Abstract
Introduction
The Fontan operation for single ventricle palliation has evolved since its introduction in 1971. 1 Multiple techniques for channeling systemic venous return from below the diaphragm to the pulmonary arteries have been developed. Operation selection is based on ease of surgical approach, possible avoidance of postoperative complications, and surgeon preference. Support for these various operations fluctuates as we continue to gather outcome data from around the world. 2
As debate continues between the lateral tunnel Fontan (LTF) and extracardiac conduit Fontan (ECCF) operations, the external pericardial LTF (EPLTF) remains another viable option. The EPLTF technique, first described by Gundry in 1997, is a modification of Hvass’ extracardiac tube of pediculated pericardium.3,4 A flap of posteriorly based, viable pericardium is sewn to the lateral atrial wall, creating an extracardiac tunnel from the inferior vena cava (IVC) to the central pulmonary arteries (Figure 1). This technique eliminates the use of thrombogenic foreign material in the Fontan pathway, does not require intra-atrial suture lines, eliminates the need for foreign material in the pulmonary venous side of the circulation, does not require cardioplegic arrest and typically allows for shorter bypass times. 3 Hasaniya and colleagues have shown the EPLTF to be a relatively simple, durable, and safe operation with low morbidity and mortality. 5 We hypothesize that a unique feature of the EPLTF may be the ability of a viable pericardial pathway to adapt to the increasing physiologic demands of the growing patient. In this preliminary study, we investigated the ability of the EPLTF pathway to enlarge with increasing physiologic demands of somatic growth.
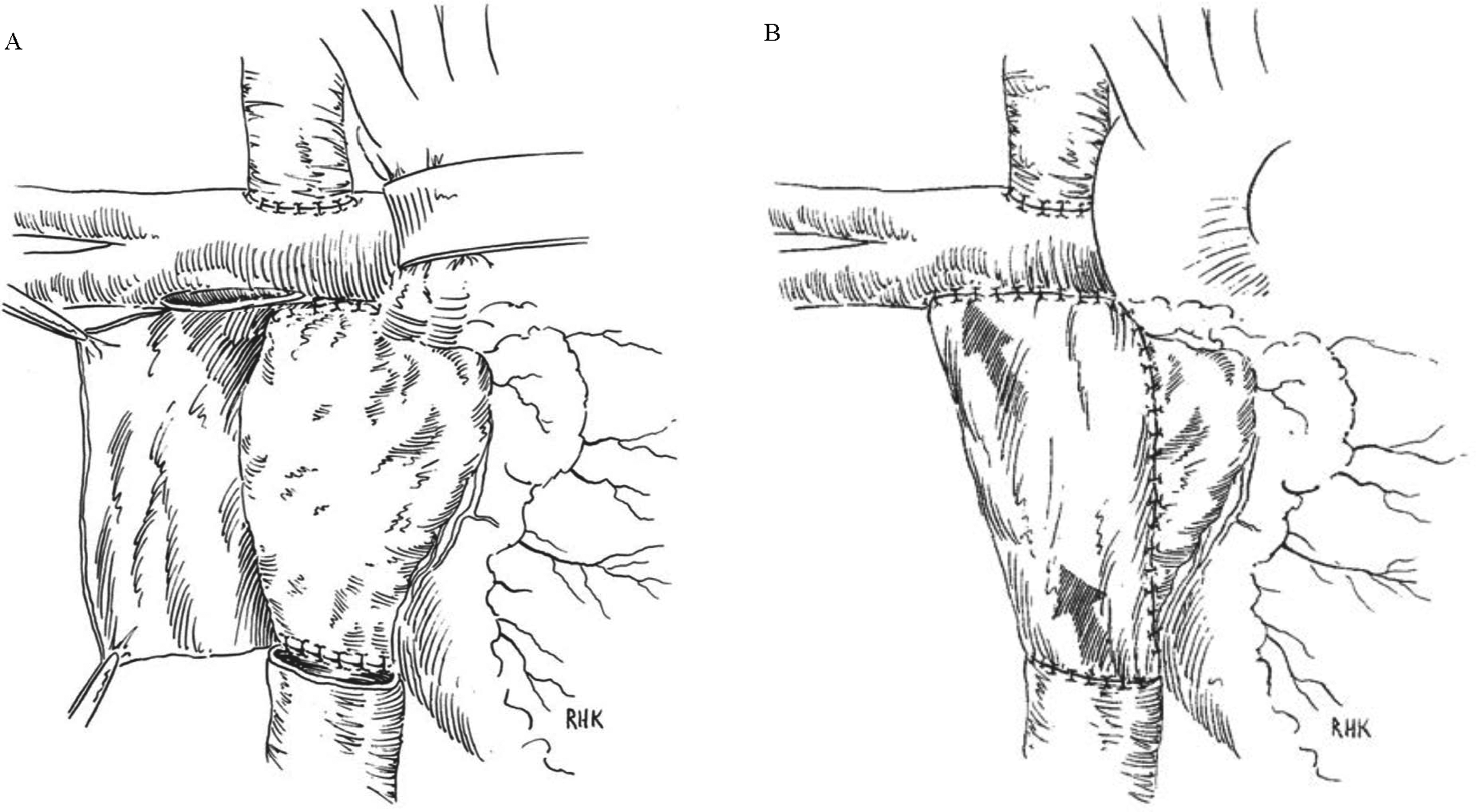
Extracardiac pericardial lateral tunnel Fontan (EPLTF) surgical technique. A flap of posteriorly based, viable pericardium is sewn to the lateral atrial wall, creating an extracardiac tunnel from the inferior vena cava to the central pulmonary arteries. (Reprinted with permission from the Journal of Thoracic and Cardiovascular Surgery. Figure 1A has been altered from the originally published drawing to more accurately illustrate the central location of the Fontan pathway connection to the pulmonary arteries.).
