D whole heart MRI or MRA obtained in a single heart phase or a non-ECG gated scan may not accurately reflect the maximal RVOT diameter. In contrast, patients with a transannular patch may have a systolic outward bulging of the RVOT inferior to the annular plane, due to scar tissue or fibrosis, and the maximum dimension may be in the systolic phase. For this reason, both systolic and diastolic phases of the dataset should be evaluated to find the maximal dimensions in the native RVOT and MPA. If a single cardiac phase is acquired, the phase of acquisition should be individualized and based on underlying anatomy to capture the maximal MPA and RVOT dimension. For stenosis, the maximum dimension at the narrowest point of the RVOT often determines how aggressive preprocedural angioplasty and stenting may need to be and whether TPVR is feasible. For large native and patched RVOTs, the maximum dimension in the cardiac cycle along with the length of the potential landing zone determines if a balloon expandable or self-expandable TPVR is feasible with stable device position. 3D datasets allow for virtual valve implantation prior to the procedure (Figure 4) that can enhance understanding of device-anatomy interactions and be used for procedural planning (Figure 9).
Analysis of the Native and Patched Right Ventricular Outflow Tract Landing Zone: Standardized Method of Measurement
A dedicated analysis should be performed to quantify cross-sectional dimensions of the RVOT/MPA landing zone and landing zone length. Evaluation for surgical PVR and balloon expandable TPVR should be done from an ECG-gated CCT scan, and the phase with the maximal cross-sectional area and length should be chosen for analysis. This is best performed using a center line technique/curved planar reformats (CPR) along with the long axis of the RVOT. Alternatively, angulated multiplanar reformats (MPRs) can be used (4D/multiphasic data set). Often, MPRs are made in both systole and diastole, and data from both datasets are superimposed to illustrate RVOT dimensions throughout the cardiac cycle. The cardiac phase used for self-expanding TPVR evaluation depends on the valve type and is determined by how the evaluation was performed in the clinical trials (ie, 30% systole for Alterra Adaptive Prestent, 70% diastole for Harmony TPV) (Image 5).
Evaluation of RVOT Centerline: When using a centerline technique/CPRs, the centerline should be carefully aligned with the MPA, beginning in the RVOT and extending to the PA bifurcation and to the posterior wall of the pulmonary artery (ridge of bifurcation). Extending the centerline to the ridge of the bifurcation pertains to both conventional and crisscross branch PA anatomy with symmetric branch PAs. In the case of markedly asymmetric branch PAs, the centerline can be extended into the dominant branch PA.
Differences in MPR versus centerline technique: Importantly, the nature of a centerline technique with cross sections aligned in orthogonal fashion to the centerline will commonly result in potentially slightly different orientations of the cross-sectional plane than a manually oriented MPR. This is most noticeable at the annular level with the centerline often not being exactly perpendicular to the annular plane. Nevertheless, the centerline technique takes the entire length of the landing zone into account.
Measurement of RVOT Cross-sectional dimension: Cross-sectional dimensions should be assessed by means of a contouring tool, yielding perimeter and area, as well as minimum, maximum, and perimeter-derived average diameter. Importantly, the contour should be drawn in a smooth fashion to avoid accidental increase in the assessed perimeter due to oblique orientation.
Annular level: The annular/valve level can be identified based on visualized native leaflet tissue. If no native leaflet tissue can be visualized, wall characteristics (eg, myocardium, primarily posteriorly) can be used to determine the annular/valve level as the transition between RVOT and MPA.
Mid-MPA: An additional cross-sectional assessment should be performed approximately half-way in-between the annular level and bifurcation and in any area of special anatomic interest specific to each patient.
Landing zone length: The landing zone length should be assessed beginning at the annular plane to the beginning of the bifurcation (ie, first PA branch take-off). When using a centerline technique, the distance is measured along with the centerline itself, commonly using a function of the employed post-processing platform. When using a manual MPR technique, landing zone length quantification can be more challenging, as it is difficult to visualize the necessary annular and bifurcation levels simultaneously. For self-expanding valves, the landing zone length should include the distal contractile tissue in the RVOT and the distance to the back (posterior) wall of the PA bifurcation.
PA Bifurcation: The level of the bifurcation measurement is defined by the beginning of the first branch PA take-off. Care should be taken not to measure landing zone diameters beyond the first branch PA take-off, as the cross-sectional dimensions are not relevant. The distance from the first branch PA take-off to the posterior wall of the bifurcation (ridge of the bifurcation) is measured along with the centerline. It is an important variable for determining how far distal a self-expanding valve can be positioned and if there is a risk for branch PA occlusion. When using an MPR technique a caliper measurement can be used to measure distance between these two location (Figure 5). There is significant variability of branch pumonary artery anatomy, illustrative examples are shown in Figure 6.
Determine MPA shape: The gross anatomy of the MPA should be qualitatively graded into the categories listed below (adopted from Schievano et al). 83 This grading provides a high level summary of the MPA anatomy and defines a potential landing zone (Figure 7). Recommended reporting elements for the native or patched RVOT are listed in Figure 8.
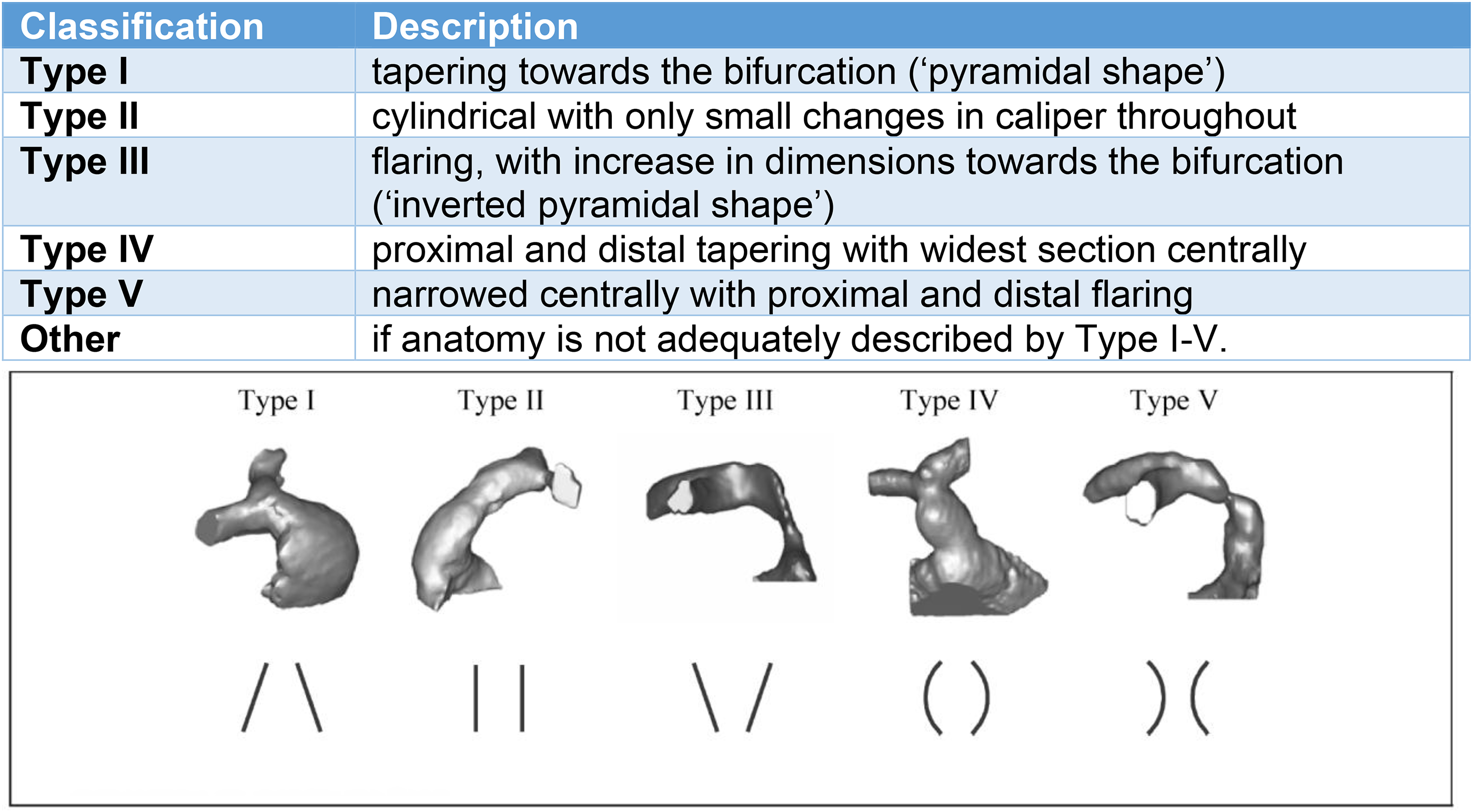
Classification Native RVOT/MPA Anatomic Shape (Source: Schievano et al). 83 Understanding the anatomic substrate of the native RVOT helps determine the optimal landing zone for the proposed TPVR. RVOT, right ventricular outflow tract; TPVR, transcatheter pulmonary valve replacement.
