Abstract
Background:
Despite the improved outcome in complete atrioventricular septal defect (AVSD) repair, reoperations for left atrioventricular valve (LAVV) dysfunction are common. The aim of this study was to evaluate the effect of fresh untreated autologous pericardium for ventricular septal defect (VSD) closure on atrioventricular valve function and compare the results with the use of treated bovine pericardial patch material.
Methods:
Clinical and echocardiographic data were collected of patients with complete AVSD with their VSD closed with either untreated autologous pericardial or treated bovine pericardial patch material between January 1, 1996, and December 31, 2003. Evaluation closed in September 2019.
Results:
A total of 77 patients were analyzed (untreated autologous pericardial VSD patch: 59 [77%], treated bovine pericardial VSD patch: 18 [23%]). Median age at surgery was 3.6 (interquartile range [IQR]: 2.7-4.5) months, and median weight was 4.5 (IQR: 3.9-5.1) kg. Trisomy 21 was present in 70 (91%) patients. Median follow-up time was 17.5 (IQR: 12.6-19.8) years. Death <30 days occurred in two (3%) patients. Reinterventions occurred in eight patients (early [within 30 days] in two, early and late in one, and late in five), all in the autologous pericardium group. Log-rank tests showed no significant difference in mortality (
Conclusions:
In AVSD, the VSD can safely be closed with either untreated autologous pericardium or xeno-pericardium. We found no difference in LAVV regurgitation or the need for reoperation between the two patches.
Introduction
Despite the improved early outcome in primary infant complete atrioventricular septal defect (CAVSD) repair, left atrioventricular valve (LAVV) regurgitation is the most common indication for reoperation with reported rates between 10% and 20%. 1 -3
Most LAVV reoperations are performed within the first year after correction, 4,5 but there is a consistent risk at follow-up which is associated with late mortality. 6 Risk of LAVV reoperations has been related to valve morphology 7 as well as the timing of repair 8 but was irrespective of repair mode (single or double patch), 1 whereas the mechanism of valve failure is often caused by suture dehiscence. This failure has also been related to low bodyweight and atrioventricular valve (AVV) tissue fragility. 9 When delicate bridging leaflets are immobilized on stiff artificial patch or septal crest, it may become the “Achilles heel” of atrioventricular septal defect (AVSD) repair.
We changed our repair strategy by choosing fresh autologous pericardium for septal defect repair, avoided artificial pledgets, and left the bridging leaflets intact. We assumed that the elastic properties of untreated autologous pericardium could help to preserve the natural dynamics of the AVV when both atrial and ventricular septal defects (VSDs) are closed with two of these patches. The purpose of this study was to evaluate the effect of this modification on AVV function and compare the results with those from patients in whom the VSD was closed with treated bovine pericardial patch material.
The current COVID-19 pandemic may place surgeons in a situation where the patch material of preference (xeno-pericardium, Gore-Tex, Dacron) is not available because of supply chain interruption. This study provides insights into the use and outcomes of fresh autologous pericardium in the VSD position, which could be a valuable patch material option.
Material and Methods
Patient Population
Patients who underwent CAVSD repair with a VSD component that needed patch closure were included. Operations were performed between January 1, 1996, and December 31, 2003, by two surgeons at Leiden University Medical Center. Patients with associated tetralogy of Fallot or major other cardiac anomalies were excluded. This study was performed with institutional review board approval.
Surgical Technique
Standard bicaval cannulation and moderate hypothermic bypass and crystalloid cardioplegia were used. The common AVV was approached via the right atrium. The atrial septum between primum atrial septal defect (ASD) and open foramen ovale was cut and retracted for exposure. The central coaptation point of both bridging leaflets was marked with a stay suture. The VSD was closed using an untreated autologous pericardial patch in 59 (77%) patients and a treated bovine pericardial patch in 18 (23%) patients. Based on preoperative echo and direct inspection, a boat-shaped patch was tailored and stretched out between stay sutures. Currently, the patch is fixed on the drapes with two mosquito clamps on each corner of the patch. The convex side was fixed on the septal crest with running 6/0 Prolene. The straight side was attached to the bridging leaflets with separate 6/0 Prolene U-stitches (leaflets left intact). After the first few 6/0 Prolene stiches, the patch is advanced into the heart to complete suturing of the patch to the leaflets with interrupted U-stitches. These sutures were also used to fix the patch for ASD closure (autologous pericardium: 59 [77%], xeno-pericardium: 18 [23%]).
