Abstract
Objectives:
Restenosis after aortic arch reconstruction is a known complication in neonates and infants. Homograft is the most commonly used patch material for aortic arch reconstructions in our center. Since 2014, tissue-engineered bovine pericardium (CardioCel) has been used as an alternative. The aim of our study was to determine whether the choice of material affected the development of restenosis in these patients.
Methods:
Data of all neonates and infants who underwent aortic arch reconstruction with the use of any patch material between 2005 and 2016 were analyzed. Restenosis was defined by the need for reintervention, either percutaneous or surgical.
Results:
Forty-one patients underwent aortic arch repair. Excluding the 30-day mortality, 36 patients represented the study population. At primary repair, the aortic arch was reconstructed with homograft (n = 26) or CardioCel (n = 10). Restenosis was documented during the first year of life in 13 patients: Six (23%) patients in the homograft group and seven (70%) patients in the CardioCel group (P = .01). In the homograft group, the median time from operation to first intervention for restenosis was 22.0 (range: 14-32) weeks, as compared to 14.0 (range: 7-21) weeks in the CardioCel group (P = .04).
Conclusion:
We conclude that choice of patch material is likely to be an important determinant for the risk of restenosis needing reintervention following reconstruction of the aortic arch in neonates and infants.
Introduction
Restenosis after aortic arch reconstruction is a known complication in neonates and infants. 1 Various materials can be used in aortic arch surgery such as homograft or pericardial patches, either homologous or from animal tissue. 2,3 It has been postulated that the patch material used for aortic arch reconstruction is an important factor in the development of restenosis. 1,2 Homograft (usually pulmonary artery) is the most frequently used patch material for aortic arch reconstructions in our center. Because homograft availability is limited in the Netherlands, we searched for an approved commercially, readily available alternative. Since 2014, tissue-engineered bovine pericardium (CardioCel, Admedus Regen Pty Ltd) became available and was approved for use in congenital heart defects. 4,5 In contrast to homograft, there is no waiting time for CardioCel, which otherwise might delay the operation. The aim of our retrospective study was to determine whether the patch material used for aortic arch reconstruction in neonates and infants had an influence on the development of significant restenosis.
Patients and Methods
Data of all neonates and infants who underwent aortic arch reconstruction with use of homograft or bovine pericardium (CardioCel), in either univentricular or biventricular repairs, at the Erasmus Medical Centre Rotterdam, between 2005 and 2016, were reviewed retrospectively. Data were extracted from the local surgical database and electronic patient notes. Echocardiographic images and reports, operation reports, and available angiographic images of these patients were reviewed. Demographics included cardiac diagnosis, age, sex, weight at the time of procedure, serum creatinine, birth term, birth weight, and diameter of the transversal aortic arch and ascending aorta. Operative variables included the surgical procedure performed, as well as the material used for aortic patching, duration of the operation and the extracorporeal circulation and selective cerebral perfusion. Postoperative variables included length of both hospital stay and intensive care unit (ICU) stay.
Reasons to reintervene, either by percutaneous or surgical approach, included noninvasive blood pressure gradient (defined as a pressure gradient >20 mm Hg between arm and legs), the presence of upper limb hypertension, left ventricular hypertrophy, echocardiographic data, and/or clinical symptoms if present. The local medical ethics commission waived the need for individual consent, granting approval for retrospective review and analysis of deidentified data.
Surgical Repair
Aortic arch reconstructions were performed through a midline sternotomy with standard bypass cannulation. Operations were performed under deep hypothermia with circulatory arrest and antegrade cerebral perfusion utilizing balloon catheters in the innominate artery and/or the left carotid artery, with temporary clamping of the left subclavian artery. During the procedure, near-infrared spectroscopy was used to monitor brain oximetry. The arch was incised along the inner curvature into the descending aorta. Ductal tissue was resected. In the case of a coarctation shelf, this was also resected (n = 14), with direct anastomosis of the aortic arch and descending aorta at the outer curvature, and patch augmentation of the aorta at the inner curvature from the descending aorta into the ascending aorta. This was performed in all cases in order to provide an adequate lumen with continuity of native aortic tissue at the outer curvature to provide potential for growth. The same technique for arch reconstruction was applied in cases of interrupted aortic arch. All operations were performed by the same two surgeons. Furthermore, the same surgical technique was used throughout the whole study period, with either patch material.