Methods
Study Population
A retrospective review of all patients undergoing EPLTF at Loma Linda University Children's Hospital (LLUCH) from January 2012 to December 2017 was performed. Routine echocardiographic surveillance imaging of Fontan pathway dimensions was initiated in January 2012. A minimum of five years surveillance post-EPLTF was required for inclusion in this study. Patient demographics were obtained from the electronic medical record. Fifty-one subjects were identified; 13 were excluded for insufficient echocardiographic data. The study protocol was approved by the LLUCH Institutional Review Board. A waiver of informed consent was granted.
Echocardiography
Echocardiogram images and reports were reviewed using Syngo Dynamics (Siemens Healthcare/Siemens Medical Solutions USA Inc). Three distinct Fontan circuit dimensions were measured on individual echocardiographic images for the study. The inferior vena caval junction dimension and midsection dimension were measured in centimeters on a standard subcostal bicaval view (Figure 2). The cross-sectional area of the Fontan pathway was measured in centimeters squared in a standard apical four-chamber view (Figure 3). Height, weight, body surface area (BSA), and date of study completion were obtained from finalized echocardiogram reports for each corresponding image.
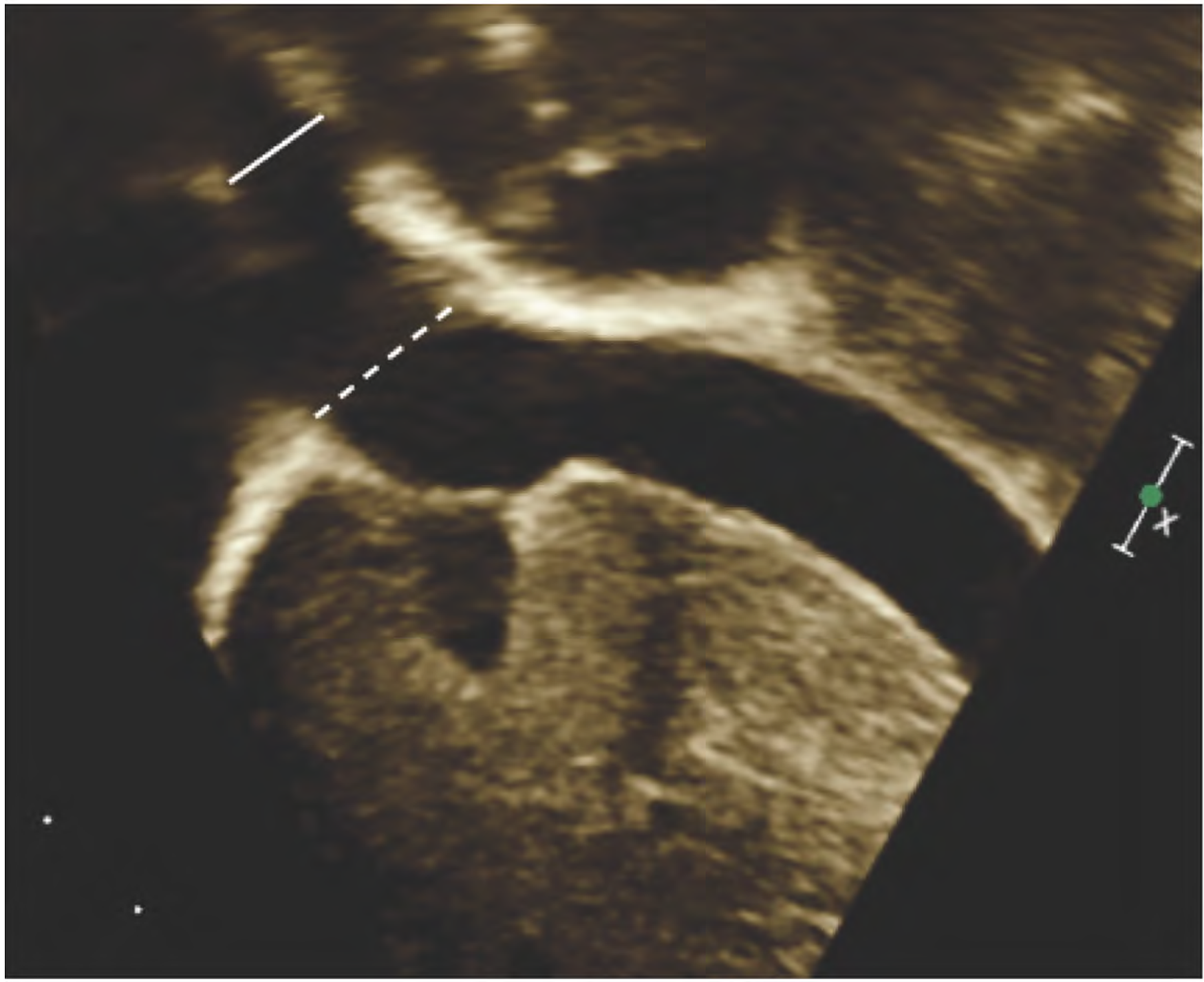
Echocardiogram measurements from bicaval view. The solid white line shows the inferior vena cava dimension and the dotted white line shows the midsection dimension.
