Abstract
Background
Although considering the pathophysiology of post-coarctectomy hypertension, β-blockers should be effective, experience with labetalol for treatment is limited in the literature.
Methods
Retrospective collection and analysis of data in children aged ≤6 years following coarctectomy in our tertiary care university medical center between January 2009 and June 2018.
Results
96 patients were included, 45 were treated with intravenous labetalol and 51 received no treatment. Median time to maximum dose received (median 1.1 mg/kg/h) was 2.7 h, and median time to the reduction of labetalol dose was 8.3 h. No antihypertensives had to be added. In one child, labetalol was switched to nitroprusside due to bronchoconstriction. Of patients receiving intravenous labetalol, 48% had been switched to oral labetalol at discharge.
Conclusions
Intravenous labetalol is a fast, effective, and safe drug to treat hypertension following aortic coarctation repair. Labetalol is easily converted to oral therapy when the continuation of treatment is considered necessary.
Introduction
Paradoxical hypertension is common and well recognized after aortic coarctation repair, occurring in about 40% to 72% of cases.1,2 Antihypertensive treatment options are numerous. Pharmacological agents most frequently used worldwide are intravenous nitroprusside and esmolol. 3 However, evidence-based guidelines are not available to date. Insufficient therapeutic effect of a single agent or possible side effects can be reasons to change or add medication. Some treatment strategies appear to cause an increase in health care–related costs without evidence of superiority. 4
Previous studies have shown hypertension in the first day following aortic coarctation resection to be caused by the initial increase in catecholamines due to increased sympathetic output by the bulbar vasomotor centers to compensate for the decreased proximal blood pressure. The second phase of hypertension is caused by activation of the renin–angiotensin system (RAS). 5 This early postoperative hypertension may be harmful to the aortic anastomosis and may be the cause of post-coarctectomy syndrome. From a pathophysiological point of view, the initial use of a β-blocker such as labetalol should have a satisfactory therapeutic effect on hypertension. Labetalol is a combined alpha 1 and nonselective beta-receptor blocker with a half-life of 3 to 5 h. Studies on the efficiency of labetalol, however, are limited.
In our tertiary care hospital, paradoxical hypertension post-coarctectomy is treated with intravenous labetalol, with the continuation of therapy by oral labetalol when required. The aim of this study is to describe our 9-year experience in the efficiency and safety of this treatment strategy.
Patients and Methods
We retrospectively obtained data on all children following coarctation repair in our tertiary care university medical center from January 2009 until June 2018. The study was approved by the accredited Medical Ethical Committee of our institution, the MREC LDD, which determined that the study did not fall under the Medical Research Involving Human Subjects Act. Direct approval by parental informed consent was waived due to the pseudo-anonymous retrospective data extraction. Data were retrospectively collected from our digital patient data management system (MetaVision, iMDsoft, version 5.46.44).
Children up to the age of 6 years at the time of surgery, who were diagnosed with coarctation of the aorta without other major concurrent anatomical lesions, were eligible for inclusion in our study. The performed surgery had to be registered as coarctectomy with an (extended) end-to-end anastomosis of the aorta, subclavian flap, or patch aortoplasty. Patients were excluded if they had used antihypertensive medication prior to surgery. In general, all children are postoperatively admitted to the pediatric intensive care unit (PICU). However, some children were admitted to the neonatal intensive care unit (NICU) because of capacity issues and were excluded due to missing data as the NICU had no electronic patient data management system at the time.
Ventricular function was evaluated by echocardiography by the attending pediatric cardiologist preoperatively before the start of inotropic support. A fractional shortening of less than 28% was considered a reduced LV function.
The choice to start antihypertensive treatment was left at the discretion of the attending pediatric intensivist in agreement with the operating cardiothoracic surgeon. Children who received an initial antihypertensive medication other than labetalol were excluded. Generally, systolic blood pressure threshold to start treatment was set at or above the 95th percentile for age for children aged older than 12 months and between 90th and 95th percentile when aged 12 months or less. In our PICU intravenous labetalol is the first-line treatment, according to our local protocol, unless there is bradycardia or a history of severe asthma. The goal of antihypertensive treatment is to lower blood pressure at or below the 90th percentile. We do not use a loading dose.
