Abstract
We describe the perventricular device closure of a large mid-muscular ventricular septal defect (VSD) in a 2.9 kg infant born with hypoplastic aortic arch and VSD using an Occlutech perimembranous occluder. In this case, the anatomy required a short low-profile device and hence a perimembranous occluder was used. To our knowledge, this is the first described use of this device for hybrid closure of a muscular VSD and the application of this technique in a patient <3 kg.
Introduction
Percutaneous closure of ventricular septal defects (VSDs) has been performed for over two decades. 1 Following FDA approval of the muscular VSD device in 2007, 2 devices designed for perimembranous (pm) VSD closure were introduced. An initially high risk of conduction abnormalities led to improvements in delivery systems and device design, such as the Occlutech pmVSD Occluder. 3 , 4 Occlutech introduced its muscular VSD occluder, a double-disc, braided Nitinol device, in 2016. 5 Muscular VSD closure via standard percutaneous interventional routes has now been well described; with many physicians advocating for hybrid perventricular approaches in smaller patients. 6
Case
The patient was born full term in Trinidad, West Indies, and postnatally diagnosed with a large mid-muscular VSD and coarctation of the aorta with hypoplasia of the distal transverse arch. The patient developed signs of heart failure within the first two weeks of life, which persisted despite aggressive medical management.
The patient underwent hypoplastic aortic arch repair at six weeks of life via a left lateral thoracotomy with a reverse left subclavian flap to enlarge the transverse arch below the left carotid artery. This was followed by an end-to-end anastomosis to repair the aortic coarctation segment. Postoperative echocardiogram and clinical course revealed no evidence of residual coarctation. The VSD was not addressed at this time with a plan for pulmonary artery (PA) banding versus delayed primary VSD closure depending on the efficacy of medical management. The patient was extubated on postoperative day one but had persistent decompensated heart failure over the subsequent days.
