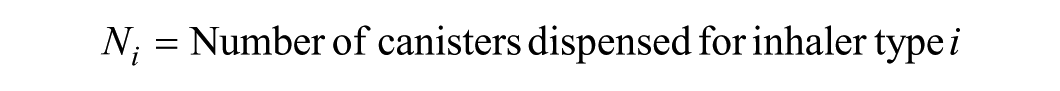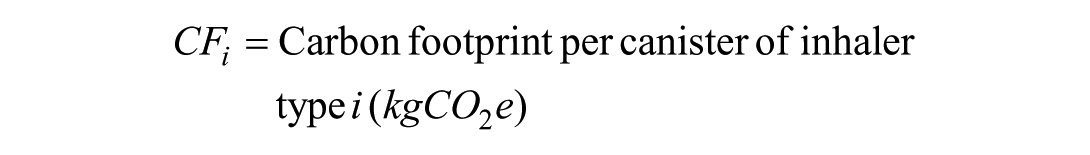Abstract
Background:
Global Initiative for Asthma (GINA) has recommended inhaled corticosteroid (ICS)-Formoterol as Track-1 treatment for patients. Using ICS and SABA (Short-Acting Beta-Agonist) as a reliever is an alternative Track-2 option. Both modalities are tied to type of inhaler use, dry powder inhalers (DPI) and propellent-containing metered-dose inhalers (pMDI). Compared to propellent-free DPI, pMDI have much higher carbon footprint (CF) detrimental to the environment. Leveraging on dispensed inhaler data from the electronic medical records of patients managed in primary care, the study aimed to quantify their CF using Budesonide-Formoterol (BUD-FOR) DPI alone, compared to those who were treated with BUD-FOR DPI + pMDI as a reliever.
Methods:
Electronic medical records from 8 public primary care clinics were analysed, covering adult asthma patients (aged ≥21) between 2018 and 2023. Data on inhaler dispensing, asthma control test (ACT) scores and rescue therapy (RT) needs were assessed. CF was computed based on inhaler canisters dispensed. Associations between treatment modality, asthma control, RT and CF were analysed using Generalized Estimating Equations.
Results:
A total of 5634 patients using BUD-FOR DPI were included. Over the study period, Track-1 usage increased substantially from 466 to 2317 patients, while Track-2 rose modestly from 628 to 758. In 2023, 78.5% of patients achieved good asthma control compared to 68.7% in Track-2. The total CF per patient was substantially lower in Track-1 compared to Track-2 (3.3 vs 62.4 kgCO₂e). Patients in Track-1 had a significantly lower CF by 60 kgCO₂e (P < .001), had 1.5 times higher odds to achieve good asthma control based on ACT scores (P < .001) and had approximately 30% lower odds of receiving RT (P < .001). The average number of SABA-pMDI canisters dispensed per patient declined from 2.8 to 2.1 over the study period. BUD/FOR inhalers use per patient per year was consistently higher in Track 2 compared to Track 1, with an average difference of 1.2 canisters (5.3 vs 4.1).
Conclusion:
Patients managed under Track-1 treatment approach demonstrated significantly better asthma outcomes and lower CF. These findings highlight potential of Track-1 treatment as the preferred strategy, enabling better clinical outcomes and reduced environmental impact.
Introduction
Climate change represents a critical threat to global public health, with cascading effects that compromise human health outcomes by undermining both the social determinants of health and environmental conditions fundamental for well-being. 1 The World Health Organization 1 reports that 3.6 billion people live in areas highly susceptible to climate change, which is expected to cause approximately 250 000 additional deaths per year between 2030 and 2050. The direct damage costs to health are estimated to be between US$ 2 and 4 billion annually by 2030.
Climate change occurs mainly from excessive emissions of greenhouse gases (GHG) such as carbon dioxide to the environment. 2 GHG emission can be quantified using carbon footprint (CF), which is a measure of the amount of carbon dioxide released into the atmosphere because of activities of individuals, organizations and communities. Urgent and prompt measures to reduce CF are pertinent to mitigate the effects of global warming, which is detrimental to life on earth, including humans. 3
Healthcare systems are responsible for 4% to 5% of the GHG emissions worldwide. 4 According to the Greenhouse Gas Protocol, healthcare’s carbon footprint is predominantly driven by Scope 3 emissions (50%-75%) from disposables, equipment and pharmaceuticals, with the remainder (15%-50%) attributed to direct and indirect energy use under Scopes 1 and 2 of the Greenhouse Gas Protocol.4,5 In UK healthcare system, primary care is responsible for over 20% of GHG emissions. 5
Inhalers are commonly prescribed as evidence-based effective treatment for asthma and COPD. In such therapeutics, Hydrofluroalkane (HFA), a potent GHG, may be released into the environment from pressurized meter-dosed inhalers (pMDI). Propellent-containing metered-dose inhalers (pMDIs) alone account for 22% of primary care’s carbon footprint. Changing to dry powder inhalers (DPIs) can potentially decrease the CF of an inhaler by up to 98% but it should be grounded in evidence-based clinical practice guidelines. 6
In 2024, Global Initiative for Asthma (GINA) updated its recommendation for Track-1 treatment with Inhaled corticosteroid (ICS)-Formoterol (FOR) as a Single Maintenance And Reliever Therapy (SMART) for patients with mild to moderately severe asthma. 7 However, GINA also recommends the use of ICS with as-needed Short-Acting Beta-Agonist (SABA) due to limited access to SMART in certain communities and healthcare system. In Singapore, a developed and urbanized nation with a dual public-private healthcare system, the 2 treatment modalities are available to patients who receive asthma treatment in public primary care clinics (polyclinics) even before the latest GINA recommendations were issued.