Data were analyzed using SPSS (IBM SPSS® Statistics, version 25). Depending on the normality of distribution, variables are presented as mean/standard deviation or median/interquartile ranges. In statistical analyses, Mann-Whitney U/Wilcoxon rank-sum test was used for numerical/continuous variables, Pearson χ2 test was used for nominal variables with the Fisher exact test in case of small numbers. A binary logistic regression analysis was done with variables that were significantly different between both groups to define predictors of the need for labetalol treatment, taking multicollinearity into account. Multiple regression analysis was done to assess the association between PICU or hospital length of stay with age, left ventricular function, and labetalol treatment. A P value <.05 was considered statistically significant.
Results
A total of 136 patients had undergone coarctectomy. Twenty-nine were excluded because of missing data due to postoperative admission to our neonatal intensive care unit. Other reasons for exclusion were: initial treatment with another antihypertensive medication than labetalol (mainly nitroprusside) in 8 patients, start of labetalol more than 24 h after surgery in 3 patients, absence of invasive arterial blood pressure (ABP) measurement in 2 patients, and concomitant treatment with amiodarone in 1 patient. None were excluded due to prior antihypertensive drug use (Figure 1).
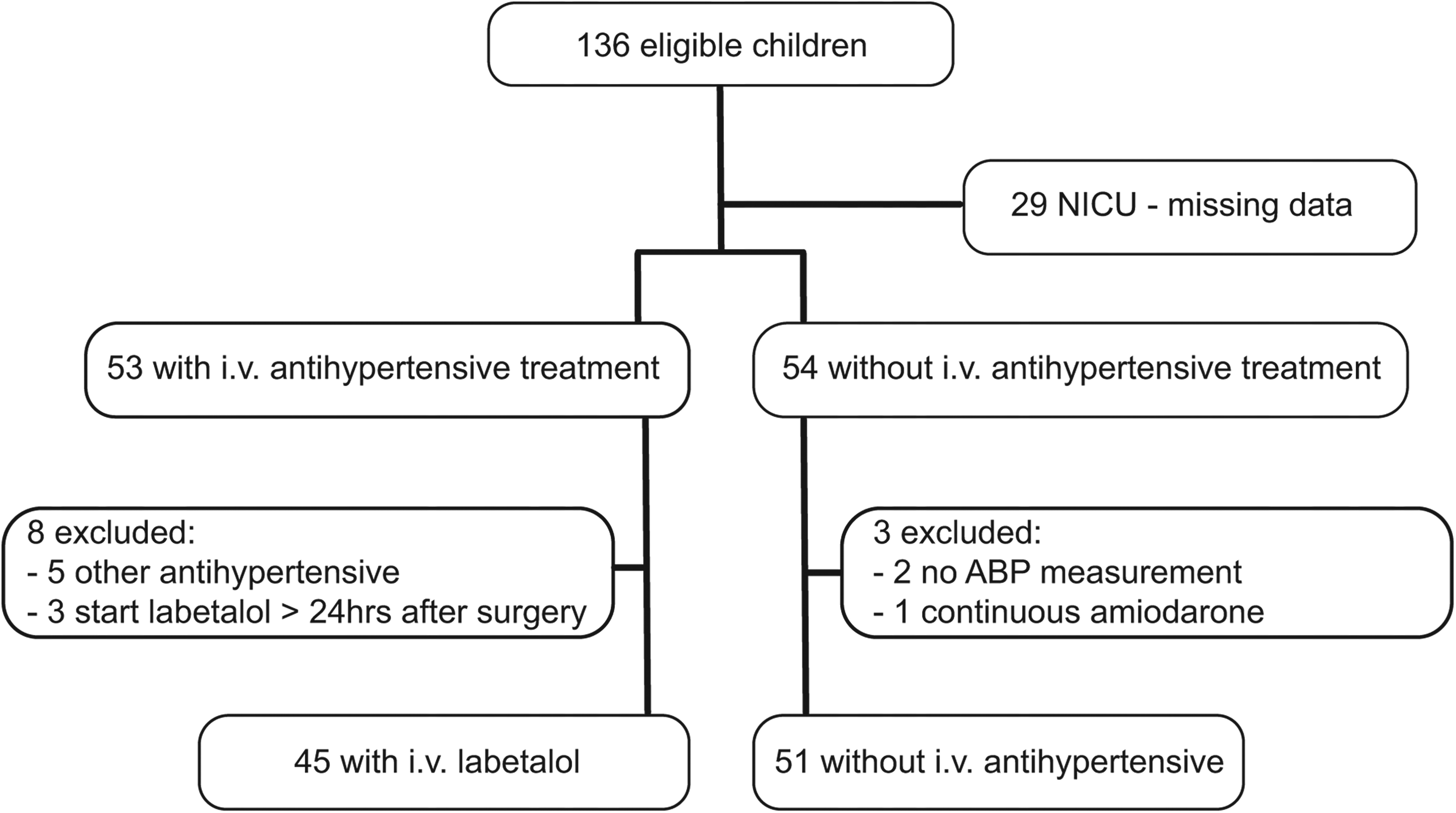
Flow diagram of patients included in the study. Abbreviations: ABP, arterial blood pressure; NICU, neonatal intensive care unit.