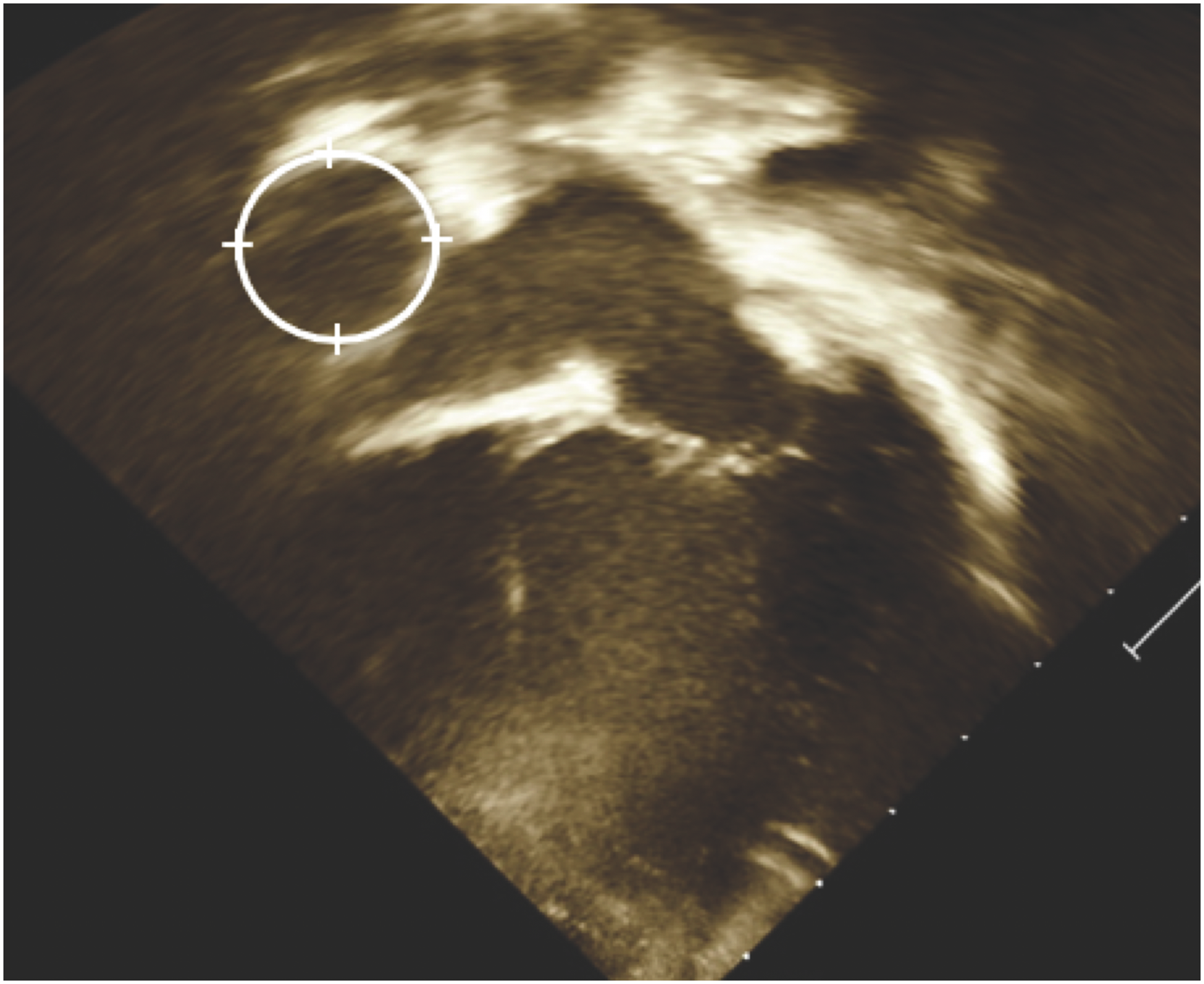
Echocardiogram measurements from apical four chamber view. The circle shows an example of a traced Fontan pathway utilized to calculate cross-sectional area.
Statistical Analysis
SPSS version 28 (IBM Corporation, 2021) was utilized for statistical analyses with statistical significance defined as P < .05. Descriptive statistics utilized means and standard deviations (SDs) for demographic and anthropometric data. Homoscedasticity was assessed using homogeneity of variance tests and regression residual plots. 6 Q-Q plots and Kolmogorov-Smirnov tests were used to assess for normality of data.7,8 Repeated measures correlation was performed using web-based rmcorrShiny.9,10 Correlation coefficients were defined using the commonly categorized values of weak, moderate, and strong corresponding to value ranges of 0 to 0.3, 0.3 to 0.7, and 0.7 to 1, respectively. 11 Repeated measures one-way analysis of variance was performed to evaluate for differences between Fontan conduit and IVC repeated measures as viewed on ECHO 4-Chamber and conduit slides. Post hoc pairwise comparisons were conducted with Bonferroni adjustment.
Results
Fifty-one subjects were identified; 13 were excluded for insufficient echocardiographic data; 332 echocardiographs from 38 patients were included in the study. Patient demographics of the study population are summarized in Table 1.
