Abstract
Background
Between 2000 and 2010, the Native Hawaiian and Pacific Islander population increased by 40% in the United States. 1 During the same time period, the Pacific Islander population in Arkansas grew by over 250%; most of the Pacific Islanders in Arkansas are Marshallese from the Republic of the Marshall Islands (RMI). 1 Marshallese adults have a high prevalence of type 2 diabetes, with levels 2 to 3 times that of the general population.2-4 Pilot data from health screenings of Marshallese adults (n = 401) conducted in Arkansas documented prevalence of type 2 diabetes at 38.4%, 4 compared with 9.4% in the general US population 5 and 8.5% worldwide. 6
Despite these disparities, there has been limited success implementing diabetes self-management education (DSME) with Marshallese in both the RMI and the United States.7,8 To address these disparities, the authors developed an Adapted-Family model of DSME through a community-based participatory research (CBPR) partnership with Marshallese stakeholders in Arkansas,9-11 and tested the intervention in a randomized controlled trial (RCT). 12 The RCT demonstrated participants in the Adapted-Family DSME arm experienced significantly greater reductions in mean HbA1c (glycated hemoglobin) at immediate postintervention and 12 months postintervention compared with participants in the standard DSME arm. 13 While the curriculum was effective at improving glycemic control, further analysis showed that the effectiveness may have been a result of receiving increased educational dosage due to the delivery of the intervention in the participants’ homes. 14 This left a significant gap in the literature regarding the effectiveness of the Adapted-Family DSME if conducted in a setting other than the home.
To help fill this gap and increase the feasibility of dissemination of the Adapted-Family DSME in real-world settings, the preliminary effectiveness and feasibility of implementation of the Adapted-Family DSME delivered in a clinic environment needed to be tested. The purpose of this study was to pilot test the preliminary effectiveness and feasibility of the Adapted-Family DSME when implemented in a real-world clinical setting.
Methods
Study Setting
Participants were recruited from the University of Arkansas for Medical Sciences (UAMS) North Street Clinic. The North Street Clinic is staffed with internal medicine physicians, internal medicine resident physicians, a doctor of pharmacy practice, certified diabetes educators (CDEs), interprofessional student learners, nurses, and bilingual Marshallese community health workers (CHWs).
Inclusion Criteria
Inclusion criteria specified that participants be Marshallese, 18 years of age or older, have type 2 diabetes (HbA1c ≥6.5%), and have at least 1 family member willing to participate in the study. Participants were excluded if they had previously participated in DSME within the past 5 years or had a condition that made it unlikely that the participant would be able to follow the protocol, such as terminal illness, severely impaired vision or hearing, eating disorder, or plans to move out of the geographic region.
Recruitment
Marshallese patients (referred to as “primary participants”) were recruited in the North Street Clinic by CHWs, who contacted potentially eligible participants to explain the inclusion criteria and scheduled consent and preintervention data collection for those expressing a willingness to participate.
Consent Process
Bilingual (English and Marshallese) research staff trained in informed consent procedures, the study protocol, and research ethics provided information about the study and completed the consent process for primary participants and their family members in a private room. The study and consent procedures were approved by the UAMS Institutional Review Board (IRB #207077).
Data Collection
The data collection instruments and protocol were developed from the Adapted-Family model of DSME RCT10,13 (UAMS IRB#203482) (Clinical Trial #NCT02407132). The instruments were developed with input from Marshallese stakeholders and translated from English to Marshallese. Demographic data (age, gender, marital status, education level, and employment status) and health-related measures (health insurance coverage, general health status, and support for managing their type 2 diabetes) were self-reported through a survey instrument administered by trained study staff at preintervention and immediate postintervention (12 weeks). Participants were also asked to complete a diabetes medication inventory at the preintervention data collection event.
Biometric data were collected by trained study staff at pre- and post intervention. Biometric measures included HbA1c, height, weight, blood pressure, and lipids. Height and weight measurements were used to compute a continuous measure of body mass index (BMI). Level of engagement for each participant was documented based on observed behavior using a tool developed in the prior RCT. CDEs were trained on the tool and rated participant engagement as fully engaged, moderately engaged, or not engaged at all based on verbal, nonverbal, and participation cues. Some exceptions to the data collection process occurred. For example, 1 participant was wheelchair bound and unable to participate in height and weight measurements. All primary and family member participants who took part in the study were provided a $20 gift card for each of the data collection events.
