Abstract
Prostate-specific antigen (PSA) screening has played a critical role in reducing prostate cancer mortality but remains controversial due to concerns regarding overdiagnosis, overtreatment, and inconsistent screening guidelines. In 2012, the United States Preventive Services Task Force (USPSTF) issued a grade D recommendation against PSA screening, leading to a national decline in PSA testing and prostate biopsy rates. This was followed by an increase in the incidence of metastatic prostate cancer at diagnosis. The USPSTF has since revised its recommendation to grade C, emphasizing shared decision-making for men aged 55 to 69 years. However, variability in screening recommendations across professional societies has resulted in clinical uncertainty, particularly for primary care physicians (PCPs), who frequently initiate PSA testing and urology referrals. This review provides an evidence-based overview of PSA screening tailored for the PCP. We performed a comprehensive literature review using Ovid MEDLINE, PubMed, Embase, and the Cochrane Library, including systematic reviews published between January 2020 and April 2025, and current guidelines from the American Urological Association (AUA), National Comprehensive Cancer Network (NCCN), European Association of Urology (EAU), and American Cancer Society (ACS). We summarize the evolving landscape of PSA screening and its integration with contemporary tools such as shared decision-making, multiparametric magnetic resonance imaging (mpMRI), and transperineally targeted biopsy. We advocate for initiating PSA screening between the ages of 45 and 49 in men with average or elevated risk, based on long-term evidence demonstrating the value of an early baseline PSA level and risk-stratified follow-up. This approach, coupled with improved diagnostic tools, can help PCPs navigate guideline variability and implement a rational, common-sense strategy to maximize early detection while minimizing harm.
Introduction
Prostate cancer is the most common non-cutaneous cancer among men in the United States and remains the second leading cause of cancer-related mortality.1,2 Prostate-specific antigen (PSA) testing was introduced in the late 1980s and was widely adopted for the early detection of prostate cancer.
While PSA screening led to a substantial decline in prostate cancer mortality in the 1990s and early 2000s, its widespread use also resulted in overdiagnosis and overtreatment of indolent disease. 3 Although population-level trends suggested a mortality benefit, these data were largely observational and confounded by contemporaneous advances in therapy.
These concerns, together with inconsistent trial evidence, culminated in the 2012 recommendation from the United States Preventive Services Task Force (USPSTF) against PSA-based screening, citing limited mortality benefit in randomized trials and substantial harms related to overdiagnosis, biopsy complications, and overtreatment. 4 Following the 2012 grade D recommendation by the U.S. Preventive Services Task Force, prostate-specific antigen screening and prostate biopsy rates declined across the United States. 5 And in the subsequent years, the incidence of metastatic prostate cancer at diagnosis increased significantly. 6 While these data are observational and do not prove causality, several large-scale and population-based analyses show that lower screening intensity coincides temporally with higher rates of advanced disease.7,8 Since then, perspectives on PSA screening have evolved. In 2018, the USPSTF updated its recommendation to grade C for men aged 55 to 69 years, encouraging individualized decision-making between patients and their physicians based on personal values and risk profiles. 9 This shift reflects growing recognition of the benefits of early detection when combined with contemporary strategies that minimize harm. Major guideline organizations now support screening in well-informed men with sufficient life expectancy, although the starting age, screening intervals, and definitions of elevated PSA vary across societies.10-13
The current landscape of prostate cancer screening has become increasingly complex. Primary care physicians (PCPs), often responsible for initiating PSA screening, must navigate a web of differing recommendations while ensuring patients receive individualized, evidence-based care. The decision to screen is no longer binary but instead involves consideration of a patient’s age, race, family history, comorbidities, baseline PSA level, and personal preferences. In addition, newer diagnostic tools such as PSA kinetics, PSA density, genetic biomarkers, multiparametric magnetic resonance imaging (mpMRI), and targeted transperineal biopsy can refine risk assessment and reduce unnecessary procedures.14-17 The advanced diagnostic modalities discussed are presented to illustrate the evolving tools available within contemporary urologic practice rather than to suggest their implementation in the primary care setting. Their inclusion serves to contextualize the continuum of prostate cancer evaluation, enhance primary care physicians’ understanding of the diagnostic pathways that may follow referral, and highlight how newer modalities reduce the risk of overtreatment and mitigate harms from PSA screening, thereby facilitating more informed counseling and shared decision making.