Data of all included patients are described in Table 1. Median age and weight were significantly higher in the children who received labetalol compared to those who did not. In addition, left ventricular failure was more prevalent in children who did not receive labetalol. Thirteen of the 16 children with decreased left ventricular function preoperatively were treated with inotropes postoperatively. Ten children received milrinone (1 was also treated with levosimendan), 2 received dobutamine, and 1 received adrenaline. In one child, the aortic coarctation was resected through a median sternotomy, the rest underwent lateral thoracotomy.
Descriptive Statistics of Demographics and Treatment.
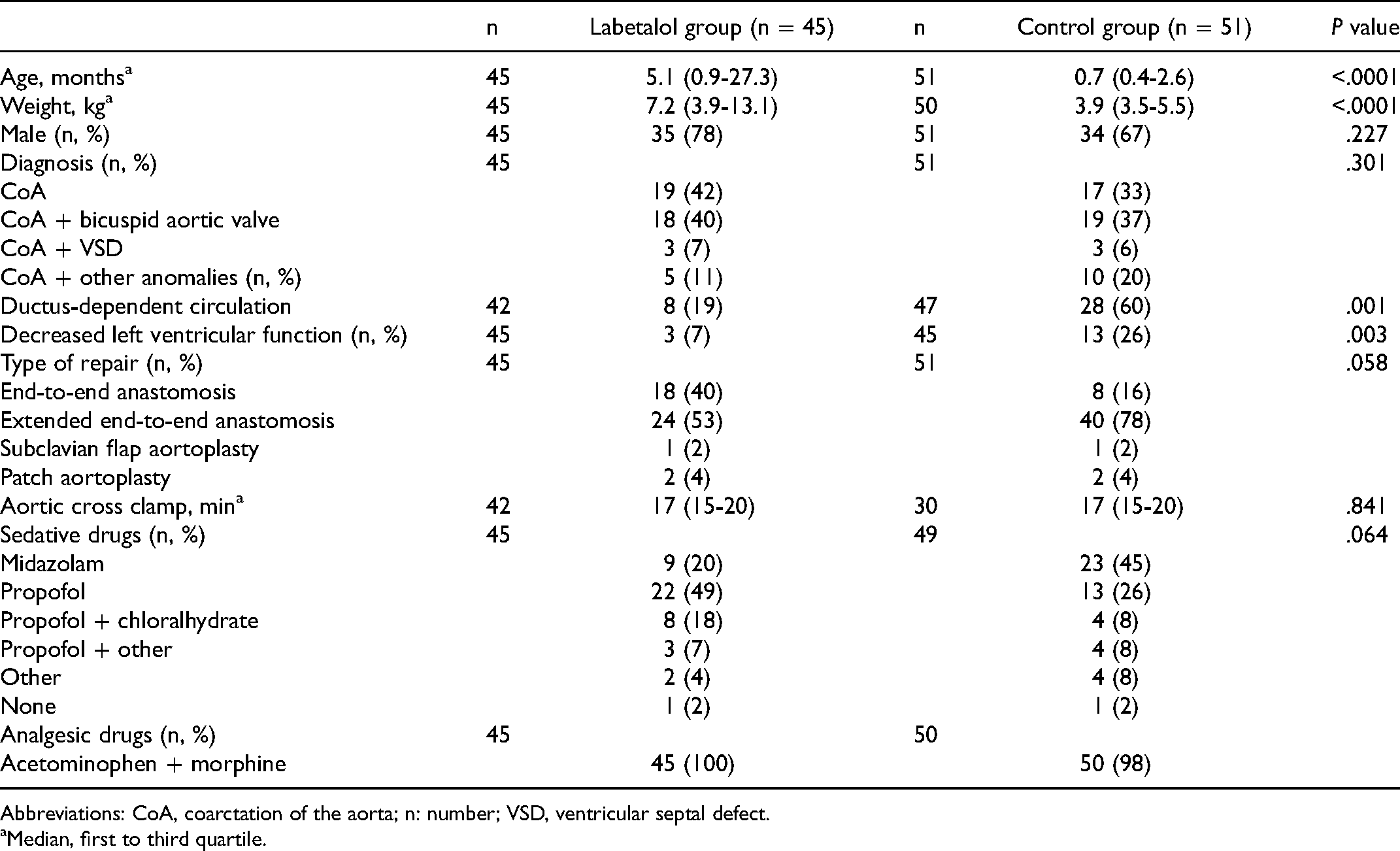
Abbreviations: CoA, coarctation of the aorta; n: number; VSD, ventricular septal defect.
Median, first to third quartile.
