Abstract
Background
Patient temperature during extracorporeal membrane oxygenation (ECMO) is commonly managed by dedicated heating units (HUs) that are integrated into ECMO circuitry. Currently, no HU has received approval for ECMO by the FDA in the United States. Older FDA-approved HUs have been implicated in life-threatening patient infections and are no longer manufactured or available for use in the United States.
Methods
We performed laboratory tests to evaluate the safety and efficacy of the Micro-Temp and the HTP-1500 HU systems that are potentially suitable for use in ECMO and describe our initial experience with the HTP-1500 HU after being placed in clinical service.
Results
Both units demonstrated similar heating efficacy, with the HTP-1500 achieving steady-state temperature approximately 5 h earlier than the Micro-Temp. Microorganisms were detected in the water compartment of all HUs prior to and after performing the manufacturer's recommended cleaning procedure, and after implementation of the HTP-1500 into clinical use we observed a decrease in the rate of bloodstream infection/ECMO days which did not reach statistical significance.
Conclusion
Based on the results of this analysis and our institutional experience, we believe that integration of the HTP-1500 HU, an easily replaceable HU, into ECMO systems may reduce the risk of bacterial contamination and thus nosocomial infection when the devices are cleaned and maintained according to manufacturer's guidelines.
Introduction
Extracorporeal membrane oxygenation (ECMO) provides cardiac and respiratory support for patients with severe, refractory cardiopulmonary injury. 1 During the past 40 years, ECMO circuitry has become increasingly miniaturized and sophisticated to provide prolonged bedside support for patients with refractory cardiopulmonary failure. Heat loss by blood flowing through extracorporeal tubing and circuitry dedicated heating units (HUs) are incorporated into ECMO circuit design to facilitate temperature management in many ECMO patients. However, HUs have been implicated as a potential reservoir for pathogens that have been linked to global infections with Mycobacterium chimaera.2–7 In addition to infections with this nontuberculous mycobacterium, cases of nosocomial infections by Ralstonia pickettii and Cupriavidus panuculus in ECMO patients have been described and further implicated HUs as reservoirs for infection.8–10
The only ECMO HU approved by the FDA for use in the United States, the ECMO-Temp (CSZ Medical) was withdrawn from the global market in 2018. This effectively eliminated approved HUs available for ECMO use in the United States. During this time, a patient at our institution developed a bloodstream infection (BSI) secondary to Ralstonia pickettii, which was ultimately found to be present in every ECMO-Temp HU in our institution. Based on this index case and growing global concern for HU-associated nosocomial infections, we designed an experimental protocol to evaluate the safety and efficacy of commercially available HUs in order to identify an alternative HU for use in bedside and transport ECMO.
Materials and Methods
Experimental Circuitry
The HTP-1500 (Adroit Medical Systems), Micro-Temp (CSZ Medical), and ECMO-Temp HUs (control units) were individually evaluated and compared using an experimental ECMO system comprised of a standard 3/8″ CardioHelp (CH) 7.0 disposable ECMO circuit (Getinge). Operational features and manufacturer descriptions of intended use and limitations for the Micro-Temp and HTP-1500 HU are described in Table 1. Manufacturer information was not publicly available for the ECMO-Temp during design of this experimental protocol.
Baseline Operational Features of Heating Units Tested.

Abbreviation: ECMO, extracorporeal membrane oxygenation.
Our institutional protocol: 500 mL sterile water with 15 mL bleach.
Comparison of Microorganism Growth in New Heaters Before and After Cleaning Protocol.