Care was taken not to oversize the VSD patch or cause any AVV retraction. When the LAVV surface area was large enough, the zone of apposition was closed with separate single 6/0 Prolene sutures. After the left AVV was tested by filling the ventricle, the right valve was tested and repaired if necessary. The opened atrial septal bridge was subsequently reconnected and the primum ASD closed leaving the coronary sinus in the right atrium. A foramen ovale was separately closed primarily.
Early Outcome Variables
The primary outcome variables studied were mortality, length of stay (LOS), and major adverse events (MAEs) including reoperation for LAVV failure (according to the Society of Thoracic Surgeons Congenital Heart Surgery Database). 5 Indication for LAVV reoperation was based on LAVV function, presence of pulmonary hypertension, and presence of clinical symptoms.
“Postoperative days on the ventilator” was defined as the total number of days on the ventilator after the index operation including all reintubation days. Postoperative intensive care unit (ICU) LOS was defined as total postoperative days in the ICU, including days readmitted to the ICU during hospitalization for the index operation.
Follow-Up
Secondary outcome variables were late mortality, late morbidity, and echocardiographic findings. Atrioventricular valve function was assessed in echocardiographic reports at discharge and 5, 10, and 15 years after surgery. Regurgitation was registered as a dichotomous variable, either moderate/severe or ≤mild.
Statistical Analysis
Patient and procedural characteristics were summarized as frequencies and percentages for categorical variables and medians and interquartile ranges (IQRs) for continuous variables. Chi-square tests and Fisher exact tests were conducted to analyze differences in categorical patient characteristics/outcomes between types of VSD patch (untreated autologous pericardium or treated bovine pericardium). Normality was assessed by the Shapiro-Wilk test (
Independent-samples
Results
Patients
There were 77 patients eligible for analysis (untreated autologous pericardial VSD patch: 59 [77%], treated bovine pericardial VSD patch: 18 [23%]; Table 1). The median age at surgery was 3.6 (IQR: 2.7-4.5) months, and the median weight was 4.5 (IQR: 3.9-5.1) kg. Forty-four (57%) patients were female. Trisomy 21 was present in 70 (91%) patients. Five (6%) patients were on ventilator support prior to operation. Preoperative moderate/severe AVV regurgitation was present in nine (12%) patients (data complete 84%). Patient and procedural characteristics were comparable in both patch groups. Median follow-up time after surgery was 17.5 (IQR: 12.6-19.8) years.
Patient and Procedural Characteristics.a,b
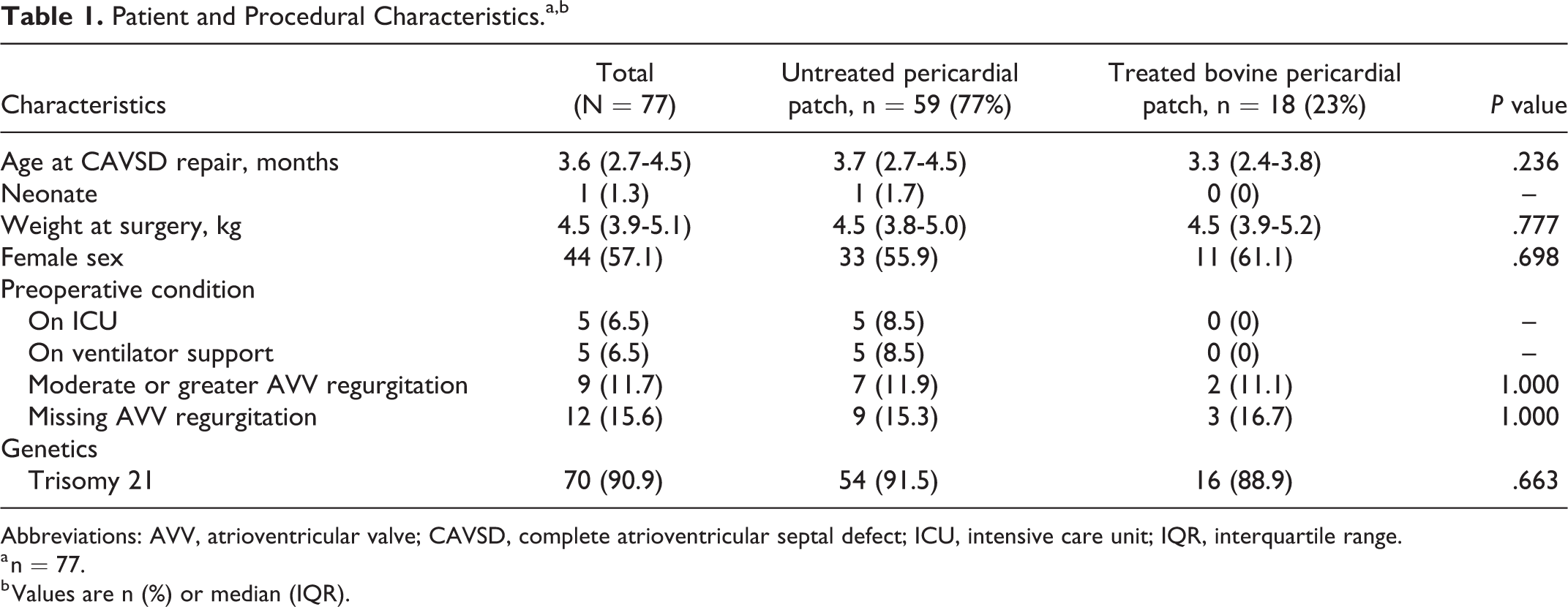
Abbreviations: AVV, atrioventricular valve; CAVSD, complete atrioventricular septal defect; ICU, intensive care unit; IQR, interquartile range.