In 45 of 96 children (47%) labetalol was started within 24 h after surgery because of hypertension. The median labetalol dose at maximum dosing to reach our therapeutic goal was 1.1 mg/kg/h (first-third quartile (Q1-3) 0.6-1.2 mg/kg/h). The median time to reach the target blood pressure, at maximum labetalol dose, was 2.7 h (Q1-3 1.3-6.8 h). The median time to start reducing labetalol dose, a sign of blood pressure control, was 8.3 h (Q1-3 4.7-12.0 h). All children had a reduction of labetalol dose within 22.7 h after the start of treatment. The median total duration of intravenous labetalol infusion was 14.6 h (Q1-3 8.4-20 h) with a maximum of 49.2 h (see also Figure 2).
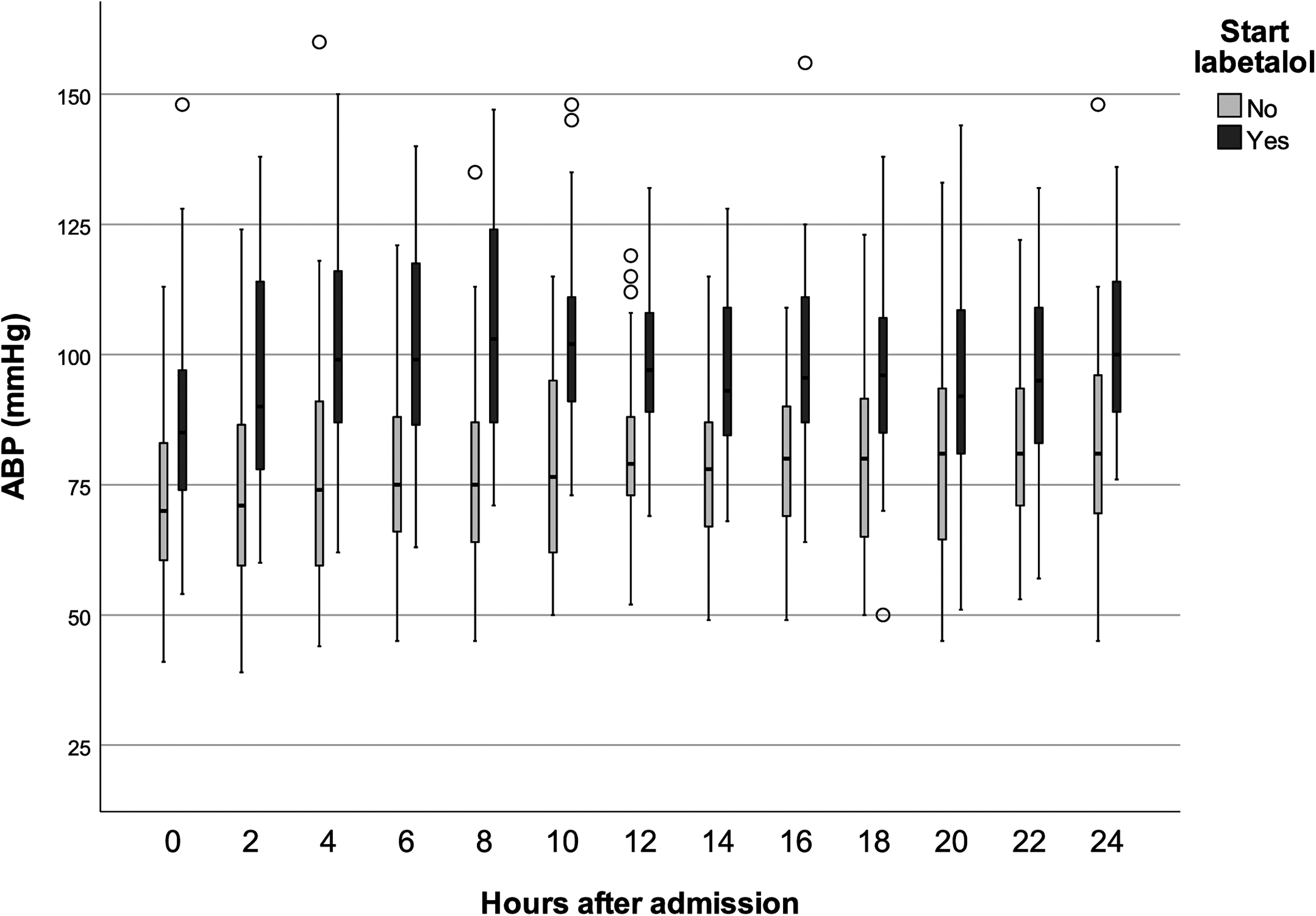
Systolic arterial blood pressure (ABP) in children with and without labetalol. The boxes represent Q1 to Q3 with the median, whiskers to Q1 minus 1.5 IQR and Q3 plus 1.5 IQR, and open dots representing the outliers.
Binary logistic regression with inclusion of age, ductus-dependent circulation, and left ventricular function showed that age and ductus-dependent circulation were significantly associated with intravenous labetalol treatment (P = .009 and P = .03, respectively) with an odds ratio for every month increase of the age of 1.07 (95% CI 1.02-1.12) and for ductus-dependent circulation of 0.41 (95% CI 0.18-0.93). The variance inflation factor (1.12) did not support multicollinearity. After adding near-significant variables (P < .065) sedation type and type of surgical repair, the association remained significant.
Treatment with intravenous labetalol was not associated with an increased PICU or hospital length of stay (LOS) as both were significantly lower in the labetalol group compared to control group (Table 2). When correcting for age on admission and left ventricular function, there still was a negative, however nonsignificant, correlation between start of labetalol and PICU LOS (B = −25.9, t = −1.63, P = .10) or hospital LOS (B = −33.2, t = −1.34, P = .19). There was no mortality in either group.
Descriptive Statistics of Outcome.
