Abstract
Many new medications for the treatment of migraine are now available on the market. In the current evolving migraine treatment landscape, an individualized treatment approach is needed. This review provides practical recommendations on how to obtain a correct diagnosis and then engage in a long-term partnership with patients with the most severe form of migraine: chronic migraine (CM). Given the need to effectively treat this complex neurological disease, clinicians in primary care, general neurologists, and headache specialists are at the forefront to ease the burden of this disease for their patients. This manuscript will review how to discuss the currently available treatment options to help control migraine attacks, manage expectations, and, together with the patient, determine the most effective and appropriate treatment. The goal is to create an environment where the clinician partners with the patient in shared decision-making to choose the most effective appropriate treatment for the individual patient.
Keywords
The most severely impacted patients with migraine are those with chronic migraine (CM), who experience 15 or more headache days (migraine-like or tension-type-like) per month for ≥3 months as defined by International Classification of Headache Disorders, third edition criteria (ICHD-3) (Table 1). 1 These diagnosis criteria for migraine include headache duration, characteristics (eg, pulsating, throbbing), and the presence of an aura, neurological symptoms occurring before, during, or after a migraine episode. American Headache Society (AHS) and American Academy of Neurology (AAN) guidelines recommend that patients with CM receive preventive medications, as lack of prevention may lead to acute medication overuse, worsening of headache, and disease progression.2-5 Historically, those with CM are often underdiagnosed and undertreated.6,7 This leads to only 4.5% to 13% of eligible patients receiving appropriate preventive medications.6,8
Diagnostic Criteria for Chronic Migraine (ICHD-3). 1

Abbreviation: ICHD-3: International Classification of Headache Disorders, third edition.
Providers who take care of headache patients can ease the burden of CM patients by adopting best practices in clinician-patient dialogue during the diagnosis and disease-management discussion. In the author’s experience, developing trustful relationships and cultivating open communication are critical to determine the patient’s health problems, obtain a correct diagnosis, and choose the most effective disease-management plan. Empathy plays a major role in eroding the stigma commonly associated with migraine. The clinician’s knowledge and compassion are important in helping patients navigate the array of sometimes misleading information about available treatment options and providing a professional, unbiased overview of the most appropriate therapies, which includes a full discussion of the different medications’ efficacy and safety data.
In the author’s experience, shared decision-making is part of a successful therapeutic relationship. Using the correct lexicon is important to demonstrate an understanding of the patient’s situation and foster a true partnership. CM patients should be educated about treatment options in a balanced and comprehensive fashion before making a joint decision about the best individualized treatment plan. Many CM patients conduct their own research before meeting with their clinician; therefore, detailed responses will facilitate a collaborative approach. Clinicians should use the diagnosis discussion to emphasize the chronic nature of the disease and explain that preventive treatment is necessary to reduce the frequency of headache days, decrease the need for acute medication, and diminish the symptom burden.