The Group Procurement Office (GPO) supports local public regional healthcare systems in bulk purchase of required drugs for better pricing, including the inhalers. Budesonide-Formoterol (BUD-FOR) is one of the ICS-long-acting beta agonist (LABA) medications procured by the GPO. BUD-FOR is dispensed as Turbuhaler, a DPI, from the in-house pharmacy within each polyclinic, while SABA is mainly inhalational Salbutamol delivered by pMDI. The asthma control and the impact on CF among patients receiving these 2 asthma treatment modalities over the years remain unclear.
The study aimed to compare the asthma control and CF among adult patients treated with BUD-FOR based on SMART compared to those using BUD-FOR DPI with add-on SABA-pMDI as a reliever. The results will be shared with policy makers in drug procurement and facilitate shared decision making between primary care professionals and their patients based on clinical effectiveness and environmental impact.
Methods
Study Design
This retrospective longitudinal study analysed data from electronic medical records (EMR) based on routine clinical asthma management. The study covered 8 public primary care clinics in Eastern Singapore from 2018 to 2023.
Study Sites and Population
The study included patients aged 21 years and above with a clinical diagnosis of asthma during the study period based on International Classification of Diseases 10th Revision (ICD-10). Clinical and administrative data comprising demographics, Asthma Control Test (ACT) scores, dispensed asthma medications and Rescue Therapy (RT) records were extracted. RT refers to the institution’s protocol-based administration of inhaled salbutamol and ipratropium via spacer device for patients presenting with wheezing and/or breathlessness with rhonchi detected on lung auscultation by healthcare professionals. The polyclinics provide walk-in access for acute medical conditions, including RT for asthma exacerbations.
Patients who had at least 1 ACT score per year, indicating active asthma follow-up were included. We excluded patients with incomplete ACT records, those who switched inhaler types (DPI or pMDI) and those not on BUD-FOR during the study period. All asthma medications were dispensed through in-house polyclinic pharmacies, with prescription and dispensing data captured in the EMR.
Data Management
In compliance with national bioethics regulations, data extracted from the Electronic Health Intelligence System (eHINTS), a database storing all clinical data, were de-identified by an independent approved third party before analysis. The variables extracted were demographic (age, gender and ethnicity), ACT records, RT records with treatment intensity, dispensed inhaled ICS or ICS-LABA medication (Beclomethasone Dipropionate, Budesonide and Fluticasone propionate, Fluticasone/Salmeterol, Budesonide/Formoterol) and dispensed inhaled beta-agonist (Salbutamol). The list of inhalers was extracted based on the institution-approved formulary and procurement records. RT utilization records were available from 2016 onwards, following the integration of these records into standardized clinical documentation. The RT utilization records capture the number of RT cycles delivered to each patient throughout their treatment course. Only de-identified data availed to the study team for analysis.
Outcome Measures
The primary outcome was inhaler usage, quantified by the number of canisters dispensed for the inhaler type. Each inhaler was assigned a carbon footprint value based on its propellant type, referenced from literature.8-10 Calculations for the carbon footprint in this study focus on the entire lifecycle of the inhalers, with the assumption that all puffs are actuated, and include the manufacturing, distribution and disposal phases of the inhalers. The carbon footprint value represents total greenhouse gas emissions per canister expressed in kilograms of carbon dioxide equivalent (kgCO₂e). More details on carbon footprint computation are provided in Supplementary Table 1. RT carbon footprint calculations were based on the standard protocol of 10 Ventolin puffs and 4 Atrovent puffs per RT cycle, in accordance with the organization clinical guidelines. Moreover, prior to inhalation of Ventolin, 10 Ventolin puffs are sprayed onto the spacer to prevent Salbutamol particles from sticking to the walls and reduce the delivered dose. Patients who need RT within the polyclinic could receive up to 3 RT cycles depending on their severity. Those whose symptoms did not resolve after 3 cycles were referred to the Emergency Department (ED) for further management. The total carbon footprint calculation was as follows:
where,
Patients were categorized based on ACT scores as either suboptimal-controlled (ACT < 20) or well-controlled (ACT ≥ 20). Patients with any ACT score below 20 within a calendar year were classified as suboptimal-controlled. Only patients who were dispensed BUD/FOR-DPI without any other ICS or ICS-LABA medications throughout the year were included in the analysis. Among them, patients who dispensed at least 1 canister of Salbutamol within the year were classified as Track-2, while the remaining patients were categorized as Track-1. We conducted comparative analyses of greenhouse gas emissions between these 2 tracks.
