Abstract
Background
Controversy regarding the optimal pulmonary valve substitute remains, with no approved surgical valve for pulmonary valve replacement (PVR). Furthermore, unfavorable anatomy often precludes transcatheter PVR in patients with congenital heart disease. We therefore sought to evaluate the feasibility of the Edwards Inspiris pericardial aortic bioprosthesis in the pulmonary position in pediatric and adult patients requiring PVR.
Methods
Data from consecutive patients who underwent PVR from February 2019 to February 2021 at our institution were retrospectively reviewed. Postoperative adverse events included paravalvular or transvalvular leak, endocarditis, explant, thromboembolism, valve thrombosis, valve-related bleeding, hemolysis, and structural valve degeneration. Progression of valve gradients was assessed from discharge to 30 days and one year.
Results
Of 24 patients with median age of 26 years (interquartile range [IQR]: 17-33; range: 4-60 years), 22 (91.7%) patients had previously undergone tetralogy of Fallot repair and 2 (8.3%) patients had undergone double-outlet right ventricle repair in the neonatal period or infancy. All patients had at least mild right ventricular (RV) dilatation (median RV end-diastolic volume index 161.4, IQR: 152.3-183.5 mL/m2) and at least moderate pulmonary insufficiency (95.8%) or stenosis (8.3%). Median cardiopulmonary bypass and cross-clamp times were 71 (IQR: 63-101) min and 66 (IQR: 60-114) min, respectively. At a median postoperative follow-up of 2.5 years (IQR: 1.4-2.6; range: 1.0-3.0 years), there were no mortalities, valve-related reoperations, or adverse events. Postoperative valve gradients and the severity of pulmonary regurgitation did not change significantly over time.
Conclusions
At short-term follow-up, the bioprosthesis in this study demonstrated excellent safety and effectiveness for PVR. Further studies with longer follow-up are warranted.
Introduction
Congenital heart disease involving the right ventricular outflow tract (RVOT), including tetralogy of Fallot with or without pulmonary atresia, double-outlet right ventricle (DORV), and pulmonary stenosis (PS), often requires iterative surgical or transcatheter interventions throughout childhood and into adulthood. 1 In such cases, surgical repair begins in the neonatal period or during infancy and may involve the use of techniques that result in significant pulmonary insufficiency. Left untreated, this can lead to chronic postoperative complications such as right ventricular (RV) dysfunction and failure, atrial and ventricular tachyarrhythmias, and sudden cardiac death. When indicated, pulmonary valve replacement (PVR) is necessary to restore valvular competence, ameliorate symptoms, and improve RV function. 2 However, there are no approved surgical valves for PVR, and prostheses approved for aortic valve replacement are often utilized in the pulmonic position. Moreover, transcatheter options are restricted by intrinsic patient anatomy, limited range of available sizes, and complications related to frame fracture and coronary compression. 3 Other viable options include pulmonary and aortic homografts, but these are usually reserved for patients without a native pulmonary artery (PA) and those requiring a complete conduit change. Although homografts work well in the pulmonic position, they are subject to structural valve denegation (usually calcific stenosis) and allosensitization that can jeopardize future options for transplantation. 4 Similarly, while mechanical valves in the pulmonic position are durable, the risks associated with thrombosis and lifelong anticoagulation have limited their widespread adoption. 5 In the current era, stented aortic bioprostheses remain the pulmonary valve substitute of choice, but data are varied with regard to durability and structural valve degeneration (SVD).6,7
The absence of a valve specifically designed for PVR and suboptimal outcomes with contemporary bioprosthetic valves have led to off-label use of various aortic valve prostheses. The decision to use a specific prosthesis or valve substitute for PVR is often based on its durability in the aortic position. 8 In recent years, the Edwards Inspiris Resilia pericardial bioprosthesis (Edwards Lifesciences) has gained popularity for surgical aortic valve replacement given its safety, hemodynamic performance, expandable stent design that facilitates future valve-in-valve interventions, and resistance to early thrombosis and noncalcific degeneration. 9 This prosthesis incorporates an anticalcification process with bovine pericardial tissue preserved with glycerol and has been purported to have longer durability and enhanced resistance to SVD compared to other commercially available bioprostheses. 10 However, data on its use in the pulmonic position are scarce. We, therefore, sought to evaluate the safety and effectiveness of the Inspiris Resilia valve in children and adults requiring PVR.