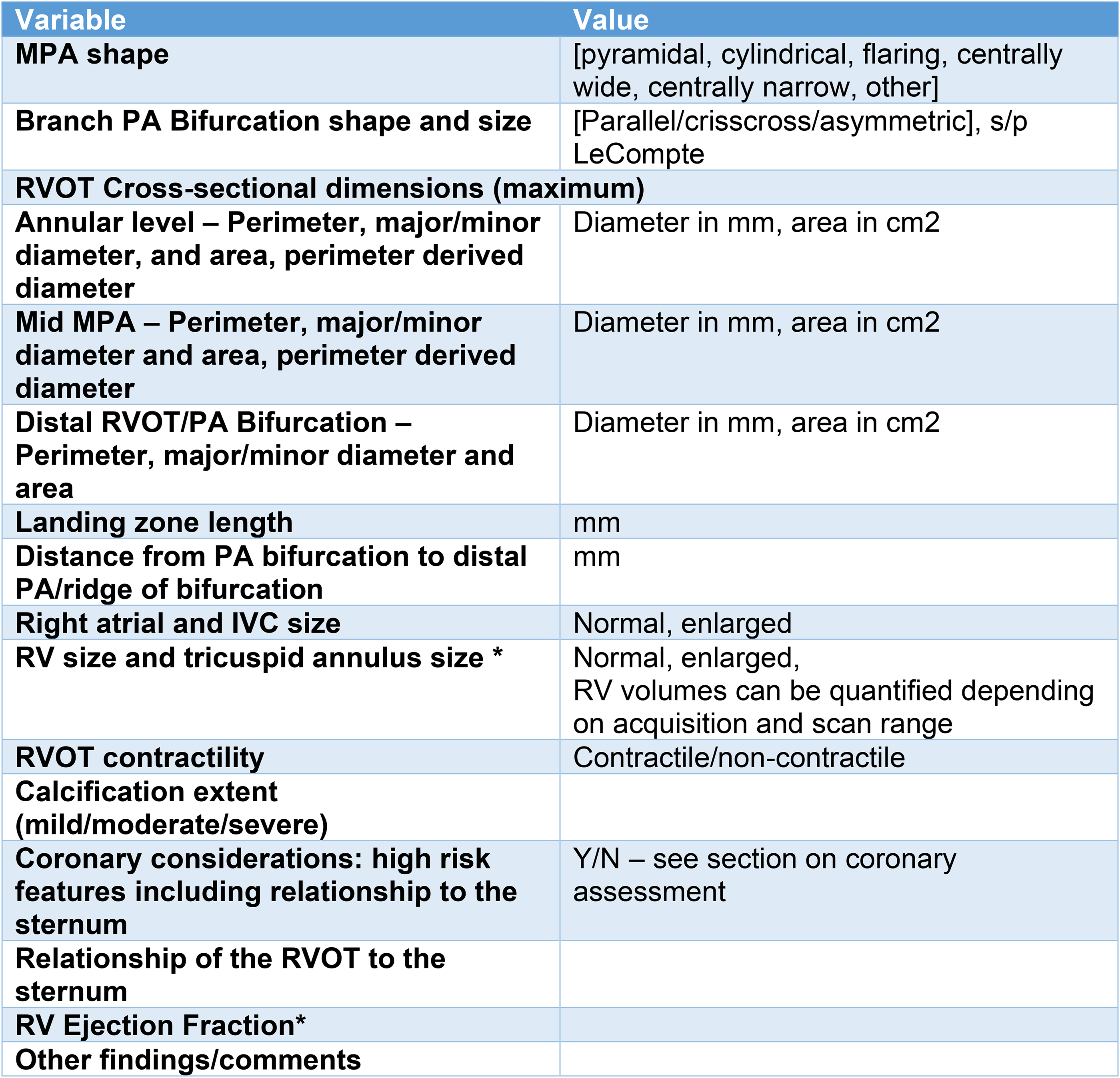
Recommended native RVOT reporting elements. *Many patients will have prior advanced imaging, and the CT is done specifically to assess the RVOT. Some centers may use an ECG-gated cardiovascular computed tomography (CCT) scan to both assess RV size and function in addition to planning for percutaneous PVR. If so, the RV volumes and EF can be reported. Abbreviations: EF, ejection fraction; IVC, inferior vena cava; MPA, main pulmonary artery; PA, pulmonary artery; PVR, pulmonary valve replacement; RVOT, right ventricular outflow tract.
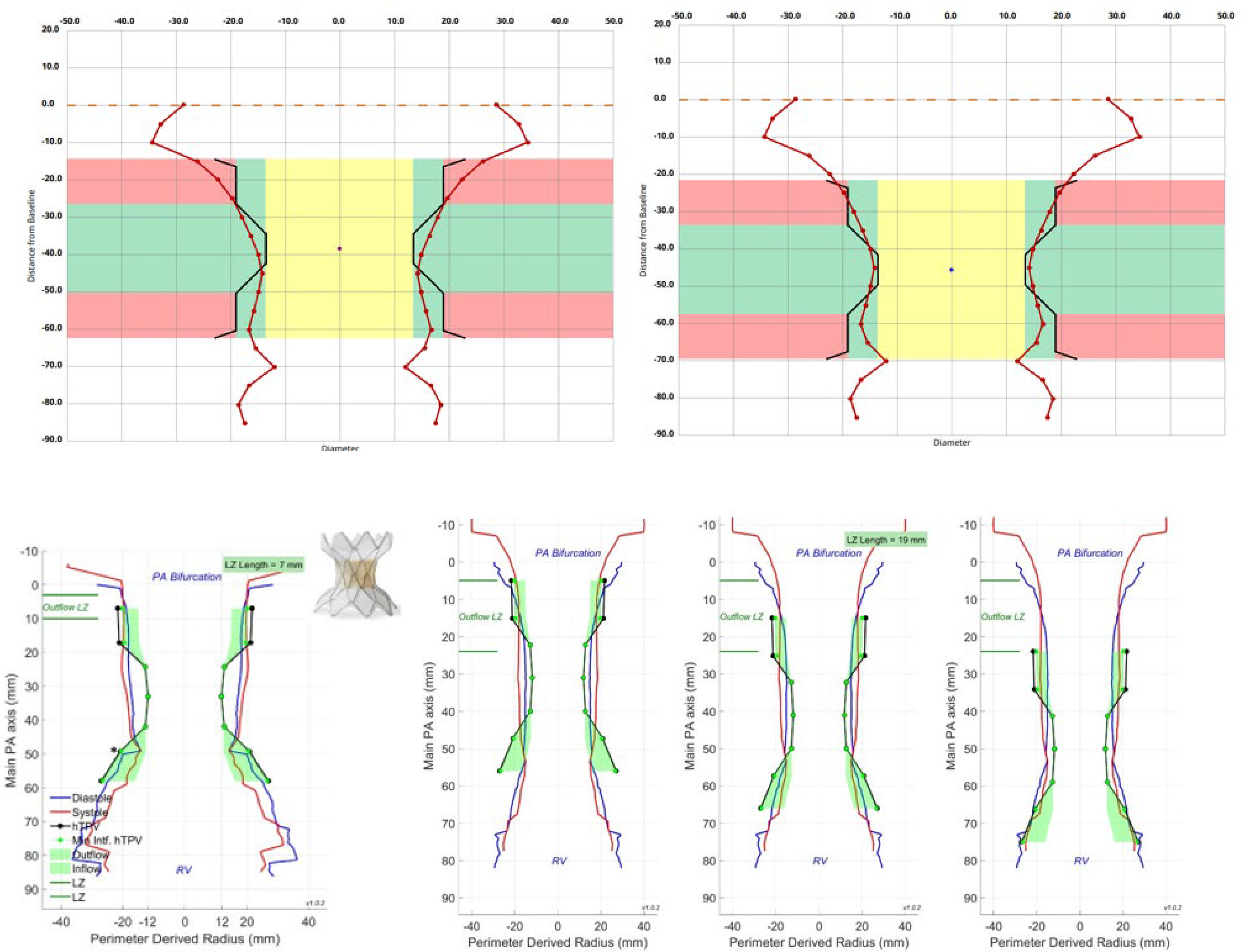
Example native RVOT perimeter plots. Example perimeter plots with the potential valve positions for Alterra (top) during systole (left) and diastole (right) and for Harmony (bottom) displaying both diastolic (blue line) and systolic (red line) measurements. The black lines represent the unconstrained Alterra and Harmony valve within patient plots with the perimeter-derived radius on the x-axis. The perimeter plots are constructed to assess device fit and to evaluate different potential landing zones within the RVOT. The green areas in the inflow and outflow of the THV device represent device interaction (oversizing) with the anatomy, which is needed for anchoring. The valve housing unit is located in the waist of the device. RVOT, right ventricular outflow tract; THV, transcatheter heart valve.
Conduit and Bioprosthetic Valve Landing Zone
Patients who have previously undergone RVOT bioprosthetic valve or surgical conduit placement should have an evaluation of the RVOT, the conduit or bioprosthetic valve, and the relationship of the distal conduit to the branch pulmonary arteries. There is not significant dynamism in a fixed-dimension surgical conduit or bioprosthetic valve and measurements in systole and diastole are less likely to have significant variation. There is dynamism of the RVOT, however, that can be assessed by echocardiography, MRI, CCT, or conventional angiography. The valve-in-valve (ViV) app can provide important information regarding true internal dimension, fluoroscopic marker location, and sizing for ViV procedures. 84
The presence of calcification should be assessed and can be described using semiquantitative grading scales as none, mild, moderate, and severe, as well as protruding and nonprotruding and whether the calcification is circumferential. Further, any symmetric, bulbous-like outpouchings or focal narrowing should be documented. The RVOT should be evaluated for proximal muscle bundles or distal obstruction that may not be relieved by TPVR placement.
Determining Etiology of Bioprosthetic Valve Dysfunction
The etiology of conduit or bioprosthetic valve dysfunction should be determined, if possible. In many patients, it will be patient-prosthesis mismatch due to somatic growth. Hypoattenuated leaflet thickening can be the cause of early valve gradient when associated with restricted leaflet motion, which can respond to anticoagulation obviating the need for valve replacement. 33 Endocarditis or thrombus can result in RVOT obstruction or new valvular regurgitation, and vegetations can be difficult to visualize by transthoracic or transesophageal echocardiography. Calcified or noncalcified valve leaflet thickening due to bioprosthetic valve degeneration may also cause obstruction and would not be amenable to medical therapies. Figure 1 illustrates Halt, endocarditis and both calcified and non calcified leaflet degeneration. Figure 10 illustrates extensive and circumferential RVOT conduit and branch pulmonary artery calcification. Recommended reporting elements for a RVOT conduit or bioprosthetic valve are listed in Figure 11.