Given the patient's size (2.9 kg) and frailty, the lack of access to a neonatal bypass circuit, and the position of the VSD in the septum, we rejected the idea of a PA band or conventional surgical patch repair in favor of a hybrid VSD closure. The septum in the region of the VSD was only 2 mm thick, and there was minimal space from the ventricular septum to the right ventricular (RV) free wall (RV lateral dimension). We were concerned that the closure of the defect with a standard muscular VSD device or an Amplatzer duct occluder device would create an undesirable interaction with the RV free wall (Figures 1 and 2). Reviewing the available occlusion inventory, we determined that the low profile and short length of a pmVSD device may be better suited in this case (Figure 1).
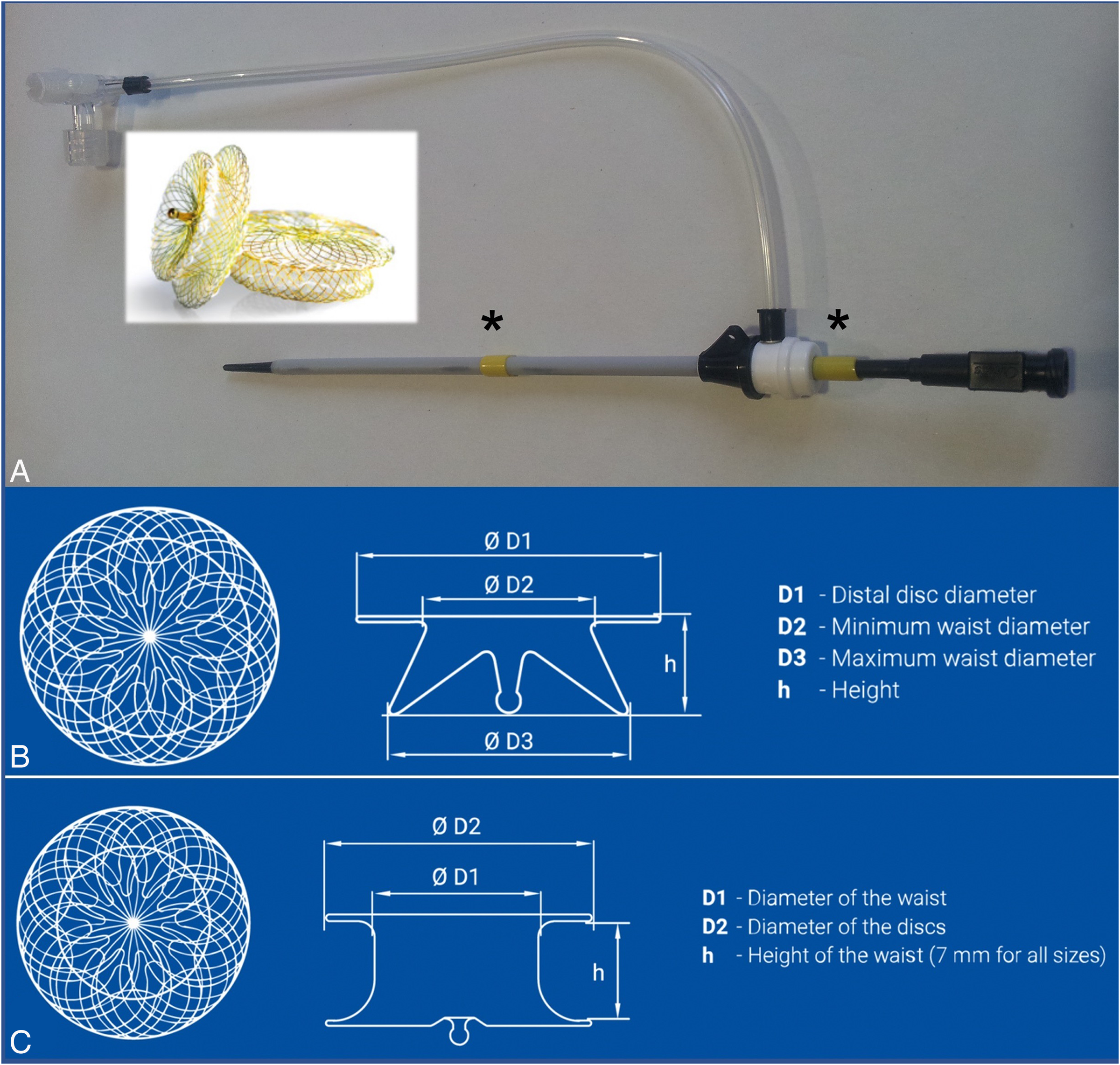
(A) The modifications made to delivery sheaths for hybrid perventricular access. The two 1 cm long pieces of yellow rubber tubing marked by * can be positioned to limit the progress of the sheath into the ventricle and also to decrease the length of dilator which protrudes from the sheath tip. These two modifications decrease the risk of posterior left ventricular (LV) wall trauma and help stabilize the sheath's position in the right ventricular (RV) free wall (these modifications were made to the 6F sheath). (B) A schematic of the Occlutech perimembranous VSD device. “h” denotes the device’s height or length. For the device we chose in this case (an 8 mm pmVSD device), the length (5 mm) incorporates the entire excursion of the right-sided disc, minimizing the RV profile of the device. This can be further appreciated when compared with the muscular VSD device in (C), which has a height of 7 mm, not including the excursion of its right-sided disc. The schematics in B and C are reproduced with permission from Occlutech GmbH.
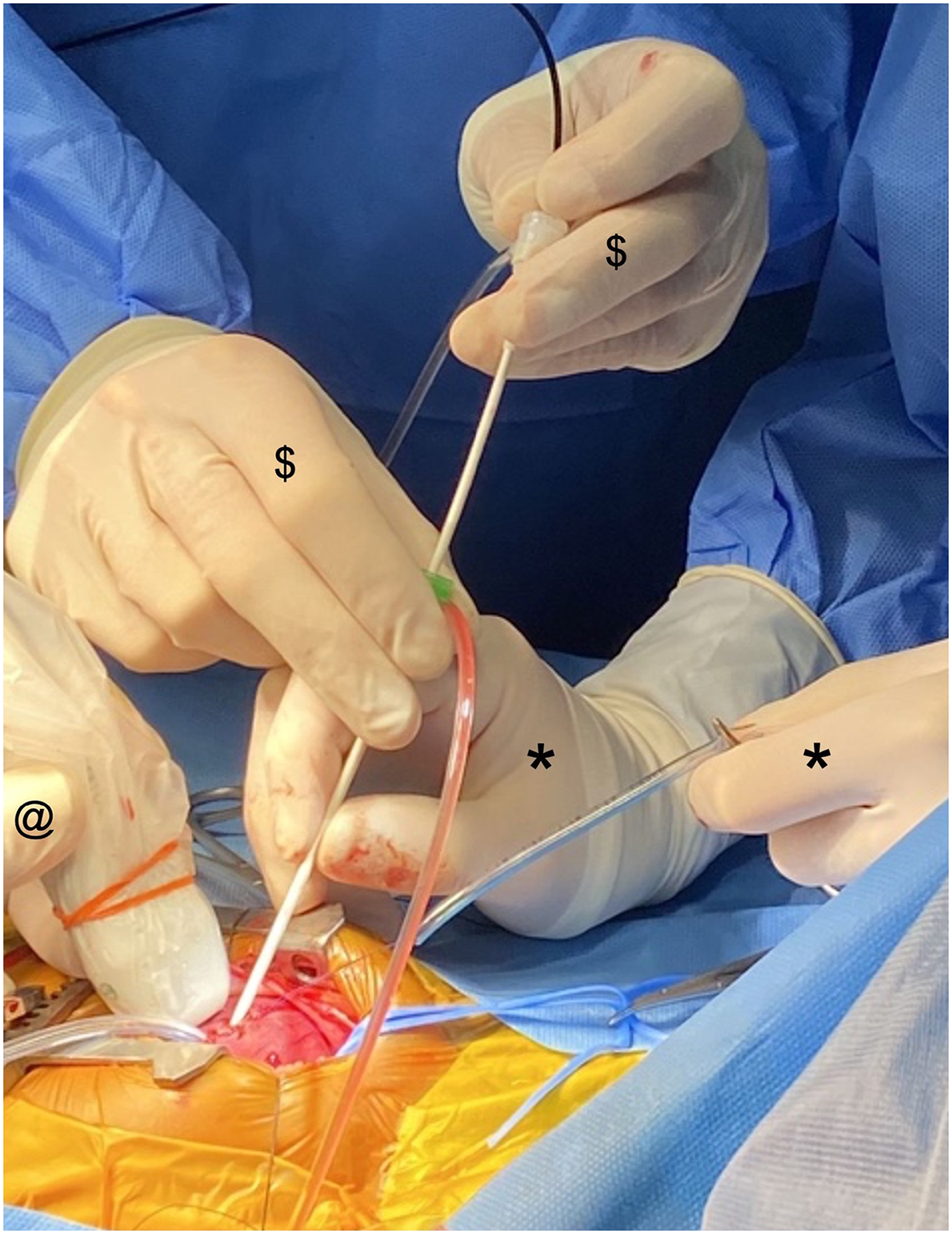
This image shows the 5F sheath loaded with the device within the 6F sheath. It shows the interaction required between the pieces of equipment and the operators. The surgeon is controlling the purse-string (*); the interventional cardiologist ($) is manipulating the sheath, the introducer for the device and the device itself; the echocardiographer (@) is manipulating the imaging probe.