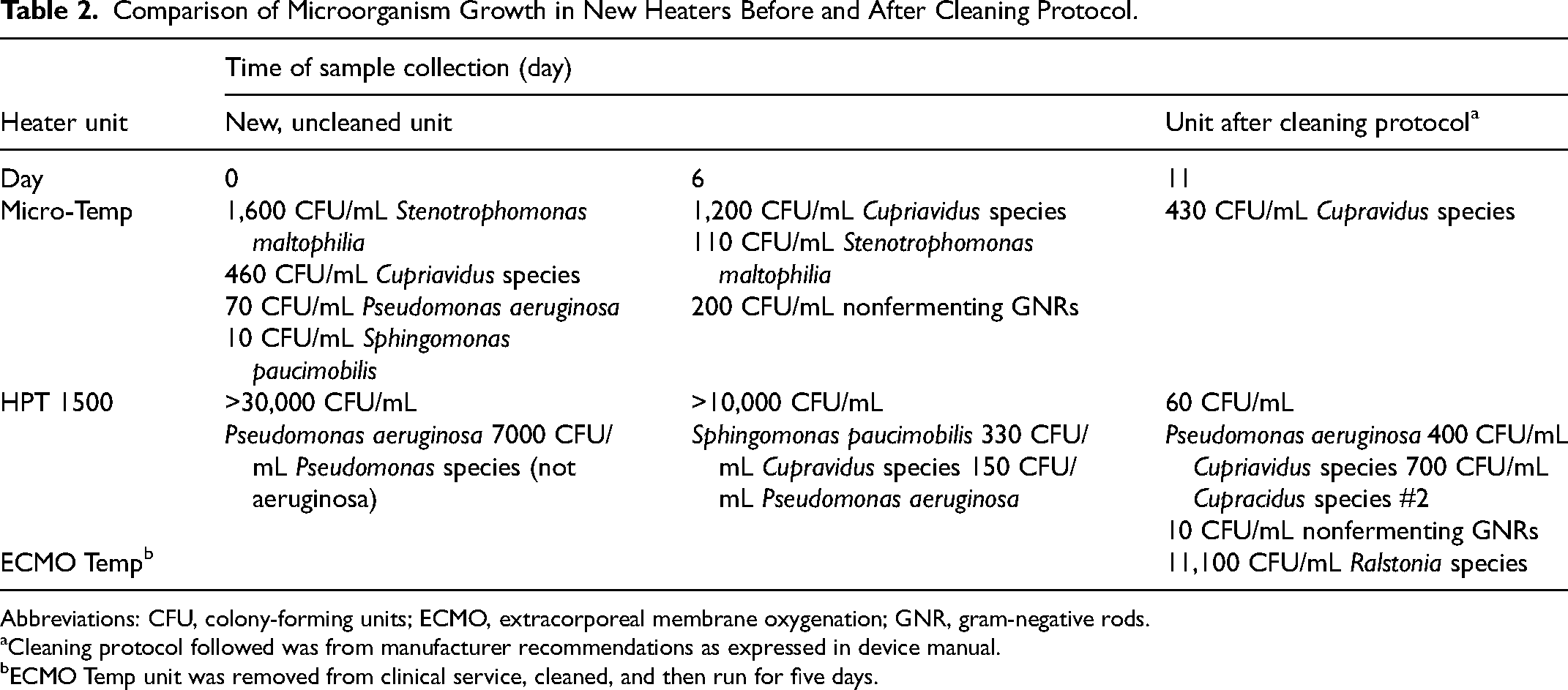
Abbreviations: CFU, colony-forming units; ECMO, extracorporeal membrane oxygenation; GNR, gram-negative rods.
Cleaning protocol followed was from manufacturer recommendations as expressed in device manual.
ECMO Temp unit was removed from clinical service, cleaned, and then run for five days.
Testing of Heating Efficiency
Heating efficiency and durability of each experiment HU was tested using two distinct circuit designs. The design of circuit 1 was constructed to evaluate the ability of each HU to warm and subsequently maintain all prime fluids from a room temperature of 20.2 °C to 37 °C. The design of circuit 2 was constructed to evaluate each HUs ability to maintain a steady-state temperature of 38 °C in the setting of conductive heat loss.
Design of Circuit 1
For test circuit 1, the ability of each HU to warm and then maintain all prime fluids (both HU and ECMO circuit) to 37 °C from room temperature (20.2 °C) was modeled utilizing a modified 7.0 CH disposable roughly simulating our ¼ inch infant ECMO circuit. The main modification was a 1/4 arterial/venous (A/V) loop cut to relative clinical length (about 4 feet) each side and connected to a 3-L normal saline bag. The CH console was set to a flow of 1 lpm, with a line pressure of 150 mm Hg and RPM of 3,000. At time 0, all fluids including the internal water bath of the HUs were room temperature as measured by the CH console and disposable system. Next, the HU was set to warm all fluids to 37 °C. The time until all fluids reached maximum steady-state temperature was measured as well as the ability of each HU to maintain this temperature over five days.