a n = 77.
b Values are n (%) or median (IQR).
Surgical Technique
Procedural details and outcomes are outlined in Table 2. Second bypass run was used in ten (13%) patients (residual lesion: eight, low cardiac output syndrome [LCOS]: two). Duct closure (30), clipping of left superior vena cava (1), and coarctation repair (1) were performed as additional procedures in 31 (40%) patients. The zone of apposition was closed in all but three patients.
Operative and Outcome Data.a

Abbreviations: AV, atrioventricular; ICU, intensive care unit; IQR, interquartile range; LAVV, left atrioventricular valve; RAVV, right atrioventricular valve; VSD, ventricular septal defect.
a Values are n (%) or median (IQR).
b Included variables are patent ductus closure (30), clipping persistent left superior vena cava (1), and coarctation repair (1).
c Included cardiac arrest requiring resuscitation, bleeding requiring reoperation, unplanned reoperation prior to discharge, and low cardiac output syndrome requiring reoperation.
Early Mortality
Death <30 days occurred in two (3%) patients (both autologous pericardium) due to gastrointestinal bleeding 1 and infant respiratory distress syndrome at three and two weeks after uncomplicated AVSD repair, respectively.
Early Reoperations
Atrioventricular valve reoperations prior to discharge (within one month) were performed in three patients: one for right atrioventricular valve (RAVV) repair and two for LAVV repair with residual VSD/ASD closure. Details on reoperations are outlined