Statistical Analysis
Statistical analysis was performed using Statistical Package for the Social Sciences (SPSS) software, version 24.0 (SPSS Inc). Frequencies were given as absolute numbers and percentages. For normally distributed continuous data, mean and standard deviation (mean ± SD) were used. For not normally distributed continuous data, the median with data range (minimum to maximum) were employed. For discriminating between normally and non-normally distributed variables, the Kolmogorov-Smirnov test was applied. For normally distributed continuous data, the unpaired t test was used, for non-normally distributed continuous data the Mann-Whitney U test was used. We applied the χ2 test to compare frequencies in the two groups. The Kaplan-Meier method was applied to estimate freedom from reintervention. For all tests, a P value of less than .05 was considered significant.
Results
During the study time frame, 41 patients underwent aortic arch repair either as part of complex cardiac surgery or as single intervention. Five patients died within 30 days after the operation. As they were unlikely to have developed restenosis, they were excluded from the analysis. All of them had undergone Norwood palliation for univentricular physiology. Four received homograft and one received CardioCel for arch reconstruction. None of these deaths were related to the failure of the graft used for arch reconstruction. None had signs of immediate postoperative aortic arch obstruction. The cause of early death was hemodynamic instability and cardiac failure in all of them.
Therefore, the study population comprised 36 patients (22 males [61%], 14 females [39%]). In all, 26 underwent arch repair with homograft and 10 with CardioCel.
Twenty-five patients had a functionally univentricular heart and were diagnosed with hypoplastic left heart syndrome (n = 22), double inlet left ventricle with aortic arch hypoplasia (n = 2), and double outlet right ventricle of Taussig Bing type with aortic arch hypoplasia (n = 1).
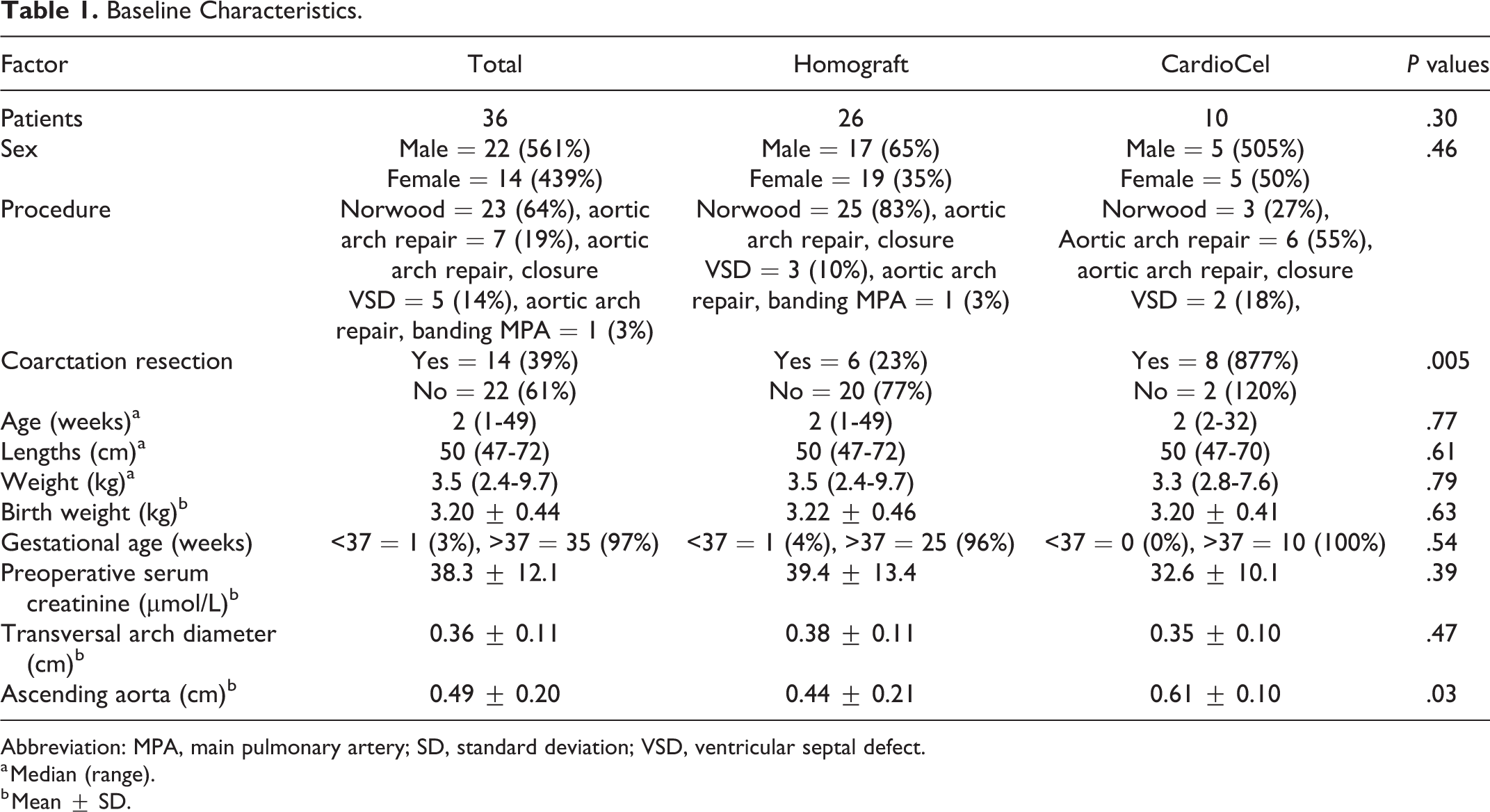
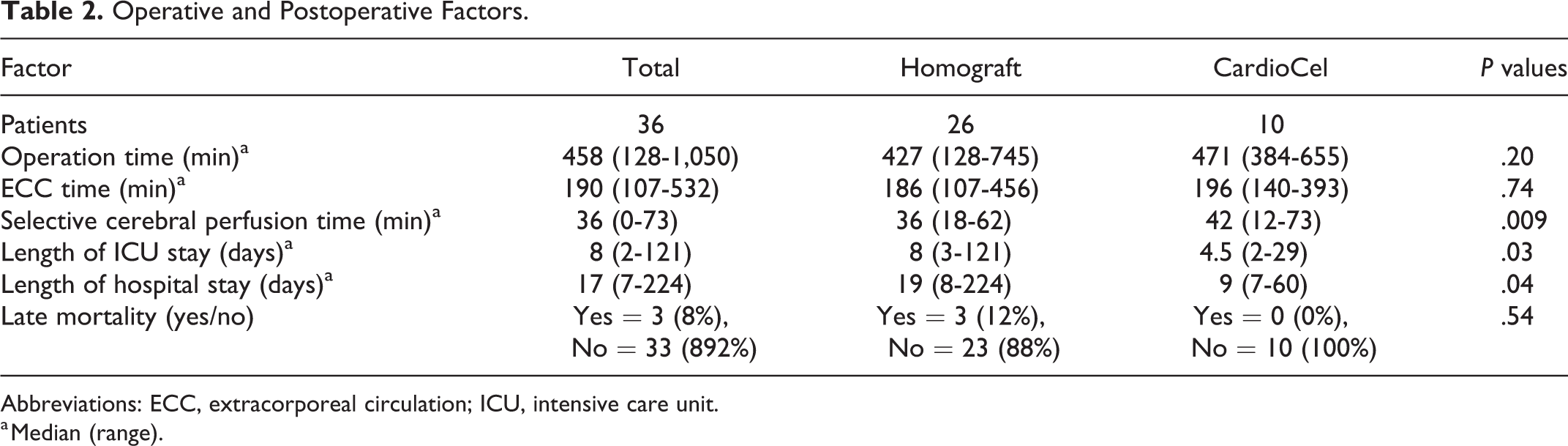
Ten of the remaining 11 patients presented with biventricular hearts with aortic arch hypoplasia, including coarctation associated with ventricular septal defect (VSD)s (n = 5), bicuspid aortic valve (n = 3), and partial anomalous pulmonary venous return (n = 1). One biventriculair heart patient had interrupted aortic arch type B with a perimembranous VSD. Patient characteristics, surgical data, and perioperative data are presented in Tables 1 and 2.