Design of Circuit 2
After this basic HU functionality test, a more robust environment was constructed and tested using circuit 2. Each HU was connected to an unmodified 7.0 CH 3/8 circuit. During this testing, the full 3/8 tubing (about 6 feet each side) was used and connected into a closed loop thereby eliminating the priming bag. This tubing loop served as the test “vasculature” and was fully submerged in a covered 45-L water bath. This 45-L bath was chosen to roughly simulate a 100 lb patient (1 L = 2.2 lbs.). The entire test circuit and water bath were cooled to 15 °C using the CardioQuip 1000i heater/cooler (HC) (CardioQuip LLC) which is a large capacity HC typically used during cardiopulmonary bypass (CPB). Once 15 °C was reached the CPB HC was replaced with the test HU which was set to warm the entire 45L to 38 °C with a CH pump flow set at 7 lpm (±0.5). The temperatures of 15 °C and 38 °C were chosen to necessitate greater heat exchange between each respective HU and the simulated patient to redemonstrate efficacy and create a more challenging environment prior to consideration of implementation for patient care. Serial temperatures were recorded with an Intellivue 8000 ICU monitor (Philips Healthcare) and a 9F DeRoyal general purpose temperature probe (Powell) over the following 14 h for each HU.
Microbiology
New, unprocessed HTP-1500 and Micro-Temp HUs were tested by the Seattle Children's Bioengineering Department to confirm compliance with institutional electrical safety standards. Heating units were then filled with sterile water and then activated and heated to 37 °C. Water was sampled from each HU water reservoir and tested for the presence of microorganisms using standard microbiology culture protocols (day 0). Each HU was then connected