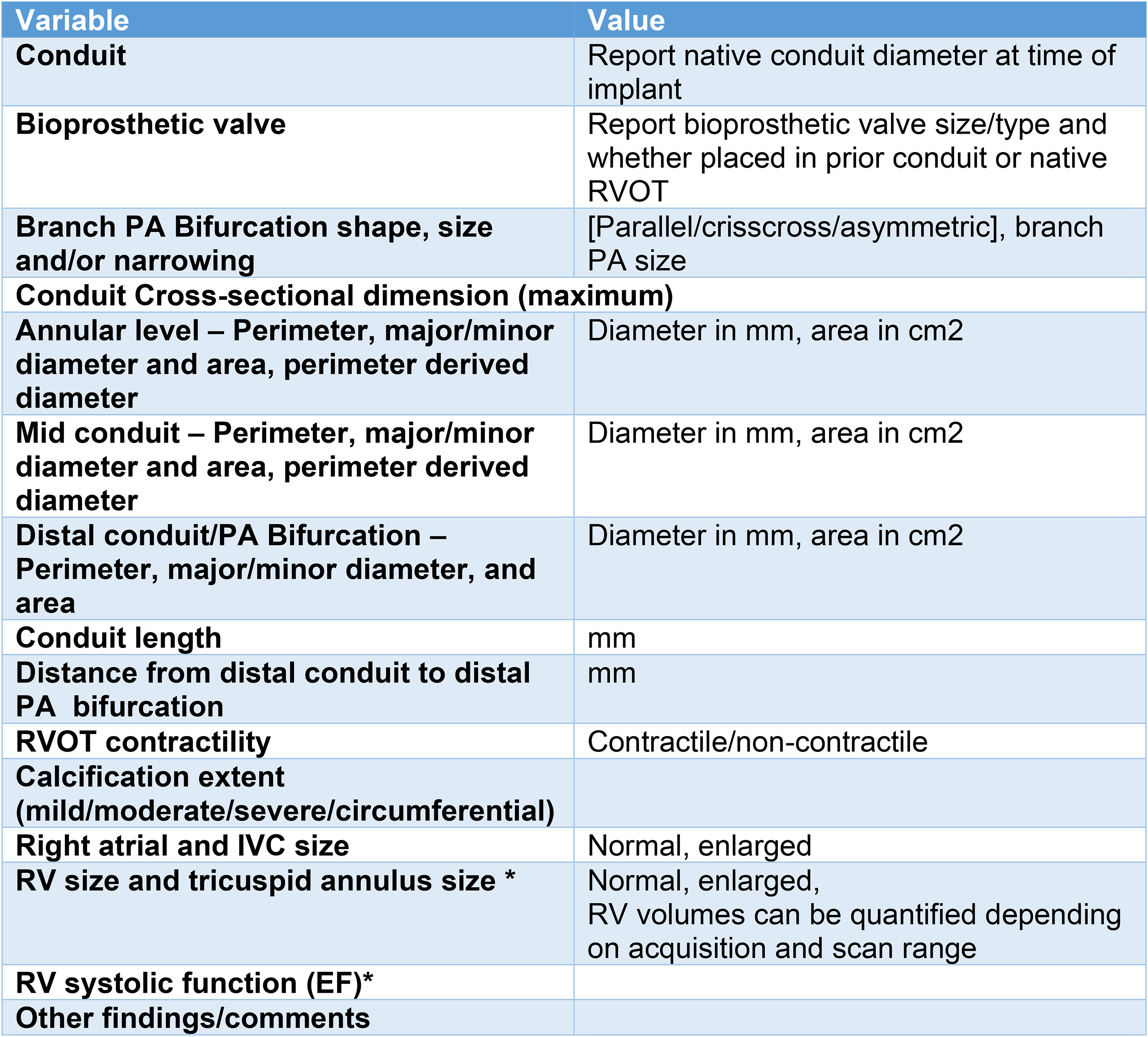
Recommended RVOT reporting elements (bioprosthetic valve or conduit). *Many patients will have had prior advanced imaging, and the CT is done specifically to assess the RVOT. Some centers may use an ECG-gated cardiovascular computed tomography (CCT) scan to both assess RV size and function in addition to planning for percutaneous PVR. If so, the RV volumes and EF can be reported. Abbreviations: IVC, inferior vena cava; MPA, main pulmonary artery; PA, pulmonary artery; PVR, pulmonary valve replacement; RVOT, right ventricular outflow tract.
Standardized Approach for Cardiovascular Computed Tomography Assessment of Coronary Anatomy in the Dysfunctional Right Ventricular Outflow Tract in Preparation for TPVR or Surgical Intervention
A recognized risk of TPVR is coronary artery compression, which is a rare but potentially catastrophic procedural complication. The risk of coronary artery compression has been shown to be approximately 5% to 6% among patients evaluated with simultaneous RVOT balloon inflation and aortic or coronary angiography performed prior to valve implantation.85–87 Coronary artery compression has been reported from RVOT conduit stenting, from branch pulmonary artery stenting, as a complication of conduit rupture, and with TPVR implantation. Both the right and left coronary artery systems can be compromised with acute or subacute clinical presentation. Coronary compression has resulted in rare sudden death, need for urgent surgical conduit replacement, and percutaneous coronary intervention.88–99 Balloon expandable valves may have a higher risk profile than self-expanding devices, which have not been shown to cause coronary compression to date. Pulmonary hypertension is common in ACHD patients and is an independent risk factor for coronary compromise in a dilated RVOT.100,101
The 2018 ACHD recommendations from the American College of Cardiology state that before any surgical or percutaneous intervention in adult patients with TOF, the origins and proximal course of the coronary arteries should be delineated. For patients with d-TGA, benchmark assessment of the anatomic course and patency of the coronary arteries is considered prudent, and coronary angiography is also recommended in all patients who have undergone surgical coronary reimplantation.27,102 Additionally, coronary artery imaging for evaluation of coronary lesions that may need intervention at the time of valve intervention is recommended in high-risk patient subsets, including men >40 years of age and postmenopausal women prior to surgical valve intervention. 103 Practitioners may choose to optimize CCT for the assessment of both the RVOT and coronary arteries in the same scan for these higher risk patients. An adult cardiac imager experienced in coronary CTA should over-read the coronary artery segments for atherosclerotic disease. Radiation doses for coronary angiography and for comprehensive CHD evaluation in the current era are significantly reduced over historical estimates.79,104 Invasive coronary angiogram may also be performed at the time of TPVR in selected patients.
High-risk anatomic features for coronary compression with balloon-expandable TPVR include98,105:
Anomalies in the origin and trajectory of the coronary arteries: Coronary artery anomalies have been shown to be predictive of coronary compromise, particularly if the coronary course is between the aorta and RVOT in either a vertical or horizontal orientation. This is best imaged with aorta in short axis and pulmonary artery in long axis with the potential landing zone visualized. There is a higher risk of coronary compression if the coronary artery runs between the aorta and the PA at the level of the proposed landing zone. Underlying diagnosis of conotruncal abnormality: This is associated with clockwise rotation of the coronary arteries placing the origin between the aorta and PA, even when arising from the appropriate aortic sinus. High origin of the coronary artery from the sinus of Valsalva or above, running posterior and below the conduit and landing zone. Coronary artery course adjacent to or just posterior to the landing zone, particularly if acute angulation from the aortic root. Coronary artery distance < 3mm from the RVOT landing zone, <8 mm from the sternum. Loss of intervening fat between the coronary and RVOT conduit. Coronary artery near areas of calcification and scar tissue, where conduit rupture is higher risk. Pulmonary hypertension (highest risk when the PA diameter is >4 cm or the PA/AO diameter ratio is >1.2)100,106,107
Characteristics of the Conduit and TPV that May Predict Coronary Compromise
The potential RVOT landing zone should be defined. The coronary artery course that runs directly adjacent to a landing zone (particularly if stenotic) is considered high risk. The risk of compression is determined by the underlying RVOT substrate, TPVR device, and need for preimplant RVOT rehabilitation including balloon angioplasty and stenting. For example, the presence of a stented bioprosthetic surgical valve is protective if the TPVR device is positioned within the stent frame of the surgical valve and not fractured during the procedure. Similarly, the risk of coronary compression is thought to be very low with self-expanding valves designed to treat pulmonary regurgitation in the native RVOT, owing to less radial force. Providing a 3 and 4D (3D + motion) evaluation can be helpful to visualize the coronary artery, aorta, and pulmonary artery/conduit simultaneously. Newer software platforms can offer an “intraluminal view” that optimizes this type of visualization. Reviewing the study with the interventionalist and understanding the proposed device aids in patient and device-specific assessment of coronary compression risk (Figures 12-15).
Standardized Approach for Cardiovascular Computed Tomography Assessment of the Dysfunctional Right Ventricular Outflow Tract in Preparation for Surgical Intervention
Surgical PVR has been performed for decades, with early and late outcomes that are firmly established. Operative mortality is roughly 1% and freedom from reintervention is approximately 95% at five years.108–111 Long-term outcome data now extend well beyond 20 years. The performance of pulmonary valve substitutes is strongly influenced by patient age and prosthesis size.5,112 The emergence of TPVR has clearly transformed PVR therapy, but outcomes should be evaluated compared to surgical PVR outcomes which are still considered to be the benchmark. 113
Current surgical options for PVR include stented bioprosthetic valves,114–116 (porcine and pericardial), homografts8,117,118 (pulmonary, aortic, and femoral vein valved conduits), xenografts119,120 (bovine jugular venous valved conduits), heterografts 121 (Dacron graft stented porcine valved conduit), and surgeon-made conduits122,123 (GoreTex valved conduit).124–127 Pulmonary valve repair is appropriate for specific circumstances, such as isolated cusp injury resulting in significant pulmonary regurgitation but is otherwise rarely employed for pulmonary valve dysfunction.
Advanced imaging is a critical component of preoperative evaluation and surgical planning for PVR. 128 Specific considerations include pulmonary annular dimension, MPA dimension, and length, the presence or absence of branch pulmonary artery stenosis, the geometry and character of the proximal and distal RVOT as well as the RV body, the location and branching pattern of the coronary arteries, and the relationship of the great arteries and cardiac mass to the sternum.
Pulmonary annular and MPA dimensions are important determinants of appropriate choices for PVR. Accurate sizing determines if a stented bioprosthesis can be positioned within the RVOT with an adequate effective orifice area for acceptable hemodynamic performance. Annular size need not match prosthesis size exactly, as most stented valves are implanted with an onlay patch, which covers a portion of the anterior aspect of the prosthesis, but a small annulus will result in a large amount of anterior patch material and little native outflow tract tissue within which to seat the valve. Thus, in the setting of a smaller annulus or MPA (usually in the setting of intervention in childhood or early adolescence), the more favorable hemodynamics of homografts and xenografts make these more attractive choices for PVR. When the MPA is short, care must be taken when implanting a stented bioprosthetic valve, such that none of the struts of the valve obstruct the orifice of the right pulmonary artery, which may have an early take-off.
Branch pulmonary artery stenosis has been shown to have a significant impact on PVR longevity. 5 Complete and accurate characterization of branch pulmonary artery anatomy is, therefore, an important component of surgical planning, and the operation should effectively address any significant branch pulmonary artery obstruction. This may be addressed by patch angioplasty of the pulmonary arteries, separate from PVR, but orifical or proximal narrowing is often most efficiently treated by extension of the distal end of homograft, xenograft, or heterograft conduits.
Anatomies that require PVR are frequently associated with proximal RVOT obstruction due to RV hypertrophy or, more often, anomalous RV muscle bundles. Echocardiography may provide excellent structural and hemodynamic characterization of the presence and significance of proximal RVOT obstruction, but CCT may provide important additional details, which aid in surgical planning. Conservative division and/or resection with preservation of RV architecture are the principles of surgical therapy for proximal RVOT obstruction. Accurate characterization of RV muscle bundle origin and insertion facilitates execution of these principles. Additionally, thinning and dyskinesis of the RV free wall, aneurysm related to large transannular patch reconstruction of the RVOT, or overly aggressive RV muscle resection at the time of complete repair should be identified and addressed at the time of PVR by right ventricular remodeling. 129 This is true regardless of whether planned PVR is by traditional surgical or “hybrid” approach. 130
Coronary artery anatomy is an extremely important aspect of CCT evaluation prior to surgical PVR. Anomalies of coronary artery pattern that have a direct and major impact on the conduct and/or feasibility of surgical PVR include coronary arteries that course anterior to the RVOT such as left anterior descending from the right coronary artery, single left coronary with circumnavigation of the pulmonary root by the right coronary, dual left anterior descending arteries, and early take-off of large conal or left diagonal coronary arteries. As previously noted in the section on coronary artery assessment prior to TPVR, in TOF and other conotruncal anomalies, dextro-rotation of the aortic root results in juxtaposition of the right coronary sinus and the rightward pulmonary root. This places the proximal right coronary artery near the interarterial groove and prone to injury during preliminary dissection of the pulmonary root in preparation for PVR.