The patient was taken for perventricular device closure of the VSD with epicardial echocardiographic guidance on day of life 45. The VSD was measured on preprocedural transthoracic and epicardial echocardiograms at approximately 6 mm; and appeared quite symmetric. We, therefore, chose an 8 mm Occlutech perimembranous VSD occluder device. Two separate sheaths were used. To minimize sheath size and maintain maximum control during deployment in this small infant, we loaded the device into a 9 cm, 5F Terumo pinnacle introducer sheath. Following a midline sternotomy, the optimal position of the ventricular puncture was determined by gently digitally indenting the RV free wall, while relating this to the VSD on echocardiogram. A purse-string suture was accordingly placed on the RV free wall followed by a needle and wire insertion into the RV cavity. That allowed passage of a 9 cm, 6F Terumo pinnacle introducer sheath through the RV cavity, modified to minimize the chance of posterior left ventricular (LV) wall trauma (Figure 1A). 7 A 014 balanced middle-weight coronary wire was shaped into a gentle curve and used to cross the defect, then passed up into the LV outflow tract and through the aortic valve under echocardiographic guidance. The 6F sheath was advanced over this to lie in the LV mid-cavity. The 5F sheath loaded with the pmVSD device was then introduced into the back of the 6F sheath, and the device was deployed in the manner described in multiple manuscripts (Figures 2 and 3A-C, Video 1). 5 The device was shown to have an appropriate position within the ventricular septum (Figure 3D, Video 2). There was no significant residual shunt, nor was there any inflow or outflow obstruction. The low-profile nature of the Occlutech pmVSD device meant that the device sat flush on the septum and did not protrude through the RV cavity toward the free wall on echocardiographic assessment. The device was released and maintained an appropriate position. The chest was closed, and the patient was extubated within 24 h with immediate resolution of heart failure and rapid progress with oral feeding. Follow-up ECHO showed normal biventricular function and closure of the VSD without interference with neighboring structures.