Patients and Methods
Patient Population and Study Design
Clinical and echocardiographic data from consecutive patients who underwent PVR using the Inspiris Resilia pericardial bioprosthesis (the study valve) at a single institution between February 1, 2019, and February 1, 2021, were retrospectively reviewed following Institutional Review Board approval (Protocol Number: STUDY-22-00528; Date of Approval: April 19, 2022). The requirement for informed consent was waived during the approval process given the retrospective study design and the sole use of de-identified patient data. Patients were included if they had pulmonary valve disease requiring surgical PVR (the index operation) of their native or prosthetic valve. Prior to discussion in a multidisciplinary conference to determine suitability for surgical PVR, all patients underwent transthoracic echocardiography and cardiac magnetic resonance imaging to evaluate the severity of pulmonary regurgitation (PR) and stenosis, ventricular function, and degree of RV dilatation. Data regarding the primary diagnosis, indication for PVR, and any prior interventions were queried. The data pertaining to the present study are available upon reasonable request from the corresponding author and institutional approval.
Technique of Valve Implantation and Postoperative Thromboprophylaxis
All operations were performed via median sternotomy and beating-heart cardiopulmonary bypass with mild hypothermia. Cardioplegic arrest was instituted only if a left-to-right shunt was noted (either preoperatively or via an intraoperative bubble study) or if concomitant procedures were required. The native main PA and RVOT or transannular patch were incised, followed by the excision of any calcified patch material, the native pulmonary valve leaflets, and obstructing muscle bundles. Valves were sized intraoperatively with the RVOT opened. A generous patch augmentation of the main PA was performed with bovine pericardium. The posterior sewing ring of the study prosthesis was secured to the pulmonary annulus using a continuous polypropylene suture. Anterior annular fixation of the study prosthesis to the bovine pericardial patch was then completed, with patch augmentation of the RVOT using the remainder of the bovine pericardial patch.
To minimize the risk of prosthetic valve-related thromboembolic complications, all patients were initiated on daily aspirin prophylaxis (age <18 years: 81 mg; age ≥18 years: 325 mg) at the time of discharge.
Clinical Outcomes and Adverse Events
Postoperatively, the following adverse events were queried during the index hospitalization and throughout the course of post-discharge follow-up: mortality, reintervention (defined as any unplanned surgical or transcatheter procedure involving the pulmonary valve prosthesis), paravalvular or transvalvular leak, prosthetic valve endocarditis, valve explant, thromboembolism, valve thrombosis, valve-related bleeding, hemolysis, and SVD. Follow-up echocardiography was performed at our institutional core laboratory at discharge, 30 days, and one year, and included an assessment of prosthetic valve regurgitation and transvalvular gradients as described previously. 11
Statistical Analysis
Baseline patient-related and procedural characteristics were reported as frequency (percentage) for categorical variables and median (interquartile range [IQR]) for continuous variables. Progression of valve gradients was assessed from discharge to 30 days and one year using a repeated measures analysis of variance model. The proportion of patients with varying degrees of PR and PS (ie, none, trace, mild, moderate, and severe) at discharge was compared to that at 30 days and one year using marginal homogeneity tests for dependent samples. Statistical significance was indicated by P < .05. Stata version 15 (StataCorp LLC) was used throughout.
Results
Of 24 patients who underwent PVR with the Inspiris Resilia bioprosthesis during the study period, the median age at operation was 26 years (IQR: 17-33 years; range: 4-60 years), 14 (58.3%) patients were female, and 22 (91.7%) patients had a diagnosis of tetralogy of Fallot. All patients had undergone prior surgery (either tetralogy of Fallot repair or DORV repair) in the neonatal period or during infancy, and 6 (25.0%) patients had undergone prior transcatheter procedures on the pulmonary valve or branch PAs, including branch PA stenting or dilatation and transcatheter PVR. Indications for operation included the following: severe, symptomatic pulmonary insufficiency with progressive RV dilatation, 19 (79.2%); severe pulmonary and tricuspid insufficiency with RV dysfunction, 2 (8.3%); severe RVOT obstruction or PS, 2 (8.3%); and Melody valve endocarditis, 1 (4.2%). Pulmonary valve replacement with the Inspiris Resilia bioprosthesis was performed in all patients, and 21 (87.5%) patients required an adjunctive procedure. Cardioplegic arrest was required in 3 (12.5%) patients. Implanted valve sizes ranged from 19 to 29 mm (median: 27 mm; IQR: 25-27 mm) (Table 1).