Despite ongoing debate, there is growing consensus that PSA screening, when used judiciously, can reduce prostate cancer-specific mortality while limiting overdiagnosis. However, variability in implementation across practices and specialties persists. This review aims to clarify the evidence and offer a practical, common-sense approach to PSA screening for PCPs, incorporating the latest research, guideline updates, and diagnostic tools.
Materials and Methods
A comprehensive search was performed using PubMed, Ovid MEDLINE, Embase, and the Cochrane Library for articles published between January 2020 and April 2025. Search terms included combinations of “prostate-specific antigen,” “prostate cancer,” “screening,” “biopsy,” “risk stratification,” “shared decision-making,” and “multiparametric MRI.” Foundational studies and landmark trials published prior to 2020 were also included when relevant. Additional references were identified through manual review of citations within selected articles.
Eligible sources included randomized controlled trials, systematic reviews, meta-analyses, large cohort studies, and professional society guidelines. Emphasis was placed on high-quality evidence addressing the efficacy and harms of PSA screening, diagnostic adjuncts, and clinical implementation. The most recent guidelines from the American Urological Association (AUA), National Comprehensive Cancer Network (NCCN), European Association of Urology (EAU), American Cancer Society (ACS), and USPSTF were included.10-18
Results
Prostate Cancer Diagnosis
PSA Screening
Prostate specific antigen (PSA) is a glycoprotein secreted by the epithelial cells of the prostate gland. Under normal physiologic conditions, PSA remains confined to the prostatic ducts; however, in prostate cancer and other pathologic states, disruption of the basement membrane allows PSA to enter the systemic circulation, leading to elevated serum levels. 19 Serum PSA levels serve as a surrogate marker of prostatic cellular disruption but are not specific for malignancy.
PSA was approved by the United States Food and Drug Administration in 1994 for use alongside digital rectal examination (DRE) in the early detection of prostate cancer. 20 Widespread adoption of PSA screening in the 1990s led to a dramatic shift in stage at diagnosis, with decreased rates of metastatic disease and a corresponding decline in prostate cancer mortality. 21 Prostate cancer mortality fell by 50% between 1992 and 2016, with modeling studies suggesting that PSA screening contributed to 45% to 70% of this reduction in mortality. 22
Despite these benefits, PSA screening has substantial limitations. While highly sensitive, it is not specific for prostate cancer and may be elevated in benign conditions such as benign prostatic hyperplasia, prostatitis, urinary tract infections, ejaculation, or recent instrumentation. 23 Conversely, PSA levels can be artificially lowered by medications such as 5 alpha reductase inhibitors (such as finasteride), which reduce serum PSA by approximately 50% and may obscure the detection of clinically significant prostate cancer if not appropriately adjusted. 24
A critical limitation of PSA is its inability to distinguish between indolent and aggressive tumors. As a result, PSA screening has been associated with high rates of overdiagnosis and overtreatment. Estimates suggest that 20% to 50% of screen detected cancers are unlikely to progress or impact survival. 25 The widespread overtreatment of these low-risk cancers historically led to long term side effects such as urinary incontinence, erectile dysfunction, and bowel dysfunction. 26 Beyond physical complications, the psychological impact of a prostate cancer diagnosis should also be recognized. Large population studies have demonstrated a transient increase in suicide and accidental death shortly after diagnosis, particularly among men with advanced disease, underscoring the importance of balanced counseling and psychosocial support during screening discussions. 27
Adjunctive tools have been used to improve screening accuracy. While DRE remains a traditional companion to PSA, its diagnostic utility is limited. In men with PSA levels below 2.0 ng/ml, the positive predictive value of DRE ranges from 4% to 11%. 28 Schröder et al 29 found that DRE offered minimal additional diagnostic value for PSA levels below 3 ng/ml, calling into question its utility in low-risk scenarios. Nonetheless, professional guidelines, including the AUA and NCCN, recommend DRE as an adjunct to PSA testing rather than as an isolated screening tool, recognizing its potential value in risk assessment and in the evaluation of men with elevated PSA or abnormal findings on examination. Thus, the digital rectal exam is not recommended as a stand-alone method for prostate cancer screening.
