Abstract
Background:
Adult immunisation is a critical yet underutilised public health strategy in India, despite the growing burden of vaccine-preventable diseases (VPDs) among adults. However, previous studies have explored awareness, knowledge, and acceptance of selected adult vaccines, a comprehensive understanding of utilisation patterns and vaccine-seeking behaviour remains limited.
Methodology:
A prospective observational study was conducted from April 2023 to February 2024 (10 months) at the Adult Immunisation and Travel Medicine Centre of a tertiary hospital. Adults aged ≥16 years who received at least 1 recommended vaccine were included, focusing on those outside the age group covered by the national immunisation programme. Data on demographics, immunisation status, comorbidities, and adverse events following immunisation (AEFIs) were collected. Statistical associations were analysed using the chi-square test (P < .05) and logistical regression.
Results:
A total of 1021 vaccine doses were utilised by 819 individuals. The most commonly utilised vaccines were Hepatitis B (62.03%), Influenza (10.99%), and Pneumococcal Conjugate Vaccine (PCV; 6.72%). The majority of recipients were female (58.97%), young adults (70.7%), healthcare workers (HCWs; 70.69%), and individuals from higher socio-economic classes (61.91%). Vaccine utilisation was 2.4 times higher among HCWs than non-HCWs. Partial immunisation was more common among young males (P < .0001) from non-HCWs (47.92%, 0.03) populations and those from lower-middle (67.93%, P < .0001) socio-economic classes. Only 1 serious Adverse Event (AE) was reported, later found to be unrelated with vaccination. Chi-square analysis revealed statistically significant associations between vaccine utilisation and variables such as gender, age, population type, socio-economic status, and comorbidities (P < .05).
Conclusion:
Similar to global and national trends, adult vaccine coverage remained sub-optimal. However, among the limited users, only the economically advantaged sections of the community were able to afford and access immunisation, while vaccine-seeking behaviour was largely absent across wider segments of society.
Keywords
Introduction
India is recognised as one of the leading lower-middle-income countries with a strong track record in implementing and expanding childhood vaccination programmes. 1 Over the years, India has introduced several key immunisation initiatives, such as the Expanded Programme of Immunisation in 1978, the Universal Immunisation Programme (UIP) in 1985, and Mission Indradhanush in 2014. These programmes have significantly reduced the morbidity and mortality rates associated with Vaccine Preventable Diseases (VPDs). 2 Despite the well documented benefits of immunisation, adult vaccinations remain relatively neglected among Indian population. As per World Health Organisation’s (WHO) global vaccine market report in 2019, estimated 5.5 million vaccine doses (excluding those used for travel and military purpose) are targeted towards the paediatric poulation. 3 This indicates a similarly underutilised profile of adult vaccines globally, regardless of a country’s development status.
The concept of ‘Healthy ageing’ prompted by many developed nations, encompasses a trio of healthy diet, regular exercise, and timely vaccinations. 4 However, inadequate vaccine-literacy and increasing vaccine-hesitancy have negatively affected the targeted immunisation coverage among adults. Vaccine refusal and hesitancy are influenced by a complex mix of social, cultural, political, financial, and personal factors.5,6 In addition to the financial burdens encountered by lower and middle-income countries, there is also a low demand for adult vaccines in many developed countries. 7 Despite, various legitimate efforts by stakeholders, such as mandating vaccinations by law, these measures have often been perceived as infringement on individual’s rights. 6
Currently, immunisation efforts primarily focus on vulnerable groups such as children, pregnant women, individuals in high-risk occupation (including health-care professionals) and travellers. In absence of government-led initiatives specifically targeting the general adult population, access to adult vaccines remains largely confined to private healthcare facilities.There are no specific vaccine recommendations targeting the general adult population, as they are often considered resilient to infectious diseases. 8 However, studies have shown that adults face significant health risks due to factors such as waning immunity, age-related issues, the risk of immunosenescence, and vulnerability during outbreaks.9,10 Additionally, adults can serve as carriers of diseases, indirectly contributing to delayed herd-immunity. Although adult immunisation is not a new concept and recommended vaccines (as listed in Supplemental Table 2) have existed for decades, they remain largely inaccessible and underutilised. The recent COVID-19 pandemic and the impact of mass vaccination highlight the critical need of adult immunisation. 11
Despite the favourable environment created by the pandemic, maintaining a continuous system for adult immunisation presents several challenges. These include limited infrastructure for vaccine research, development, and implementation; the complexity of adult-vaccine schedules; lack of data on VPDs among adults, greater emphasis on children’s health; economic instability; challenges in decision-making and increased vaccine-hesitancy.2,12,13 Studies have shown that 12% to 30% of Indian women of reproductive age are susceptible to rubella infection,2,14 and there has been a 62% increase in Hepatitis A infections among adolescents and adults. 15 Additionally, the case fatality rate for influenza subtype infections (H1N1) in the country is estimated at 15.02%. 16 Overcoming the socio-economic burden imposed by these VPDs requires a comprehensive catch-up strategy.
