Abstract
Wire atrial septostomy, a new transcatheter technique, combines 2 atrial septal defects (ASDs) into a large one in a thickened atrial septum. Although the thin-wire-looped atrial septum between ASDs needs to be pulled deeply into a hard catheter to tear the septum, this action might harm the surrounding cardiac structures. We utilized radiofrequency wire energy to facilitate the tearing of the septum, making the procedure easier and safer. This technical article presents the modified procedure in detail; the procedure was performed in a 6-month-old boy with double outlet right ventricle and intact ventricular septum.
Mini-abstract
This technical article describes modified wire atrial septostomy for thickened atrial septum in detail. Radiofrequency energy facilitated a thin wire-loop in tearing the septum between 2 atrial septal defects to combine the defects into a large one. We believe that this transcatheter procedure is easier and safer than the original one not using a radiofrequency wire energy.
Keywords
Introduction
Ventriculo-coronary artery communication is often recognized in patients with either ventricular outlet atresia, such as pulmonary atresia with intact interventricular septum (IVS), 1 mitral stenosis and aortic atresia in hypoplastic left heart syndrome, 2 and, rarely, double outlet right ventricle (DORV) with IVS. 3 In addition, some patients have right ventricle- or left ventricle-dependent coronary circulation (LVDCC). When interatrial communication is restrictive in such patients, balloon atrial septostomy 4 or static balloon dilation 5 is considered. However, these procedures are often ineffective for thickened atrial septa beyond the neonatal period. In addition, surgical atrial septal defect (ASD) enlargement using a cardiopulmonary bypass may lead to myocardial ischemia because it decompresses the ventricle that directly supplies blood to the coronary arteries. 6 In such cases, wire atrial septostomy (WAS) is a reasonable treatment option to create a large ASD against thickened atrial septa without any remnant artificial material. 7 This technical article describes a modified WAS procedure using radiofrequency wire energy; this procedure was performed in an infant with DORV, IVS, LVDCC, and restrictive foramen ovale (FO). We believe that the modified WAS is easier and safer than the original procedure not using radiofrequency wire energy.
Case
We present the case of a 6-month-old male infant weighing 5.0 kg with atrioventricular discordance, mitral valve stenosis, DORV, IVS, severe pulmonary valve stenosis, patent ductus arteriosus, restrictive FO, and LVDCC. Following birth, a continuous infusion of prostaglandin E1 was started to maintain the patency of the ductus arteriosus. In addition, balloon atrial septostomy was performed for restrictive FO; however, a slight obstruction remained.
At the age of 2 days, he experienced shock due to increased pulmonary blood flow involving myocardial ischemia with serum troponin T level elevation (1.4 ng/mL). Despite medical management, he developed necrotizing enterocolitis. Therefore, bilateral pulmonary artery banding was emergently performed to regulate pulmonary blood flow at the age of 21 days. Right-ventricular function gradually improved, and the patient grew up.
At the age of 6 months, we decided to perform transcatheter ASD creation before bidirectional Glenn surgery without cardiopulmonary bypass because we considered that surgical ASD enlargement with cardiopulmonary bypass could involve fatal myocardial ischemia associated with LVDCC, similar to the previous myocardial ischemia episode. In addition, we believed that conventional septostomy would be ineffective because FO was located at the superior side of the atrial septum and the septum appeared thickened (Figure 1A, and B). Hence, modified WAS was attempted.