Intervention Protocol
Primary participants and their family members took part in 10 hours of Adapted-Family DSME delivered over 8 weeks (8 classes of 75 minutes each). The core elements of the curriculum were consistent with the recommendations regarding self-care behaviors provided by the American Diabetes Association and American Association of Diabetes Educators. 15 The curriculum was adapted using a CBPR approach, that has been described elsewhere. 9 Curriculum adaptations included culturally appropriate nature analogies, such as tide changes to explain changes in blood glucose numbers; incorporation of pictures of Pacific Islanders; integration of culturally relevant foods, such as seafood and fruits; in-depth discussions of the importance of medication adherence, with a focus on the natural plant-based properties of diabetes medications; and emphasis on engaging participants’ collectivistic, family orientation as a means of self-management. The Adapted-Family DSME curriculum incorporated family goal-setting and family motivational interviewing, and emphasized the importance of engaging the family member(s) in behavioral changes. 9 Bilingual CHWs delivered the curriculum in Marshallese, with a CDE present to provide support and answer any questions. Prior to the intervention, CHWs completed more than 60 hours of study and curriculum-specific training.
Analytical Methods
Descriptive statistics for primary participants and family members included summaries of self-reported demographic and health-related characteristics.
Nonparametric statistical tests (Wilcoxon ranked sign test) for primary participants and family members included analysis of pre- and postintervention biometric data: HbA1c, BMI, low-density lipoproteins (LDL), high-density lipoproteins (HDL), systolic blood pressure (SBP), and diastolic blood pressure (DBP). Although the study’s small sample size does not allow for formal tests of hypotheses, it was important to determine whether changes were trending significantly in the appropriate direction. In addition, individual primary participant changes on the primary outcome of interest (HbA1c) are provided to examine whether aggregate changes were consistent across primary participants, or simply magnified by one or more outliers. Information regarding retention, attendance, and engagement in educational sessions is provided to evaluate preliminary feasibility.
Qualitative Methods to Understand Facilitators and Barriers to Implementation
To capture lessons learned and implications for clinical practice, the lead author—a researcher trained and experienced in qualitative research methods—conducted interviews with the study implementation team. The implementation team included the nurse manager of the clinic, 2 CDEs, 3 CHWs who helped implement the intervention, and the primary care physician and doctor of pharmacy practice who lead the clinic. Interviews allowed the implementation team to discuss their experiences in their own words. The interviews focused on the broad topic of facilitators and barriers to implementation of the intervention. Extensive notes were taken during the interviews and notes were summarized and provided back to the implementation team for review and corrections. A brief summary of the lessons learned regarding facilitators and barriers are summarized.
Results
Twelve primary participants agreed to participate, and each had 1 family member who agreed to participate in the study. During the preintervention data collection, 2 of the primary participants were determined to be ineligible for the study based on HbA1c test results, which were less than 6.5% and therefore not indicative of type 2 diabetes. For this reason, the primary participants and their family members were unenrolled from the study. The remaining 10 primary participants and 10 family members enrolled in the study and completed the preintervention data collection event.
Retention, Attendance, and Engagement
All 10 primary participants attended all 8 classes and received all 10 hours of the educational intervention. Eight of the 10 family members completed the study and received all 10 hours (8 classes) of the educational intervention. One family member participated in half of the intervention (4 classes), and 1 family member discontinued participation before receiving any of the intervention. Among primary participants, all 10 were observed to be fully engaged in the educational intervention. Among family members who participated in the educational intervention, 7 were observed to be fully engaged and 2 were moderately engaged. Nine of the 10 primary participants provided postintervention data. Eight family member participants provided postintervention data.
Demographics and Health-Related Characteristics
As shown in Table 1, the mean age of primary participants was 59.3 years (SD = 13.6) and the mean age of family members was 55.8 years (SD = 14.1). Seven of 10 primary participants and 5 of 10 family member participants were female. The majority of primary participants (80.0%) and family members (70.0%) had less than a high school education.