Adult immunisation has become an undeniable necessity, and urgent action is required to boost the acceptance and utilisation of vaccines. Beyond raising vaccine awareness and enhancing vaccine literacy, it is equally important to understand the existing gaps and behaviours surrounding immunisation. This study objective is to explore real-world trends in adult vaccine utilisation and evaluates the safety profiles of administered vaccines through active surveillance. Additionally, it provides insight into vaccine-seeking behaviour across various population groups who accessed the study site, highlighting the disparities in reach and utilisation.
Methodology
The study was conducted prospectively over ten months at the ‘Adult immunisation and Travel Medicine Centre’ a facility exclusively dedicated to immunising the adult population. Located within a tertiary care teaching hospital, this new centre was launched on 26th April 2023, under the department of General Medicine. A trained nurse and a general practitioner (GP) specialised in immunisation were in-charge of the centre. The study included all vaccine beneficiaries immunised at the centre during 10 months (26th April to 26th February, 2024) regardless of co-morbidities. Children aged 16 years and below were excluded, as they were covered by the childhood immunisation programme according to the National Immunisation Schedule (NIS). 17 All eligible study participants were enrolled after providing informed consent, designed in accordance with the Indian Council of Medical Research guidelines for biomedical research on human subjects. This study was reviewed and approved (JSSMC/IEC/240921/01NCT/2021-22) by the ethical committee of study site.
The study included all vaccine recipients, encompassing individuals from adolscents (17 years) through to geriatric (60 years and above) age groups. Participants voluntarily sought immunisation and visited the adult immunisation centre for any vaccine. The name and type of vaccines were recorded in the patient’s medical file, which was then verified by the GP incharge at the immunisation centre. Additionally, patients had the option to consult directly with the GP at the immunisation centre before receiving the vaccination. A general check-up, including their vital signs and past medical history was documented in the file. Eligibility for vaccination was determined based on the individual’s pervious vaccination status, medical history, and medication history (such as allergy, contraindication, and immune-compromised conditions). The demographic details and medical history of each vaccine-beneficiary were also entered into the digital immunisation registry maintained at the study site.
All information related to vaccination and vaccine beneficiaries was collected at the time of enrollment. A receptionist with basic medical training managed the Digital Immunisation Registry, which was integrated with the hospital information system. Prior medical history available in the hospital database was automatically updated in the registry during each vaccination visit. Additional information, such as current medications and past medical history, was obtained directly from the vaccine recipients. Weekly data were extracted in Excel format for detailed analysis and used for vaccine safety surveillance. Any reported AEFIs were thoroughly investigated, categorised, and documented to ensure continuous safety monitoring. As this was an observational study involving all voluntary adult vaccine recipients over a 10-month period, no a priori power calculation was performed. The sample size was based on the total number of eligible individuals who received at least 1 adult vaccine during the study period.
Following immunisation, all vaccine-beneficiaries were required to remain at the centre for the first 30 min to monitor for any acute reactions. In case of an emergency, on-call medical services were available at the centre. Most recommended adult vaccines were readily available at the study site, while a few, such as yellow fever and monkey fever vaccines, required prior booking to ensure availability. Except for Tetanus vaccines administrated before surgery (administrated in respective wards), all other vaccination details were collected from the newly established immunisation-centre.