Data Cleaning
Data wrangling was performed to ensure the accuracy, reliability and consistency. Inhaler usage data was tabulated to ensure that patients remained on the same inhaler throughout the year. Duplicated ACT records from the same visit were removed. The final dataset were structured in a longitudinal format for analysis, where a single patient could have records in multiple years within the study period. Patients with missing data on demographics and ACT scores were excluded to ensure that patients who were regularly reviewed and managed by the respective polyclinics were included in the study population. Only complete case records were analysed (Figure 1).

CONSORT-style flow diagram of participant inclusion and exclusion.
Statistical Analyses
All statistical analyses were performed using R software (version 4.4.2), and data visualizations were generated using Tableau (version 2023.3). Descriptive statistics were presented to summarize carbon emissions from patients on Track-1 and Track-2. The relationship between asthma outcomes and treatment modality over the 6-year study period was analysed using Generalized Estimating Equations (GEE) with a logit link function for binary outcomes. The GEE model accounted for potential confounding variables including age, gender, ethnicity, obesity, frequency of ACT records and baseline ACT severity. An exchangeable correlation structure was selected to address within-subject correlation in the longitudinal data, accommodating irregular patterns of patient follow-up across years. For the analysis of total carbon footprint, a separate GEE model was constructed using a Gaussian distribution with identity link function, as the outcome variable was continuous. This model similarly incorporated an exchangeable correlation structure and adjusted for relevant covariates. Statistical significance was set at P < .05, and results were presented with 95% confidence interval where applicable.
Results
Data from 5634 unique Asian adult patients aged 21 years and older who were treated with BUD/FOR DPI were included. Year on year between 2018 and 2023, more patients (from 1094 to 3075) were prescribed BUD/FOR DPI, corresponding to a 2.8-fold increased. However, the total annual CF from the increasing number of BUD/FOR DPI users was similar in 2018 and 2023 (54 938 kgCO₂e) due to declining CF per patient, from 48 to 18 kgCO₂e. Table 1 presents the carbon footprint, categorized by treatment modalities, asthma control and inhaler type.