in Table 3. Indication for LAVV repair was regurgitation due to dehiscence of initially closed zone of apposition in one patient. In the other patient, the mechanism of LAVV regurgitation was not mentioned in the surgery report. Other in-hospital MAE included cardiac arrest in ICU requiring resuscitation,
5
reoperation for bleeding,
5
and LCOS.
1
Major adverse events occurred almost exclusively (all but one) in patients operated before 2000. Type of patch material was not a risk factor for MAEs (
Reoperations.a
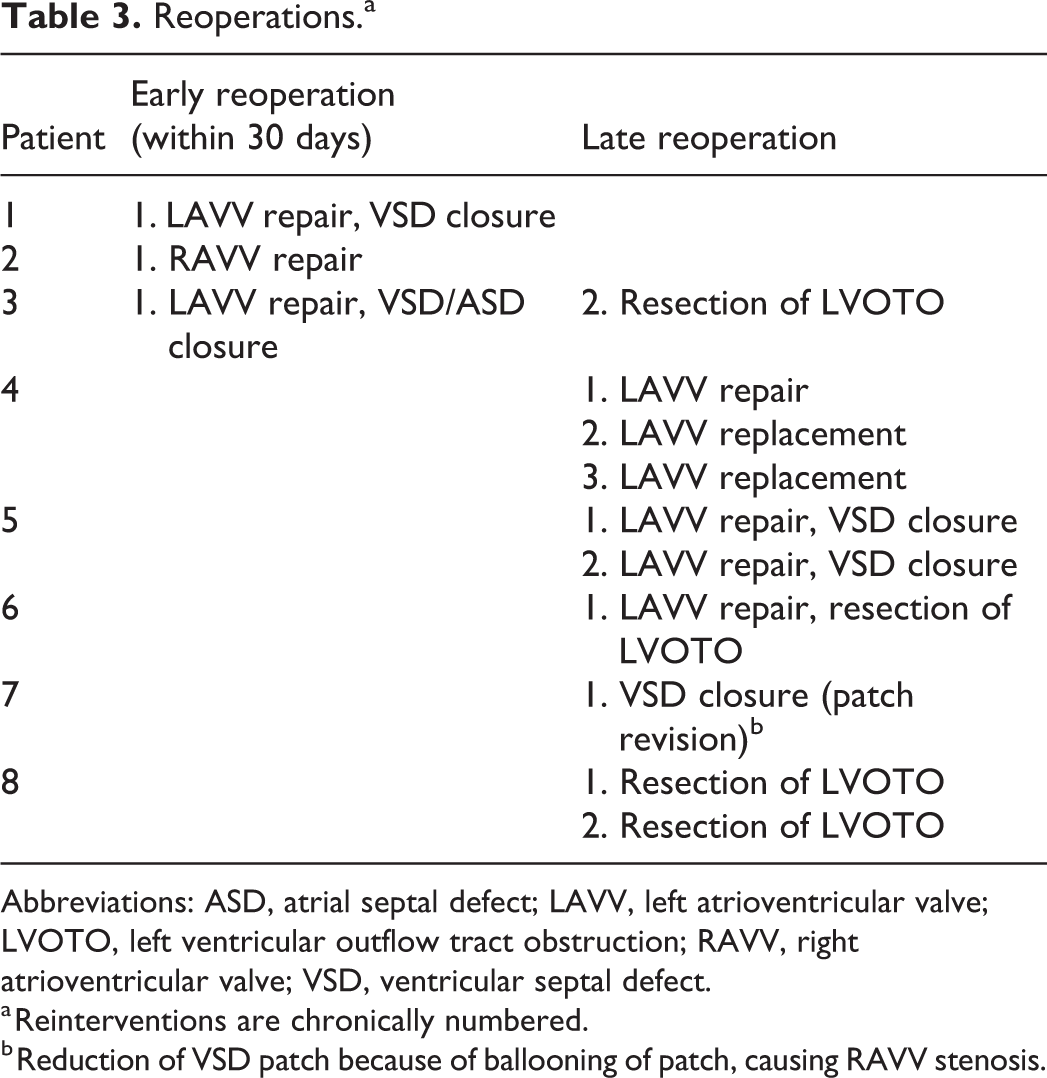
Abbreviations: ASD, atrial septal defect; LAVV, left atrioventricular valve; LVOTO, left ventricular outflow tract obstruction; RAVV, right atrioventricular valve; VSD, ventricular septal defect.
a Reinterventions are chronically numbered.
b Reduction of VSD patch because of ballooning of patch, causing RAVV stenosis.
Median duration of ventilator support was 5.0 (IQR: 3.0-8.0) days. Median ICU LOS was 7.0 (IQR: 4.0-9.3) days. Median postoperative hospital LOS was 13.0 (IQR: 9.0-18.5) days.
Follow-Up
Late Mortality
Seven (12%) patients (autologous pericardial patch) died at follow-up. Causes were cardiac related (two) (pulmonary hypertension, myocardial infarction in lymphocytic myocarditis) but unrelated to AVSD repair, and noncardiac related (five): pneumonia (one) and unknown cause (four). Two (10%) patients (treated bovine pericardial patch) died of subarachnoid bleeding (one) and unknown cause (one). Patch material was not a risk factor for late death (