Baseline Patient-Related and Perioperative Characteristics.

Summary statistics are shown here as frequency (percentage) or median (interquartile range).
Prior interventions involving the pulmonary valve or branch pulmonary arteries, including branch pulmonary artery stenting or dilatation and transcatheter pulmonary valve replacement.
Melody valve endocarditis.
Assessed by cardiac magnetic resonance imaging and indexed to body surface area.
Cardioplegic arrest was required in three patients.
There were no in-hospital mortalities or valve-related reinterventions. Prior to discharge from the index hospitalization, 1 (4.2%) patient required reexploration for bleeding, 1 (4.2%) patient developed a deep sternal wound infection requiring operative debridement, and 1 (4.2%) patient underwent a planned implantation of an automatic implantable cardioverter-defibrillator. There were no major pre-discharge complications, including refractory ventricular arrhythmia, RV failure, low cardiac output syndrome or post-cardiotomy shock, prolonged mechanical ventilation, new renal failure with or without dialysis, hepatic failure, and stroke or seizure. The median postoperative hospital length of stay was six days (IQR: 5-8 days). At a median post-discharge follow-up of 2.5 years (IQR: 1.4-2.6 years; range: 1.0-3.0 years), there were no mortalities or valve-related reinterventions. A heart failure–related readmission was required in 1 (4.2%) patient. There were no instances of paravalvular or transvalvular leak, prosthetic valve endocarditis, thromboembolism, valve thrombosis, valve-related bleeding, hemolysis, and SVD at the latest follow-up.
The average peak transvalvular gradients (PG) at discharge, 30 days, and one year were 14.6 ± 6.6 mm Hg, 9.1 ± 5.8 mm Hg, and 10.0 ± 6.0 mm Hg, respectively. Corresponding values for the mean gradient (MG) were as follows: discharge, 7.4 ± 2.3 mm Hg; 30 days, 5.8 ± 3.1 mm Hg; one year, 6.3 ± 3.5 mm Hg. There was no significant change in mean PG (P = .37) or MG (P = .14) over time. The proportion of patients with varying degrees of PR and PS at discharge, 30 days, and one year is shown in Figure 1. At the one-year mark, all patients had at most mild PR and/or mild PS. The distribution of the severity of prosthetic valve stenosis did not change significantly from discharge to 30 days (P = .15) or from discharge to one year (P = .78). Similarly, there was no significant difference in the distribution of the severity of prosthetic valve regurgitation between the predischarge and 30-day evaluations (P = .77) or between the predischarge and one-year evaluations (P = .52). However, it should be noted that the proportion of patients with mild PR increased from 10% at discharge to 11% at 30 days and 35% at one year (Figure 1).
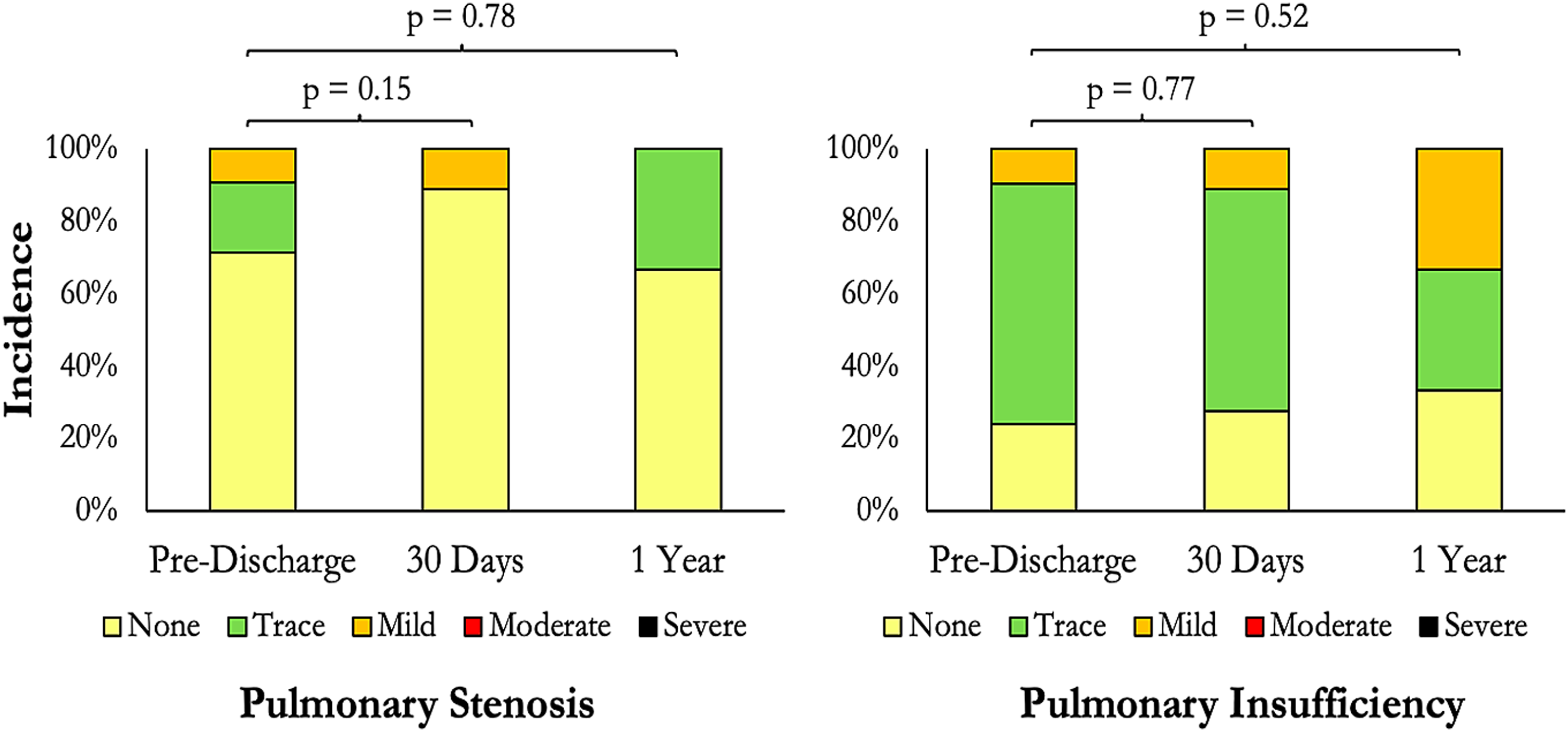