Two major randomized trials have defined the global debate on PSA screening. The European Randomized Study of Screening for Prostate Cancer (ERSPC) showed a 20% relative reduction in prostate cancer specific mortality over 13 years with organized screening. 30 This corresponds to a number needed to screen (NNS) of 781 men to save 1 prostate cancer death. 28 With longer follow-up to 16 years, this NNS has further decreased to 570. 31 This compares favorably to the NNS to prevent cancer death in other conditions such as breast cancer (NNS = 1298 for women age 50-59) and colorectal cancer (NNS = 871).32,33 In contrast, the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial reported no mortality benefit. However, the no screening control arm of the PLCO trial experienced significant PSA contamination, with up to 90% of participants undergoing opportunistic PSA testing during the study period, which likely diluted any observed screening effect size. 34
In response to concerns about overdiagnosis and overtreatment, the USPSTF in 2012 issued a grade D recommendation against PSA based screening for all men, concluding that the harms outweighed the benefits. 4 Following this guidance, PSA screening and biopsy rates declined nationally. 5 However, this was soon followed by an increase in the incidence of high risk and metastatic prostate cancer at diagnosis, especially among older men and those with limited access to care.6-8
Recognizing these consequences, the USPSTF revised its recommendations in 2018. For men aged 55 to 69 years, PSA screening now carries a grade C recommendation, indicating that clinicians should engage in shared decision making based on individual risk and patient preferences. For men aged 70 years and older, screening remains discouraged with a grade D recommendation. 9
Discrepancies among PSA screening guidelines have contributed to practice variability and uncertainty for primary care physicians. The American Urological Association recommends PSA testing for men aged 40 to 45 with elevated or average risk, with screening every 2 to 4 years through age 69. Digital rectal examination is recommended as an adjunct to PSA to establish risk. 10 The National Comprehensive Cancer Network supports shared decision making for men aged 45 to 75, with earlier initiation (age 40) in African American men and those with a family history of prostate cancer. 11 The European Association of Urology and American Cancer Society recommend screening starting at age 50 for most men, and at 40 to 45 for those at higher risk.12,13 Large population studies such as the Malmö Preventive Project demonstrate that PSA measured at ages 45 to 49 strongly predicts subsequent metastatic or lethal prostate cancer, while later analyses incorporating kallikrein markers have improved the specificity of PSA-based screening for clinically significant disease.35,36 Establishing a baseline PSA between ages 45 and 49 aligns with contemporary guideline recommendations and is supported by long-term cohort data showing that early mid-life PSA concentrations strongly predict subsequent metastatic and lethal prostate cancer, enabling risk-stratified screening intervals.35,36
The Surveillance, Epidemiology, and End Results Program Cancer Statistics Review from the National Cancer Institute reported that the implementation of PSA screening was associated with a 51% reduction in prostate cancer mortality in the United States between 1992 and 2017. 37 Supporting a more tailored screening approach, a secondary analysis of 10 968 men aged 55 to 60 years from the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial evaluated baseline PSA values in relation to the future risk of clinically significant prostate cancer. The analysis suggested that men in this age group with a baseline PSA level below 2.0 ng/ml could undergo screening less frequently, while those with levels below 1.0 ng/ml might be considered for discontinuation of screening altogether. 38
Collectively, the current consensus acknowledges that the aim of PSA screening is to detect high risk disease early in order to reduce the development of metastatic and lethal prostate cancer, while simultaneously minimizing the detection and treatment of low-risk tumors. This includes reducing unnecessary biopsies, lowering the risk of overtreatment, and preserving quality of life. Personalized, risk adapted screening that integrates age, baseline PSA, race, family history, and life expectancy has emerged as the modern paradigm for balancing benefit and harm in prostate cancer early detection.