Active vaccine safety surveillance was conducted with the assistance of Adverse Drug Reaction Monitoring Centre (AMC) as part of the Pharmacovigilance Programme of India (PvPI). In the event of any severe or Serious Adverse Events (SAEs) occurring following vaccination, the vaccinees were informed to report back or notify the GP. Additionally, the surveillance team at the study site proactively contacted each vaccine beneficiary by telephone 10 ± 2 days of post-immunisation. All AEs reported by the vaccine beneficiaries were meticulously documented by the surveillance team using a specially designed data collection form. This form included provisions to document demographic details, past medical history, co-morbidities, allergies, and vaccination specifies such as the date, type, and dose of the vaccine. Adverse Event Following Immunisation (AEFI) were reported in accordance WHO’s core variables, which included details like the onset date and time of AEFI, event description, severity, management, outcome, and cessation date and time of AEFI.
The AEFI investigation team at the study site conducted thorough investigations into each reported AEFI. In the case of any SAE, medical records and relevant information pertaining to the event were collected. The AEFI investigation team was composed of 2 senior physicians, a clinical pharmacist, and a pharmacologist with an interest in the vaccine safety. Valid diagnoses of the reported AEFIs were classified according to Brighton collaborations case definitions and relevant literature reviews. The WHO’s new causality assessment algorithm was used to assess the causality of each reported event, ensuring eligibility through a checklist and algorithm, and categorising them based on the causality assessment classification. 18 Vaccine safety surveillance was specifically incorporated as part of this study, while all other procedures reflect the routine practices followed at the Adult Immunisation Centre.
The study population was categorised by age into Adolescents (17-19 years), Young adults (20-34 years), early middle-aged adults (35-44 years), late middle-aged adults (45-59 years), and old aged (60 years and above). Vaccine utilisation was analysed across these age groups to evaluate variations in immunisation patterns. Several factors influencing vaccine utilisation was examined, including socio-economic status (assessed using the Modified Kuppuswamy scale 2022 19 ), age, Cost of the vaccine, vaccine safety, Education and occupation (Health-Care Workers (HCWs), and Non-HCWs). Additionally, immunisation status was categorised as partial or full based on the vaccine utilisation. Individuals eligible for vaccines who did not receive their next due dose were considered as partially immunised, whereas those who completed all recommended doses were considered fully immunised. However, individuals still within the dosage interval for their next scheduled dose were not classified as partially immunised.
The cost of each vaccine administered at the study site is provided in the Supplemental Table to highlight the financial burden associated with adult immunisation. The table also includes details on the recommended doses, vaccine brands used, and their contents, offering a comprehensive overview of the current adult immunisation recommendations in the country.
Statistical Analysis
Statistical analyses were carried out using IBM SPSS Statistics, version 22.0 (IBM Corp., Armonk, NY, USA; licensed to JSS University). Univariate analysis was conducted using descriptive statistics (frequencies and percentages) to summarise vaccine utilisation patterns. Bivariate analysis was performed using the Chi-square (χ²) test to examine associations between categorical variables such as gender, age group, type of population (HCWs vs non-HCWs), socio-economic class, immunisation status, and comorbidities with the utilisation of different vaccines. Logistic regression analysis was further employed to identify demographic and clinical factors independently associated with partial vaccination. The distribution of data was found to be non-uniform, with significant variations observed across multiple demographic characteristics. A P-value of <.05 was considered statistically significant.
Results
During the study period, a total of 40 268 eligible individuals visited the out-patient department for various medical consultations and treatments. Among them 819 adults (2.03%), including adolescents, received a total of 1018 vaccine doses from the Adult Immunisation Centre at the study site. Overall, vaccine utilisation was higher among females (58.97%, n = 483) compared to males (41.03%, n = 336). The majority of vaccines (70.7%, n = 579) were administrated to young adults, while adolescents had the lowest utilisation rate (1.71%, n = 14). Vaccine utilisation was notably higher among HCWs (70.69%, n = 579) than non-HCWs (29.30%, n = 240). Among the HCWs, 62.35% (n = 361/579) belonged to the young adult age group. Figure 1 presents a detailed age-wise demographic distribution of the study population who received adult vaccines.

Demographic distribution of adult vaccine recipients at the study site.