Patient Demographics.a
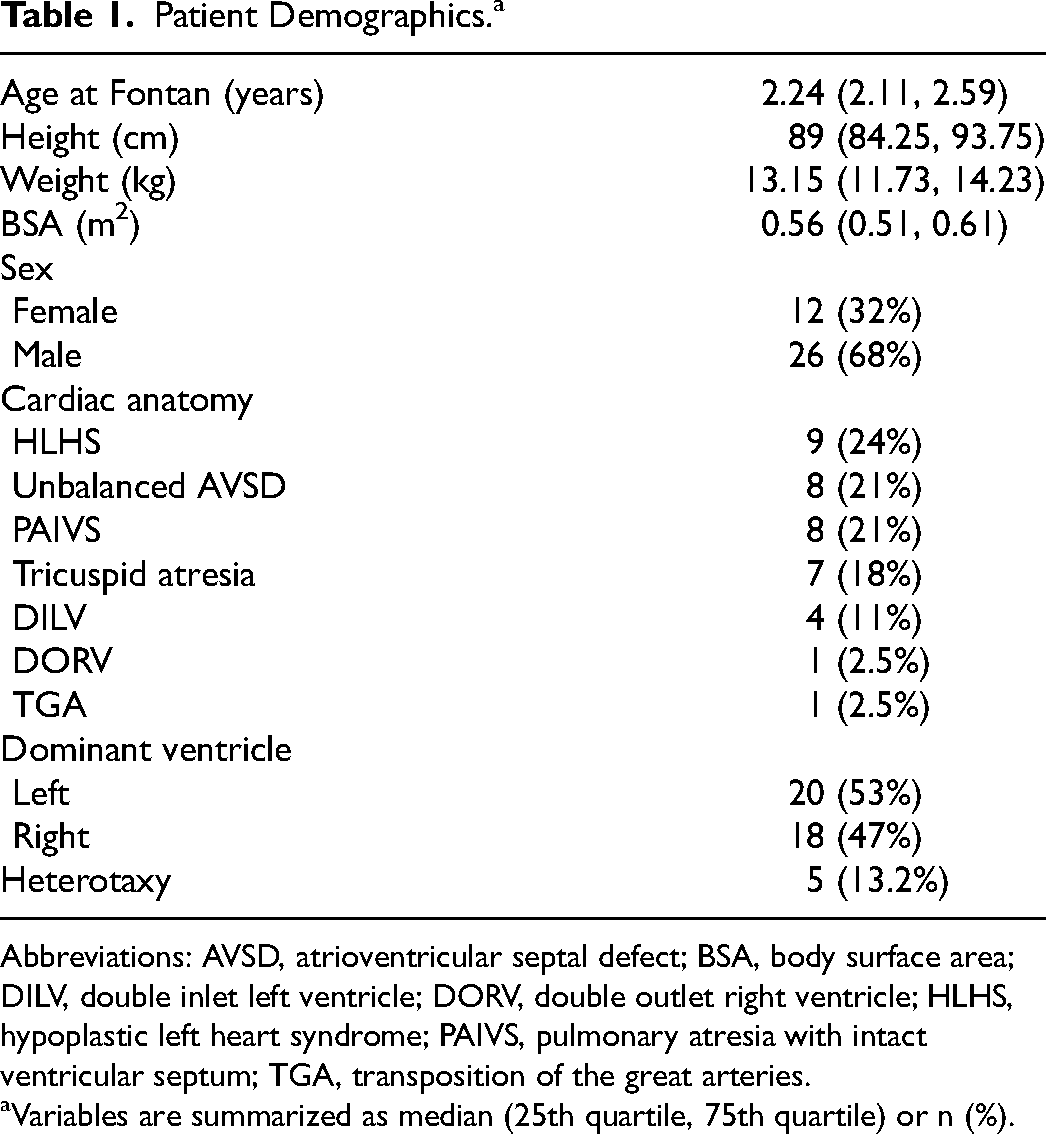
Abbreviations: AVSD, atrioventricular septal defect; BSA, body surface area; DILV, double inlet left ventricle; DORV, double outlet right ventricle; HLHS, hypoplastic left heart syndrome; PAIVS, pulmonary atresia with intact ventricular septum; TGA, transposition of the great arteries.
Variables are summarized as median (25th quartile, 75th quartile) or n (%).
The EPLTF mean inferior vena caval junction size significantly increased from 0.93 cm (SD ± 0.20) to 1.38 cm (SD ± 0.22) over ten years (P < .001). Individual patient measurements over time can be seen in Figure 4. The EPLTF mean midsection pathway size significantly increased from 1.23 cm (SD ± 0.29) to 1.78 cm (SD ± 0.28) over ten years (P < .001). Individual patient measurements over time can be seen in Figure 5. The EPLTF mean cross-sectional area significantly increased from 1.5 cm2 (SD ± 0.89) to 3.09 cm2 (SD ± 1.33) over ten years (P < .001). Individual patient measurements over time can be seen in Figure 6.