Finally, since most PVRs are performed in previously operated patients, planning for sternal reentry is critical to maximize patient safety and outcome. In the setting of right heart enlargement due to PR, the RV mass is commonly closely applied to the posterior sternal table (Figure 16). In the setting of RVOT obstruction, the RV is hypertensive, and bleeding that is incurred can be rapid and difficult to control. The appendage of the enlarged right atrium can also be situated so as to be at risk of injury. The position of the often large and dextro-rotated aorta places it in a more anterior position relative to the sternum. This is perhaps the most important risk for sternal reentry, as aortic injury can be a catastrophic complication. Previously placed conduits are frequently adherent to, or grown into the chest wall. Occasionally, such conduits have been routed in a retrosternal location, a situation that may be extremely hazardous at the time of reentry. Advanced imaging is the sole mechanism by which these relationships can be accurately assessed. All sternal reentry operations should involve preparation for femoral arterial and venous cannulation. The patency and dimension of the femoral vessels, therefore, should also be assessed (although this information is not routinely acquired in current protocols for PVR). When reentry is deemed to be potentially hazardous based upon preoperative imaging, dissection of the femoral vessels should be performed, and consideration given to cannulation and institution of cardiopulmonary bypass prior to reentry. Not only does this provide for circulatory support in the event of bleeding but also it minimizes the chances of cavitary entry, as the heart is decompressed by shifting intravascular volume to the venous reservoir of the heart-lung machine. 131
Thus, CCT informs surgical PVR in many dimensions. Optimization of outcome depends on careful characterization of each of these features, with clear and precise communication between the surgical and imaging teams regarding imaging findings. 131 Figure 16.
Transcatheter Pulmonary Valve Replacement: Procedural Overview
It is important for advanced imagers to be familiar with basic procedural and device characteristics to better inform patient and device selection.
Patient selection: Data from a large (n = 2,476) International Registry on TPVR revealed that 38% of TPVR procedures are performed to address obstruction, 30% for PR and 32% for mixed disease (obstruction and regurgitation). 52 The most common underlying diagnoses leading to TPVR are TOF (55%) and other conotruncal abnormalities (19%), such as truncus arteriosus, double outlet right ventricle, and d-TGA. At present, the majority of TPVR procedures are performed in previously implanted homograft conduits (45%) or stented bioprosthetic valves (22%). Approximately 16% of TPVR procedures are performed in native or patched RVOTs, a number that is expected to increase with the introduction of new TPV platforms specifically designed for the native/patched RVOT. The type of anatomic substrate has important implications for procedural planning and valve selection (Table 2). 3D image fusion software can be used for guidance of transcatheter interventions in CHD but is not yet routine for TPVR. 132
Vascular access and other considerations: Most TPVR procedures are performed under general anesthesia via transfemoral access 53 although other routes for valve delivery (ie, transjugular) have been used.
Conduit Rehabilitation and Coronary Compression Testing
Prior to TPVR in conduits where stenosis is the prominent pathology, a conduit typically requires rehabilitation, with serial balloon angioplasty and subsequent stent implantation. The conduit should be expanded gradually, by increasing balloon diameters in ∼2 mm increments, to at least its original size and may be expanded larger, in some circumstances. Once the conduit is expanded to the size of the desired stent implantation, the coronaries are evaluated during balloon dilation of the conduit to evaluate for compression. Prestenting is then usually performed. In the International TPVR registry, prestenting was used in 72% of cases prior to valve implantation, and covered stents were used in 20% of these cases. Common indications for prestenting include stenosis longer than the length of the TPV, prevention or treatment of conduit tears (in the case of covered stents), and use of the Melody TPV, which has a platinum-iridium stent that is prone to fracture in the absence of prestenting. 54
Transcatheter Valve Options for Treatment of Native Right Ventricular Outflow Tract
Balloon expandable valve platforms such as the Melody TPV and SAPIEN THV have been utilized off-label for implantation in the dysfunctional native and patched RVOT but are limited in the application based on size (≤24 mm for Melody valve and ≤29 mm for the SAPIEN 3 THV). This has prompted the development of self-expanding hourglass-shaped covered platforms that allow for frame fixation in the outflow and inflow regions of the native RVOT. The Harmony TPV was FDA approved in March of 2021 for treatment of PR in native RVOT anatomy. The Harmony valve is a porcine pericardial valve that is sutured into an asymmetric hourglass-shaped self-expanding fully covered stent platform. A polyester fabric is then sutured to the nitinol (nickel-titanium alloy) stent struts. The valve is treated with amino-oleic acid (AOA) to reduce calcification. The Harmony TPV platform comes in two commercially available sizes (valve sizing chart—Table 6), the TPV 22 and the TPV 25. The TPV 22 has a 22-mm porcine pericardial valve and is narrower and longer than the TPV 25, which contains a 25-mm porcine pericardial valve. Both valve platforms are self-expanding and are deployed by unsheathing the valve within the RVOT. The Alterra Adaptive Prestent, which was FDA approved in December 2021, is a partially covered laser-cut nitinol self-expanding radiopaque frame with polyethylene terephthalate fabric covering. It is designed as a docking adaptor for the 29 mm SAPIEN 3 THV. The Alterra is placed initially in the RVOT as an adaptive prestent that is followed (typically during the same procedure; however, staging is a possibility) by placement of the 29 mm SAPIEN 3 within the 27 mm diameter waist of the prestent (Figure 17; Table 2).
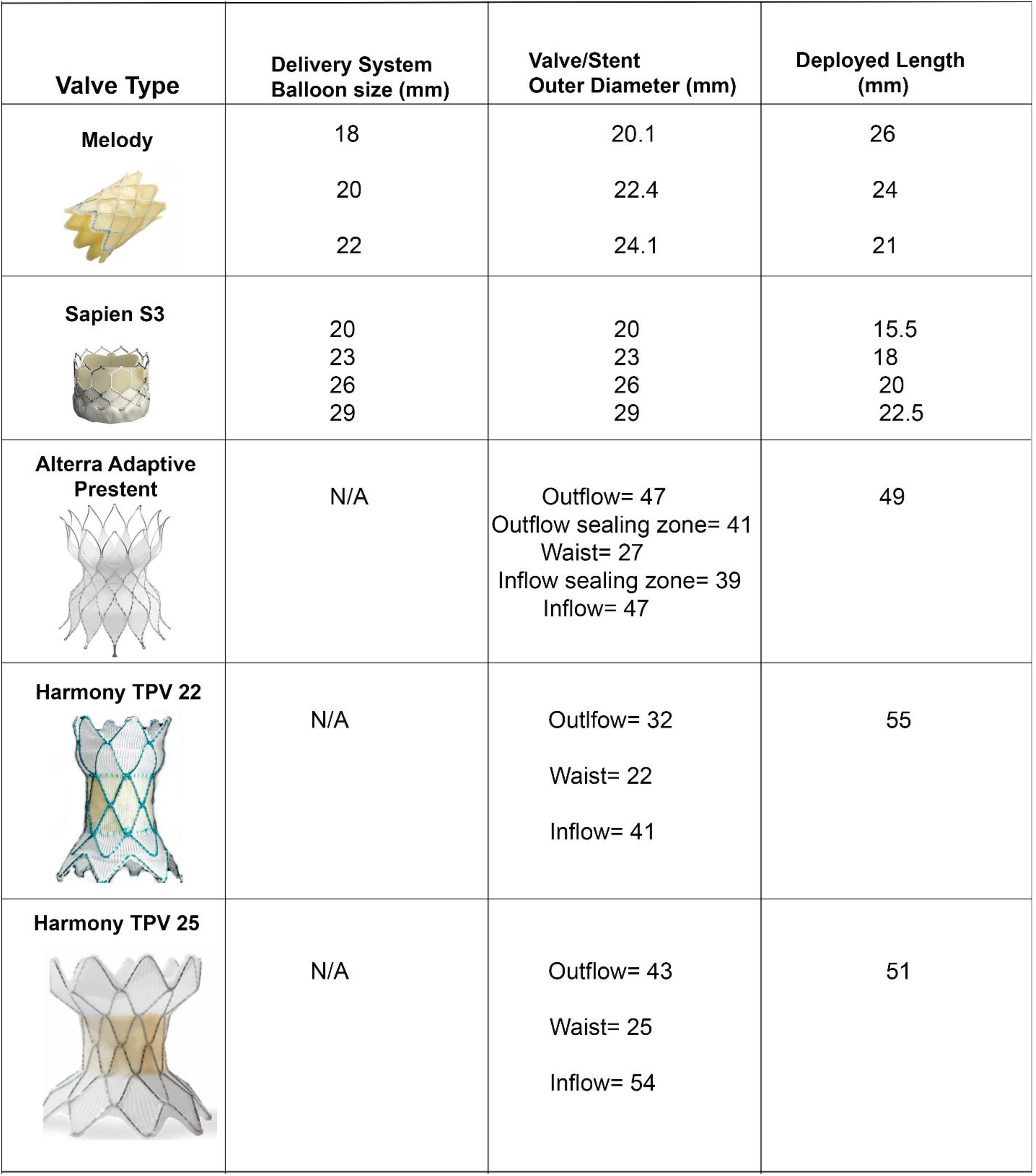
Transcatheter valve sizing chart and illustration of currently approved TPV in the US. This table lists currently approved percutaneous valves for treatment of RVOT dysfunction. The valve image, delivery system, device diameter, and deployed length are illustrated. RVOT, right ventricular outflow tract.
Conclusions
Pulmonary valve and RVOT dysfunction is common in patients with CHD, and surgical and transcatheter interventions are frequently needed to address both pulmonary stenosis and regurgitation. Cardiovascular computed tomography has emerged as an important modality in determining optimal candidates for surgical and TPVR and to guide intervention. Cardiac advanced imagers in both pediatric and adult medicine may benefit from guidance to provide a reproducible assessment of RVOT anatomy to plan and guide PVR. The present document provides a comprehensive approach to CT imaging protocols, device selection, and procedural planning.