Trends and Patterns in Adult Vaccine Utilisation
Among 819 vaccine recipients, the most commonly utilised vaccine was Hepatitis B, accounting for 62.03% of all doses, while Hepatitis A was the least utilised at 0.12%. The majority of Hepatitis B vaccine recipients were females (58.07%), primarily young adults (84.25%), HCWs (70.67%), and individuals belonging to the upper-middle socioeconomic class (39.57%). Hepatitis B was the most frequently utilised vaccine among both HCWs (70.67%) and non-HCWs (29.33%) during the study period. In addition to Hepatitis B, influenza vaccine with a utilisation rate of 34.44% was the second most commonly used adult vaccine among non-HCWs. A detailed breakdown of vaccine utilisation trends within the study population is shown in Table 1.
Detailed Breakdown of Adult Vaccine Beneficiaries and the Association Between Vaccine Utilisation With Demographic Factors.

Overall, only 10.74% of the old age population among in the study received adult vaccination. Among this group, the most commonly administrated vaccines were influenza (54.54%), followed by PCV (34.09%). Notably, utilisation of other vaccines among the old-age was minimal, with Hepatitis B and Tdap accounting for only 6.81% and 4.54% respectively. The majority of vaccine recipients belonged to upper-middle-class families, while the lowest utilisation was observed among individuals from the lower socio-economic class (1.34%). Among this group, only 2 vaccines were utilised: Tdap (n = 9) and rabies (n = 2) vaccines. In contrast, vaccines such as HPV, influenza, and PCV were predominantly utilised by individuals in the upper 3 socio-economic classes, with very limited utilisation among lower-income groups.
A low utilisation of the Hepatitis B vaccine was observed among individuals with comorbid conditions, accounting for only 1.97% (n = 10/508) of such recipients in the study population. In contrast, beneficiaries of the influenza vaccine (40%, n = 36/90) and PCV (63.63%, n = 35/55) were highly composed with comorbid conditions rather than other vaccine utilisers. Overall 4.4% of the vaccine beneficiaries with comorbid conditions had pre-existing respiratory diseases (Refer Table 1).
Partial Vaccination Coverage Among Vaccine Beneficiaries
Overall, 68.25% (n = 559/819) of the vaccine beneficiaries completed full recommended doses for each vaccine as per guidelines, while the remaining 30.89% (n = 253) had a partial immunisation status. Both Rabies and Tdap vaccine beneficiaries completed the full recommended doses. In contrast, a high proportion of partial immunisation was observed among Hepatitis B vaccine beneficiaries accounting 41.93% (n = 213). Notably, 66.20% of them were males, with 60.56% were HCWs and 50.70% belonged to the lower middle socioeconomic class.
Both the influenza vaccine and PCV showed similar rates of partial vaccination among their beneficiaries, at 21.11% and 21.81% respectively. In both cases, females were the most affected group, as they were less likely to complete the full recommended doses—accounting for 12.22% of partial influenza vaccinations and 8.74% for PCV. Similarly, the majority of partially vaccinated individuals for both vaccines were non-HCWs (influenza; 13.04% and PCV; 8.70%) and primarily belongs to the upper-middle and lower-middle classes (refer Figure 2).

Vaccination status across different population categories.