Annual Greenhouse Gas Emission Among Patients on BUD/FOR DPI.

Total: Total carbon footprint calculated for all patients under the indicated track.
In 2018, slightly more than half of the patients (57.4%, n = 1094) were treated with BUD/FOR DPI with SABA-pMDI. Dispensed SABA-pMDI (Salbutamol) was the largest CF contributor, accounting for more than 80% of total annual CF, with 44 296 kgCO₂e calculated in 2023. Consequently, from 2020 to 2023, there was an increasing proportion of patients solely treated with BUD/FOR DPI, and it peaked at 75.3% in 2023. Despite having 3-fold more patients in 2023, the total CF from Track 1 (7627 kgCO₂e) was approximately 6 times lower than that of Track 2 (47 311 kgCO₂e) (Figure 2).
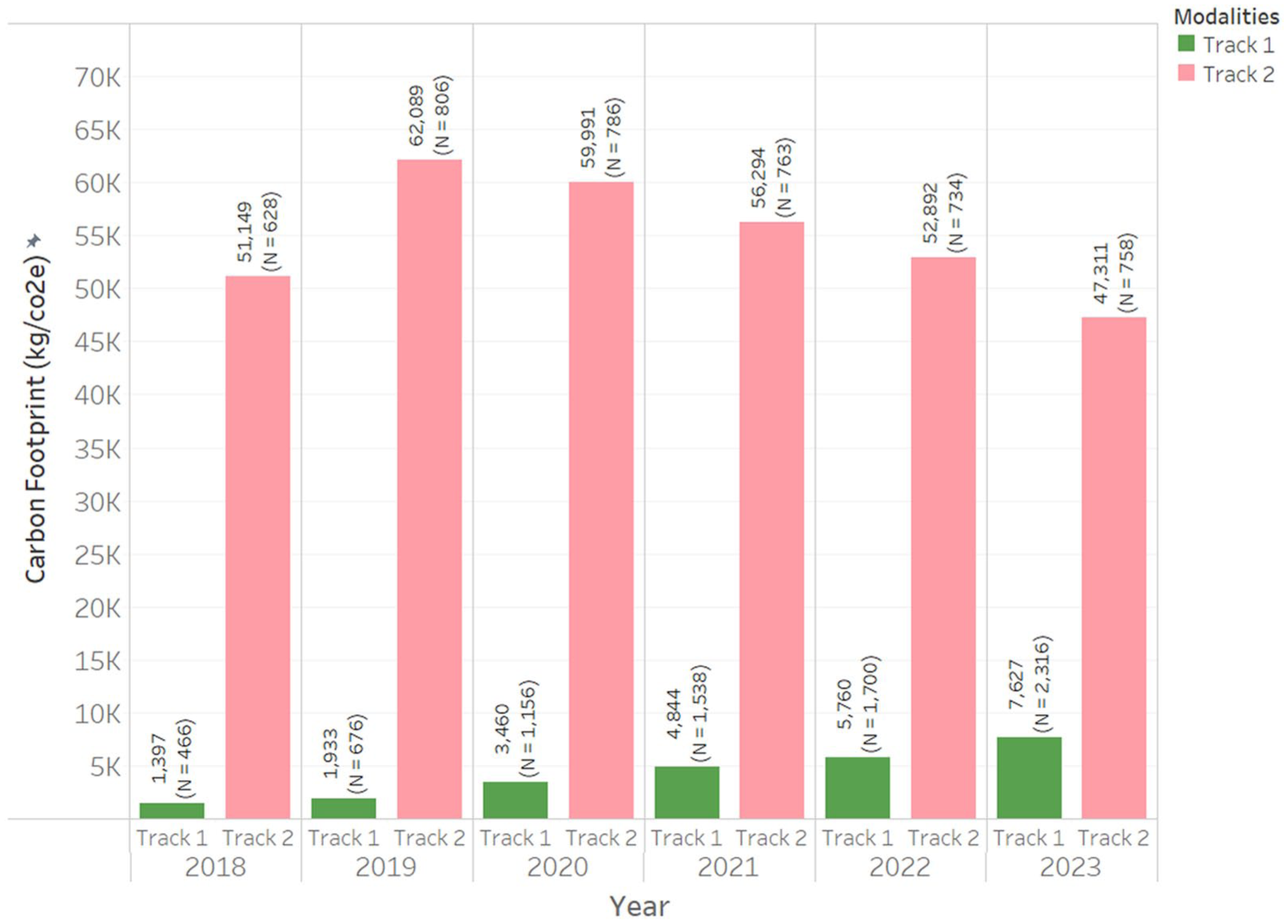
Total carbon footprint stratified by modalities.
Comparing the Asthma Control Between the 2 Cohorts of Patients
Patients on BUD/FOR DPI-only achieved better asthma control across the entire study period compared to those on BUD/FOR DPI with Salbutamol. Almost one-third of patients on BUD/FOR DPI-SABA-pMDI (31.3%) had suboptimal asthma control in 2023, compared to one-fifth in the BUD/FOR-DPI group (21.5%). Patients with suboptimal asthma control had significantly higher number of ACT records, signifying more clinic visits (3.3 vs 2.4, P < .001). GEE analysis revealed that patients on Track 1 had 1.6 times odds of achieving good asthma control (AOR = 1.63, 95% CI = 1.43-1.86, P < .001). Additionally, patients on Track 1 have lower rates of asthma exacerbation requiring RT, with mean rate of 12.3% compared to 17.7% in Track 2 (Figure 3). Further analysis with GEE modelling revealed that patients on Track 1 have a reduced odds of receiving RT (AOR = 0.7, 95% CI = 0.62-0.79, P < .001). The results are summarized in Table 2.
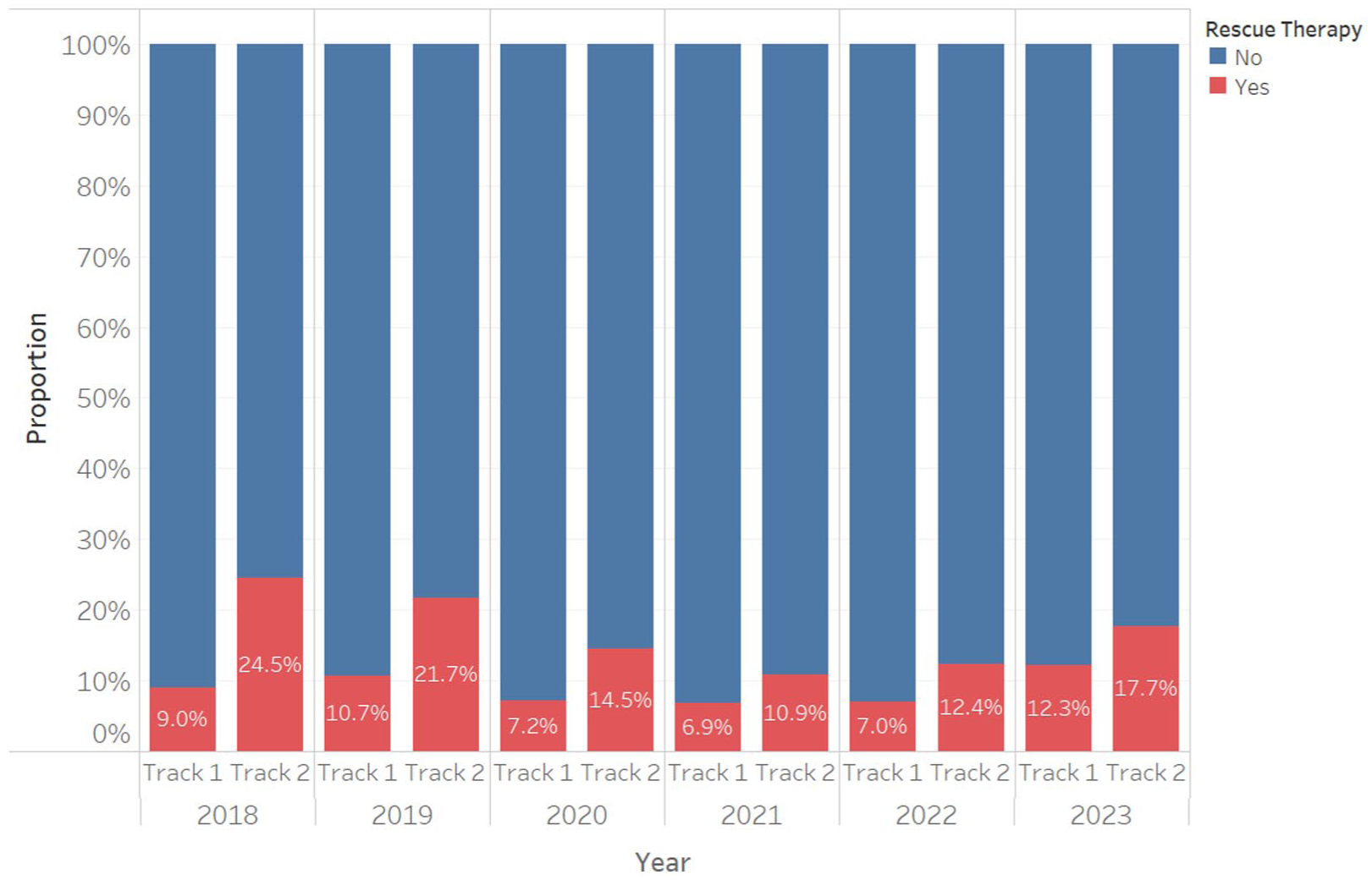
Proportion of rescue therapy utilization in track 1 and 2.
Association of GINA Track With ACT Control, Total Carbon Emission and RT Occurrence.