Late Reoperations
Reoperations after more than one month (all autologous pericardium) were performed in six patients (one late reoperation in three patients, two late reoperations in two, three late reoperations in one; Table 3). Sites addressed were LAVV: six (repair: four, replacement: two), left ventricular outflow tract obstruction (LVOTO): four, and VSD: three. One of those patients (LVOTO) had already undergone an early reintervention (LAVV repair and closure of residual VSD/ASD). All patients who underwent LAVV reoperation have undergone closure of zone of apposition at initial AVSD repair. Mechanism of LAVV regurgitation was dehiscence of initially closed zone of apposition (1) and leaflet malcoaptation (1) and additional cleft in superior leaflet that was initially missed (1). Patch material was not a risk factor for reoperation (
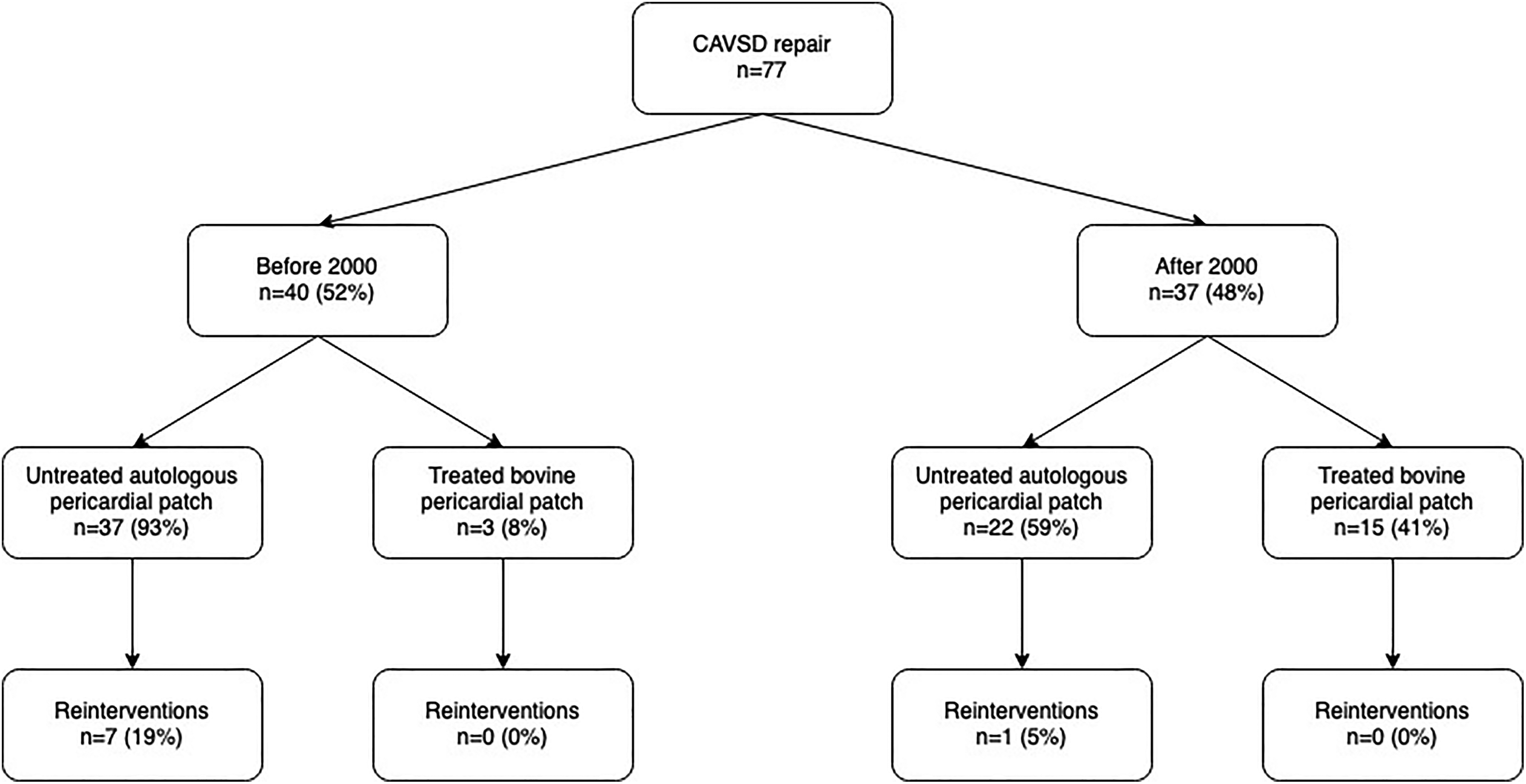
Overview of surgeries performed before and after 2000.
In one patient, we had the opportunity to study the histology of autologous pericardial VSD patch 2.5 years after implantation. This patient needed reoperation for patch aneurysm causing RAVV stenosis. The pericardial patch ballooning toward the right was already visible on intraoperative echo immediately after AVSD repair but was not addressed. Excessive patch was resected at reoperation and sent for pathology. Histology revealed viable smooth muscle cells (immunohistochemistry), collagen, and newly formed elastin structures in the matrix (Figure 2).