Safety Profile of Vaccines
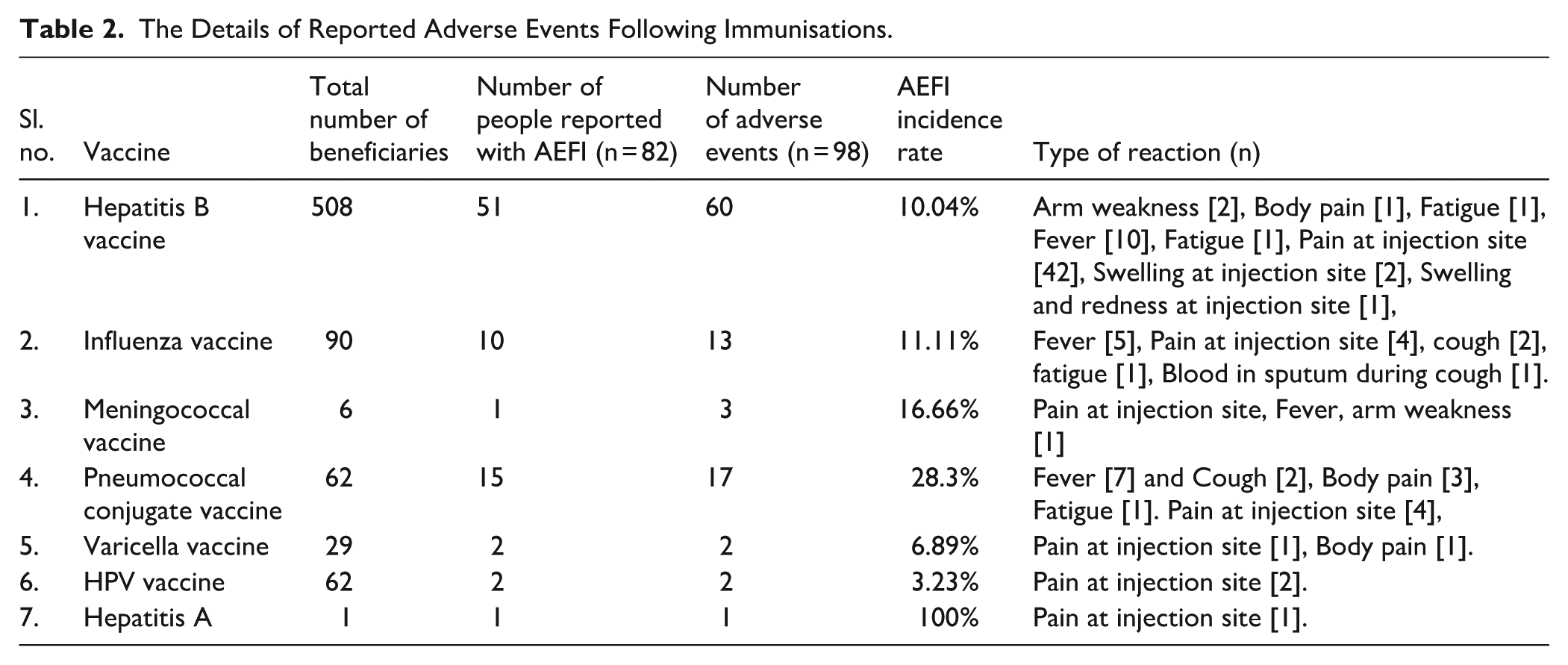
A majority of the study population 84.86% (n = 695/819) responded to the safety surveillance, while 15.14% (n = 124/819) did not respond and were categorised as dropouts. Among the respondents, 82 vaccine beneficiaries reported a total of 98 adverse events, yielding an overall AEFI incidence rate of 10.01%. The highest incidence of AEFI was observed following Hepatitis B vaccination (61.22%), followed by the Pneumococcal vaccine (17.35%) and the influenza vaccine (13.26%). The most commonly reported AE was Pain at injection site (56.12%) followed by fever (24.49%) and body-pain (8.16%). Detailed information on reported AEFIs is presented in Table 2.
The Details of Reported Adverse Events Following Immunisations.
Severity and Causality Assessment of Reported AEFIs
The majority of AEs reported during the study period were ‘mild’ (n = 77 [80.21%]) followed by ‘moderate’ (n = 19 [19.79%]). No deaths or serious adverse events were observed. Most AEs (94.79%) fell under the causality category ‘consistent with causal association to immunisation’ and 5.21% were categorised as ‘inconsistent with causal association to immunisation or coincidental events’. All AEs classified under consistent category were related to the vaccine product and were expected, as documented in the respective fact sheets. The causality assessment of reported AEFIs is shown in Figure 3.

Causality assessment of reported AEFIs as per WHO causality assessment algorithm. A: Consistent with causal association to immunisation, B: Indeterminate, C: Inconsistent with causal association to immunisation.