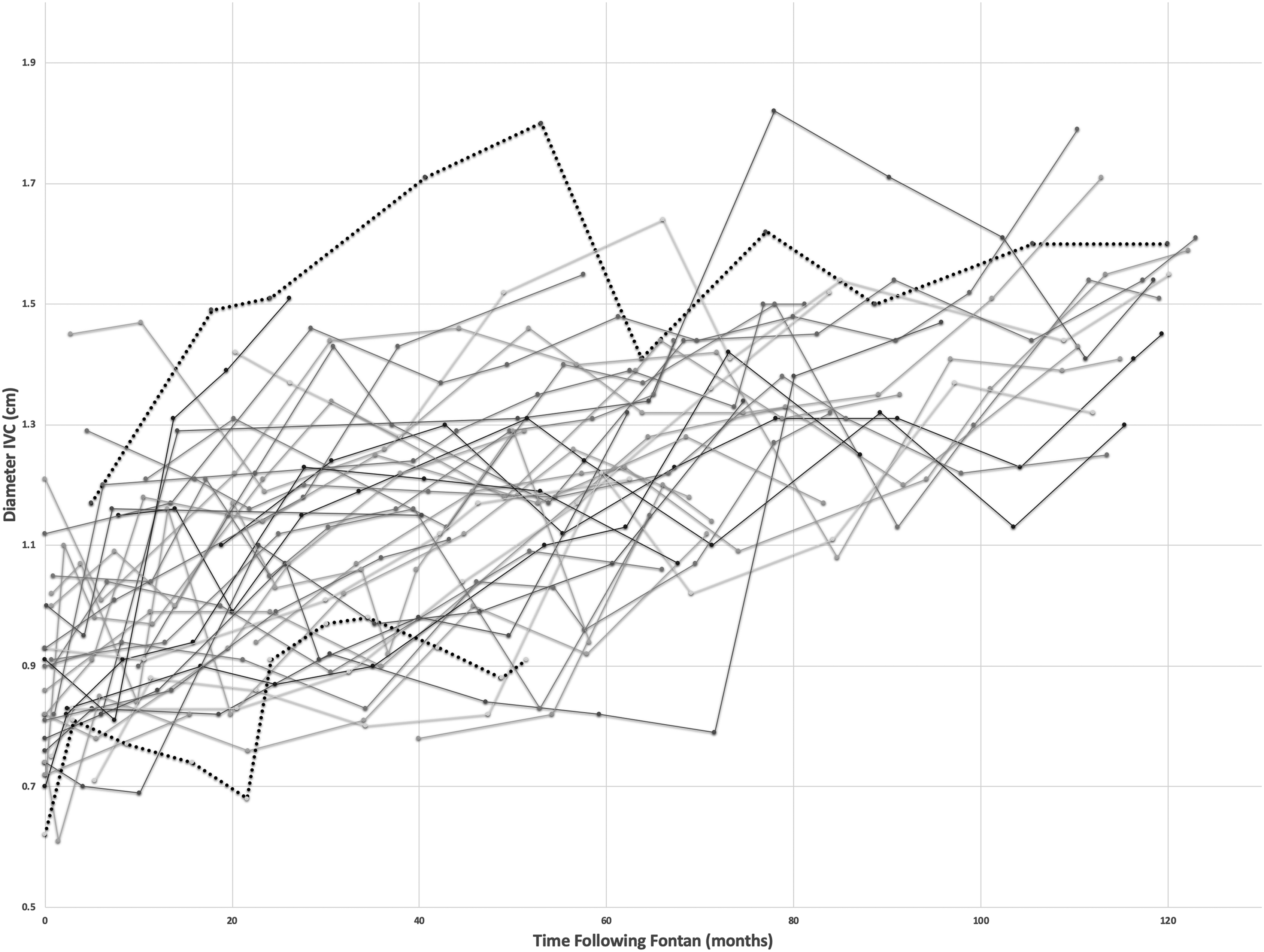
Inferior vena caval junction measurements over time for individual patients. Two patients with elevated pulmonary vascular resistance are highlighted with dotted lines.
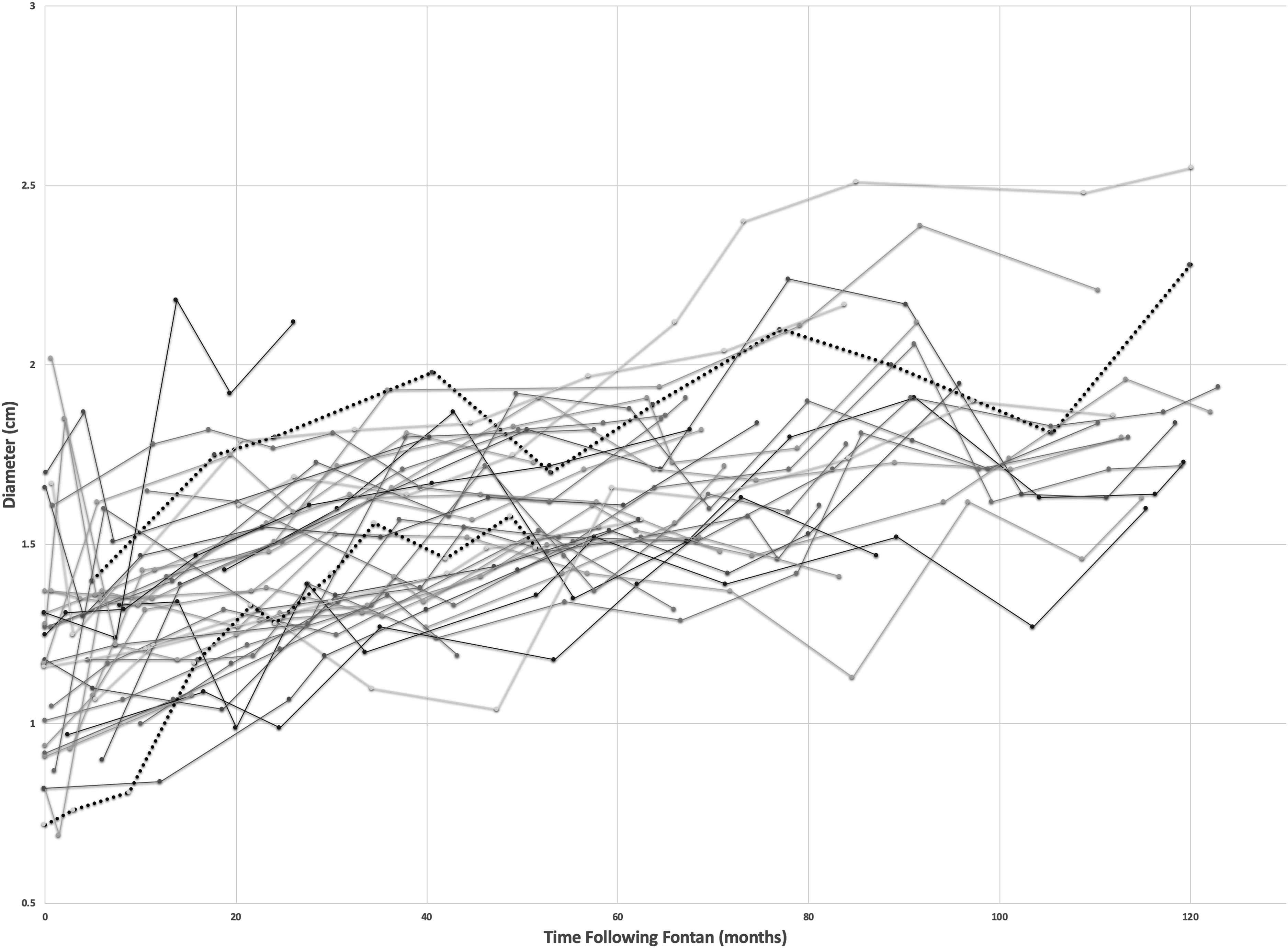
Midsection pathway measurements over time for individual