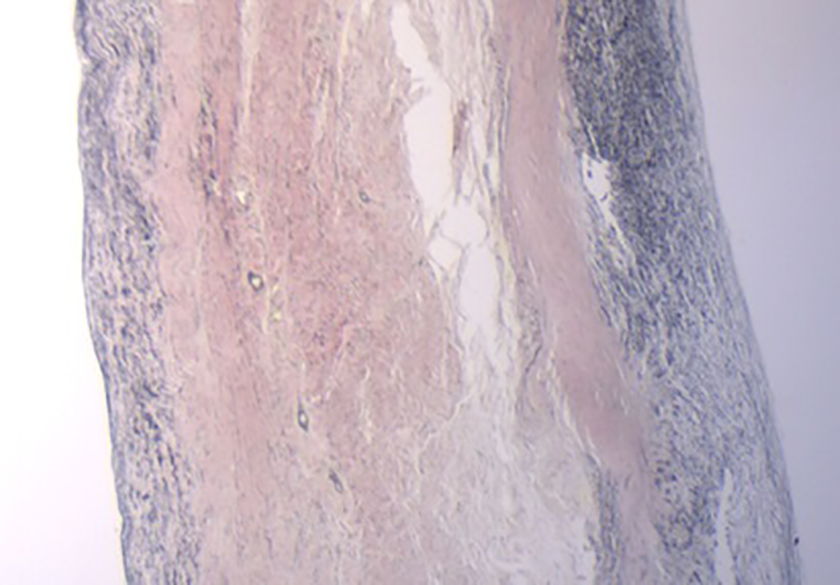
Histology image of an untreated pericardial patch 2.5 years after implantation. Hematoxylin–eosin staining. Cell nuclei are stained blue. Extracellular matrix and cytoplasm are stained pink. Newly formed elastin structures are shown. (See this image in color in the online version).
The Kaplan-Meier curve in Figure 3 shows the time to reintervention on LAVV. Similar to the survival curve, log-rank test showed no difference in time to LAVV reintervention between the two treatment groups (
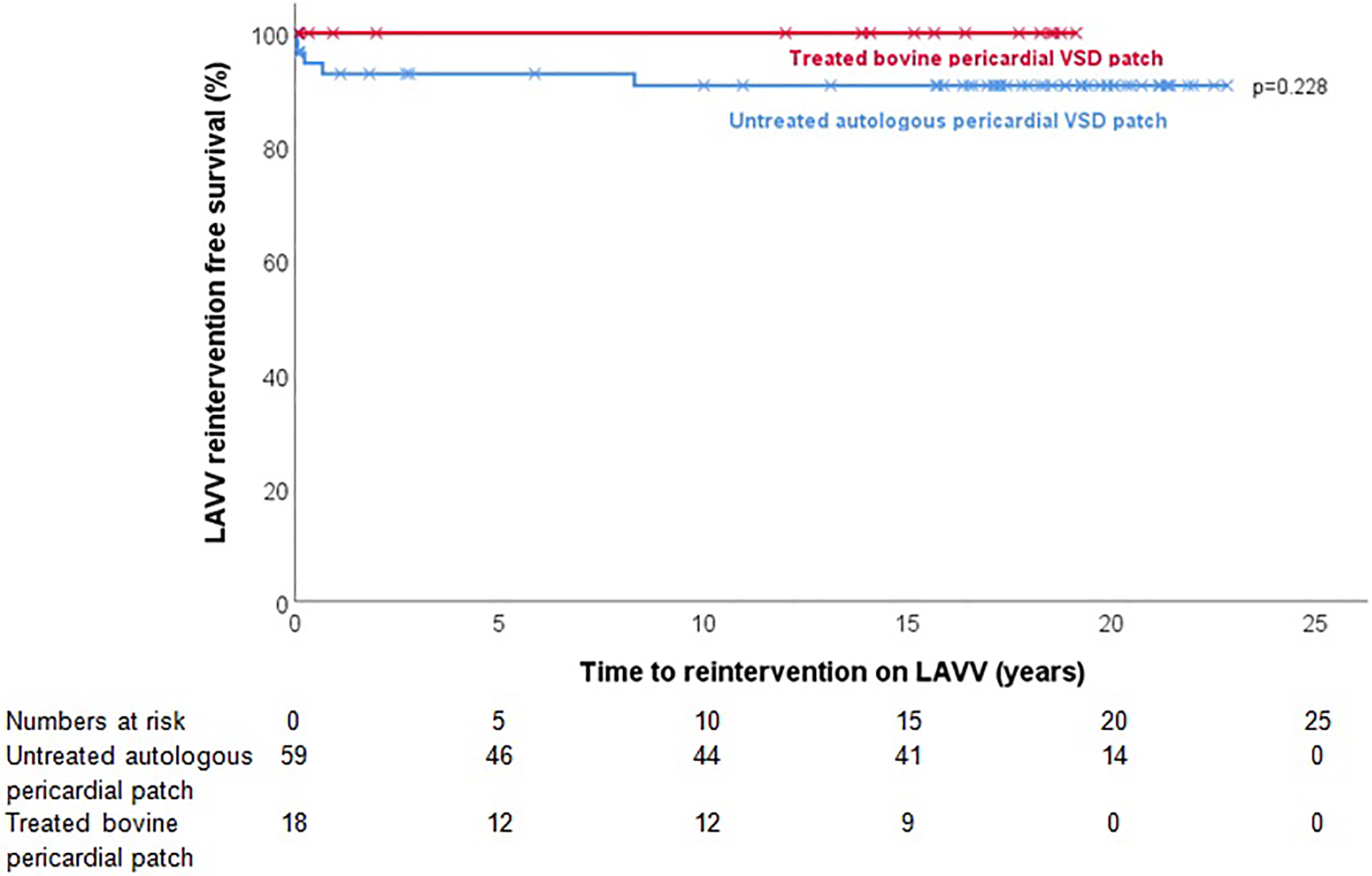
Kaplan-Meier survival estimation for time to reintervention on LAVV based on VSD patch (untreated autologous pericardium vs treated bovine pericardium). LAVV indicates left atrioventricular valve; VSD, ventricular septal defect.