Multiparametric MRI
Multiparametric magnetic resonance imaging (mpMRI) has emerged as a valuable tool in the diagnostic pathway for prostate cancer, particularly in men with elevated PSA levels or abnormal digital rectal examination. mpMRI combines anatomical and functional imaging sequences, including T2-weighted imaging, diffusion-weighted imaging, and dynamic contrast-enhanced imaging, to localize and characterize prostate lesions based on the Prostate Imaging Reporting and Data System (PI-RADS) scoring system. 39 Lesions scored PI-RADS 4 or 5 are more likely to represent clinically significant cancer and typically warrant targeted biopsy.
mpMRI is not used as a primary screening tool but rather serves to improve risk stratification in men with elevated PSA, guiding biopsy decisions and minimizing unnecessary procedures. Compared to traditional transrectal ultrasound-guided (TRUS) systematic biopsy, mpMRI offers higher sensitivity for detecting clinically significant prostate cancer and a higher negative predictive value, which helps reduce the detection of indolent, low-risk disease. 40 In the PROMIS trial, the use of mpMRI followed by targeted biopsy demonstrated a sensitivity of 93% for clinically significant prostate cancer, compared to 48% for TRUS biopsy alone. 41 Importantly, up to 27% of men with a negative mpMRI in the PROMIS trial could have safely avoided biopsy, highlighting its utility in reducing unnecessary interventions.
The PRECISION trial further validated the role of mpMRI in biopsy-naïve men. In this multicenter randomized trial, men undergoing mpMRI followed by targeted biopsy had a significantly higher rate of clinically significant cancer detection (38% vs 26%) and a lower rate of clinically insignificant cancer detection (9% vs 22%) compared to those undergoing systematic TRUS biopsy alone. 42 These results support the use of mpMRI to both improve diagnostic accuracy and reduce overdiagnosis.
Professional guidelines increasingly endorse mpMRI as part of the diagnostic workup. The American Urological Association and the National Comprehensive Cancer Network recommend mpMRI prior to initial or repeat biopsy in men with elevated PSA.10,11 The European Association of Urology further emphasizes its role in biopsy decision-making, particularly in men with prior negative biopsy or clinical suspicion despite normal findings. 12 In clinical practice, mpMRI is most effective when interpreted by radiologists experienced in prostate imaging and when used in conjunction with targeted biopsy platforms.
Despite its benefits, mpMRI is not without limitations. Image quality and interpretation can vary across institutions, and access may be limited in certain settings due to cost and availability. 43 Moreover, while a negative mpMRI significantly reduces the likelihood of clinically significant cancer, it does not eliminate the risk entirely. However, factors, such as PSA density (PSA/prostate volume in mL), can help to additionally risk stratify the need for biopsy, with data showing a very low 2.4% risk of missing clinically significant cancer in patients with a PSA density < 0.15 and an MRI without visible lesions. 44 Therefore, mpMRI findings must be interpreted within the context of the patient’s clinical risk factors, including PSA kinetics, family history, race, and prior biopsy history.
Transrectal and Transperineal Biopsies
Prostate biopsy remains the definitive method for diagnosing prostate cancer following abnormal PSA levels or suspicious imaging findings. Two main approaches are used in clinical practice: transrectal ultrasound guided (TRUS) biopsy and transperineal (TP) biopsy.
Transrectal biopsy involves advancing a biopsy needle through the rectal wall under ultrasound guidance to sample the posterior prostate. It has been the most widely used method due to its procedural familiarity and convenience. However, TRUS biopsy carries limitations, including suboptimal sampling of the anterior and apical zones and a risk of infection. Because the needle traverses the rectal mucosa, there is potential exposure to rectal flora, which can result in urinary tract infections or, less commonly, sepsis. Reported post-TRUS biopsy sepsis rates range from 0.5% to 1.5%, even when prophylactic antibiotics are administered. 45
Transperineal biopsy, by contrast, involves percutaneous access to the prostate through the perineum and avoids transgression of the rectal mucosa. While this approach reduces the risk of infection, recent studies suggest that infection rates may not differ significantly between TRUS and TP approaches when effective antibiotic prophylaxis is employed prior to TRUS biopsy. However, TP biopsy does not require any antibiotic prophylaxis and in large trials has reduced the risk of sepsis to <0.3%. 46 Overall, with modern augmented antibiotic prophylaxis TRUS biopsy, or TP biopsy, both methods have shown comparably low sepsis rates.47,48
An additional advantage of the transperineal approach is its improved access to the anterior and apical regions of the prostate, which are sometimes under sampled using the transrectal route. This enhanced anatomical coverage may improve detection of clinically significant cancers, especially in patients with anteriorly located lesions, rising PSA after negative biopsy, or suspicious findings on MRI. 49 TP biopsy is also increasingly adopted in MRI targeted strategies and is well suited for integration with fusion or in bore guidance platforms.