to a test ECMO circuit, which was run continuously for six days. Reservoir water samples were then collected and tested for the presence of microorganisms using standard microbiology culture protocols (day 6). Heating units were cleaned and disinfected according to individual manufacturer recommendations. After cleaning, each HU reservoir was refilled with sterile water and ran for an additional five days at 38 °C. Reservoir water samples were then collected and tested for the presence of microorganisms (day 11).
Statistical Analysis
Data were analyzed and graphed using Stata 17 (StataCorp. 2021. Stata Statistical Software: Release 17: StataCorp LLC.) and GraphPad Prism 9.2.0 for Mac (GraphPad Software, Inc). Discrete and continuous data were summarized using mean and standard deviations, differences between groups were analyzed using unpaired two-tailed t tests, and variance compared using Levene's robust test statistic, with statistical significance defined as P < .05.
Results
The Micro-Temp, HTP-1500, and ECMO-Temp achieved similar water bath temperatures at 500 min, with the ECMO-Temp demonstrating a slightly more rapid rate of heat exchange (rate) than the Micro-Temp (rate) and HTP (rate) units (Figure 1). The HUs were next compared according to arterial steady state temperature (Figure 2), with mean temperature found to be 38.4 ± 0.9, 38.4 ± 0.1, and 38.7 ± 0.1 for the Micro Temp, HTP-1500, and ECMO-Temp units, respectively (P < .0001). The test statistic for equality of variance was found to be 4.72 (P = .009), supporting that there was inequality of variance between samples.
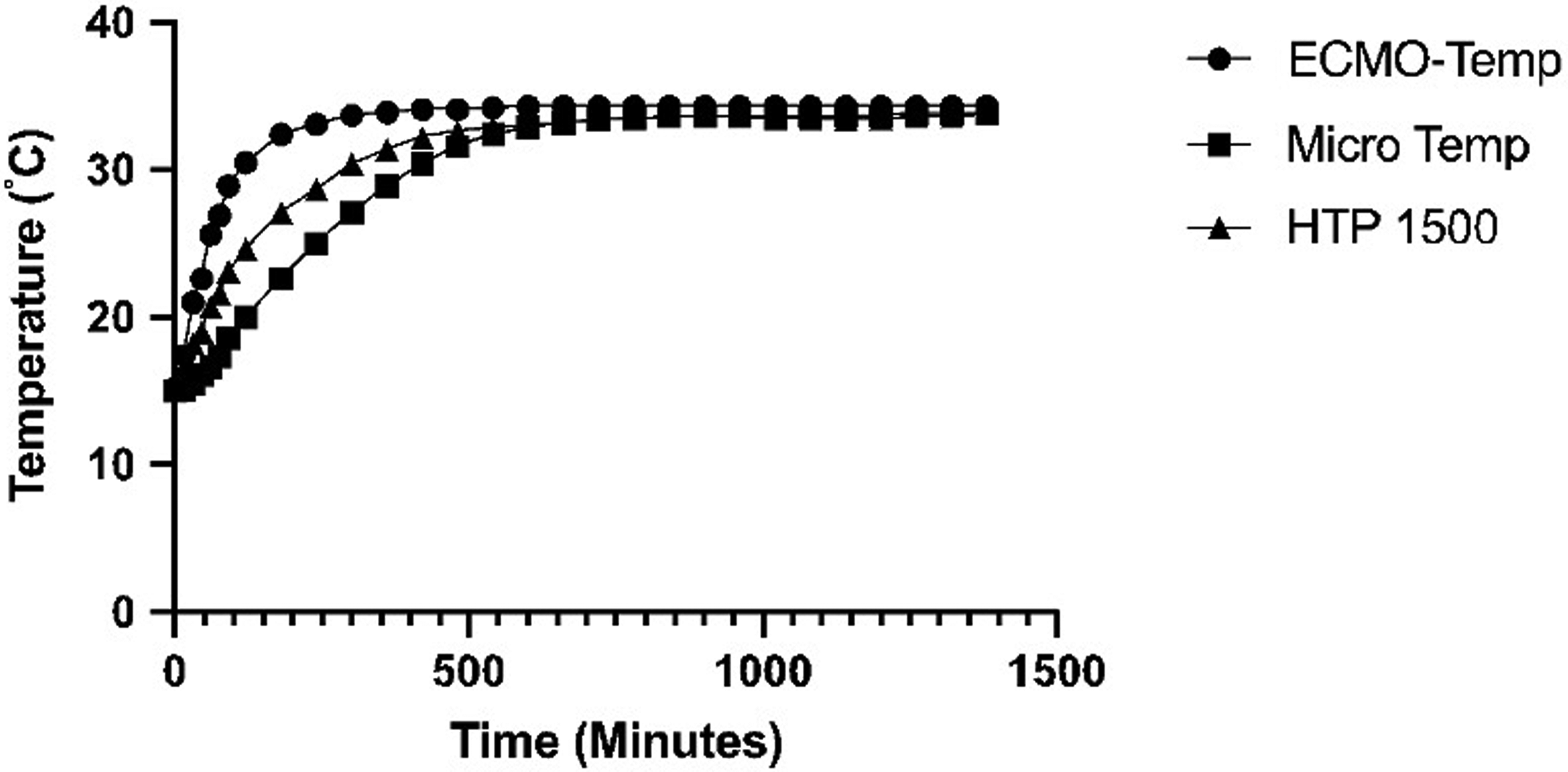
Comparison of heater unit warming curves.
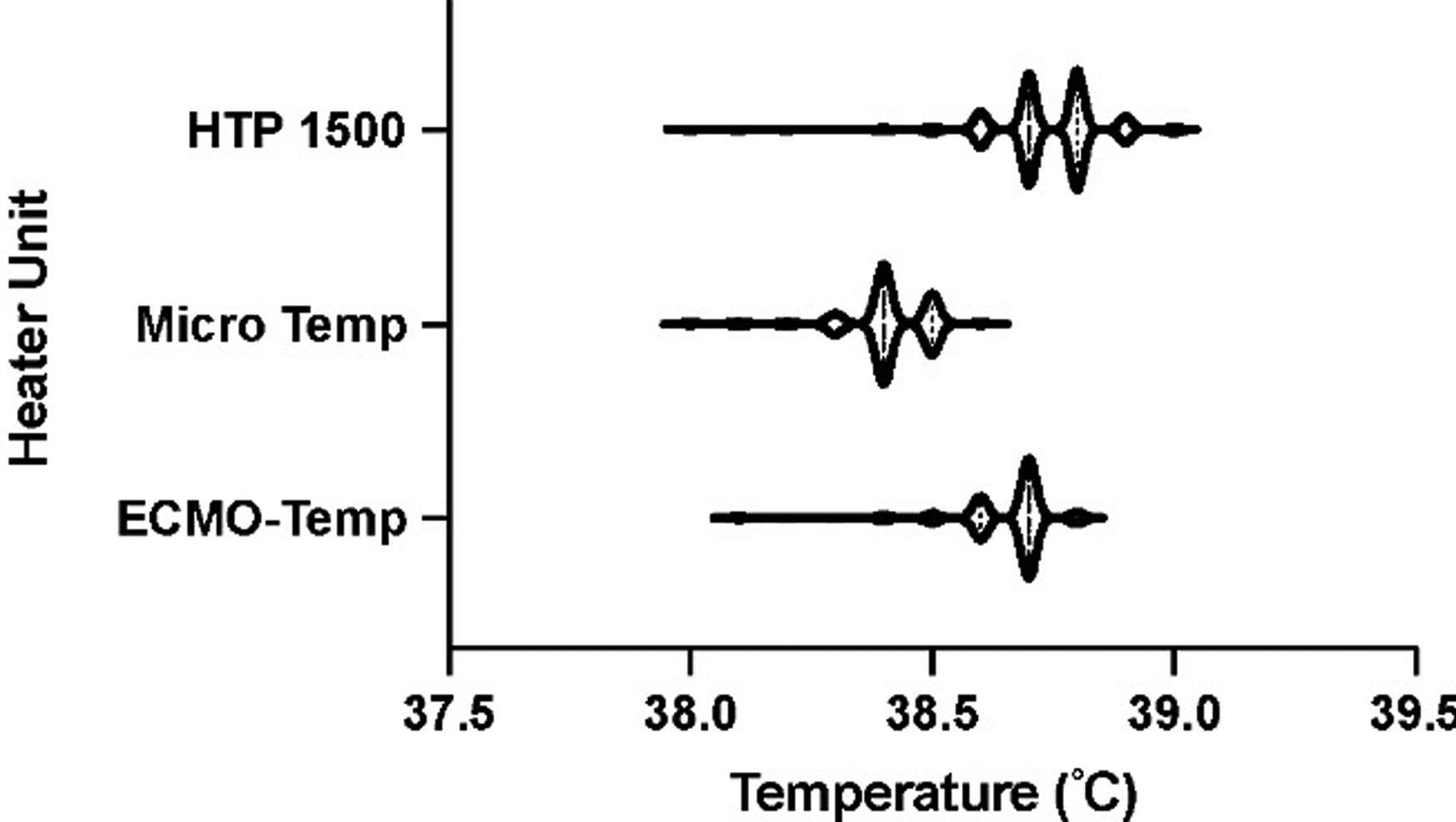
Comparison of arterial steady state temperature.