Progression of prosthetic valve regurgitation and stenosis over time. The proportion of patients with none, trace, mild, moderate, and severe pulmonary stenosis (left) and regurgitation (right) is depicted here. P values are derived from the comparison of pulmonary stenosis or regurgitation severity at discharge to that at 30 days and one year.
Comment
Patients requiring PVR for sequelae of congenital pulmonary valve disease or RVOT obstruction constitute a challenging population given the need for reoperative surgery and lack of a dedicated bioprosthesis for the pulmonic position. 12 This is of particular relevance given the growing population of patients with adult congenital heart disease. 13 Although aortic bioprostheses are most commonly used for PVR, the limited durability of currently available valves continues to plague outcomes. In contrast to adult patients with calcific aortic stenosis requiring aortic valve replacement, the adult congenital heart disease population requiring PVR (usually with aortic bioprostheses) is comparatively younger. Given the relatively lower pressures of the right heart, earlier prosthetic valve dysfunction is not uncommon with the off-label use of aortic bioprostheses. 14 In this observational report, we reviewed our institutional experience with PVR using the Inspiris Resilia pericardial aortic bioprosthesis and demonstrated excellent safety and valve hemodynamics at short-term follow-up. Specifically, there were no mortalities or valve-related reinterventions or complications at a post-discharge follow-up of up to three years. Furthermore, valve gradients remained stable from discharge to 30 days and one year, and the distribution of the severity of prosthetic valve regurgitation and stenosis did not change significantly over time. The limited duration of clinical and echocardiographic follow-up notwithstanding, our outcomes compare favorably to those reported in the literature.15,16
Although there is a dearth of data regarding the durability of the Inspiris Resilia bioprosthesis in the pulmonic position, various retrospective studies have validated its precursor, the Carpentier-Edwards Perimount valve, as a suitable alternative for PVR. Taken together, these studies indicate that stented bioprosthetic valves have become the favored valvular replacement option given optimal rates of survival and freedom from reintervention (Table 2).17,18 Specifically, freedom from reintervention or reoperation among patients receiving the Perimount bioprosthesis is estimated at 100% at 2.5 years, 17 50% to 98% at five years,19,20 75% at seven years,21,22 and 79% to 94% at ten years. 