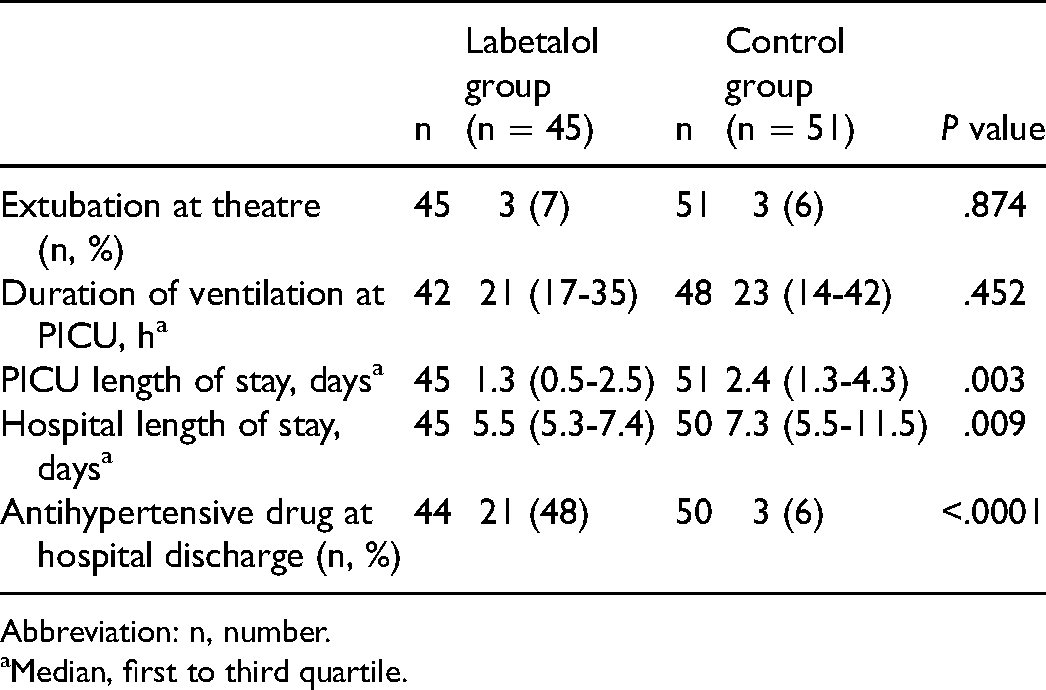
Abbreviation: n, number.
Median, first to third quartile.
With regard to safety, there were no episodes of severe bradycardia during intravenous treatment despite a lower mean heart rate in the labetalol group compared to the nonlabetalol group (Figure 3). The mean heart rate at maximum dose of labetalol was 120 beats per minute (SD 18 bpm). In a subgroup receiving ≥1.5 mg/kg/h, heart rate at maximum dose was 114 beats per minute (SD 13 bpm). One boy, aged one month at the time of surgery, developed bradycardia after intravenous labetalol was switched to oral labetalol. The oral treatment was ceased, and no further antihypertensive treatment was necessary. In another boy, aged 11 months, labetalol was replaced by nitroprusside because of the occurrence of bronchoconstriction during intravenous labetalol treatment. There were no cases of intracerebral hemorrhage nor bleeding at the surgical site.
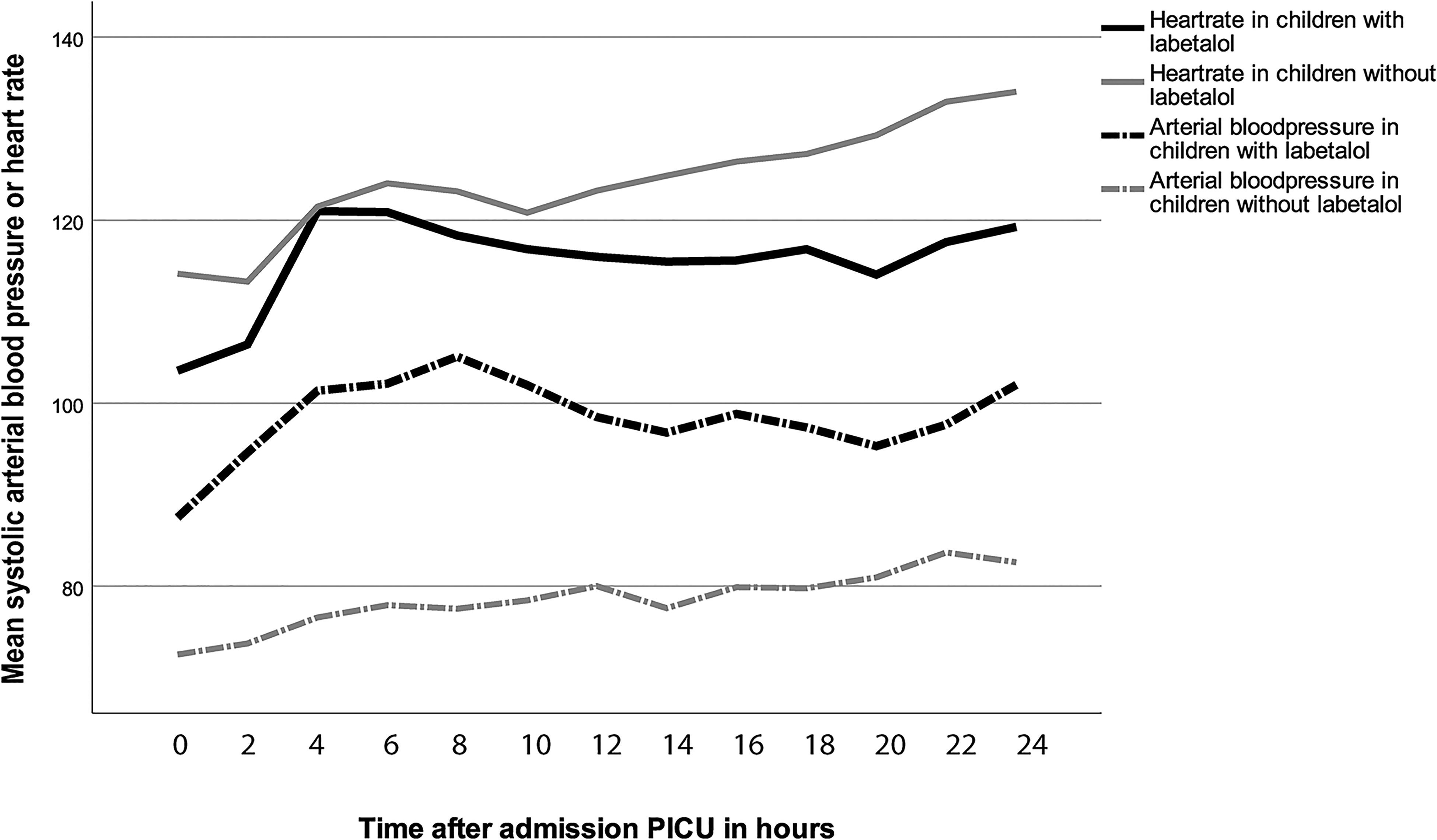
Mean systolic arterial blood pressure (ABP) and heart rate in children with and without labetalol.
Two female children received a second episode of treatment with intravenous labetalol. One girl, aged 19 days at coarctation repair by subclavian flap, was also diagnosed with bicuspid aortic valve and decreased left ventricular function prior to surgery. She was first treated with low-dose labetalol (0.3 mg/kg/h for 4.4 h). The second girl was aged 22 days at coarctation repair by end-to-end anastomosis with a decreased right and left ventricular function prior to surgery. During the first episode, she was treated with labetalol for 16.6 h with a maximum dose of 1 mg/kg/h.