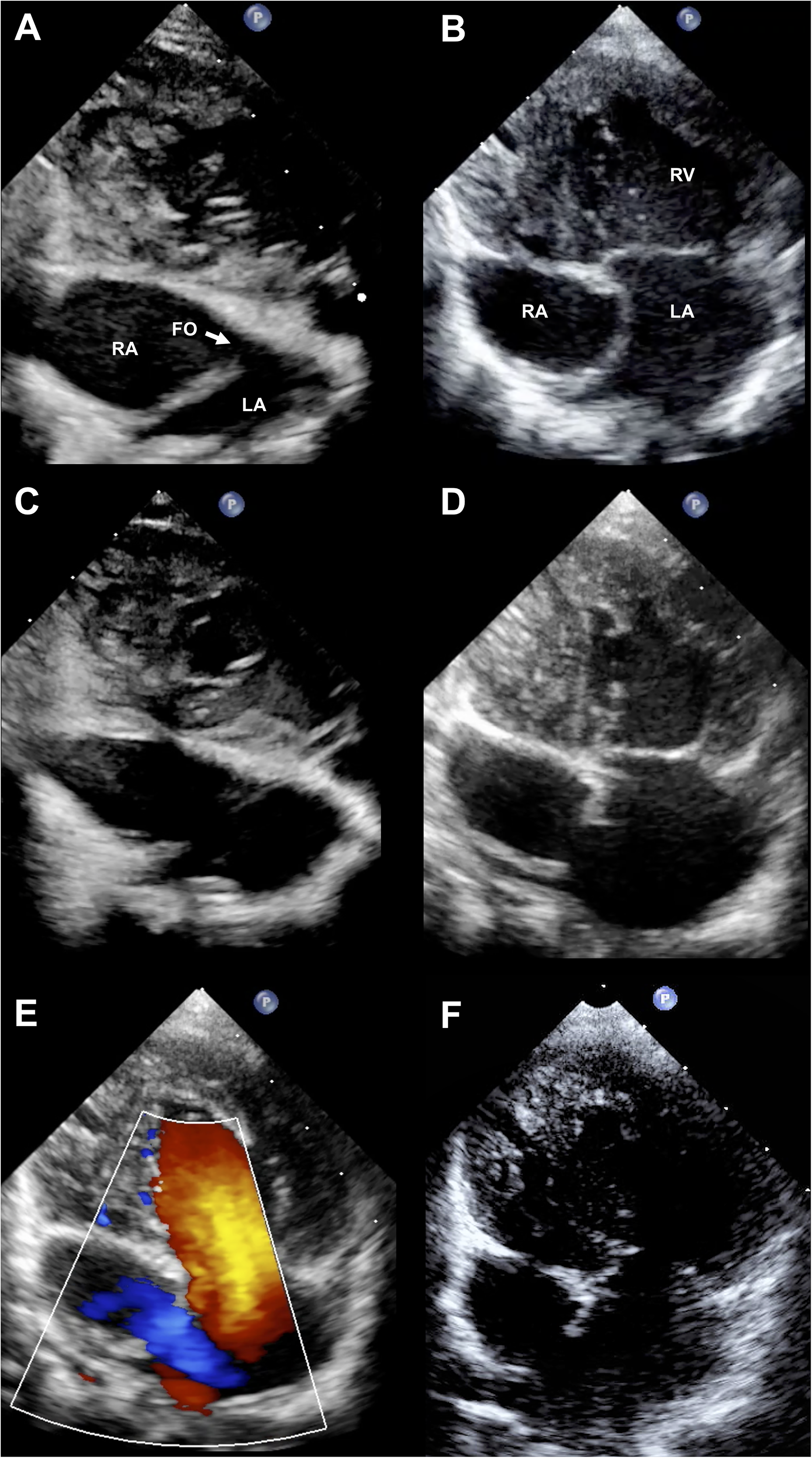
Echocardiograms before and after modified wire atrial septostomy: A, subxiphoid view before the procedure, B, four-chamber view before the procedure C, subxiphoid view after the procedure, D and E, 4-chamber view after the procedure, F 4-chamber view 8 months after the procedure.
Modified WAS Procedure
A catheterization study showed that the right and left ventricle pressures were 75/7 and 70/8 mm Hg, respectively, and the right atrium (RA) and left atrium (LA) mean pressures were 9 and 8 mm Hg, respectively. Left ventriculography and aortography confirmed that the left circumflex coronary artery originated only from the left ventricle, indicating LVDCC. A 6 Fr sheath was inserted into the right femoral vein, and the following procedures were performed under fluoroscopy and transesophageal echocardiography guidance. First, a 6 Fr Judkins right guiding catheter (Vista Brite Tip, Cordis, Ohio, USA) was advanced into the RA. After setting a 2.6 to 2.8 Fr microcatheter (Masters PARKWAY HF, Asahi, Japan) and Nykanen radiofrequency wire (Baylis Medical, Mississauga, ON, Canada) in the guiding catheter, we pushed it to the center of the atrial septum with tenting and perforated the site with the Nykanen wire at 10 W for 1 s (Figure 2A). After advancing the microcatheter into the LA, a 0.014 inch hard guide wire (Spindle XS, Asahi, Japan) was placed there. Second, for the perforated hole, we performed serial balloon dilation using a 5 × 15-mm NC Emerge (Boston Scientific, Marlborough, MA, USA) at 18 atm and an 8 × 20-mm Sterling (Boston Scientific, Marlborough, MA, USA) at 12 atm (Figure 2B). Third, via the dilated ASD, we advanced the guiding catheter into the LA and directed the tip upward. From this catheter, we advanced a 4 Fr pigtail catheter (Hanaco Medical, Japan). Whose tip was cut in a J-shape, and directed it toward the FO. From the pigtail catheter, we further advanced a 0.025 inch soft wire, which was attached to the 2.7 Fr microcatheter (Progreat, Terumo, Japan), into the inferior vena cava (IVC) via FO as gently as possible (Figure 2[C]). The microcatheter was subsequently advanced into the IVC, and the wire was exchanged for a 0.01 inch soft wire (RG3, Asahi, Japan). After pulling the guiding catheter to the IVC, we removed the pigtail and microcatheters out of the body, advanced a 4 mm Amplatz Goose Neck Snare (Medtronic, Minneapolis, MN, USA) from the guiding catheter, caught the RG3 