Abbreviation: OR, odds ratio.
Model 1: ACT control status in current year as outcome. Adjusted for age, gender, ethnicity, obesity, total ACT records, baseline ACT control.
Model 2: Total carbon emission from BUD/FOR DPI + Salbutamol as outcome. Adjusted for age, gender, ethnicity, ACT control status, year, obesity, total ACT records, baseline ACT control.
Model 3: Rescue therapy occurrence in current year as outcome. Adjusted for age, gender and ethnicity, ACT control status, year, obesity, total ACT records, baseline ACT control.
Comparing the CF Between the 2 Cohorts of Patients
Patients on Track 1 emit an average of 3.1 kgCO₂e annually. Comparatively, those on Track 2 demonstrated substantially higher CF, with approximately 18-fold greater emissions (Figure 4). In the Track 2 cohort, patients with suboptimal asthma control exhibited a slighter higher CF compared to those with good asthma control, with convergence observed in 2022 and 2023 at 62 kgCO₂e. (Figure 5). The difference in CF was minimal between patients with suboptimal and good control asthma in Track 1, with a maximum difference of 0.6 kgCO₂e observed in 2018.

Average carbon footprint per patient stratified by modalities.

Average carbon footprint per patient stratified by asthma control and modalities.
The CF associated with inhaler use was significantly lower among patients in Track 1 compared to those in Track 2, based on the inhalers dispensed (β = −60, P < .001) (Table 2). Among patients in Track 2, those with suboptimal asthma control had a higher calculated carbon emissions compared to with good control (β = 9.4, P < .001). CF from rescue therapy (RT) in the primary care clinics were significantly higher in patients with BUD/FOR-SABA pMDI group compared to those on BUD/FOR DPI only (β = 0.4, P < .001). The annual average CF per patient was higher in Track 2 (0.9 kgCO₂e) compared to almost half (0.5 kgCO₂e) in the solo BUD/FOR-DPI group (Figure 6). Patients from both groups had the lowest CF from RT in 2021, with 1.2 kgCO₂e for suboptimal patients in Track 2 (Table 3).

Carbon footprint generated from rescue therapy in primary care clinics.
Annual Greenhouse Gas Emission for Rescue Therapy Among Patients on BUD/FOR DPI.