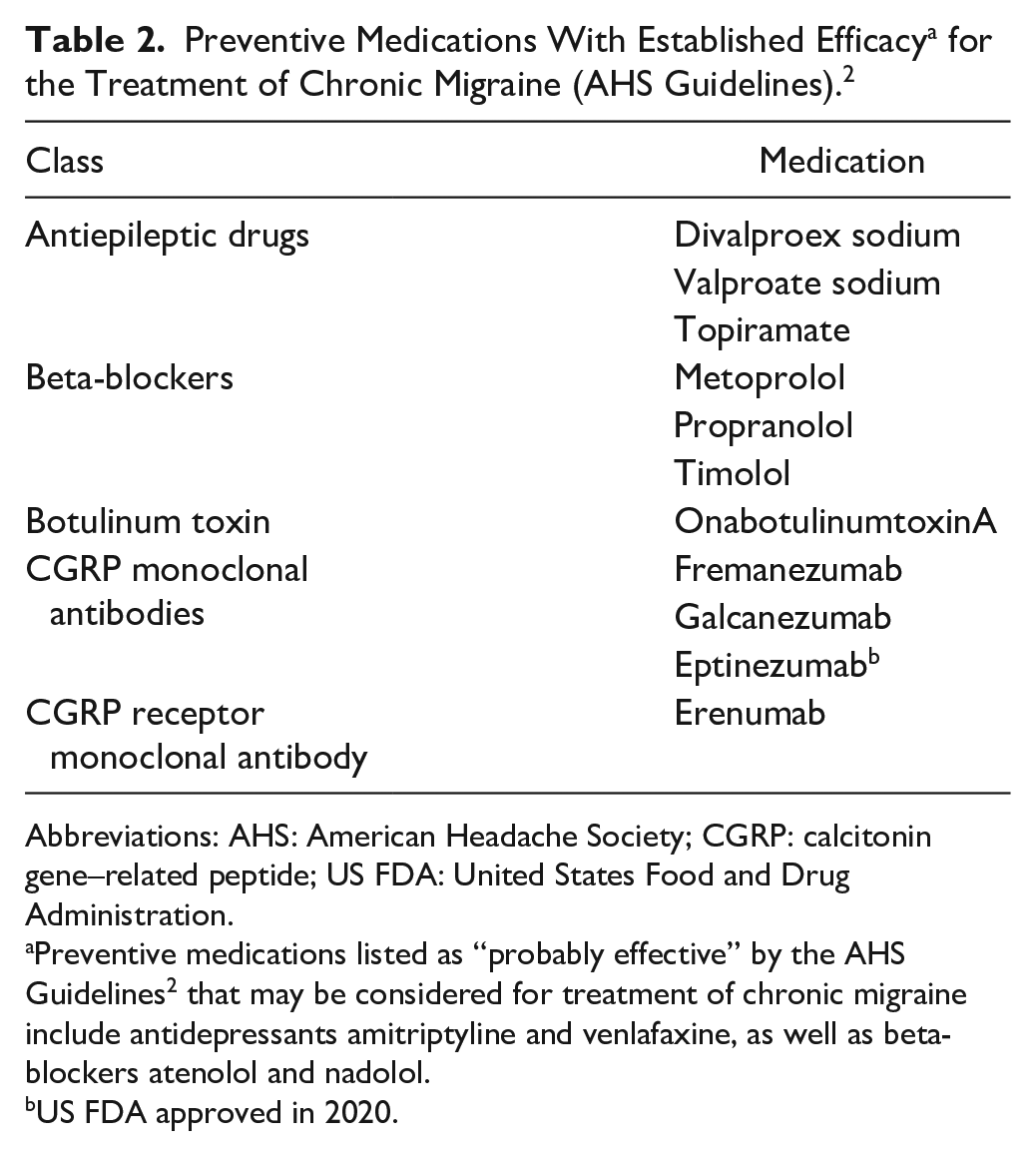
Multiple treatment options are currently available to reduce/prevent migraine attacks for those with CM (Table 2).2,9 Because medication response is highly individualized and variable, clinicians should explain that a therapy that works for one patient may not work for another. Insurance providers may influence treatment decisions through reimbursement policies that often require failure of ≥2 different medication classes (antihypertensives, antidepressants, anti-epileptics 10 ) before approving the use of migraine-specific treatments. It is also important to set clear treatment expectations for CM patients: explain that current therapies are not a cure, and effectiveness is currently measured as the decrease from pretreatment baseline in the number of monthly headache and/or migraine days over time. Use of oral preventive medications may be limited by frequent side effects or lack of efficacy. 11 AHS consensus guidelines recommend that CM patients initiate therapy with ≥2 oral preventives or with Botox® (onabotulinumtoxinA). If these treatments fail, patients should initiate therapy with a monoclonal antibody directed against calcitonin gene–related peptide (CGRP) or its receptor. 2
Abbreviations: AHS: American Headache Society; CGRP: calcitonin gene–related peptide; US FDA: United States Food and Drug Administration.
Preventive medications listed as “probably effective” by the AHS Guidelines 2 that may be considered for treatment of chronic migraine include antidepressants amitriptyline and venlafaxine, as well as beta-blockers atenolol and nadolol.