23 In addition, freedom from SVD, at least moderate prosthetic valve insufficiency, or valve explant ranges from 89% to 92% at five years19,24 and 79% to 98% at ten years.22,23 Conversely, the use of stentless porcine valves, compared to stented bovine valves, has been associated with an increased risk of reoperation and SVD. 25 Interestingly, in a retrospective study of 122 patients with tetralogy of Fallot requiring PVR, Tweddell et al did not find a difference in survival or freedom from reintervention between heterograft and homograft valves. However, the authors concluded that heterograft valves are a better option given the potential advantages of lower cost and increased availability. 26 Finally, in one of the largest comparative studies to-date, Neukamm et al demonstrated that the use of the Perimount valve was associated with the lowest rate of pulmonary insufficiency and stenosis at mid-term follow-up (compared to homografts, monocusp valves, and the ContegraTM pulmonary-valved conduit). 27 Of note, there have been no randomized controlled trials evaluating the merits and drawbacks of the various prostheses (the COMMENCE-P trial [NCT02656290], a prospective, nonrandomized, single-arm study designed to assess the safety and durability of the Inspiris Resilia bioprosthesis for PVR, has recently completed recruitment). The differential effects of prosthetic valve function in the pulmonic position as compared to the aortic position also remain incompletely characterized. 14 Nevertheless, contemporary data suggest that the use of stented bovine pericardial valves, especially the Perimount bioprosthesis, results in acceptable hemodynamics and clinical outcomes at mid-to-long-term follow-up. It is plausible that improvements to the Perimount valve, conferred by the Edwards Integrity-PreservationTM technology and features that facilitate future transcatheter pulmonary valve-in-valve interventions, will lead to enhanced outcomes with the Inspiris Resilia bioprosthesis.
Clinical and Echocardiographic Outcomes of Selected Studies of Pulmonary Valve Replacement.
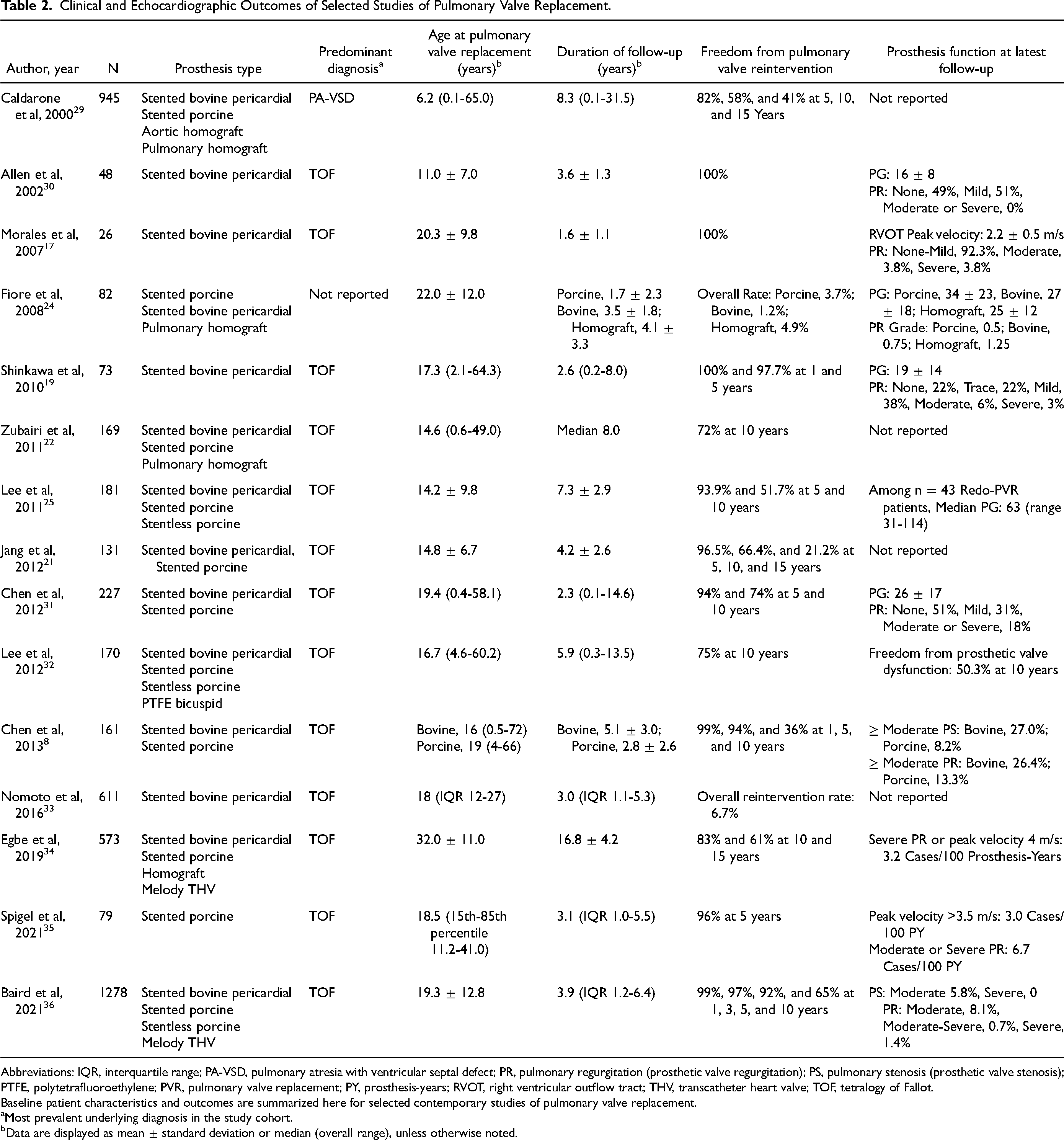
Abbreviations: IQR, interquartile range; PA-VSD, pulmonary atresia with ventricular septal defect; PR, pulmonary regurgitation (prosthetic valve regurgitation); PS, pulmonary stenosis (prosthetic valve stenosis); PTFE, polytetrafluoroethylene; PVR, pulmonary valve replacement; PY, prosthesis-years; RVOT, right ventricular outflow tract; THV, transcatheter heart valve; TOF, tetralogy of Fallot.
Baseline patient characteristics and outcomes are summarized here for selected contemporary studies of pulmonary valve replacement.
Most prevalent underlying diagnosis in the study cohort.
Data are displayed as mean ± standard deviation or median (overall range), unless otherwise noted.