Preintervention Demographics and Health-Related Characteristics of Primary Participants and Family Members.

Only 4 primary participants (40%) reported having any kind of health insurance coverage, while 7 family members (70%) reported having any kind of health insurance coverage. Eight primary participants (80.0%) and 7 family members (70.0%) reported being in good or very good health. All 10 primary participants reported receiving support from family or friends to help manage their diabetes. Nine primary participants reported being previously prescribed a diabetes medication, primarily metformin.
Biometric Outcomes: Primary Participants
As shown in Table 2, primary participants had a mean decrease in HbA1c of 0.7%, a mean increase in BMI of 0.2 kg/m2, a mean decrease in LDL of 2.1 mg/dL, and a mean increase in HDL of 2.7 mg/dL. Primary participants also experienced a mean decrease in SBP and DBP of 10.9 mm Hg and 1.0 mm Hg, respectively. Overall blood pressure improved, with the number of participants with stage 2 hypertension decreasing from 5 to 1. While most biometric measures were improved from pre- to postintervention, none of these changes were statistically significant (Ps > .05).
Primary Participants’ Biometric Measures at Pre-and Postintervention With Test of Change.
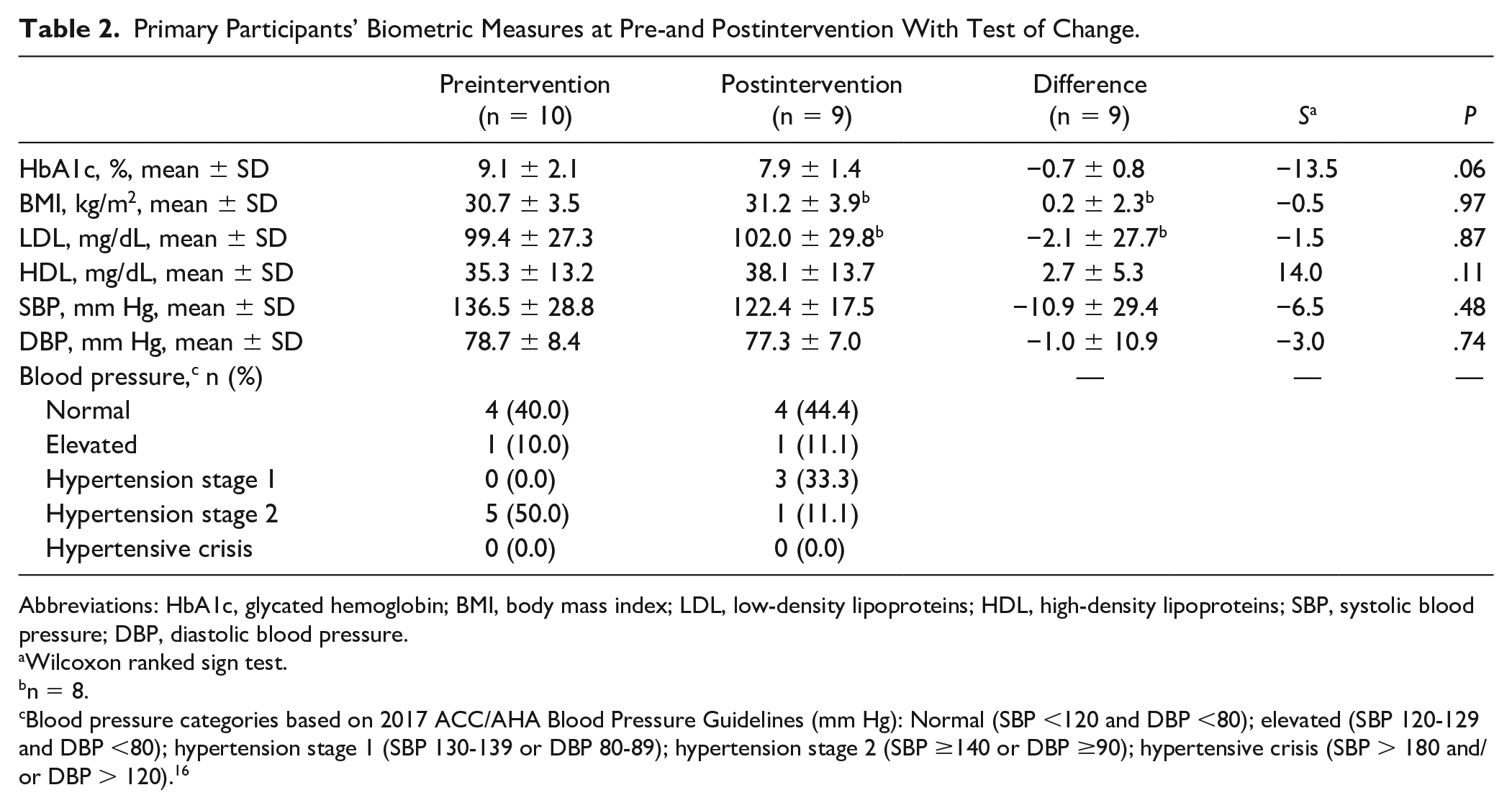
Abbreviations: HbA1c, glycated hemoglobin; BMI, body mass index; LDL, low-density lipoproteins; HDL, high-density lipoproteins; SBP, systolic blood pressure; DBP, diastolic blood pressure.