In 13 children (30%) of the labetalol group and 3 children in the control group, oral antihypertensive drugs were started in PICU. At hospital discharge, 21 children (48%) in the labetalol group versus 3 children (6%) in the control group were still treated with antihypertensive drugs. The duration of intravenous labetalol treatment (hrs) appears to be a predictor of oral antihypertensive treatment at hospital discharge (odds ratio = 1.16, 95% CI 1.05-1.29, df 1, P = .006), whereas maximal dose of intravenous labetalol was not (odds ratio = 0.60, 95% CI 0.26-1.40, P = .24).
Comment
In our cohort, almost 50% of children received antihypertensive treatment to correct systemic hypertension after aortic coarctation repair. With intravenous labetalol, sufficient blood pressure reduction was achieved after a median time of 2.7 h with limited side effects and without the requirement to add second-line medication. Treatment with intravenous labetalol was not associated with increased PICU or hospital LOS.
According to our national formulary, the therapeutic range of intravenous labetalol is 0.25 to 3.0 mg/kg/h. 6 As reported, we required a relatively low median dose (1.1 mg/kg/h) to reach an effective decrease in blood pressure. In the study by Charlton et al, 5 labetalol dose was increased up to a maximum of 1.2 mg/kg/h according to their study protocol. This relatively low maximum dose may partly explain why in 12 of their 15 children (80%) nitroprusside was added to achieve adequate blood pressure control.