Regardless of approach, biopsy specimens are graded using the Gleason scoring system, which evaluates glandular architecture. The 2 most prevalent histologic patterns are summed to yield a Gleason score ranging from 6 to 10. Gleason 6 denotes low grade cancer, Gleason 7 represents intermediate risk disease, and scores from 8 to 10 indicate high grade, aggressive tumors. This system is central to risk stratification and treatment planning. 50
Prostate Cancer Treatment
Active Surveillance
Active surveillance is a conservative management strategy for men with low-risk prostate cancer. Treatment is deferred while patients are monitored through periodic prostate-specific antigen (PSA) tests, digital rectal examinations, multiparametric magnetic resonance imaging, and repeat biopsies as indicated. This approach aims to avoid overtreatment in men whose cancers are unlikely to progress during their lifetimes and to preserve quality of life.
Commonly, active surveillance is offered to men with Grade Group 1 (Gleason score 6) disease, PSA below 10 ng/ml, and clinical stage T1c or T2a. Long-term data from the Prostate Cancer Research International Active Surveillance (PRIAS) cohort showed prostate cancer–specific survival at 10 years for men meeting these criteria, with very low rates of metastasis. 51 Such outcomes support the safety of active surveillance when used in the appropriate clinical setting. Figure 1
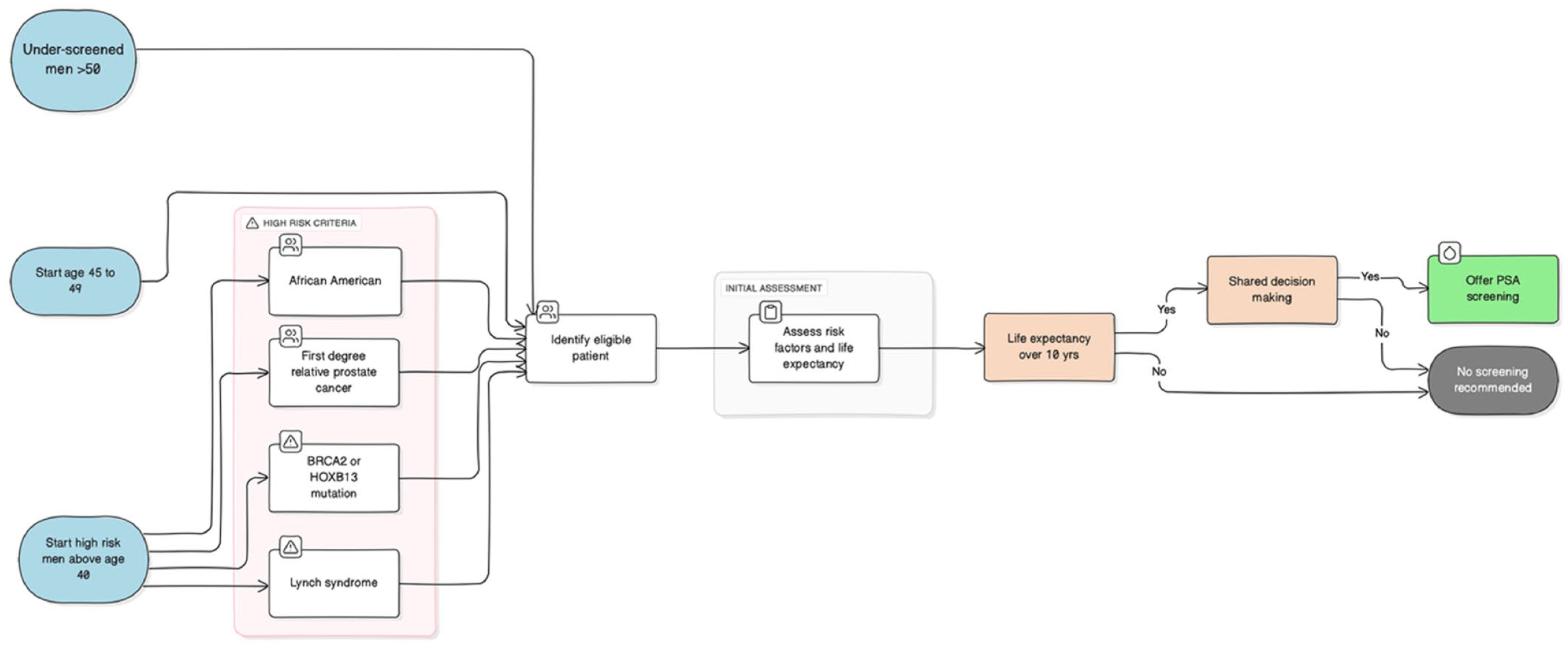
PSA screening algorithm: identifying high-risk and eligible men.