patients. Two patients with elevated pulmonary vascular resistance are highlighted with dotted lines.
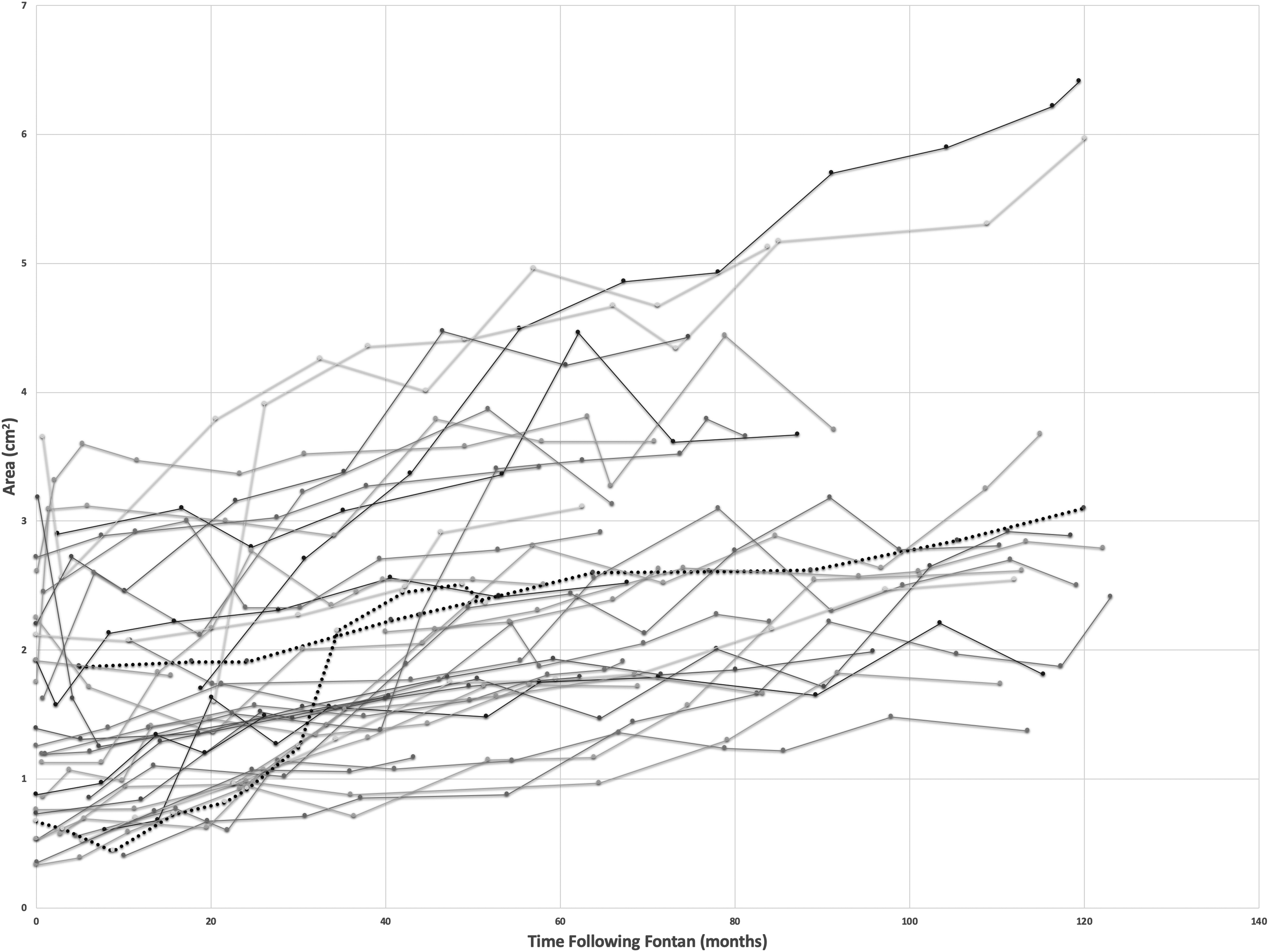
Cross-sectional area pathway measurements over time for individual patients. Two patients with elevated pulmonary vascular resistance are highlighted with dotted lines.