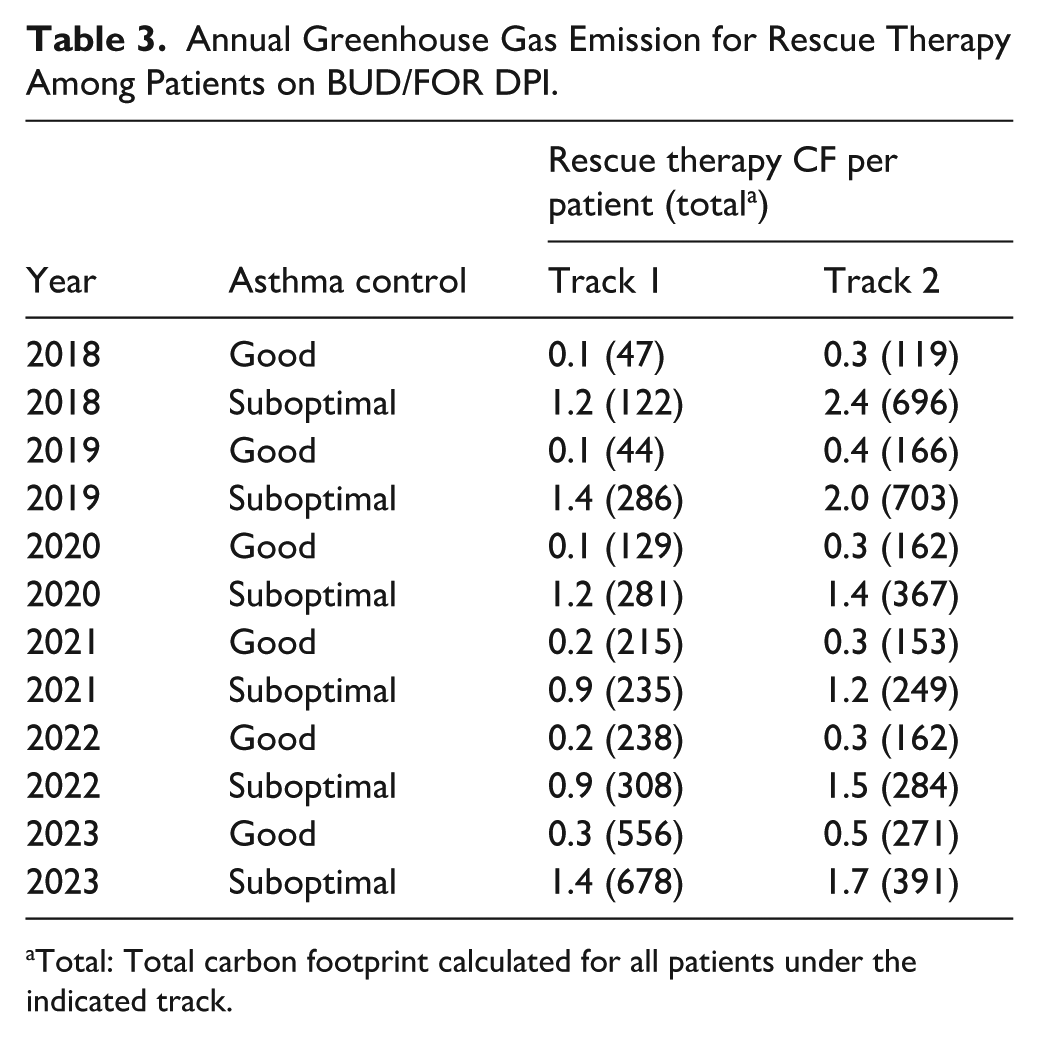
Total: Total carbon footprint calculated for all patients under the indicated track.
Comparing Canisters Dispensed Between the 2 Cohorts of Patients
Figure 7 shows that the average number of SABA-pMDI dispensed per patient decreased by about 1 from 2.8 to 2.1 between 2018 and 2023. The number of BUD/FOR inhalers dispensed per patient per year was consistently higher in Track 2 compared to Track 1, with an average difference of 1.2 canisters. Among Track 1 patients, the annual number of canisters dispensed increased slightly over the years, from 4 in 2018 to 4.4 in 2023.

Average inhaler canisters dispensed in Track 1 and 2.
Discussion
GINA strategy first recommended the 2-track system for asthma treatment in its 2021 report to use either ICS-formoterol alone (Track-1) or with add-on SABA (Track-2). The updated GINA 2024 Report recommends as-needed low dose ICS-formoterol as the reliever at all steps, with the option of adding daily maintenance ICS-formoterol (MART) for steps 3 to 5, as the preferred Track-1 treatment for adults and adolescents.
Using real-world data in primary care where most patients with asthma are treated, this study provides additional evidence to support the GINA 2024 preferred strategies for adults to use Track-1 treatment approach. The results show significantly better asthma control in patients who were treated based on GINA Track-1 recommendation using BUD/FOR DPI alone compared to those with add-on SABA-pMDI in Track-2 (Table 1). The odds of achieving good asthma control based on ACT scores was 1.6 times in patients receiving Track-1 treatment in primary care (Table 2). This is also supplemented by a 30% reduction in likelihood of seeking treatment for RT for patients on Track-1. It is aligned to evidence gathered by GINA, which reported that patients on MART had 33% fewer severe exacerbations resulting in hospitalization than those treated with ICS/LABA + SABA. 11 A meta-analysis of randomized controlled trials similarly showed that MART using ICS/FOR significantly decreased risk of exacerbations compared with those treated with ICS ± LABA + SABA in 22 748 patients aged 12 years and older. 12
Aside from asthma control, the findings reveal that using GINA treatment strategies led to less adverse environmental impact, with an average 18-fold reduction in CF annually. 7 In a Singapore-based study comparing CF across different ICS-LABA inhalers, BUD/FOR was found to have both the lowest and a declining annual CF per patient. This trend aligns with the increasing uptake of patients on Track 1 treatment, thereby contributing to a lower per patient CF. 13 In contrast, more patients in Track 2 had rescue therapy in the primary care clinics, which routinely entails delivery of SABA via pMDI and spacer to patients experiencing acute exacerbation from bronchoconstriction, and hence further increased their CF. Additionally, a previous study that examined treatment by ICS + SABA demonstrated substantial CF of 140 kgCO₂e per patient, with SABA accounting to almost 80% of the emission despite similarity in asthma outcomes. 14 These findings further support the environmental benefit of optimizing asthma care with Track-1 treatment, reducing reliance on SABA in favour for MART therapy.