The ProtecT trial provides further high-quality evidence. In this randomized study of 1643 men aged 50 to 69 with localized prostate cancer, participants were assigned to active monitoring, radical prostatectomy, or radiation therapy. At 10 years, prostate cancer–specific mortality was low across all groups (1.5%, 0.9%, and 0.7%, respectively), with no significant difference in all-cause mortality despite a small increase in metastases and disease progression in the active monitoring arm. 52 This finding underscores that delayed intervention through surveillance does not compromise survival, while potentially sparing men from immediate treatment side effects.
Active surveillance significantly preserves quality of life by avoiding or delaying treatment-related harms. The ProtecT trial’s patient-reported outcomes after 12 years demonstrated better urinary and sexual function in men on active monitoring than in those receiving immediate surgery or radiation. 53 Moreover, surveillance may be appropriate for select men with favorable intermediate-risk cancer (Grade Group 2, Gleason 3 + 4) when disease volume is low and close follow-up is feasible.
It is important to distinguish active surveillance from watchful waiting, as these strategies differ significantly in intent, intensity, and clinical application. Active surveillance is a curative-intent approach used primarily in younger, healthier men with low-risk or favorable intermediate-risk disease. It involves structured monitoring with serial PSA testing, digital rectal examinations, multiparametric MRI, and periodic biopsy to detect signs of progression in time to offer curative treatment if needed.51,52
In contrast, watchful waiting is a palliative strategy intended for older men or those with limited life expectancy who are unlikely to benefit from definitive therapy. Rather than actively monitoring for disease progression, this approach delays intervention until the development of symptoms from metastatic or locally advanced disease, at which point systemic therapies such as androgen deprivation therapy may be initiated for symptom control. 54 Unlike active surveillance, watchful waiting does not include regular biopsies or imaging, and it does not offer the possibility of cure. For primary care physicians, understanding this distinction is critical to ensure that patients are referred for the appropriate management pathway based on age, comorbidities, cancer risk, and personal goals. 55
Surgery
Radical prostatectomy is a definitive treatment option for localized prostate cancer, particularly in patients with intermediate- or high-risk disease who have a life expectancy of 10 years or more. 54 It is typically offered to men who are good surgical candidates and prefer active treatment over surveillance or radiation. The procedure involves removal of the entire prostate gland and seminal vesicles, often with pelvic lymph node dissection in patients with elevated risk of nodal involvement (Figure 2).

Clinical workflow for PSA testing and urology referral.
Most radical prostatectomies in the United States are now performed using a robot-assisted laparoscopic approach. Compared to open surgery, robotic-assisted techniques are associated with less blood loss, reduced hospital stays, and faster recovery, though long-term oncologic outcomes appear similar. 49 Surgical management allows for accurate pathologic staging, which can help inform prognosis and decisions regarding adjuvant or salvage therapies. In particular, information such as Gleason grade, margin status, extra-prostatic extension, and lymph node involvement is critical in guiding postoperative care. 56 In patients with adverse features, early salvage radiation therapy may offer improved long-term outcomes. 57
The long-term cancer-specific survival benefit of surgery has been demonstrated in randomized trials. The Scandinavian Prostate Cancer Group Study Number 4 (SPCG-4) reported improved overall and prostate cancer specific survival and reduced metastasis among men undergoing prostatectomy compared to watchful waiting, particularly among younger patients and those with high-grade disease. 58
Functional outcomes remain a key consideration. Urinary incontinence and erectile dysfunction are the most common adverse effects. While most men regain urinary control within a year, persistent leakage may occur in some cases. Erectile function recovery varies based on preoperative status, patient age, and the ability to preserve the neurovascular bundles. Nerve-sparing prostatectomy techniques can improve recovery, but outcomes remain variable. 59
Radiation
Radiation therapy is a curative option for localized prostate cancer and an alternative to surgery in men with low-, intermediate-, or selected high-risk disease. 58 It is particularly appropriate for patients who prefer non-surgical management or are poor surgical candidates due to age or comorbidities.