Hemodynamic data from cardiac catherization are available for 25 of 38 patients (66%). Fontan pressures average 12 ± 1.8 mm Hg (range, 9-16 mm Hg). Pulmonary vascular resistance (PVR) averages 1.26 ± 0.69 Wood units (range, 0.4-3.7 Wood units). Two patients have elevated PVR >2.5 Wood units (2.7, 3.7 Wood units). The single patient with an elevated Fontan pressure >15 mm Hg also had an elevated PVR of 2.7 Wood units. The second patient with an elevated PVR of 3.7 Woods units had a low Fontan pressure of 9 in the context of a fenestration and systemic oxygen saturation of 90% on room air. The increase in Fontan pathway size for these two patients was similar to the overall group (highlighted by dotted lines in Figures 4-6).
The relationship between measurements of the EPLTF pathway and BSA at annual time points over ten years was highly significant (P < .001). Repeated measures correlation between BSA and inferior vena caval junction was rrm(287) = 0.60, 95% CI [0.524, 0.672], P < .001. Repeated measures correlation between BSA and midsection was rrm(287) = 0.65, 95% CI [0.579, 0.713], P < .001. Repeated measures correlation between BSA and cross-sectional area was rrm(287) = 0.71, 95% CI [0.646, 0.762], P < .001. The relationship between mean BSA and mean dimensions at the inferior vena caval junction, midsection, and cross-sectional area can be seen in Figure 7.
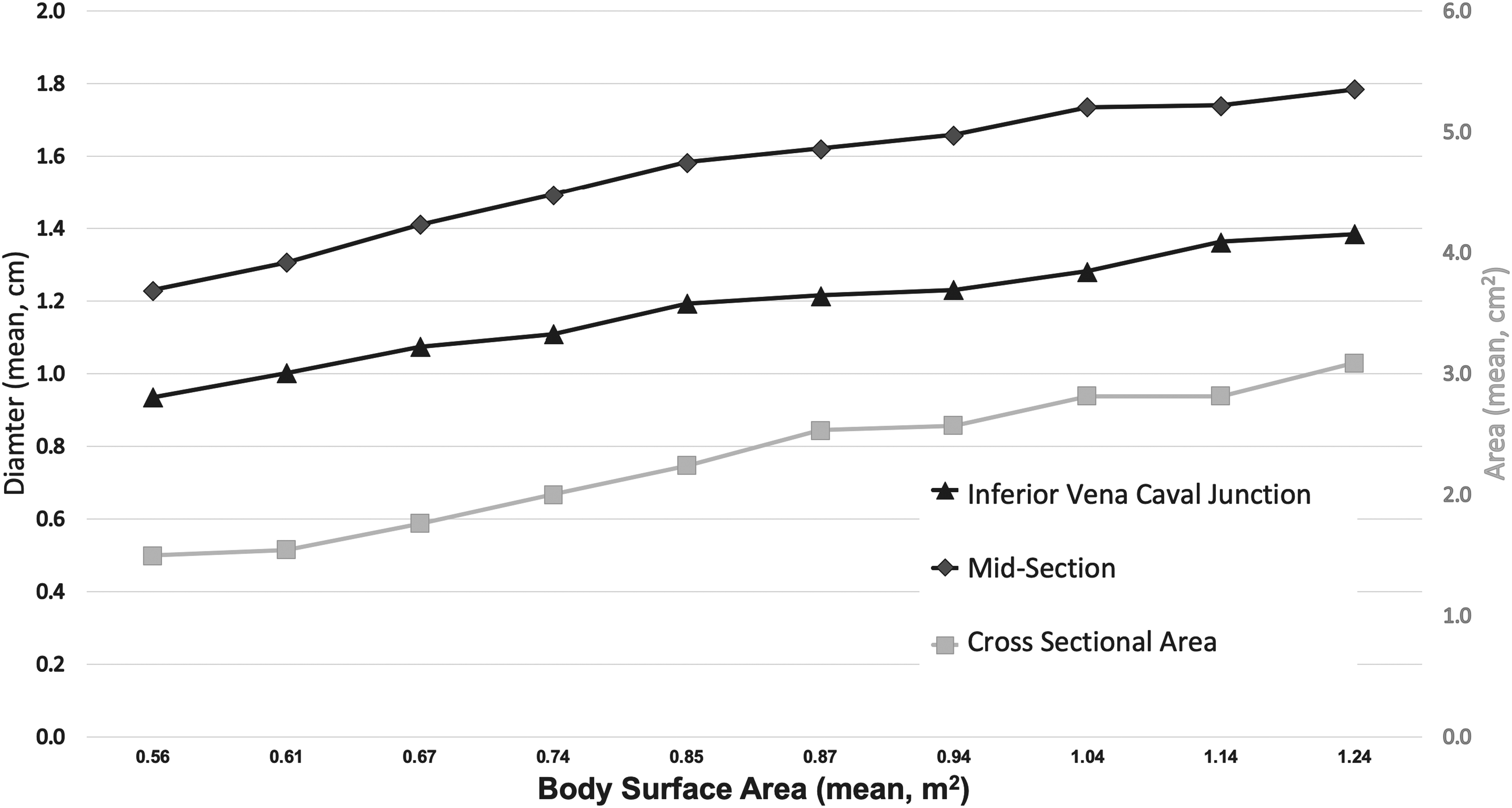
Annual mean body surface area (BSA) in relation to mean pathway dimensions.