Wilcoxon ranked sign test.
n = 8.
Blood pressure categories based on 2017 ACC/AHA Blood Pressure Guidelines (mm Hg): Normal (SBP <120 and DBP <80); elevated (SBP 120-129 and DBP <80); hypertension stage 1 (SBP 130-139 or DBP 80-89); hypertension stage 2 (SBP ≥140 or DBP ≥90); hypertensive crisis (SBP > 180 and/or DBP > 120). 16
Table 3 shows disaggregated results, providing each primary participants’ changes in HbA1c from pre- to postintervention. Six of the primary participants showed improvements from pre- to postintervention, while 1 showed no change and 2 worsened very slightly. Improvements ranged from 0.1% to 2.0% and 5 of the 6 participants had improvements of 0.5% or higher.
Detailed Changes in HbA1c (Glycate Hemoglobin, %) for Primary Participants.

Participant 3 failed to complete postintervention data collection.
Biometric Outcomes: Family Members
As shown in Table 4, family members experienced a mean reduction in HbA1c of 0.4%, a 0.2 kg/m2 mean increase in BMI, a mean increase in LDL by 16.6 mg/dL, and a mean increase in HDL of 4.0 mg/dL. Family members experienced a mean decrease in SBP of 2.6 mm Hg and a mean decrease in DBP of 2.6 mm Hg. Overall blood pressure improved, with the number of family participants who had elevated blood pressure decreasing from 2 to 0 and the number in hypertensive crisis decreasing from 1 to 0. While most biometric measures improved between pre- and postintervention, none of the changes were statistically significant (Ps > .05).
Family Members’ Biometric Measures at Pre- and Postintervention With Test of Change.

Abbreviations: HbA1c, Glycated hemoglobin; BMI, body mass index; LDL, low-density lipoproteins; HDL, high-density lipoproteins; SBP, systolic blood pressure; DBP, diastolic blood pressure.
Wilcoxon ranked sign test.
n = 7.
Blood pressure categories based on 2017 ACC/AHA Blood Pressure Guidelines (mm Hg): Normal (SBP <120 and DBP <80); elevated (SBP 120-129 and DBP <80); hypertension stage 1 (SBP 130-139 or DBP 80-89); hypertension stage 2 (SBP ≥140 or DBP ≥ 90); hypertensive crisis (SBP > 180 and/or DBP > 120). 16
Lessons Learned and Facilitators and Barriers to Implementation
The primary facilitators identified by the implementation team centered on the clinical setting. Conducting the educational sessions in the clinic seemed to reduce distractions, which in turn increased participants’ attention and engagement in the class sessions. The clinical setting also allowed patients who needed medical attention to receive that care during the same visit. Another facilitator identified by the implementation team was the full involvement of clinical staff. Staff expressed excitement about providing a new type of service to patients, in addition to routine clinic services. Having a private room for data collections was also identified as a facilitator for securing quality data. Bilingual staff members were essential for the Marshallese participants who may not be fluent in English. The implementation team noted the primary barrier to implementation was transportation. The lack of reliable transportation made it difficult for participants to attend classes as scheduled. As a result, many of the educational sessions and data collections had to be rescheduled to help overcome these transportation barriers. The regular changing of schedules made it harder for the clinic staff to balance study activities with usual clinic activities.