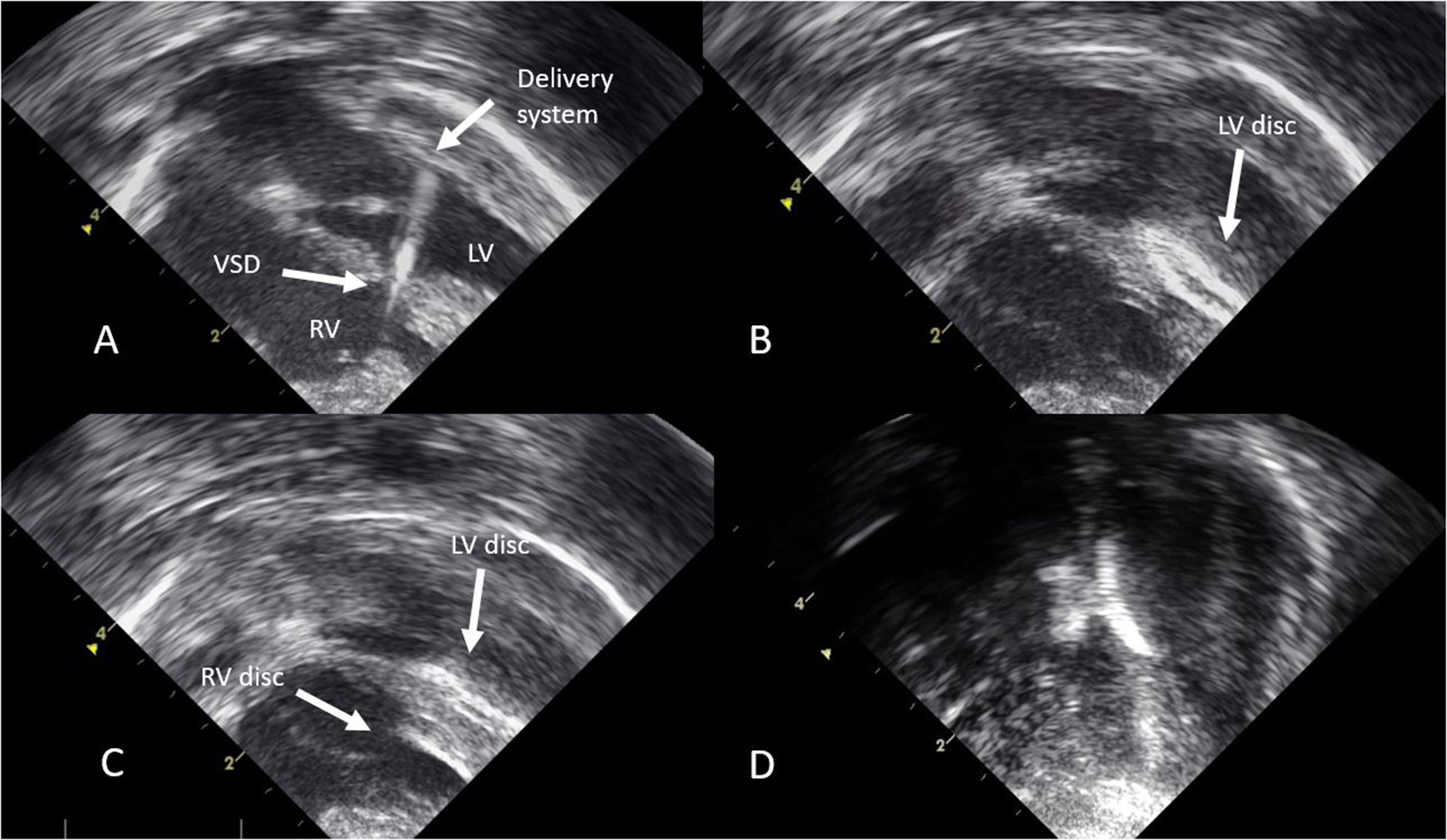
(A-C) Intraoperative epicardial echocardiogram images with the deployment of the device at different stages. (D) The final configuration of the device on postoperative transthoracic echocardiogram. LV left ventricle; RV, right ventricle; VSD, ventricular septal defect.
Discussion
The Occlutech perimembranous VSD device has a soft Nitinol weave structure. Rather than a separate waist and RV disc, its structure integrates these two components, rendering it short with a low profile. This was felt to be ideal in this small infant for closure of the muscular defect. In many cases of apical or mid-muscular VSD, the RV aspect of the defect may be complex, with trabeculations and the presence of the moderator band causing problems with the deployment of the right-sided portion of the device. In this case, the anatomy looked as though a short low-profile device would be beneficial; hence, the choice of a perimembranous occluder.
Specifically in this case, we were faced with limited resources. The lack of availability of a neonatal bypass circuit was an important factor in our decision-making. Hence, nonbypass approaches were primarily considered including perventricular device closure of the VSD versus PA banding. We recognize that PA banding would have provided a reasonable intermediate approach to addressing the left-to-right shunt from the VSD. However, we believed that a definitive approach to the VSD would be ideal for the patient since surgical and procedural expertise was only available on a periodic basis by visiting teams.
Conclusion
This case highlights that VSD closure via a hybrid, perventricular approach can be achieved safely with echocardiographic guidance in low-weight neonates and infants. To our knowledge, is the first described use of the Occlutech perimembranous VSD occluder for the closure of a muscular VSD and reported application of this device and technique in a patient <3 kg. In patients with thin septums in whom the anatomy of the RV aspect of the VSD is suitable, consideration of pmVSD devices in the muscular septum may be reasonable.
Footnotes
Authors’ Statement
Consent for publication was granted by the patient's parent.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