US FDA approved in 2020.
Topiramate is the most frequently used oral preventive treatment, with efficacy in CM demonstrated in 2 double-blind, placebo-controlled trials.12,13 However, side effects frequently limit long-term use, and claims data suggest that a majority of patients discontinue within 12 months. 10 Gabapentin also has a single, controlled study showing efficacy for prevention in CM patients. 14 Most of the other oral preventive treatments have not been specifically studied in CM or have no positive evidence for their use, despite reimbursement guidelines.
Although many CM patients are familiar with onabotulinumtoxinA due to widespread aesthetic use, there is less awareness of its efficacy as a preventive treatment for CM. Based on thorough investigation,15,16 onabotulinumtoxinA was the first treatment approved 17 by the US Food and Drug Administration (FDA), in 2010, for the prevention of headache in CM. OnabotulinumtoxinA has well-established efficacy in patients with CM2,5 and has demonstrated effectiveness (including sustained reductions in headache frequency, improvements in quality of life, reduction in migraine-related symptoms, and decreases in common psychiatric comorbidities, such as depression and anxiety, associated with CM)15,16,18-26 in more than 5000 patients treated in sponsored clinical and observational studies. OnabotulinumtoxinA has been used for CM prevention longer than any other preventive treatment option, and a large body of evidence supports its real-world efficacy and safety. 27 It is important to explain the data supporting onabotulinumtoxinA use in CM to help clarify common patient misconceptions.
OnabotulinumtoxinA administration should be explained to patients as injections to 7 head and neck muscles that require only 10 to 15 minutes.17,28 The injector needles are relatively small, feel like a small pinch, and do not restrict activity. OnabotulinumtoxinA is typically administered every 12 weeks and generally has no systemic effects; the most common adverse events with onabotulinumtoxinA are neck pain and ptosis. 17 Patients may notice improvement as early as the first week after treatment and a more robust treatment effect after 2 or 3 cycles. 25
Monoclonal antibodies against CGRP or its receptor have recently emerged as targeted therapies for migraine, providing a novel treatment alternative and increasing awareness of this disease. In 2018, Aimovig® (erenumab), Ajovy® (fremanezumab), and Emgality® (galcanezumab) were approved as subcutaneous treatments for migraine prevention. In 2020, Vyepti™ (eptinezumab) was approved as an intravenous treatment for migraine prevention. In pivotal trials, these CGRP monoclonal antibodies have demonstrated efficacy in reducing migraine days and other clinically important outcomes in both episodic and CM patients,29-33 and in long-term clinical trials they have demonstrated few systemic side effects; the most common adverse events are injection site reactions and constipation for the 3 subcutaneous antibodies and nasopharyngitis and hypersensitivity for eptinezumab.33-36 However, given that CGRP monoclonal antibodies have been on the market a relatively short time, we are still gaining an understanding of their real-world safety and tolerability profiles. Communicating what is known and what questions remain about treatment options is important to achieve the goal of shared decision-making.
The CGRP monoclonal antibodies erenumab, fremanezumab, and galcanezumab are injected subcutaneously either monthly or quarterly, and can be either self-injected or provider-injected, whereas eptinezumab is infused intravenously every 3 months.33-36 Reimbursement may play a significant role in how a patient selects a specific CGRP monoclonal antibody, but if all options are available, a patient can select which administration route is preferred.
Medication overuse is a frequently occurring complication in patients with CM. 37 This can lead to a worsening of headache frequency and refractoriness to preventive treatments. 38 As a result, patients should be educated to limit all of their acute medication use for migraine attacks (eg, triptans, ditans, and over-the-counter analgesics, and/or caffeine compounds) in combination to ≤2 days per week.2,9 Topiramate 100 mg per day is probably effective in the treatment of medication overuse headache, and there is some evidence to support that corticosteroids and amitriptyline may also treat withdrawal symptoms. 39 The recent introduction of gepants (Ubrelvy™ [ubrogepant] and Nurtec™ [rimegepant]) may change this paradigm, as medication overuse is not listed as a warning in their FDA-approved labels.40,41 The triptans and ditans (Reyvow™ [lasmiditan]) provide warnings about medication overuse.2,42,43 Evidence supporting efficacy as preventive treatment in patients with CM and medication overuse is shown in clinical trials with topiramate 43 and onabotulinumtoxinA24,43 and real-world studies with onabotulinumtoxinA.44,45 Table 3 presents a list of approved acute medications to treat migraine attacks.