Factors associated with the utilisation and partial utilisation of adult vaccines
The study identified several factors that may potentially influence vaccine utilisation among the study population. Chi-square test results revealed statistically significant associations between various demographic and socio-economic parameters and vaccination trends, as detailed in Table 1. A gender-based trend in vaccine utilisation patterns was observed. The HPV vaccine was administered exclusively to females (P = .00001), while higher utilisation among females was also noted for vaccines such as Hepatitis B (P = .0007), MMR (P = .02). In contrast, PCV vaccine’s utilisation slightly higher among males, although this difference was not statistically significant (Table1). Overall, adult vaccine utilisation was predominantly among HCWs, with the exception of the Tdap vaccine, which was more frequently utilised by non-HCWs.
Utilisation patterns of all adult vaccines varies significantly across different age group. Young adults demonstrated the highest utilisation for most vaccines, including Hepatitis B, HPV, MMR, rabies, Tdap, and varicella. Notable exceptions to this trend were observed for influenza and PCV vaccines, which showed higher utilisation among older adults. Additionally, vaccine utilisation differed across socio-economic strata, HPV and Influenza vaccines were predominantly utilised by individuals from upper socio-economic classes, while Tdap and Rabies vaccines saw higher utilisation among lower socio-economic groups. Vaccines such as MMR and Varicella were more commonly used among individuals from middle socio-economic classes. However, beneficiaries of influenza, PCV, and Tdap vaccines were significantly associated with the presence of comorbid conditions, whereas no such associations was observed for vaccine beneficiaries.
Overall the study identifies 30.89% (n = 253) incomplete vaccination among adult vaccine utilisers. Notably, partial immunisation was more common among young males, non-HCWs, and individuals from upper and lower middle socio-economic classes, without comorbidities. In addition, beneficiaries of Hepatitis B, influenza, and PCV vaccine were less likely to complete the full recommended dose schedules. A detailed representation of various factors relating to partial immunisation among adults were provided in Table 3.
Analysis of Predictors Associated With Partial-dose Adult Vaccine Utilisation.

Discussion
India reports an overall adult immunisation coverage of approximately 2%, 2 which aligns with the 2.03% coverage observed in our study population. Although several studies have extensively discussed various barriers to widespread adult vaccination in the country, the actual patterns of acceptance and utilisation in real-world settings remained underexplored.2,9,12,13 Similar to global trends, India witnessed a larger utilisation of COVID-19 vaccines among adults, with coverage exceeding 70% to 80% for at least 1 dose. 20 This represents one of the most extensive instances of adult immunisation coverage ever documented in the country. In contrast, developed countries such as the United States reported adult vaccination coverage for individual vaccines ranging from 23% to 43%, based on national survey data from 2017 to 2018. 21
The study population primarily consisted of HCWS and individuals from the upper 3 socio-economic strata. This reflects a concerning concentration of adult vaccine coverage within a limited segment of the community, indicating inequitable access across boarder population groups. A supporting observation by Mathew et al reported from India, noted that adult vaccine acceptance is not guided by epidemiological evidence. Instead, vaccines are often distributed through commercial channel or made available to physician at prices lower than the retail rate. 21 This suggests that only those who could afford vaccines were able to access them, while low awareness and limited knowledge about adult immunisation remain critical challenges to vaccine utilisation in India. 22
In the study population, HCWs comprised a significantly larger proportion of vaccine utilisers—approximately 2.4 times more than non-HCWs. This disparity was primarily driven by the high utilisation of the Hepatitis B vaccine among HCWs. Studies across India have reported high Hepatitis B vaccine coverage among HCWs, particularly among young medical students and newly recruited staffs23-25 This study aligns with these findings, showing elevated vaccine utilisation among younger age group, residing in hostel accommodations, likely due to institutional vaccination drives. Interestingly, high Hepatitis B vaccine utilisation was also observed among non-HCWs, particularly in younger age groups. This trend may be attributed to the vaccine’s affordability and (cost of the vaccines provided in the Supplemental Material File) proactive initiative by educational institutions, especially for students residing in hostels. However, 41.93% (60.56% vaccinated with 2 doses and 39.33% only with 1 dose) of them were found to be partial utilisers of the Hepatitis B vaccine, with the majority being HCWs from the upper 3 socio-economic classes. This suggests that negligence or the inability to adhere to the 3-dose schedule within a 1-month interval may contribute to incomplete vaccination, a finding consistent with concurrent studies.23-25
The utilisation patterns of vaccines other than Hepatitis B differ significantly, with affordability emerging as a major influencing factor. Excluding vaccines required for migration purposes, such as MCV and Hepatitis A, most other vaccines are predominantly utilised by the more advantaged segments of society. Recent emphasis on cervical cancer prevention has led to increased HPV vaccine utilisation, primarily among young adult female HCWs aged 20 to 34 years. Previous studies have highlighted that the high cost of the vaccine (limits the utilisation to financially weaker sessions) and lack of awareness among males were widely noted. 26 However, the voluntary utilisation of the HPV vaccine among the HCWs and their adherence to the complete vaccination schedule can enhance the public confidence in the vaccine.