Baseline Characteristics.
Abbreviation: MPA, main pulmonary artery; SD, standard deviation; VSD, ventricular septal defect.
a Median (range).
b Mean ± SD.
Operative and Postoperative Factors.
Abbreviations: ECC, extracorporeal circulation; ICU, intensive care unit.
a Median (range).
All patients, except one, were managed on prostaglandin to maintain ductal patency until the operation. The operations performed were the Norwood procedure (n = 23, 64%; isolated aortic arch repair, n = 7, 19%; aortic arch repair with VSD closure, n = 5, 14%; and aortic arch repair with pulmonary artery banding, n = 1, 13%).
Twenty-six patients underwent arch repair with homograft patch material, and ten with CardioCel. Coarctation resection was carried out in 6 (23%) of 26 cases in the homograft group and in 8 (80%) of 10 in the CardioCel group.
Patient characteristics were similar between the two groups, except for a significantly (P = .03) larger ascending aorta in the CardioCel group (0.61 cm ± 0.10 cm) versus the homograft group (0.44 ± 0.21). Most of these patients had a biventricular repair. We observed a significantly longer ICU stay (P = .005) and hospital stay (P = .001) for the univentricular repairs compared to the biventriculair repairs, regardless of the patch material used. Late mortality did not differ significantly between the two groups (P = .54). In total, 13 patients underwent reintervention for restenosis in the first year of life: Six (23%) of 26 in the homograft group and seven (70%) of ten in the CardioCel group. Freedom from reintervention was estimated as 77% and 30% at one year, for the homograft and CardioCel groups, respectively (P < .001). Figure 1 illustrates freedom from reintervention.
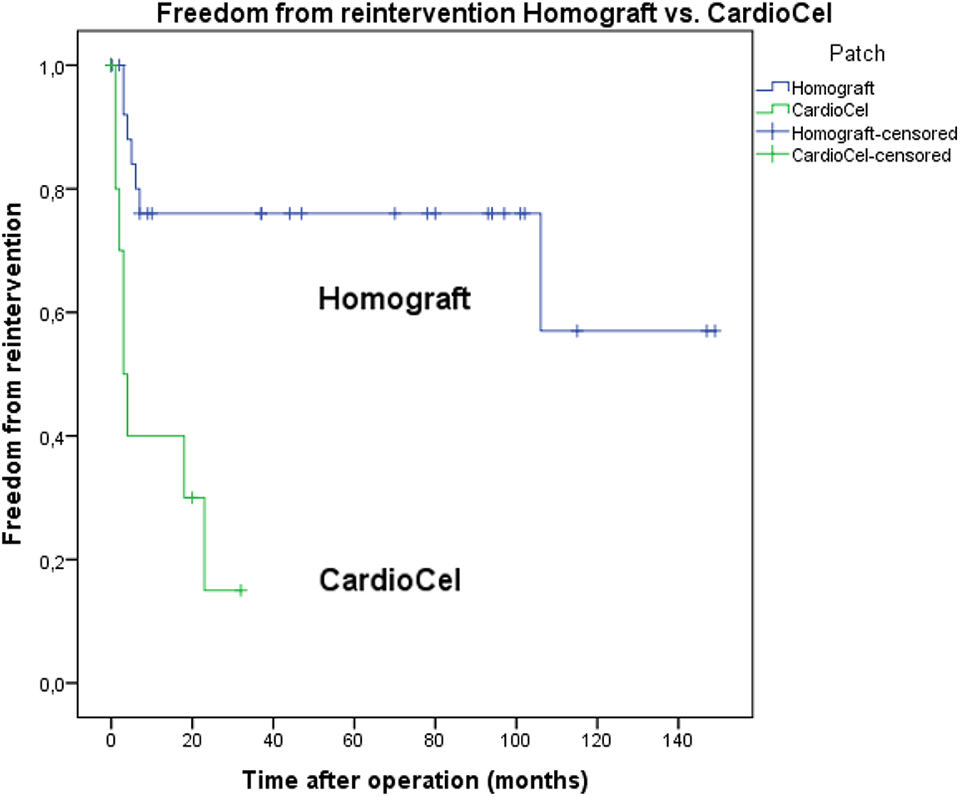
Kaplan-Meier plot for freedom from reintervention. P < .0001.