There are 2 primary forms of radiation therapy: external beam radiation therapy (EBRT) and brachytherapy. EBRT involves the use of high-energy X-rays targeted at the prostate over multiple treatment sessions, typically over 4 to 8 weeks. Technological advances such as intensity-modulated radiation therapy (IMRT) and image-guided radiation therapy (IGRT) have improved the precision and safety of EBRT. 60 Brachytherapy delivers radiation directly into the prostate via radioactive seeds (low-dose rate) or temporary catheters (high-dose rate), offering high local control in selected patients. 61
Side effect profiles differ between radiation and surgery. Radiation therapy is more commonly associated with bowel and urinary irritative symptoms, such as frequency, urgency, and proctitis, which typically improve over time. 62 Erectile dysfunction is also common and may develop more gradually compared to post-surgical cases. The risk of late complications such as hematuria, rectal bleeding, or strictures exists, particularly in high-dose treatments, but can be minimized with modern planning techniques. 60
Androgen deprivation therapy (ADT) is often used in combination with radiation for men with unfavorable intermediate- or high-risk prostate cancer, as it improves biochemical control and survival. 63 ADT is typically administered for 6 months to 3 years depending on risk stratification, but its side effects, including fatigue, hot flashes, bone loss, and metabolic changes, should be discussed with patients prior to initiation.
Focal Therapy
Focal therapy represents an emerging treatment modality for localized prostate cancer aimed at eradicating the dominant tumor focus while preserving uninvolved prostate tissue to minimize side effects. 64 This approach is typically considered in men with intermediate-risk prostate cancer who have unifocal or unilateral disease identified on imaging and targeted biopsy.
Several ablative technologies are used in focal therapy, including high-intensity focused ultrasound (HIFU), cryotherapy, irreversible electroporation, photodynamic therapy, and focal laser ablation. 65 Among these, HIFU and cryotherapy are the most studied and implemented in clinical practice. These techniques induce cellular destruction via mechanical or thermal energy, targeting the cancerous area under image guidance while sparing surrounding structures such as the neurovascular bundles, urethra, and rectum. 66
Early results from observational studies and phase II trials suggest that focal therapy offers favorable intermediate-term (5-8 years) outcomes in terms of cancer control, with reduced rates of erectile dysfunction, incontinence, and bowel dysfunction compared to whole-gland treatments. 67 However, long-term oncologic outcomes remain uncertain, and robust comparative data with surgery or radiation are lacking.
One of the major challenges with focal therapy is accurate disease localization and patient selection. Multiparametric MRI, combined with targeted biopsy, plays a crucial role in identifying appropriate candidates and defining the treatment zone.39,42,68 Despite advancements in imaging, multifocality remains common in prostate cancer, and there is a risk of under-treatment if significant lesions are missed.
Professional guidelines currently consider focal therapy investigational. The European Association of Urology acknowledges its potential but emphasizes the need for long-term outcome data before recommending routine use. 12 Similarly, the American Urological Association does not currently include focal therapy in its guideline recommendations for standard management of localized prostate cancer. 10
Recommendation to Synchronize Guidelines
Despite broad agreement that PSA screening can reduce prostate cancer mortality when applied judiciously, major professional societies continue to differ in their specific recommendations. The USPSTF recommends shared decision-making for men aged 55 to 69 years (Grade C) and advises against routine screening in those aged 70 and older (Grade D). 9 The American Urological Association (AUA) suggests initiating screening at age 40 to 45 for men at average or increased risk, with biennial or quadrennial testing until age 69. 10 The National Comprehensive Cancer Network (NCCN) recommends shared decision-making from age 45 to 75, with earlier screening in high-risk individuals, such as African American men or those with a strong family history. 11 The European Association of Urology (EAU) and American Cancer Society (ACS) propose beginning screening at age 50 for most men and earlier in those with elevated risk.12,13
The variability among major professional societies has led to inconsistent screening practices, particularly in the primary care setting where clinicians often must reconcile competing guidelines in the context of time constraints and limited resources. As a result, many men at risk for clinically significant prostate cancer may either be screened too late or not at all, while others undergo unnecessary testing and biopsies.