Discussion and Conclusion
Discussion
The purpose of this study was to pilot test the feasibility and preliminary effectiveness of the Adapted-Family DSME when implemented in a real-world clinical setting. All 10 primary participants completed all 10 hours (100%) of the educational intervention. Furthermore, eight of the 10 family members completed all 10 hours (100%) of the educational intervention. One family member completed more than half of the intervention, and one family member dropped out, completing none of the intervention. These retention rates are consistent with those documented in the prior fully powered RCT, 13 and suggests that high levels of retention in a full study conducted in a clinical setting may be feasible. Qualitative feedback from the implementation team highlighted the primary facilitators to be the clinical setting, full involvement of clinical staff, having a private room for study activities, and having bilingual staff. These findings are consistent with studies of DSME with other populations.17-19
The preliminary results of this pilot study are also encouraging. The mean decrease in HbA1c of 0.7% was not statistically significant, likely due to the small sample size. However, those participants with improvements in HbA1c equal to or greater than 1.0% are likely to experience clinically meaningful benefits.20-22 For example, each 1% reduction in HbA1c has been associated with a 14% decrease in risk for heart attack, a 12% decrease in risk for stroke, a 37% decrease in risk for microvascular complications, and a 21% decrease in risk for death related to diabetes. 23 Additionally, it has been shown that even a change in HbA1c as small as 0.5% can potentially lead to clinically meaningful outcomes.21,22,24 The mean changes documented in this study are slightly lower than those found in the prior RCT, which showed an adjusted reduction in mean HbA1c of 1.2% at immediate postintervention for those in the Family DSME arm. This study also showed improvements for participants already being treated by a physician and already on medication therapy.
Family members achieved slight improvements in HbA1c and blood pressure. This is in contrast to the RCT that did not show family member improvements with regard to clinical indicators. 25 This finding demonstrates that clinical implementation has the potential to improve family member outcomes, as well as engagement in the intervention.
Limitations
The results of this pilot study should be interpreted in light of its limitations. The inclusion of only Marshallese adults utilizing a clinic in northwest Arkansas limits the generalizability of the results to patients belonging to other racial/ethnic groups and patients in other geographic locations. Because this study was designed to test feasibility, the sample size (n = 10) limits the validity of the observed results, as the study was not adequately powered to detect statistically significant changes in clinical outcomes. Finally, the study did not include a control group for comparison; therefore, the observed changes cannot be attributed solely to the Adapted-Family DSME intervention.
Conclusion
This study extends prior findings by showing that the Adapted-Family DSME intervention can be successfully delivered in a real-world clinical setting. This clinic-based pilot study makes a significant contribution to the literature because it may be more feasible and cost-effective compared to delivering the Adapted-Family Model DSME in patients’ homes. The study’s results and lessons learned will be used to inform a fully powered RCT of the Adapted-Family Model DSME in clinical settings.
Footnotes
Author Contributions
PAM, AJS, and ROW wrote the initial draft of the manuscript. PAM, JPS, JSH, AH, LHD, TKS, and CRL designed the study. CRL, JPS, and AJS designed the analysis plan and analyzed data. JSH, AH, LHD, and TKS ensured clinical relevance to the study design, analysis and critically revised the manuscript for clinical relevance and approved the final version. All authors substantially participated in the design, implementation, and interpretation of research. All authors critically revised the manuscript for intellectual content and approved the final version. PAM is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of data analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This pilot study was supported by the Translational Research Institute (TRI; Grant No. 1U54TR001629-01A1) through the National Center for Advancing Translational Sciences of the National Institutes of Health (NIH). This study was also funded in part by a grant from the Sturgis Foundation, and the DSME adaptation was funded by the Patient-Centered Outcomes Research Institute (PCORI; Grant No. AD-1310-07159). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH, the Sturgis Foundation, or PCORI.