Although HPV vaccines were primarily concentrated among young female HCWs, MMR emerged as one of the most commonly used adult vaccines among female HCWs of reproductive age. This pattern likely reflects their awareness of occupational exposure risks and the importance of completion all recommended doses. In contrast, vaccine utilisation among non-HCWs and individuals from lower socio-economic groups was notably limited, highlighting disparities in access and awareness. Despite the higher cost of varicella vaccines (refer Supplemental Material File), their utilisation rates were similar to MMR (3.78% and 3.54% respectively), suggesting that awareness and knowledge of infection complications significantly contribute to vaccine coverage. 27
Influenza and PCV vaccine utilisation was highest among older adults (53.33% and 54.54% respectively), followed by late-middle aged adults (26.675 and 21.82%).This trend aligns with the increased burden of Influenza (7.8% case fatality rate) 27 and Pneumococcal (8.8/100 000) 28 infections among the Indian population underscores the need for effective adult-immunisations. However, age-related immune decline and co-morbidities in elderly reduce vaccine-efficacy (30%-50% for influenza and 50% for pneumococcal infections). 29 Hence, promoting adult immunisation in middle-adulthood is advisable. Affordability remains a barrier, with vaccine access predominantly limited to higher socio-economic groups. Both Tdap and rabies vaccines are administrated for specific purposes; Tdap is often given preoperatively, while rabies vaccines are primarily used post-exposure following animal bites. Consequently, higher utilisation rates are observed among non-HCWs and from lower socio-economic backgrounds (statistically significant).
Vaccine safety is widely discussed topic, with studies reporting rare but notable AEs such as seizures and Guillain-Barre Syndrome followed influenza vaccines, encephalitis, and immune thrombocytopenia purpura after MMR and disseminated varicella infection.30,31 However, in this study, no deaths or serious AEs were reported, likely due to the smaller number of vaccine beneficiaries. Commonly observed AEs were mild to moderate, including injection site pain and fever. One serious incident—blood in sputum after influenza vaccination—was later identified as pneumonia and unrelated to the vaccine. Active surveillance helped distinguish such coincidental events, reinforcing that adult vaccines are generally safe, even for elderly or comorbid individuals.
Overall, gender, age, and population type showed significant variations in vaccine utilisation, with partial usage requiring closer attention. The study identified several contributing factors, including younger age, male gender, and middle-income status (upper and lower), without comorbid conditions, as being associated with incomplete vaccination. Notably, a high proportion of partial vaccination was linked to Hepatitis B vaccine, which was predominantly utilised by young adults. This finding highlights young males as potential predictor of partial vaccination. This trend may be attributed more to negligence and requirement of 3 consecutive doses for Hepatitis B, rather than financial constricts. A similar pattern of negligence was reported by Shrestha DB et al, where incomplete vaccination among medical students, occurred regardless of financial or other contributing factors. 32 In contrast, among non-HCWs partial vaccination was higher for influenza (16.67%) and PCV (18.18%), suggesting financial barriers. Conversely, HPV—despite being more expensive -had better completion rates, primarily among HCWs and individuals upper socio-economic strata, indicating vaccine access is heavily influenced by affordability, along with awareness and health literacy. Previous studies at the same study site on childhood vaccines revealed similar patterns,33,34 reinforcing that financial constraint remains a key barrier to equitable and complete access to adult immunisation.