Treatment for restenosis consisted of percutaneous balloon angioplasty in 11 patients, stent implantation in one, and reoperation in one. The mean invasive pressure gradient at the first catheter intervention (n = 12) was 37 ± 20 mm Hg. In the homograft group, the mean invasive pressure gradient was 29 ± 17 mm Hg, slightly lower (P = .17) than the Cardiocel group (45 ± 22 mm Hg). The median time from operation to the first reintervention was 22.0 weeks (range: 14-32) for the homograft group and 14.0 weeks (range: 7-21) for CardioCel. This difference reached the level of significance (P = .035). A second reintervention was performed in six patients, one in the homograft group and five in the CardioCel group. Two patients, one in each group, underwent a third reintervention. Of these, one patient in the CardioCel group needed a fourth reintervention. Almost all catheter-based reinterventions consisted of balloon angioplasty (n = 19). In only two patients, stents were implanted. Among patients with restenosis, the lesion had to be reintervened more frequently in the CardioCel group (mean of 2.0 reinterventions per patient) as compared to the homograft group (mean of 1.3 reinterventions per patient), but the difference was not significant (P = .2). There were neither intervention-related complications nor deaths.
Discussion
The patch material used for aortic arch repair is considered a potential factor in the occurrence of restenosis. Biological materials available for aortic arch reconstruction in children include pulmonary artery homograft, aortic homograft, autologous pericardium, and equine or bovine pericardial patches. Pulmonary homograft is the most commonly used patch material for aortic arch reconstructions but is not always available.
Patches are frequently used in surgery for congenital heart defects. The ideal patch material should fulfill the following requirements: resistance to infection, calcification and shrinkage, good biocompatibility and durability, and a predictable outcome. 3 In 2013, a first study demonstrated the safety and efficacy of the tissue-engineered ADAPT bovine pericardial patch (CardioCel) in pediatric patients with a range of congenital cardiac anomalies. 5 Neethling and associates demonstrated that the use of this bovine pericardial patch was safe and efficient in 30 patients with atrial septal defect (ASD), VSD, atrioventricular septal defect, aortic root enlargement, and right ventricular outflow tract reconstruction. An interesting application of CardioCel is in heart valve replacement. Labrosse et al demonstrated that CardioCel patches are mechanically close equivalents to young aortic valve leaflets. 6 Konstantinidou and Moat used CardioCel to repair the tricuspid valve after trauma-induced tricuspid regurgitation. 7 CardioCel is considered a good patching material with respect to avoidance of calcification and inflammation. 4,5
As a consequence of the present study findings, we have stopped using CardioCel for aortic arch augmentation in neonates and infants. We continue to favor homograft use for aortic patch reconstruction in our center.
In the Single Ventricle Reconstruction Trial, interventions for restenosis after aortic arch repair in neonates reached a cumulative incidence of 18%, ranging from 0% to 50%, at 12 months. 8 Especially in this patient group, any aortic arch (re-)obstruction is considered an important risk factor for ventricular dysfunction and worse outcomes. 8
Several factors, including both patient characteristics and surgical technique, are hypothesized to contribute to recurrent aortic obstruction. 9 Although some groups report excellent results with the interdigitating technique 10 for aortic reconstruction, this is not universally applied. Residual ductal tissue and incomplete resection of discrete coarctations are recognized risk factors for residual or recurrent obstruction. In our patients, all macroscopically visible ductal tissue as well as all coarctation shelves was completely excised. Considering that coarctation resection was more frequently (80%) performed in the CardioCel group than in the homograft group (23%), we found it worrisome that the restenosis rate was significantly higher in the CardioCel group. CardioCel has only been used for approximately three years (since 2014) for this indication in our center, compared to homograft, which was introduced in 2005. As a consequence, the follow-up period of patients who received homograft is much longer compared to the CardioCel patients. However, our focus was on reinterventions within the first postoperative year.