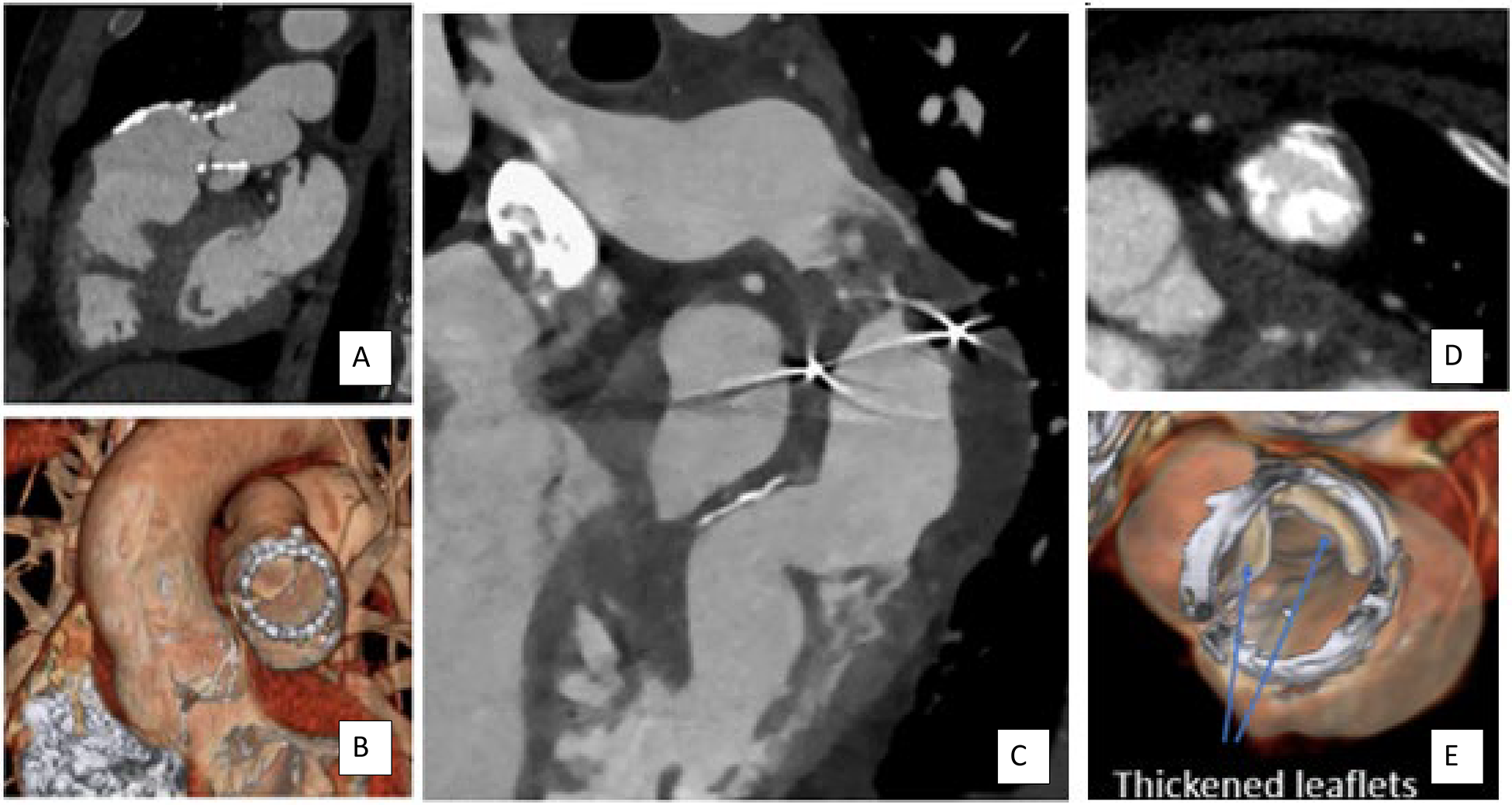
Determining Etiology of RVOT Dysfunction. (A) Hypoattenuated leaflet thickening (HALT) with restricted leaflet motion (B) causing increased gradient after TPVR. This can be progressive but may be treatable medically. (C) An example of endocarditis with vegetation in an RVOT conduit after Rastelli procedure. Calcified (D) and noncalcified (E) leaflet thickening are seen within a bioprosthetic pulmonary valve. RVOT, right ventricular outflow tract; TPVR, transcatheter pulmonary valve replacement.
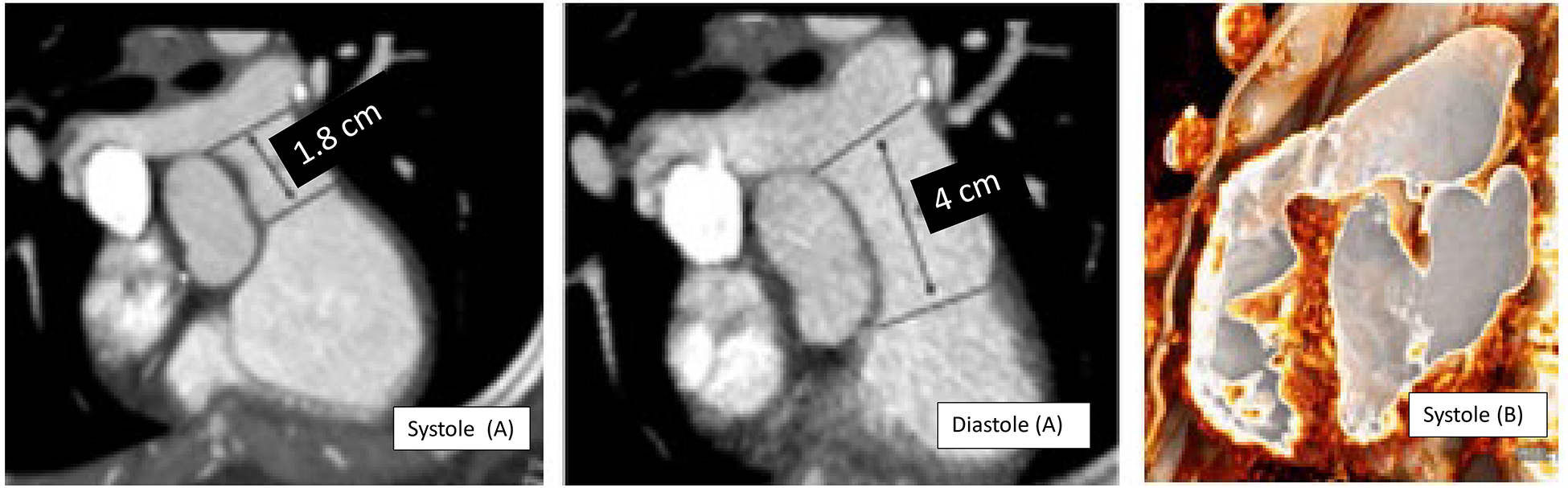
Dynamism of the Native and Patched RVOT: Longitudinal Shortening. Longitudinal shortening describes the change in pulmonary annulus to pulmonary bifurcation length in the long axis of the pulmonary artery during the cardiac cycle. On the left (A), 2D images in systole and diastole illustrate longitudinal shortening of the MPA in a patient after transannular patch with an aneurysmal RVOT. The contractile portion of the RVOT is smaller in systole but the patched RVOT and MPA are often enlarged. The RVOT in a patient with an intact annulus tends to be smaller in systole (B). These images illustrate the importance of using the longest linear dimension to determine potential landing zone length and the maximal RVOT dimension to assess feasibility of TPVR. RVOT, right ventricular outflow tract; TPVR, transcatheter pulmonary valve replacement.
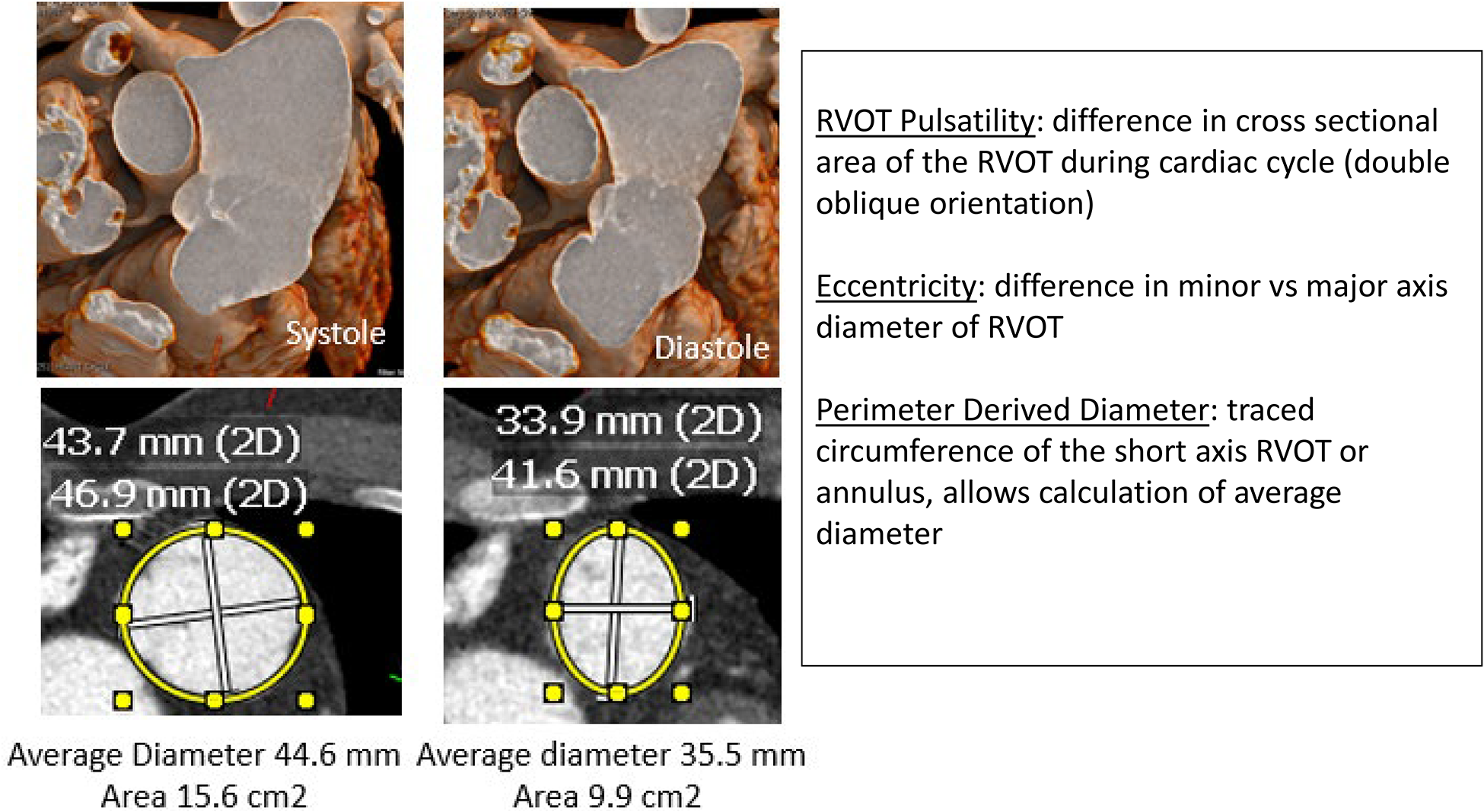
Dynamism of the Native and Patched RVOT: Pulsatility and Eccentricity/Ellipticity. This case illustrates circumferential pulsatility and ellipticity of the short axis of the annular plane of a dilated patched RVOT. Note the difference in short axis area and in minor and major diameter during systole and diastole. Major and minor diameter, area, perimeter, perimeter-derived (average) diameter should be measured at the distal RVOT, the annular plane, and the mid MPA. The maximal dimensions at the potential landing zone are used to assess feasibility of TPVR. RVOT, right ventricular outflow tract; TPVR, transcatheter pulmonary valve replacement.