Both cohorts of patients had declined CF from RT during the COVID-19 pandemic from 2020 to 2022 (Figure 6). Infection control measures such as personal protective equipment, mask, physical and social distancing, contact tracing and isolation significantly reduced acute respiratory viral infection, an established asthma trigger. 15 In turn, patients had fewer acute exacerbations and less usage of reliever therapy. In addition, a proportion of them might have avoided medical treatment in healthcare facilities for mild exacerbation for fear of cross-infection with COVID-19. 16
Patients in the BUD/FOR-SABA-pMDI group were dispensed an average of 2.1 pMDIs in 2023. Despite a declining trend from 2018 to 2023, the CF was high in this group, even if these patients might not use up the SABA entirely. While this average of 2.1 canisters per year falls below the threshold of 3 or more canisters typically defined as over-use, it still represents substantial SABA reliance with both environmental and clinical implications. In the Swedish nationwide SABINA cohort of 365 324 patients, SABA over-use was associated with a 26%-77% higher risk of exacerbations and up to a 2.35-fold higher mortality risk. 17 Over-dependency among OECD countries is common, ranging from 16% in Germany to 38% in Sweden among patients who use 3 or more SABA canisters annually. 17
Based on morbidity, mortality and environmental risk factors, it is pertinent for clinicians to embrace the preferred Track-1 treatment in their prescription for their patients. However, modifying clinicians’ prescribing behaviour is challenging as they are influenced by multiple individual, institution and system factors. 18 It takes time to induct them to the growing evidence of distinctive difference in clinical effectiveness and environmental hazard between Track 1 and 2 treatment modality before considering to change their practice. Additionally, GINA modifies their clinical practice guidelines after 3 years based on expanding literature from 2021 to 2024. 7
Changing treatment regimen without shared decision-making between doctor and patient can adversely impact their relationship and potentially worsen the patient’s asthma control due to poor adherence to the new treatment modality. 19 Patient’s understanding of the basis and practicality of SMART by using a single inhaler can help to relieve their impending asthma exacerbation without the need for a separate reliever inhaler is crucial for the successful switch from Track-1 to Track-2 treatment. A recent UK survey revealed that a majority (81%) of patients were involved or partially involved in their inhaler choice. 20 However, only 62% of them reported willingness to switch to a more environmentally friendly inhaler with equal effectiveness. 21 A vast majority (91%) of them would like further information about their inhalers and their impact on the environment, 22 suggesting a scope for more public education on environmentally friendly management of asthma in the community.
GINA recommends that all individuals with asthma should be provided with an asthma action plan, which is a personalized step-by-step guide to assist them to manage their asthma, recognize worsening symptoms and educate them on the appropriate use of medications. 7 Incorporating Track-1 treatment details to reframe a patient’s personal asthma action plan is a measure for doctors to assist them to understand and adhere to the treatment. Adding CF information of the respective inhalers in these personalized asthma action plans is a method to raise the awareness of environmental harm among patients, especially their use of SABA-pMDI. Further research is needed to evaluate the effectiveness of such measure to facilitate the switch to Track-1 for patients currently using Track-2 treatment modality.
Successful adoption of preferred asthma treatment modality requires a comprehensive review of the local healthcare ecosystem, from national drug formulary, medication supply chain to procurement procedure and healthcare financing framework and policies. Drug procurement focuses on achieving value for money, with consideration for both clinical effectiveness and cost of the drugs. 22 A paradigm shift in drug procurement policy is necessary to take into the account the long-term financial cost resulting from environmental damage from the pMDI. Lessons can be gained from the economic and CF analysis of the inhaler prescription in National Health Service (NHS), which embarked on a mission to achieve net zero future. 23 It showed cost-savings can be achieved by switching to low GWP (Global Warming Potential) inhalers such as the BUD/FOR-DPI23. Countries that use group procurement such as Singapore with its local Group Procurement Office (GPO), could similarly incorporate environmental impact and balance it against potential long-term cost savings. The shift could be relevant despite higher up-front price of BUD/FOR-DPI even after official drug subsidies. With Track-1 uptake at approximately 13% and Track-2 uptake at 4%, assuming similar pattern nationwide, an estimated 220 kgCO₂e could be avoided yearly if patients on Track-2 BUD/FOR-SABA-pMDI were switched to Track 1. The authors recommend that patients, prescribers and policymakers carefully consider the CF of the inhalers and where they are likely to equally effective, prioritize the low GWP inhalers. 24
Limitations
This retrospective database study has several limitations. Firstly, there is a possibility of carbon emission uncertainty. The carbon footprint calculated were based on reported carbon footprint by the pharmaceutical companies at a single time point and their processes could get refined along the way to minimize environmental impact. The values presented represents estimates and actual emission might differ according to the manufactured year. Secondly, SABA usage outside the primary care clinics were not accounted for. The results were computed based on dispensed inhalers from the polyclinic pharmacies. Additional purchase of SABA-pMDI from other prescribers and community pharmacies are not accounted for, which would under-estimate the CF. Moreover, the assumption is that each dispensed 200-puff pMDI was used up entirely by patient as those unconsumed inhalers were not returned to the pharmacies. However, the HFA in these inhalers would still be released to the environment when they are disposed by incineration. Thirdly, ACT measurement bias may be present, as the ACT relies on patient self-reporting and recall, which can be subject to recall bias and may not fully capture objective measures of asthma control and severity. Lastly, our dataset lacked information on potentially important confounders including smoking status, medication adherence and socioeconomic factors. These variables may influence asthma severity and prescribing patterns, thereby affecting both clinical outcomes and carbon footprint estimates.