As opposed to our study, in which we evaluated outcomes associated with patch material only in the context of aortic reconstruction, Pavy et al demonstrated good results with CardioCel in low-pressure areas, such as in the septal, valvar, and pulmonary artery positions. However, they experienced early graft failures in infants after aortic repair, similar to our findings. This workgroup hypothesized that CardioCel early graft failure under exposure to intraluminal high pressure was related to inadequate elastic properties. 11 In their study, histological examination of the explanted patch material showed an enormous neointimal proliferation (11). They considered this to be a result of the elasticity mismatch between the native aorta and the CardioCel patch. The shear stress generated by the pulsatile systemic blood flow may predispose to greater reactive intimal hypertrophy in the blood flow path area, thus leading to severe stenosis. Vitanova and associates studied 145 neonates with hypoplastic left heart syndrome after Norwood Stage I operation and observed that 65% of the patients in whom the arch was augmented with equine pericardium developed restenosis, a rate significantly higher than those related to the use of homograft or autologous pericardium patch. 1 The higher restenosis rates reported by Vitanova et al 1 with CardioCel aortic patching were similar to ours. In addition, we observed that the invasively measured pressure gradient (45 ± 22 vs 29 ± 17 mm Hg) at percutaneous reintervention was higher in the Cardiocel group compared to the homograft group, albeit not significantly so (P = .11). Furthermore, we noticed that the number of subsequent reinterventions for restenosis tended to be higher in this group. It can be speculated that restenosis after CardioCel use is more resistant to the usual interventional methods of balloon dilatation. This could be related to intrinsic properties of CardioCel patch material that contribute to early restenosis, or it may be that this material is associated with the development of neointimal proliferation which is less amenable to balloon dilatation. Bell et al reported good durability when CardioCel was used for the repair of congenital heart defects at many different cardiac sites, including aortic root, valve, and arch repairs, across a range of ages. The specific number of neonates in which CardioCel was used for aortic arch repair was not mentioned. However, they reported one patient who underwent a Yasui operation for repair of type B interrupted aortic arch who subsequently required two balloon dilations and one reoperation for restenosis of the aortic arch. 12
Limitations
Our investigation has the limitation inherent to retrospective studies. Aortic arch augmentation was performed both in neonates and infants, as well as in univentricular and biventricular repairs. Our study was not powered for further multivariable analysis, and the numbers were too low to perform a subgroup analysis to draw firm conclusions.
Restenosis could be related to a suboptimal initial surgical result. For the earliest portion of our study cohort, patients were subjected to meticulous care, but there was not a standardized protocol for postoperative surveillance, this being developed later and therefore not applied to the whole study population. The likelihood that some instances of suboptimal immediate postoperative results were “missed” is very unlikely, but it cannot be completely excluded. In addition, the CardioCel patch material was used during the later portion of the study period, during which the standardized protocol for postoperative surveillance was utilized. It would be very interesting to obtain histopathological tissue of explanted material to better understand the underlying mechanism(s) of the higher rate of restenosis that was observed with the use of CardioCel. Since most instances of restenosis were treated percutaneously with balloon angioplasty alone, it was not possible to obtain intraoperative photographs or explanted material.
Conclusion
We conclude that choice of patch material is likely to be an important determinant for the risk of restenosis needing reintervention following reconstruction of the aortic arch in neonates and infants and the number of reinterventions needed to treat them. Based on our own observations and in accordance with the findings of previous studies by other investigators, we favor the use of homograft patch material for aortic arch augmentation in neonates and infants, and we no longer use CardioCel patch material for this application.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