Recently, Said et al reported their experience of 27 patients who underwent PVR with the Inspiris Resilia bioprosthesis between August 2019 and October 2021. 28 The majority of these patients (∼80%) underwent PVR using techniques similar to that described in the present study (ie, the standard implant group); the remainder had the prosthesis implanted in a conduit. In contrast to our findings, the study authors noted several early concerns. At a mean follow-up of 1.3 ± 0.7 years, de novo prosthetic valve regurgitation was observed in 48% of patients in the standard implant group; 11% of these patients required subsequent transcatheter valve-in-valve PVR. Moreover, 22% and 11% of patients developed moderate and severe regurgitation, respectively. Interestingly, none of the patients in the valved-conduit group developed PR. 28 This report raises concerns about the durability of the Inspiris Resilia bioprosthesis in the pulmonic position. In addition, unanswered questions remain regarding whether standard implantation techniques distort the framework of the valve, leading to early regurgitation and valve failure. Our experience has been markedly different, despite comparable patient populations and length of follow-up. Specifically, all patients in the present study had at most mild prosthetic valve stenosis and/or regurgitation at one year. Moreover, there was no significant difference in the distribution of prosthetic valve stenosis or regurgitation between the discharge and 30-day evaluations or between the discharge and one-year evaluations. Nevertheless, it should be emphasized that the proportion of patients with mild regurgitation steadily increased over time. This finding may herald disappointing outcomes of PVR with the study bioprosthesis in the future, similar to those described by Said et al. 28 The present study may also have been underpowered to detect statistically significant differences in the progression of pulmonary insufficiency over time. Appraisal of valve performance at mid- to long-term follow-up, especially within the cohort of patients with progressive prosthetic valve regurgitation, is therefore imperative.
Limitations
Some limitations are worth noting. This was a single-center, retrospective review with bias inherent to missing data and patient loss to follow-up. Furthermore, the present study represents an observational report with a relatively small number of cases. Objective evaluation of the Inspiris Resilia bioprosthesis in the pulmonic position requires a comparison of outcomes with an appropriate control group (ie, randomized controlled trial, observational study employing propensity-matched cohorts, etc). Our follow-up also did not extend beyond the short-term mark, and long-term longitudinal data are crucial to objectively assess the durability of the study valve. Prospective multicenter studies, evaluation of valve performance using varying implant techniques, long-term follow-up data, and results from the COMMENCE-P trial are all eagerly awaited.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