Microorganisms were detected in the water compartment of all HUs prior to and after performing the manufacturer's recommended cleaning procedure (Table 2). Both the Micro-Temp and HPT-1500 had evidence of polymicrobial growth, including Stenotrophomonas maltophilia, Pseudomonas aeruginosa, Pseudomonal subspecies, Cupriavidus, nonfermenting gram-negative rods, and Sphingomonas paucimobilis. The ECMO-Temp unit, which had been removed from clinical use for the purpose of this study, was found to have persistent growth of Ralstonia despite the successful completion of the manufacturer's recommended cleaning protocol.
Annual Blood Stream Infection Rate and ECMO Days Over Past Ten Years.

Abbreviations: BSI, bloodstream infection; ECMO, extracorporeal membrane oxygenation; MDR, multidrug-resistant organism; MRSA, methicillin-resistant Staphylococcus aureus; MSSA, methicillin-sensitive Staphylococcus aureus; spp, subspecies.
BSI was counted if infection prevention adjudicated a CLABSI or LCBI using NHSN criteria for a patient on ECMO for two consecutive days prior to BSI date.
Annual institutional BSIs and ECMO days over the past ten years are shown in Table 3. There was a total of 15 BSIs over the course of 3981 ECMO days, with BSI/ECMO days ranging from 0 to 0.011. Bloodstream infections were noted to be secondary to Enterococcus fecalis, Ralstonia, Candida albicans, Rotia spp, Staphylococcus aureus (both methicillin-sensitive and resistant), Pseudomonas aeruginosa, Escherichia coli, Candida parapsilosis, and Candida albicans. We next compared the rate of BSI/ECMO days prior to and after the implementation of the HTP-1500 at our institution in 2018, and although we observed a decrease in rate this did not reach statistical significance (P = .36).