Echocardiographic Follow-Up
Expert echo assessment was performed up to 15 years after surgery. Moderate LAVV regurgitation at last follow-up was present in 15 (19%) patients with no difference between both patch groups (untreated autologous pericardium: 11 [19%], treated bovine pericardium: 4 [22%]). There were no patients with severe LAVV regurgitation. Time-related incidence of moderate LAVV regurgitation was equal in both groups (
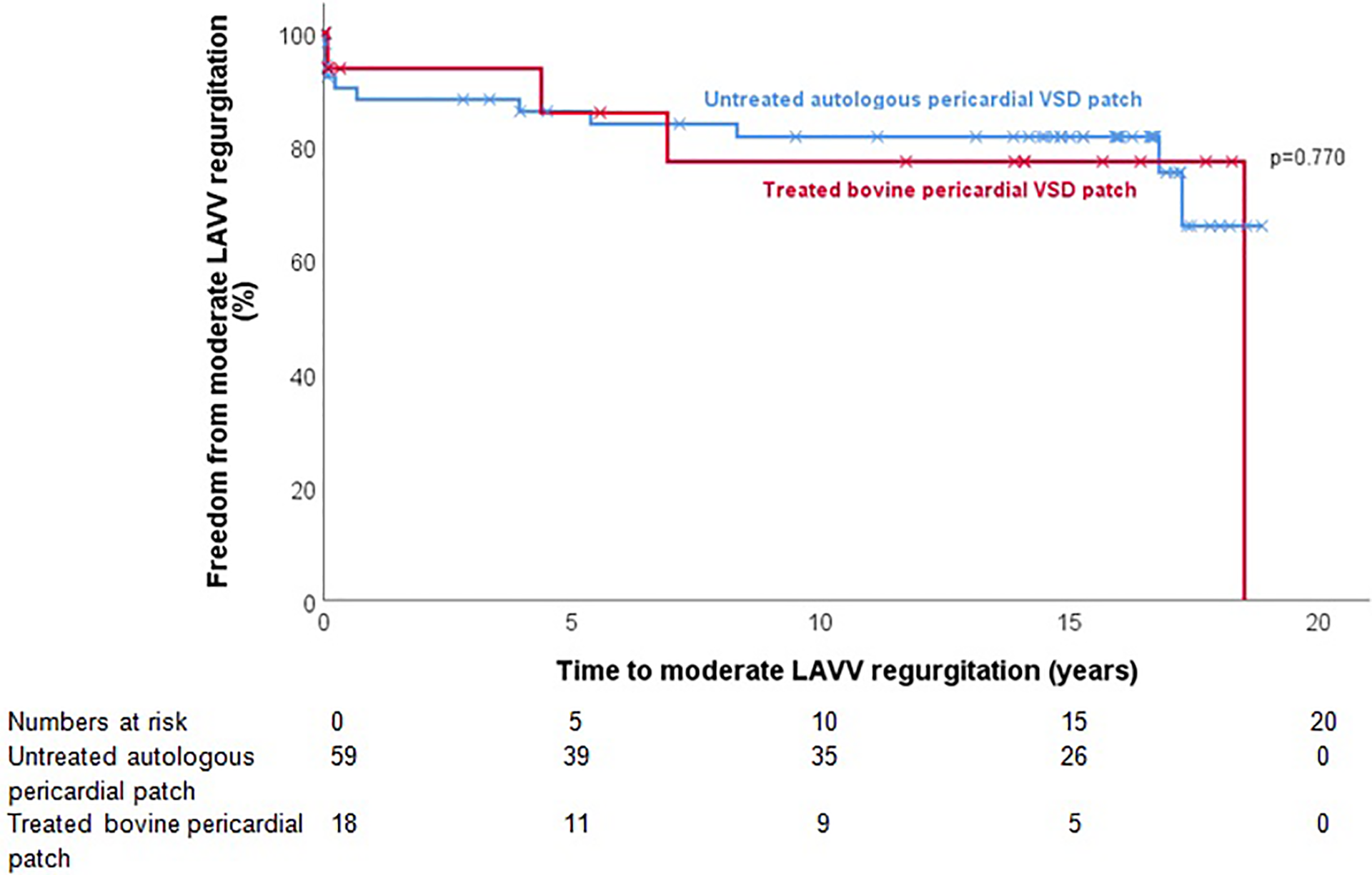
Kaplan-Meier survival estimation for time to moderate LAVV regurgitation based on VSD patch (untreated autologous pericardium vs treated bovine pericardium). LAVV indicates left atrioventricular valve; VSD, ventricular septal defect.