tip with the snare, and pulled the tip out of the body. Consequently, the RG3 wire was looped in the order of guiding catheter, RA, ASD, LA, FO, RA, and guiding catheter. Finally, we advanced the guiding catheter to the atrial septum between the 2 defects, set the Nykanen wire in the top of the guiding catheter (Figure 2D), pulled the wire-looped septum into the guiding catheter to touch the Nykanen wire, and energized it at 10 W for 1 s (Figure 2E, Supplemental video S1). The energy by itself did not tear the septum completely; however, a gentle pulling of the wire loop tore it. Hence, the 2 defects were combined into a large ASD with a diameter of 13 mm, which was confirmed by echocardiography (Figure 1C–E). Although serum troponin T level was elevated (0.45 ng/mL), there were no complications, such as obvious ischemic findings and arrhythmia. Oral low-dose aspirin was then initiated to prevent thrombosis.
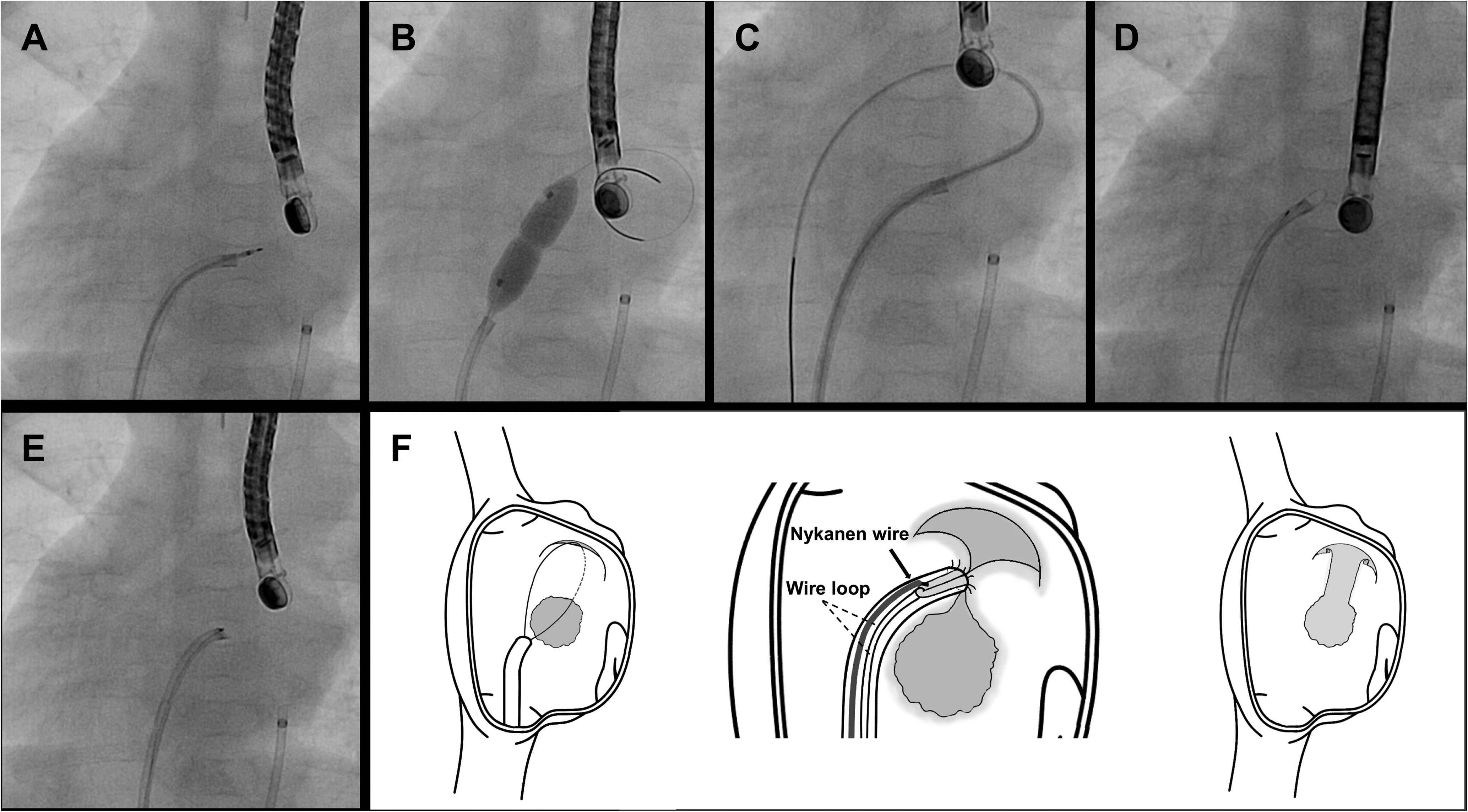
Angiograms during modified wire atrial septostomy (WAS): A, atrial septal perforation with Nykanen radiofrequency wire. B, static balloon dilation of the perforated hole, C, a microcatheter was advanced into the inferior vena cava via the foramen ovale from the 4 Fr tip-cut pigtail catheter advanced into the left atrium, D, the guiding catheter was advanced into the atrial septum between the 2 defects on the wire loop, and the Nykanen wire was set in the top of the guiding catheter, E, after pulling the wire-looped septum into the guiding catheter to touch the Nykanen wire, it was energized at 10 W for 1 s to tear the septum, F, schema indicating the principal of the modified WAS procedure and the defects on the atrial septum before and after the procedure.
Clinical Course
After 1 month, an off-pump bidirectional Glenn procedure was performed without any myocardial ischemic event. The patient was discharged 18 days postoperatively. The ASD remains large enough 8 months after modified WAS (Figure 1F), and the patient awaits undergoing Fontan procedure.
Discussion
In patients with restrictive interatrial communication and thickened atrial septum, blade atrial septostomy creates a large ASD with a diameter of 10 to 16 mm. However, this can lead to serious complications, such as cardiac perforation (3.8%). 8 In addition, although atrial septal stenting is effective, it can lead to the complications of thrombosis and stent embolization. 9 Some patients with ventricle-dependent coronary circulation, such as the present case, prefer to avoid open heart surgeries with cardiopulmonary bypass. In this situation, WAS is a good treatment option to create a large ASD without any remnant artificial material. 7 It involves a combination of perforation, static balloon dilation and merging the created ASD and FO into a single, large defect.