Discussion
Management of patient temperature during ECMO due to heat loss from tubing and circuitry remains an integral facet of patient care, both at the bedside and during transport of patients with refractory cardiopulmonary failure. Despite HUs being a fundamental component of ECMO care, the ECMO-Temp (CSZ Medical) was withdrawn from the global market, thus eliminating approved HUs for ECMO use within the United States. Heating units have additionally been identified to serve as potential reservoirs for nosocomial infections, with multiple global reports of infections with Mycobacterium chimaera.2–8 This lack of an approved HU along with a nosocomial BSI secondary to Ralstonia pickettii at our institution, served as an impetus for this study in order to identify an alternative HU for use in bedside and transport of ECMO patients.
Given that the size of many commercially available HUs preclude use for aeromedical transport, we sought to identify smaller and easily replaceable HUs to address both our institution's mobile ECMO program and address concerns for nosocomial infection after discovery that all HUs at our institution were contaminated with Ralstonia. We thus investigated the Micro-Temp and HTP-1500 as possible replacements for the ECMO-Temp due to their size, weight, portability, ease of cleaning, and operational cost. In our experimental design, both HU systems were found to have similar heat exchange efficacy in both experimental circuit designs. There was, however, noted to be unequal variance of arterial steady-state temperatures, which is likely reflective of the slightly skewed temperature data collected for each unit and not clinically significant, as temperatures for both experimental units remained within 1 °C of the set point.
Microbial growth with potentially pathogenic bacteria was observed in samples obtained from the water compartment of new, unused Micro-Temp and HTP-1500 HU. Although subsequent samples obtained after performing manufacturer-specified cleaning protocols contained fewer colony-forming units in culture, both units continued to have persistent evidence of microbiological contamination. Based on these findings, we recommend performing manufacturer-recommended cleaning processes prior to clinical use as an ECMO HU. Despite subjecting the ECMO-Temp HU to repeated cleaning according to the manufacturer-recommended cleaning protocol, samples obtained from the water compartment had persistent evidence of Ralstonia contamination, highlighting the refractory nature of colonization with this species in HUs. After reviewing the results of this study, we made a programmatic decision to remove the ECMO-Temp from clinical service at our institution. We elected not to use the Micro-Temp HU in our program because the manufacturer guidelines specifically indicate “Not for Use in ECMO” but included it in this analysis because it is currently being utilized as an ECMO HU in a small number of institutions.
We made a programmatic decision to use the HTP-1500 HUs for all bedside and mobile ECMO at our institution in May 2018. Based on microbiological data, we additionally instituted a protocol, in which all new, unboxed units are cleaned per manufacturer specifications prior to being placed in clinical use in addition to performing the recommended cleaning protocol during clinical use. Since incorporating the HTP-1500 into our ECMO circuitry, no ECMO patient has developed an HU-associated infection, including a patient who received 28 days of continuous ECMO support. Although there is not a direct causal relationship between HTP-1500 implementation and the rate of BSI/ECMO days, as this is likely multifactorial, we did not observe a statistically significant change in the rate of BSI/ECMO days at our institution after implementation of the HTP-1500 into routine clinical use. Furthermore, the rate of ECMO-associated BSI at our institution remained comparable to rates described at other institutions.11,12 The data presented is intended only to illustrate the need for further research in order to inform institutions about the potential for water-based nosocomial infections from HUs within ECMO circuity, and that there remains a need for a federally investigated and approved HU.
An important consideration of this study is the off-label use of the HTP-1500 in ECMO. Further product testing for safety and efficacy is needed prior to the widespread adoption of any medical device, and this limited-efficacy evaluation does not adequately replace more rigorous testing methods. A significant limitation of this study is that the experiments were performed in a laboratory setting that may not accurately reflect a clinical critical care environment, with associated bedside care and other sources of nosocomial infection. Additionally, the study was conducted at a single institution and included a small number of HUs, which increases the potential for type II error. Further investigation is needed to determine whether device contamination occurs during the manufacturing process, product storage, and shipping, or subsequent storage at the destination hospital.
We recommend institutions consider routine testing of HU devices prior to use in patient care. Based on the results of this analysis and our institutional experience, we believe that a universal cleaning protocol for water-based HUs should be integrated into existing ECMO systems to reduce the risk of bacterial contamination and nosocomial infection. Maintenance strategies to reduce the risk of HU-associated infection in the setting of prolonged ECMO are lacking and warrant further investigation.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Bilodeau was supported by a research training grant from the National Institute of General Medical Sciences Postdoctoral Research Fellowship of the National Institutes of Health [grant number T32GM121290].