Examples of Virtual Valve Placement for TVPR. These two images are examples of a virtual valve placement within the 3D data set of the native RVOT for the Alterra pre-stent (right) and the harmony valve (left). RVOT, right ventricular outflow tract; TPVR, transcatheter pulmonary valve replacement.
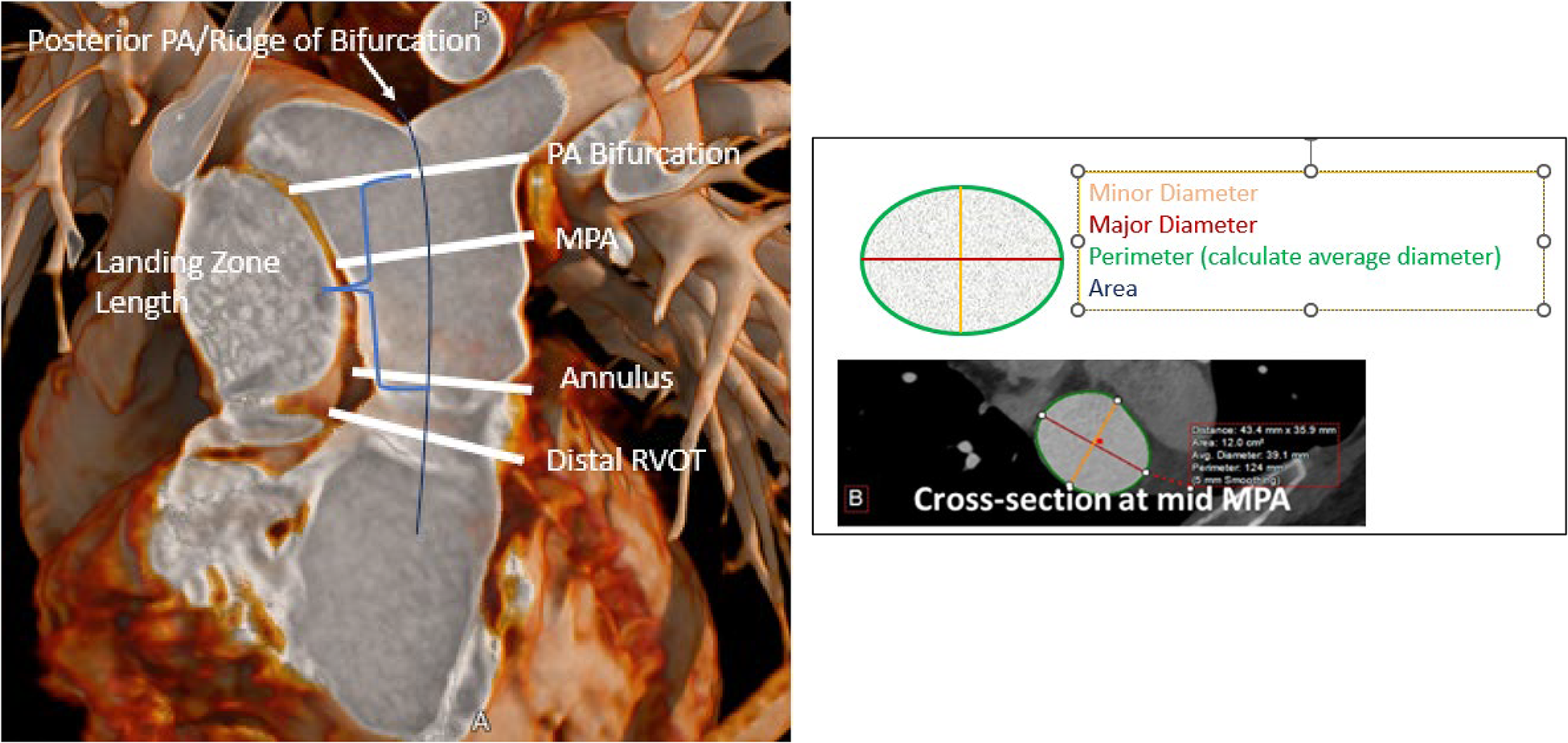
Standard Measurement Planes in the RVOT. This 3D reconstruction illustrates the standard measurement planes in the RVOT and example measurements from a short-axis image of the RVOT. The landing zone of TPVR valves will be between the annulus to the PA bifurcation, or from the distal RVOT past the PA bifurcation for self-expanding valves. Note the eccentricity of the mid-MPA measurement. RVOT, right ventricular outflow tract; TPVR, transcatheter pulmonary valve replacement.
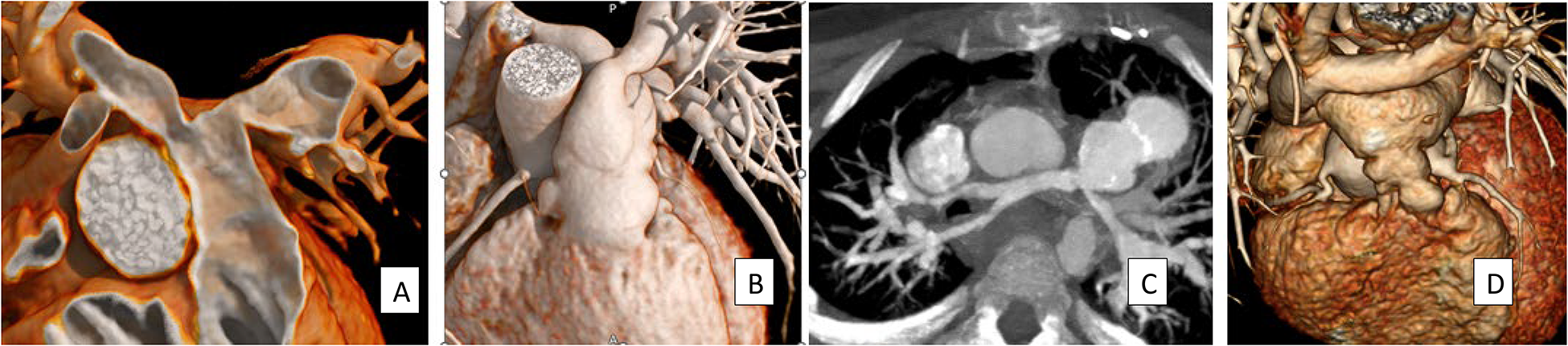
Examples of Variation in Branch Pulmonary Artery Anatomy. (A) A 3D reconstruction illustrating narrowed branch pulmonary arteries from the distal RVOT, (B) Criss-cross branch pulmonary arteries and (C) diffusely narrowed pulmonary arteries from the distal conduit after unifocalization in a patient with major aortopulmonary collateral arteries (MAPCAs). In these cases, there is no significant distance between the branch pulmonary artery take-off and the posterior pulmonary artery (ridge of bifurcation), which has implications for placement of self-expanding valves. (D) The usual orientation of the branch pulmonary arteries after the arterial switch operation and the LeCompte maneuver with the innominate vein overlying the branch pulmonary arteries. There is a relatively short neo-RVOT directly anterior to the neo-aorta and branch pulmonary arteries on the right and left side of the aorta. Note the origin of the coronary arteries from the right and left sides of the neo-aorta. RVOT, right ventricular outflow tract.
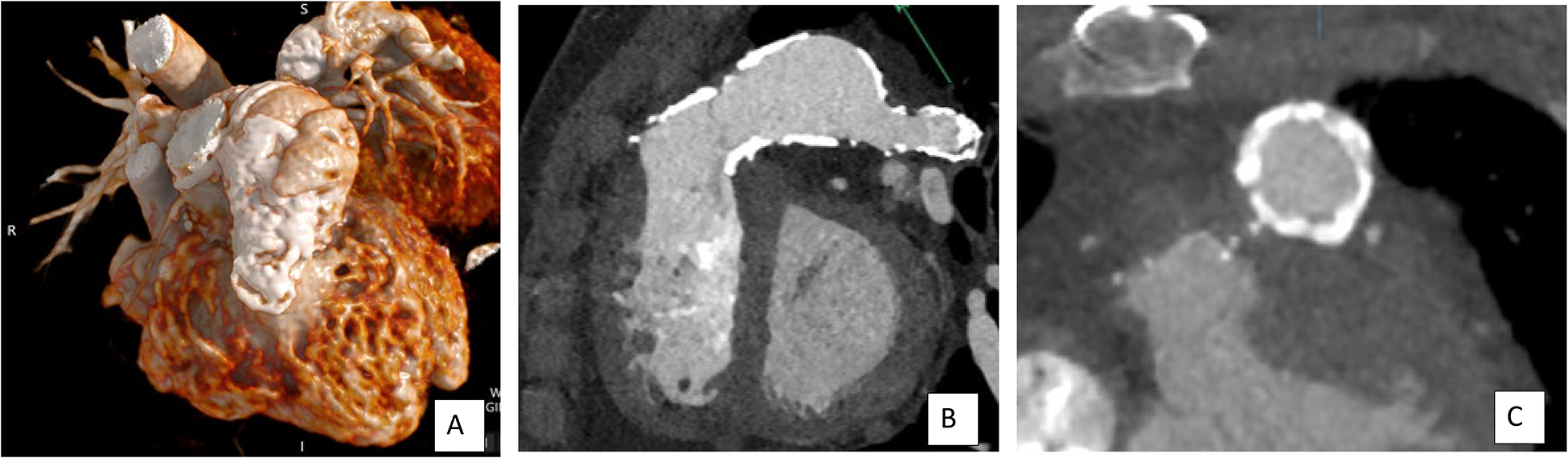
Evaluation of RVOT Calcification. The image on the left (A) is a 3D reconstruction showing extensive conduit calcification, which is also visualized on both the short axis and long axis of the RVOT 2D images (B/C). The calcification is noted to be circumferential in the proximal conduit and remains extensive through the lateral conduit and into the branch pulmonary artery, which has been patched. RVOT, right ventricular outflow tract.