Conclusion
Higher proportions of patients in the Track-1 treatment group had good asthma control and lesser CF compared to those in Track-2. Clinicians should consider treating more patients using Track-1 modality due to its higher clinical effectiveness and lower environmental risks. They have to engage and educate their patients to raise their awareness of the health and environment benefits and for shared decision making before embarking on the treatment switch.
Supplemental Material
sj-docx-1-jpc-10.1177_21501319251411430 – Supplemental material for Comparing 6-Year Carbon Footprint Between GINA Track 1 and 2 Asthma Management for Adults: A Real-World Primary Care Report from Singapore to Advocate for Policy Change in Inhaler Procurement
Supplemental material, sj-docx-1-jpc-10.1177_21501319251411430 for Comparing 6-Year Carbon Footprint Between GINA Track 1 and 2 Asthma Management for Adults: A Real-World Primary Care Report from Singapore to Advocate for Policy Change in Inhaler Procurement by Ngiap Chuan Tan, Ding Xuan Ng, Mabel Qi He Leow and Yi Ling Eileen Koh in Journal of Primary Care & Community Health
Supplemental Material
sj-pdf-2-jpc-10.1177_21501319251411430 – Supplemental material for Comparing 6-Year Carbon Footprint Between GINA Track 1 and 2 Asthma Management for Adults: A Real-World Primary Care Report from Singapore to Advocate for Policy Change in Inhaler Procurement
Supplemental material, sj-pdf-2-jpc-10.1177_21501319251411430 for Comparing 6-Year Carbon Footprint Between GINA Track 1 and 2 Asthma Management for Adults: A Real-World Primary Care Report from Singapore to Advocate for Policy Change in Inhaler Procurement by Ngiap Chuan Tan, Ding Xuan Ng, Mabel Qi He Leow and Yi Ling Eileen Koh in Journal of Primary Care & Community Health
Supplemental Material
sj-pdf-3-jpc-10.1177_21501319251411430 – Supplemental material for Comparing 6-Year Carbon Footprint Between GINA Track 1 and 2 Asthma Management for Adults: A Real-World Primary Care Report from Singapore to Advocate for Policy Change in Inhaler Procurement
Supplemental material, sj-pdf-3-jpc-10.1177_21501319251411430 for Comparing 6-Year Carbon Footprint Between GINA Track 1 and 2 Asthma Management for Adults: A Real-World Primary Care Report from Singapore to Advocate for Policy Change in Inhaler Procurement by Ngiap Chuan Tan, Ding Xuan Ng, Mabel Qi He Leow and Yi Ling Eileen Koh in Journal of Primary Care & Community Health
Supplemental Material
sj-pdf-4-jpc-10.1177_21501319251411430 – Supplemental material for Comparing 6-Year Carbon Footprint Between GINA Track 1 and 2 Asthma Management for Adults: A Real-World Primary Care Report from Singapore to Advocate for Policy Change in Inhaler Procurement
Supplemental material, sj-pdf-4-jpc-10.1177_21501319251411430 for Comparing 6-Year Carbon Footprint Between GINA Track 1 and 2 Asthma Management for Adults: A Real-World Primary Care Report from Singapore to Advocate for Policy Change in Inhaler Procurement by Ngiap Chuan Tan, Ding Xuan Ng, Mabel Qi He Leow and Yi Ling Eileen Koh in Journal of Primary Care & Community Health
Footnotes
Ethical Considerations
The study was conducted in accordance with the Declaration of Helsinki and was approved by the SingHealth Centralized Institutional Review Board (2023/2362) on Jul 10, 2023.
Consent to Participate
Written informed consent was waived for all participants due to the retrospective nature of the study.
Author Contributions
T.N.C. conceived and designed the study. N.D.X. conducted data cleaning, analysis and data visualization. N.D.X. and L.Q.H. drafted the original manuscript. L.Q.H., K.Y.L. and T.N.C. edited and reviewed the manuscript. All authors critically revised and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The publication cost is supported by SingHealth Polyclinics - Centre Grant (CG21APR3006 (NMRC/CG3/001/2022-SHP).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analysed during the current study are not publicly available due the institution’s policy but are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.