Although a 12-mm-diameter balloon catheter was used for static balloon dilation in the original WAS, we used an 8-mm-diameter balloon catheter, that can pass through the 6 Fr guiding catheters, in this case, resulting in an adequate effect. It is believed that when the ASD size created by static balloon dilation is not large enough, the distance between the 2 defects greatly affects the final combined ASD size. Hence, as the distance becomes greater, the combined ASD size becomes larger. However, greater distances between the defects can increase the difficulty of tearing. By contrast, as observed in the present case, Nykanen radiofrequency wire energy helped tear the septum between the 2 defects in the guiding catheter and reduced the traction stress of the septum on the surrounding cardiac structures.
Similarly, the modified WAS may be effective in elderly patients with thickened atrial septum. Also, the original 7 and modified WAS both have shown durable results. We speculate the reason is because in WAS, the septum between the 2 defects is strongly squeezed before cut and parts of the tissue may be dead and absorbed. Furthermore, the modified WAS procedure is a broadly contained transcatheter electrosurgery. The “Flying V” technique is another transcatheter electrosurgery technique that lacerates leaflets by concentrating current at the inner lacerating surface of a kinked guidewire. 10 Applying the “Flying V” technique may make our modified WAS procedure simpler.
Conclusion
We believe that the modified WAS using Nykanen radiofrequency wire energy is a safe and extremely useful treatment option to create a large ASD for thickened atrial septum, particularly in patients who prefer to avoid open heart surgery.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Ethical Approval
The ethics committee of Okinawa Prefectural Nanbu Medical Center & Children's Medical Center approved this report (No. R2-101).
Informed Consent
We confirm that written informed consent for publication was provided by the patient's guardians.
Supplemental Material
Supplemental material for this article is available online.