Fontan pathway measurements normalized to BSA generally decrease over time. Normalized IVC measurements (IVC diameter/BSA), midsection measurements (midsection diameter/BSA), and cross-sectional area measurements (cross-sectional area/BSA) are downward trending in 79%, 89%, and 53% of patients, respectively.
Discussion
The Fontan operation has undergone multiple changes in hopes of improving short and long-term outcomes. The LTF (introduced in 1988) 12 and ECCF (introduced in 1990) 13 have their followers just as the decision on Fontan operation timing has various camps of allegiance.2,14,15 The majority of centers complete the Fontan pathway between two and four years of age.16,17 At this age, the IVC diameter is approximately 80% of the eventual adult size. 18 The LTF with growth potential can be performed at the earlier end of this range without risk of limited IVC growth while the ECCF with a fixed conduit size is typically performed later. The differences between the LTF and ECCF techniques have been extensively compared with most studies showing equivocal results and others showing only a mild advantage for one or the other technique.19–25 With the Boston publication in 2020 showing better short and midterm outcomes for the LTF over the ECCF technique, the best Fontan pathway debate has been reinvigorated. 2 As the search for the ideal Fontan surgical technique continues, we continue to present an alternative option.
We chose the EPLTF as our preferred technique in 1993 for several reasons. It is constructed of viable, autologous tissue, eliminating the need for thrombogenic foreign material. It can be constructed without cardiac ischemia or atrial incisions thereby lessening the risk of low cardiac output and atrial arrhythmias. 3 It also can be fenestrated in the postoperative period and allow transvenous access to the atrium for pacemaker insertion, if needed.
In this single-center retrospective study of patients undergoing Fontan completion with an EPLTF, we observed statistically significant pathway enlargement over a 5- to 10-year period. The inferior vena caval junction and midsection showed significant enlargement over time with minimal SD. Although the cross-sectional area also showed significant enlargement over time, there was a larger SD noted in the measurements. This variation may be due in part to changes in the three-dimensional shape of the pathway with changes in respiration during echocardiogram measurements. 3
The ability of the Fontan pathway to enlarge with continuing somatic growth is a major advantage of the EPLTF technique. Similar findings of Fontan pathway enlargement correlating with BSA have been reported for the LTF.26,27 The ECCF, in comparison, does not have growth potential and has been shown to have a significant decrease in a cross-sectional area within six months of Fontan completion.14,28,29 However, measurements of the EPLTF pathway normalized to BSA are downward trending over time in many of our patients. This suggests that the enlarging Fontan pathway may not be “keeping up” with somatic growth. Similar concerns have been raised with the LTF. 27
The anticoagulation protocol utilized by most centers to reduce the risk of post-Fontan thromboembolism is low-dose Aspirin; Coumadin is administered with or without Aspirin for patients with arrhythmias or a history of thromboembolic events.2,30–32 In regard to the thrombogenic nature of the EPLTF pathway, we have previously reported a low risk of thrombosis and the presence of reendothelialization of the EPLTF pathway based on pathologic evaluation.3,5 Given the lack of thrombogenicity and postoperative atrial arrhythmias observed with the EPLTF technique,3,5 our post-Fontan anticoagulation protocol consists of low-dose Aspirin only.
A conduit that enlarges in correlation with somatic growth (indicated by positive BSA correlation) may be able to adapt to patient physiology with improved outcomes. Additional studies are needed to correlate changes in pathway size with hemodynamic data from cardiac catheterization, computational fluid dynamic studies, and cardiopulmonary exercise testing results over time until somatic growth is complete. This information would help determine the difference between normal physiologic growth and pathologic enlargement. We hypothesize that patients with favorable hemodynamics, flow, and exercise data may have an idealized pathway size based on individual physiology, although measurements of the pathway size may vary between patients. Although this preliminary study only evaluated changes in pathway size, our previously reported 15-year follow-up morbidity and mortality data 5 compares favorably to the 2020 Boston publication results 2 specifically in relation to atrial arrhythmias, thromboembolisms, and survival. The EPLTF technique should be considered among viable surgical options for Fontan completion.
Study Limitations
Although we have been performing the EPLTF technique since 1993, only patients with serial echocardiographic data since 2011 were included in the analysis, thereby limiting the number of patients in this initial study. Due to the retrospective study design, there are incomplete data (eg, initial EPLTF pathway size; less than five years of echo data points) in a subset of patients, and the number of echocardiographic data points per year per patient is variable. Furthermore, data are only presented for patients up to 13 years of age and includes hemodynamic data on a limited subset of patients. Although pathway enlargement has been shown, further work is needed to distinguish the difference between physiologic growth and pathologic enlargement as well as elucidate how the pathway will continue to change into adulthood. To that end, future studies are needed to correlate changes in pathway size to hemodynamic data from cardiac catheterization, computational fluid dynamic studies, and cardiopulmonary exercise testing results over time until somatic growth is complete.
Conclusions
In this single center retrospective study of patients undergoing an EPLTF, we observed significant enlargement in all pathway measurements over a ten-year period following the completion of the Fontan. The pathway enlargement was also found to have a positive correlation with increasing BSA. Further research is needed to differentiate normal physiologic growth from pathologic enlargement and to correlate changes in pathway size with physiologic studies and clinical outcomes.
Footnotes
Authors’ Statement
Consent was obtained for the use of all illustrations included in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