Comment
This study reports on the long-term (median follow-up: 17.5 years) outcomes of CAVSD double patch repair comparing untreated autologous pericardium with bovine pericardial patch material for VSD closure. Our report demonstrates that untreated autologous pericardium is as safe as treated bovine pericardial patch material. There was no significant difference in mortality, reintervention, or late AVV function between either patch.
Reoperations for LAVV regurgitation were few and limited to the autologous pericardium group, although the difference with the bovine pericardial patch group was not significant. Left atrioventricular valve reoperation rate for the autologous pericardium group (8%) and for the cohort as a whole (6%) did not exceed the incidence reported in the literature (11.4%). 10 In our opinion, VSD closure with the pericardial patch should be considered as an entirely new category which shows promising results when compared with the use of the modified single-patch technique and double-patch technique with synthetic VSD patch.
Interestingly, all reoperations except one were performed on patients operated (AVSD repair) before 2000, suggesting a possible learning effect. Autologous pericardium is less user-friendly than other patch material because it is sticky and lacks stiffness. Moreover, it has to be properly sized to prevent aneurysmal dilatation. A single case of a patient with pericardial patch ballooning made us decide to avoid the use of a patch larger than the size of the ventricular defect itself. It may take the surgeon some time to learn how to handle untreated pericardial tissue. We continued its use nevertheless encouraged by the demonstrated viability of the patch. This viability may potentially allow the patch to adapt to growth and to the complex spatial dynamics required for AVV function. Long-term echo however could not confirm any difference in LAVV regurgitation between both groups. With respect to reintervention for LVOTO, both groups were not statistically different.
Operating AVSD patients for LAVV regurgitation can be technically challenging. As our data support, surgeons should be aware of the mechanism of LAVV regurgitation being possibly related to the cleft that was left open at initial repair or that reopened due to suture dehiscence. Besides, the use of Teflon or other synthetic patch material for septal defect closure can cause calcification of the septum or AV valve annulus. Calcification of LAVV was not seen at reoperation in patients who underwent septal defect closure with two untreated autologous pericardial patches. Using fresh pericardium may prevent calcification and may leave the valve better accessible for future surgery.
Since all reoperations for LAVV regurgitation were performed in patients operated before 2000, it could be valuable to separately analyze the outcome of patients operated after 2000 to exclude a possible effect of learning.
The teams from the Royal Children’s Hospital in Melbourne and Paediatric Cardiac Centers in Brisbane recently reported their long-term outcomes using the double-patch technique for the repair of CAVSD. Xie et al (n = 138) reported a probability of freedom from moderate or worse LAVV regurgitation of 68.7% at eight years of follow-up. 11 Bell et al (n = 188) reported a probability of freedom from moderate or worse LAVV regurgitation of 85% at a median follow-up of 10.8 years. 12
Reported LAVV regurgitation rates in our study are similar or lower compared to previously reported findings, but the median follow-up in our study is much longer (17.5 years). One surgeon is still using the untreated autologous patch. The other surgeon abandoned autologous pericardium—in preference of xeno-pericardium following LAVV reoperations.
Limitations
This study is limited by its retrospective and monocentric nature with the inherent problems of missing and incomplete data especially in patients with Down syndrome who were relatively difficult to follow-up. Furthermore, the control group was relatively small.
Conclusion
In AVSD, the VSD can safely be closed with either untreated autologous pericardium or xeno-pericardium. We found no difference in LAVV regurgitation or the need for reoperation between the two patches.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