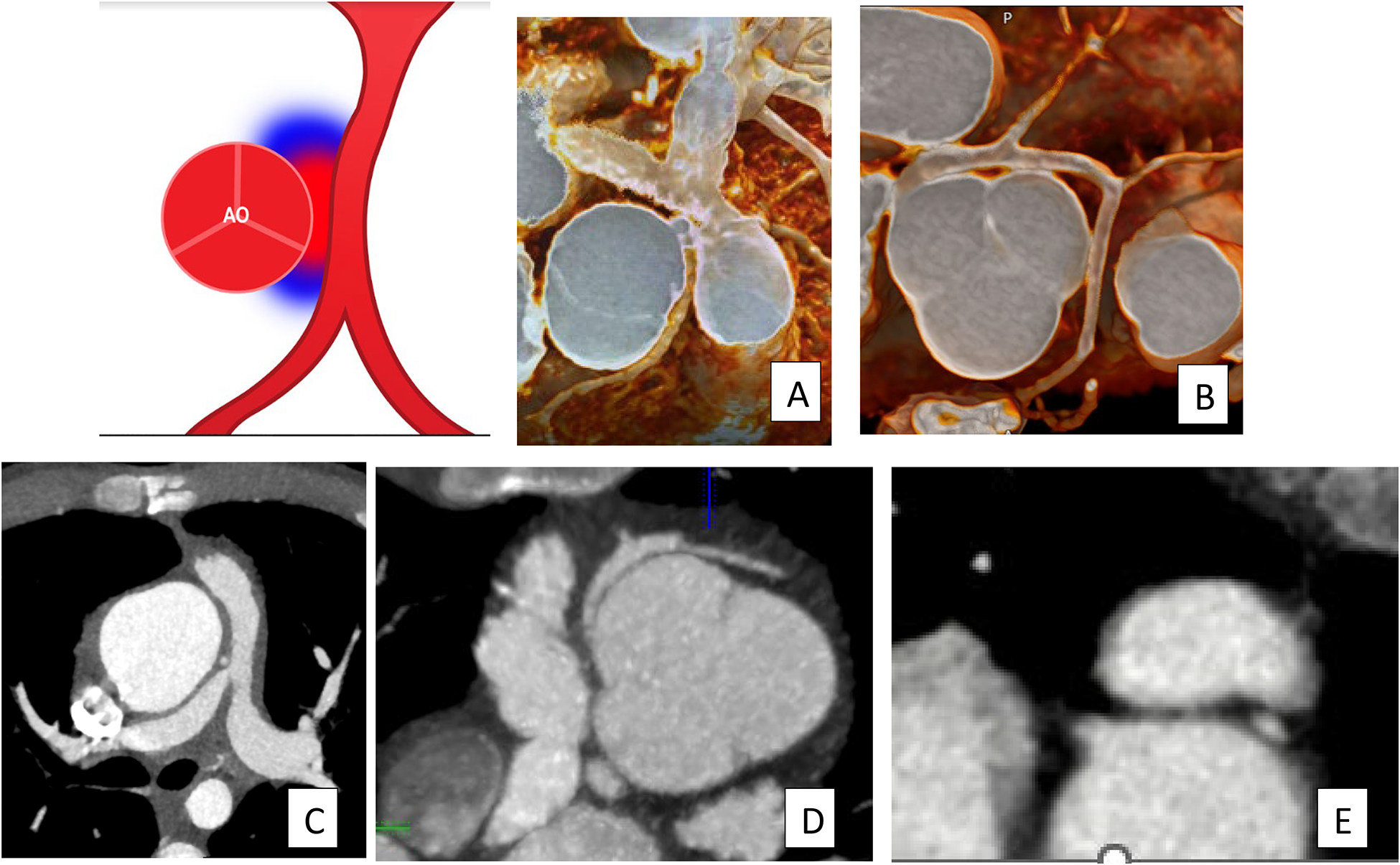
Potential for Coronary Artery Compression Based on Coronary Course Related to the RVOT. These images show coronary arteries coursing between the aorta and the RVOT, with potential for compression between the great arteries (A/B), or adjacent to a narrowed conduit (C/D). A coronary artery reimplanted posterior to the neo-RVOT after arterial switch operation may also be at risk for compression from the anterior vessel (E). RVOT, right ventricular outflow tract.
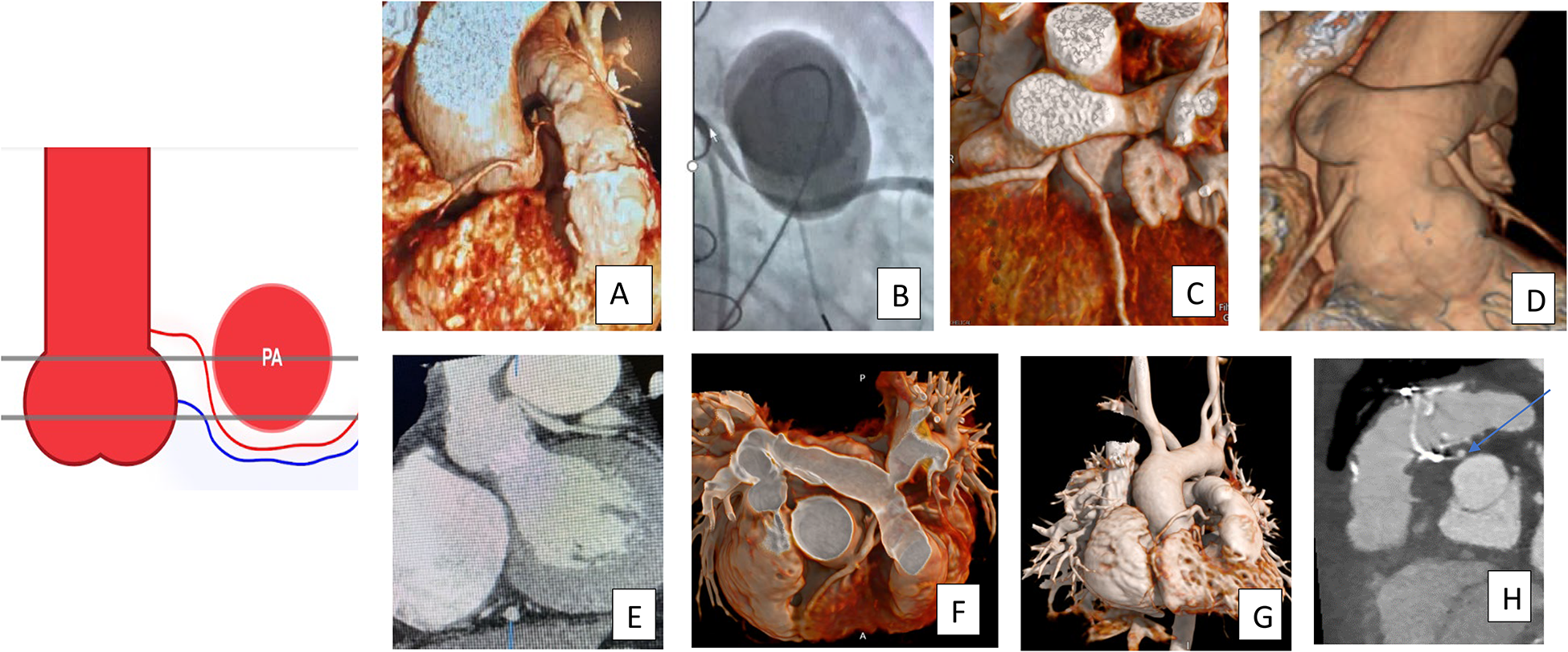
Potential for Coronary Compression Based on Coronary Course and Height of Coronary Origin. These images show coronary artery course from the superior sinus of Valsalva or ascending aorta, coursing inferior to the RVOT. (A/B) The CTA and catheter correlation with coronary compression during balloon testing. (C and D) A coronary artery reimplanted to the ascending aorta, directly posterior to the neo-RVOT after arterial switch operation. The bottom images are after repair of tetralogy of Fallot with patch (E), conduit (F/G), or bioprosthetic valve (H). These coronary arteries are at risk for compression due to height of the coronary origin relative to the RVO and/or proximity to the potential landing zone in the case on anomaly. If a coronary artery crosses the RVOT, conduits are often used in the initial repair (F/G). RVOT, right ventricular outflow tract.
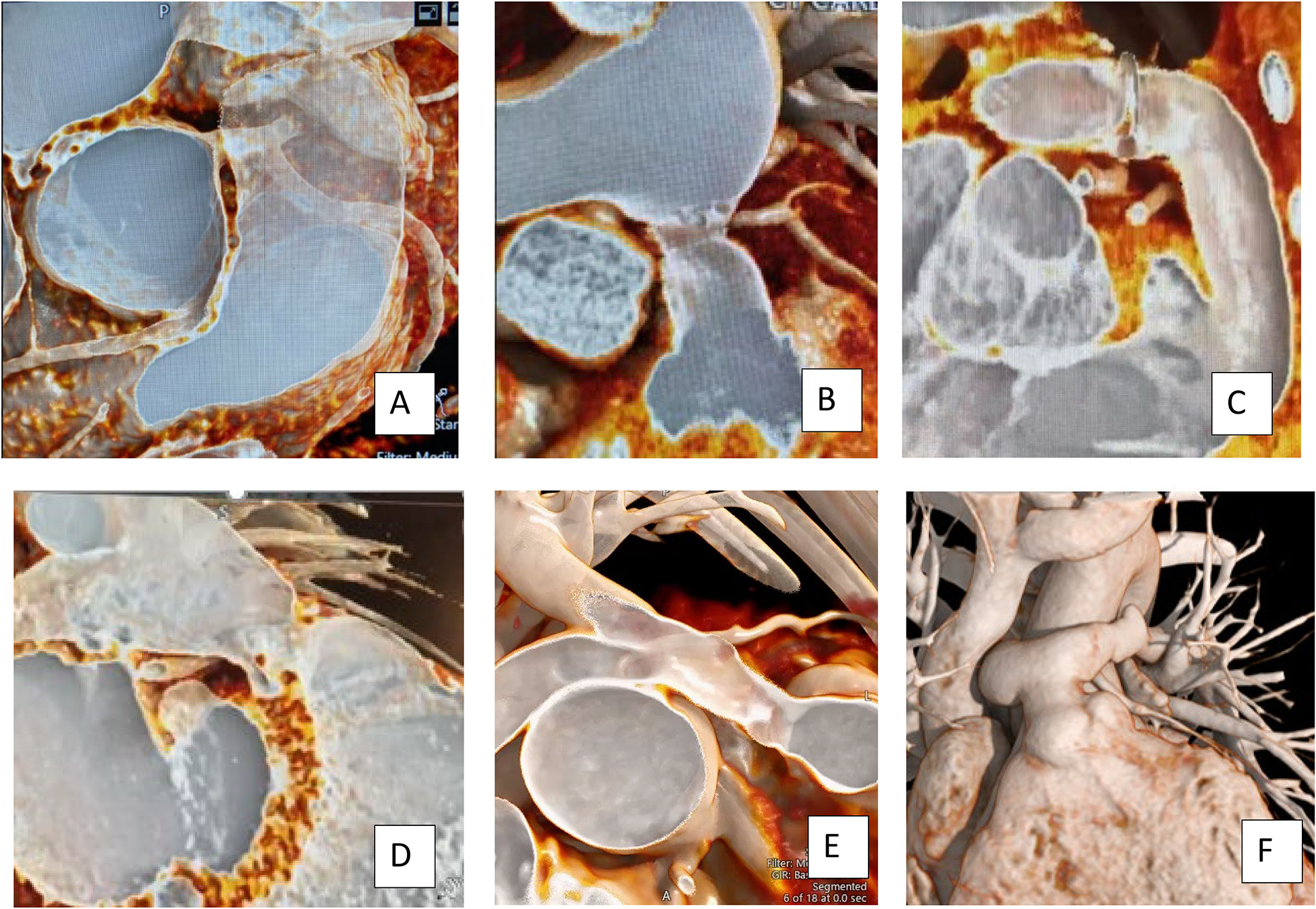
3D and ”Intraluminal“ Evaluation of the Coronary Artery Relationship to the RVOT. These 3D and 3D “intraluminal” view cardiovascular computed tomography (CCT) reconstructions allow visualization of the coronary course related to the native RVOT (A), an obstructed distal conduit (B), a bioprosthetic valve (C, D), an obstructed proximal conduit (E), and the proximal LPA after arterial switch operation with potential for coronary obstruction with ROT or LPA intervention (F). Whether or not the coronary is at risk will depend on the proposed valve type and length, and whether there will be aggressive RVOT rehabilitation including angioplasty, stenting, or bioprosthetic valve fracture prior to TPVR. RVOT, right ventricular outflow tract; TPVR, transcatheter pulmonary valve replacement.