To address this gap, we advocate for a harmonized, risk-adjusted screening framework. Specifically, we recommend establishing a baseline PSA between ages 45 and 49 in men with average or elevated risk. This initial value can help tailor future screening frequency and intensity. Screening should continue only in men with a life expectancy greater than 10 years and may be discontinued by age 70 through a shared decision-making process. In the absence of concerning PSA trends, screening is not recommended beyond age 75. In this manuscript, the term older men refers to individuals aged 70 years and above or those with a life expectancy under 10 years, consistent with recommendations from the USPSTF and AUA.9,10 This definition aligns with contemporary guideline thresholds for discontinuing routine PSA screening while emphasizing individualized decision making in men with excellent health and longer life expectancy.
We also recommend that any elevated PSA level be confirmed with a repeat test before proceeding with further diagnostic evaluation for possible prostate cancer. In parallel, clinicians should perform a history, examination, and urinalysis to assess for possible confounding factors which elevate PSA such as urinary tract infection, urinary retention, acute or chronic prostatitis, benign prostatic hypertrophy, or inflammation. While ejaculation, bicycle riding, long car rides, and digital rectal examination can cause transient elevation of PSA, it is rarely clinically significant and should not preclude initial screening. 69 Nevertheless, it is prudent to counsel patients to avoid these activities for at least 72 h prior to repeat confirmatory PSA testing. If the confirmatory PSA returns to the patient’s previous baseline or to a normal value, we recommend a shorter interval recheck at 6 months to ensure appropriate PSA monitoring.
It is important to distinguish PSA screening as discussed from diagnostic PSA testing. Diagnostic PSA testing is warranted for any male with lower urinary tract symptoms (obstructive or irritative), frequent urinary tract infections, hematuria, hematospermia, an abnormal DRE, or unexplained systemic symptoms including bony pain and weight loss. Likewise, PCPs should recall that 5-alpha reductase inhibitors, commonly prescribed for benign prostatic hypertrophy, reduce PSA values by approximately 50% and the upper limit of normal should be adjusted accordingly. 24
In summary, synchronizing PSA screening recommendations across societies and applying a common-sense, individualized approach can help reduce unnecessary testing, improve early detection of high-risk disease, and reduce confusion among primary care physicians. A uniform strategy that integrates clinical context, baseline PSA, and patient preferences will improve consistency and optimize outcomes.
Conclusions
Prostate specific antigen (PSA) screening remains a critical yet complex component of early prostate cancer detection and management. Recent evidence and updated guidelines have reaffirmed the life saving value of judicious screening. Early concerns over overtreatment have been mitigated by the broader adoption of active surveillance for low-risk disease, with recent trends showing more appropriate risk stratification and reduced overtreatment. It is also important to distinguish between dying with prostate cancer and dying from prostate cancer. Many screen detected cancers represent indolent lesions that would never have affected survival, whereas prostate cancer specific mortality reflects the smaller subset of biologically aggressive tumors responsible for most deaths. This distinction reinforces that the primary objective of screening is to prevent death from prostate cancer, not merely to increase detection rates. However, variability among national and international guidelines continues to create uncertainty in clinical practice.
A harmonized, risk adapted framework is needed, 1 that integrates modern diagnostic tools and emphasizes shared decision making as a central element of patient centered care. In this context, shared decision making represents a structured, evidence informed dialog in which clinicians explain the potential benefits and harms of screening, consider patient values and health priorities, and reach a joint decision about testing. This approach, endorsed by both the USPSTF and AUA, ensures that screening is aligned with individual risk and life expectancy rather than applied uniformly. Within this framework, we advocate for initiating PSA screening between the ages of 45 and 49 in men with average or elevated risk, guided by baseline PSA levels and individualized follow up strategies.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