We were able to reduce labetalol dose after a median of 8.6 h, which we interpreted as a sign of recovery of postoperative catecholamine-related hypertension, whereas Charlton et al 5 only were able to start dose reduction after 18 h. The higher mean age of their patients may explain both differences as there is a known correlation between age and start as well as the duration of antihypertensive treatment. 7
In a multicenter study by Tabbutt et al 7 using esmolol, 118 of 164 children (72%) received antihypertensive treatment within 30 min of cross-clamp release. The higher percentage of children in their study receiving antihypertensive treatment is probably due to the higher number of children already extubated in the operating room and their lower blood pressure thresholds. Although the actual mean systolic blood pressures measured during 24 h post-surgery in their study were significantly higher than these thresholds. Their median dose of esmolol at maximum dosing was 521 μg/kg/min. However, they required the addition of sodium nitroprusside in 59% of their patients, and the median duration of esmolol (19.9 h) was longer compared to the median duration of labetalol in our study (14.6 h).
From binary logistic regression analysis, it appeared that age was positively related to treatment with intravenous labetalol. Correction at an older age may lead to upregulation of renin–angiotensin system, altered vasoreactivity, dysfunctional baroreceptor mechanism, and abnormal aortic compliance and thereby increased requirement of postoperative antihypertensive treatment. This might also explain the longer treatment duration in the older children in prospective studies.5,7 On the other hand, the young age of infants presenting with left ventricular failure upon closure of the ductus arteriosus due to prenatal unknown critical aortic coarctation may also add to this relation. They may be less likely to develop hypertension either due to their decreased ventricular function or because of treatment with afterload-reducing inotropes.
Ductus-dependent circulation was negatively associated with intravenous labetalol treatment. Although this may seem explainable as children with a ductus-dependent circulation are diagnosed at a younger age and more often in circulatory failure, the absence of multicollinearity suggests an, at least partially, independent association.
The use of labetalol was found to be safe. In only one child, intravenous labetalol was ceased because of bronchial constriction, in all other children, we did not find any significant side effects. Heart rate was lower in the children treated with intravenous labetalol, but did not decrease below a clinically significant threshold, as is shown in Figure 3. Charlton et al 5 did not report any adverse effects with the use of labetalol.
In our study, 21 of 44 children treated with intravenous labetalol (48%) were discharged with oral antihypertensive treatment with labetalol. Since up to 83% of children post-coartectomy receive oral antihypertensives at discharge from the hospital,4,7,8 intravenous labetalol treatment has the advantage of facile conversion to oral treatment.
Study Limitations
Since this is a retrospective study, possible confounding factors, and bias that we did not correct for may have influenced our results. Interventions such as intermittent sedation boluses or fluid boluses that may have influenced blood pressure were not recorded in the database. However, these interventions are uncommon and in case hypertension is treated by antihypertensives, sedation boluses have limited effect. Due to the relatively low sample size, statistically significant differences could possibly be missed.
In our study population, antihypertensive treatment was left at the discretion of the attending pediatric intensivist. Therefore, there was no uniform systolic blood pressure at which treatment was started, although in younger children 100 mm Hg and in older children 120 mm Hg was mostly used as a threshold level. These threshold levels could be interpreted as relatively high, although the threshold levels for the younger children match the 90th to 95th percentile for that age. Besides, these levels aim at preventing postoperative hemorrhage at the surgical site, whereas in addition, older patients may have adapted preoperatively to high cerebral and cardiac blood pressures. The lack of international guidelines with regard to blood pressure targets limits the comparability of the various studies.
In our pediatric ICU, we do not use a loading dose of labetalol, which might delay reaching a steady-state blood level. This may have led to prolonged duration in achieving target blood pressure. Since the median and maximum age of our study population was relatively young, our results may only be applicable to this age group. And finally, the single-center design may limit generalizability of the findings.
This study focusses on postoperative treatment of post-coarctectomy hypertension. We have not studied the effect of prophylactic antihypertensive treatment despite the fact that previous studies have shown a possible positive effect of prophylactic use of beta-blockers prior to coarctectomy on postoperative hypertension. However, these were small studies in older patients with higher blood pressure limits than is common today, which is probably why prophylactic treatment is not standard care nowadays, especially in the current younger age group. Larger prospective randomized controlled trials on this approach may be interesting.9,10
Conclusion
This retrospective study supports the use of intravenous labetalol to treat post-coarctectomy hypertension as an effective, efficient, and safe single-drug treatment with the advantage of facile conversion to oral treatment. Future prospective studies should aim at uniformity in patient selection and blood pressure targets to develop age-specific treatments comparing different drug regimens.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