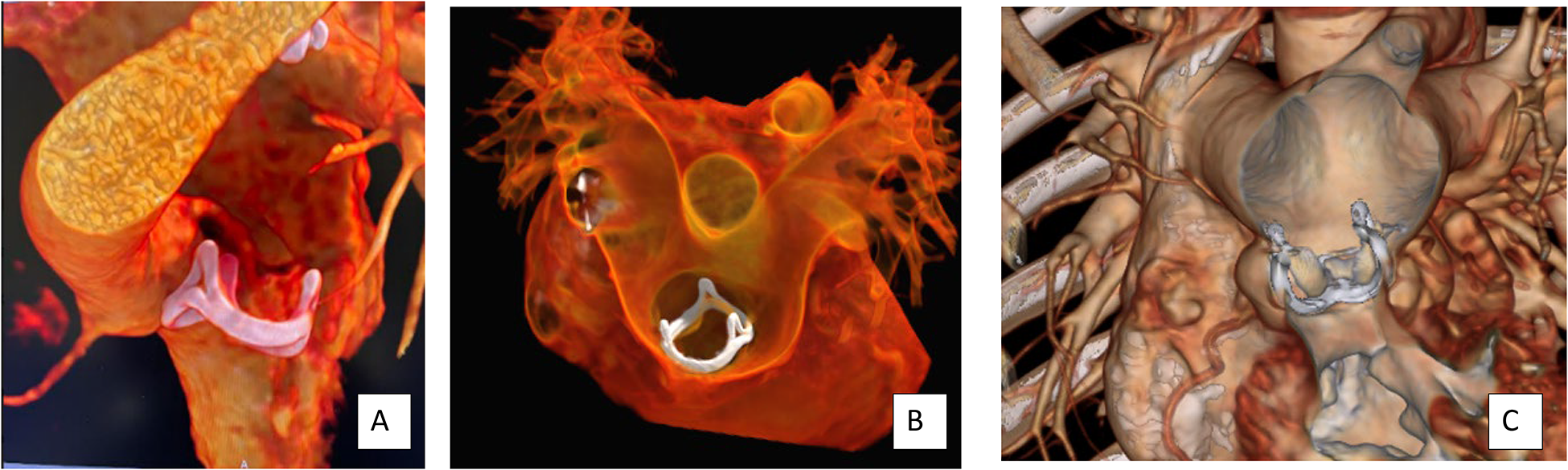
Evaluation of Bioprosthetic Valve Position within the RVOT. Visualization of a bioprosthetic valve and the position within the RVOT and the relationship to the coronary arteries can be determined prior to intervention. A bioprosthetic valve is visualized in a normally positioned RVOT (A) and in the dilated neo-RVOT after LeCompte maneuver (B/C). In both cases, the relationship of the coronary arteries to the valve ring is illustrated. RVOT, right ventricular outflow tract.
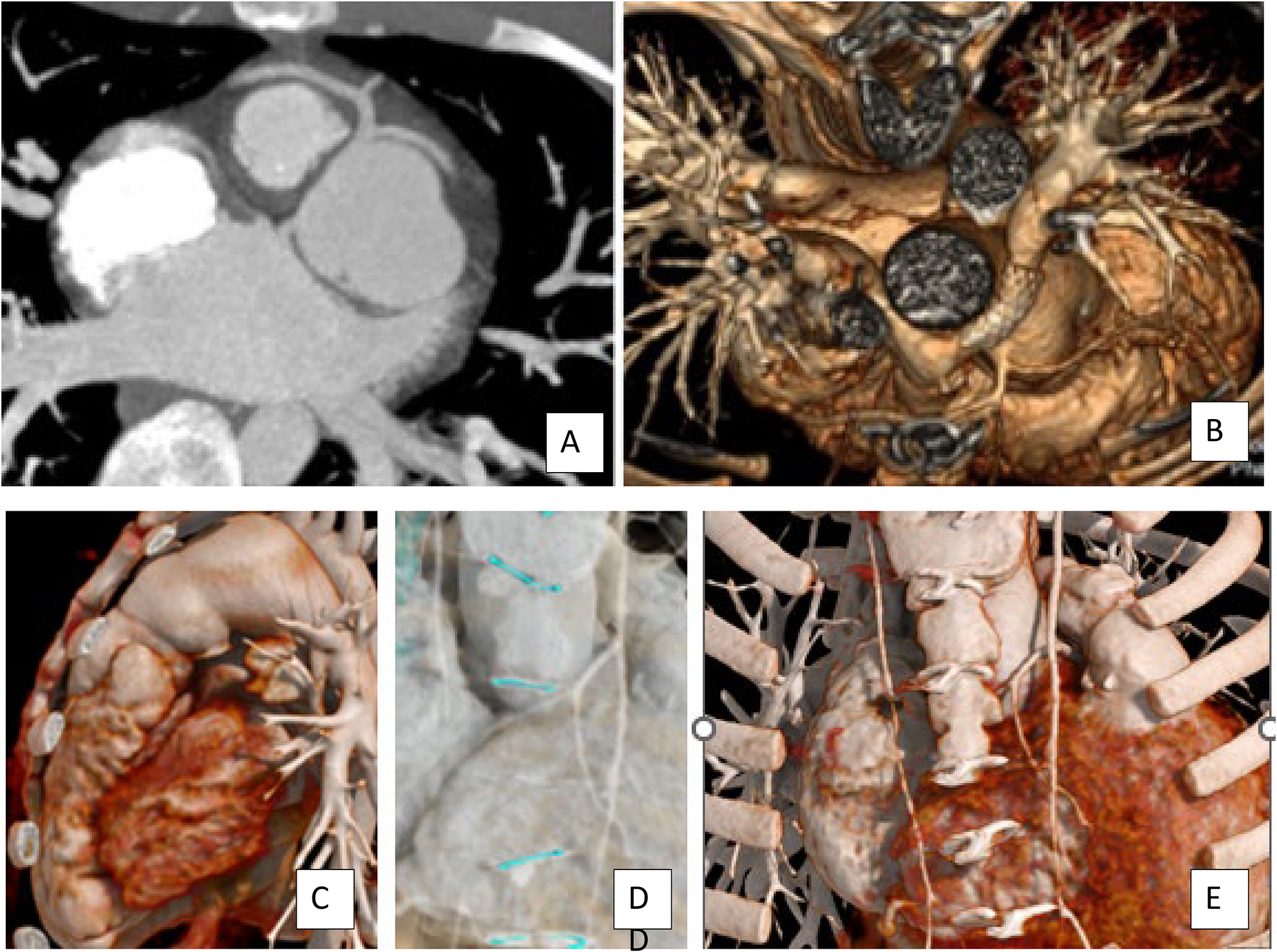
Relationship of the RVOT, Aorta, and Coronary Arteries to the Sternum. These images illustrate the relationship of the coronary artery and great arteries to the sternum. For sternal entry, coronary arteries, the RVOT, or the aorta may be directly substernal. Visualization of the sternal wires, sternum, and ribs and their relationship to the underlying cardiac structures aids in surgical planning. (A and B) Substernal coronary arteries after the arterial switch operation. (C) A dilated native RVOT directly substernal, and (D/E) different reconstructions of the aorta and coronary arteries directly posterior to the sternum and the relationship to sternal wires to guide reentry. RVOT, right ventricular outflow tract.
Supplemental Material
sj-docx-1-pch-10.1177_21501351231186898 - Supplemental material for Technical Recommendations for Computed Tomography Guidance of Intervention in the Right Ventricular Outflow Tract: Native RVOT, Conduits, and Bioprosthetic Valves
Supplemental material, sj-docx-1-pch-10.1177_21501351231186898 for Technical Recommendations for Computed Tomography Guidance of Intervention in the Right Ventricular Outflow Tract: Native RVOT, Conduits, and Bioprosthetic Valves by B. Kelly Han, MD, Santiago Garcia, MD, Jamil Aboulhosn, MD, Phillip Blanke, MD, Mary Hunt Martin, MD, Evan Zahn, MD, Andrew Crean, MD, David Overman, MD, C. Hamilton Craig, MD, Kate Hanneman, MD, Thomas Semple, MD, and Aimee Armstrong, MD in World Journal for Pediatric and Congenital Heart Surgery
Footnotes
Abbreviations
Acknowledgments
Images are from Minnesota Children's Hospital, Primary Children's Hospital, and the Minneapolis Heart Institute at Abbott Northwestern Hospital. Graphic Images (E1, E2): Kiahltone Thao, Minneapolis Heart Institute Foundation. The authors thank fantastic imaging techs who are the heart of any congenital CT imaging program (Jana, Dave, Lynn, Nicole, Megan, Alyece, Bonnie, Casey, Daren, Katie, Corrinne) KH.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online. Writing Group members' affiliations and any relevant relationships with industry: See Supplemental Appendix A. List of reviewers and their affiliations and any relevant relationships with industry disclosures: See Supplemental Appendix B.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