Approved Acute Medications for Migraine Attacks.

Women of childbearing age should consider potential pregnancy before starting any treatment, and the benefits and risk profile of the treatment to both the mother and child. There is evidence to support the safety of onabotulinumtoxinA in pregnancy.46,47 Caution should be given to avoid becoming pregnant while taking a CGRP monoclonal antibody due to its long elimination half-life. To date, no teratogenic effects have been reported, but immunoglobulins are known to be transported across the placenta.
Erenumab had a recent FDA label change and now carries a warning for de novo or worsening of pre-existing hypertension. 35 when starting this medication. There are currently no published double-placebo controlled studies available showing that combination of erenumab with gepants is effective. However, anecdotal case reports suggest that gepants are being coadministered in clinical practice with efficacy in patients receiving monoclonal antibodies.48,49 Furthermore, data from a phase 1b, open-label pharmacokinetic trial in adults with migraine (NCT04179474) suggest that no safety concerns were identified when ubrogepant was coadministered with erenumab or galcanezumab, and the pharmacokinetics of ubrogepant were not significantly changed with coadministration. 50 However, a longer-term evaluation of concomitant use in broader populations is needed and, therefore, this author recommends blood pressure monitoring if this combination is used in practice until such evidence is established.
There is also growing evidence that onabotulinumtoxinA in combination with a CGRP monoclonal antibody might benefit some CM patients, supported by preclinical evidence suggesting a synergistic effect based on different mechanisms of action. Two retrospective chart analyses in patients with intractable or refractory CM from real-world practice demonstrated a reduction in mean monthly migraine days from baseline when erenumab 70 or 140 mg was added to onabotulinumtoxinA, 51 and a reduction in headache days, intensity, severity, and patient perceptions when onabotulinumtoxinA was added following initiation of erenumab 70 mg. 52 A prospective study in CM patients who had failed ≥3 preventive treatments found that 65% of patients (n = 45) experienced a reduction in monthly headache days with onabotulinumtoxinA and erenumab 70 or 140 mg, and that the combination was more effective than erenumab alone or in combination with other oral preventives. 53 Future prospective studies evaluating the mechanisms underlying potential synergistic effects of onabotulinumtoxinA and CGRP monoclonal antibodies will be informative and help guide treatment decisions. However, it is difficult to determine if the added CGRP monoclonal antibody or the combination is responsible for the patient’s improvement. One can assume that onabotulinumtoxinA is providing added benefits to the monoclonal antibody if a wearing-off effect is noted 10 to 12 weeks after onabotulinumtoxinA treatment.
CM is a complex neurological disease associated with significant disability and requiring an individualized treatment approach. In the current evolving CM treatment landscape, the clinician should partner with the patient on decision-making rather than dictate their own opinion or leave responsibility of choosing the most effective appropriate treatment to the patient. A balanced discussion of the efficacy, adverse events, long-term safety, delivery method, and mechanism of action of each medication is necessary for the patient to make an informed decision.
Footnotes
Acknowledgements
Writing and editorial assistance was provided to the author by Peloton Advantage, LLC, an OPEN Health company, Parsippany, NJ, and was funded by AbbVie. The opinions expressed in this article are those of the author. The author received no honorarium/fee or other form of financial support related to the development of this article.
Declaration of Conflicting Interests
The author declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AMB has served on advisory boards for AbbVie, Amgen, Alder, Teva, Supernus, Promius, Eaglet, and Lilly; and has received funding for speaking from AbbVie, Amgen, Pernix, Supernus, Depomed, Avanir, Promius, Teva, and Eli Lilly and Company.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Allergan (prior to its acquisition by AbbVie).