Asaria et al evaluated wealth-specific mortality rates in India and found a 7.6-year life expectancy gap between the richest and poorest, reflecting deep-rooted health inequalities despite efforts towards Universal Health Coverage. 35 The push for adult immunisation during COVID-19 distributing in Low and Middle Income countries (LMICs) was suggested. 36 However, rising safety concerns post COVID-19 vaccines have fuelled adult vaccine hesitancy. Rebuilding public confidence requires transparent safety monitoring and effective communication. In 2018, an initiative to set-up a dedicated adult vaccination centre by All India Institute of Medical Sciences (AIIMS), Jodhpur, reported greater utilisation of post-exposure vaccines (rabies and tetanus vaccines) than pre-exposure vaccines. 2
At our study site, the primary beneficiaries of adult vaccination were females, young healthcare workers, individuals from higher socioeconomic classes, and those with comorbidities. While the high participation of HCWs is commendable and reflects voluntary initiative, the limited vaccine literacy among lower socioeconomic segments remains a significant concern. Apart from the Hepatitis B vaccine, the other 2 most utilised vaccines—Influenza and PCV—were predominantly accessed by economically advantaged individuals. This underscores the urgent need for government-led efforts, including vaccine subsidies, to reduce economic disparities and promote equitable access to adult immunisation.
Conclusion
This study highlights that the under-utilisation of adult vaccines remains a significant challenge in the current healthcare landscape. Among the limited number of adult vaccine users, the majority belonged to economically advantaged sections of society. A notable trend observed was the higher vaccine literacy and utilisation among healthcare workers (HCWs), whereas the general population (non-HCWs) largely remained under-reached and under-immunised. Beyond voluntary vaccine utilisation, the study identifies affordability as a key barrier limiting access, particularly among underprivileged populations.
In response to the question, ‘What is safe?’, the study suggests that adult immunisation—including among the elderly—demonstrated a favourable safety profile. Finally, the study raises a broader question: ‘What holds us back?’ Real-world utilisation patterns revealed that even among advantaged groups (such as HCWs and upper-class individuals), vaccine utilisation was often partial. This indicates that financial barriers and low vaccine literacy may continue to hinder broader utilisation. To comprehensively address these gaps, further behavioural studies among non-utilisers are essential to identify and understand the underlying factors contributing to vaccine hesitancy and inaccessibility.
Limitation of the Study
This study is limited to a population of individuals who voluntarily received recommended adult vaccines at a tertiary care hospital, by paying out of pocket. As a result, the finding may be subject to referral bias and may not be generalisable to the broader population. While a conventional sample size calculation was not applicable due to the observational design, all eligible beneficiaries during the study period were included. The study offers real-world insights into adult immunisation patterns; however, it does not explore physician-related factors that might influence vaccine utilisation, which is particularly relevant given the absence of a uniform adult immunisation policy in India. Additionally, the distribution of vaccine utilisation was heterogeneous—some vaccines like Hepatitis B were widely used, while others had limited utilisation—restricting uniform statistical methods across all groups. Nevertheless, appropriate statistical methods, including logistic regression, were applied to identify patterns among partial vaccinators and associated demographic factors. Being conducted at a tertiary care hospital, financial constraints may have deterred lower socioeconomic groups from accessing immunisation services, potentially contributing to underrepresentation. Larger community-based studies are needed to validate these findings and address equity gaps in adult immunisation access.
Supplemental Material
sj-docx-1-jpc-10.1177_21501319251380436 – Supplemental material for Adult Immunisation in a South Indian Tertiary Care Hospital: Who Benefits, What’s Safe, and What Holds Us Back?
Supplemental material, sj-docx-1-jpc-10.1177_21501319251380436 for Adult Immunisation in a South Indian Tertiary Care Hospital: Who Benefits, What’s Safe, and What Holds Us Back? by Merrin Mathew, Madhan Ramesh, Thippeswamy Thippeswamy, Mandyam Dhati Ravi and Yeliyur Sreerangachar Ravi Kumar in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
We sincerely express our gratitude to the staff and nurses of the Adult Immunisation Centre for their dedicated efforts in collecting essential information from vaccine recipients. We also extend our thanks to the Pharmacovigilance Centre for conducting active vaccine safety surveillance at the study site, and to the Department of General Medicine for their valuable support and cooperation throughout the study.
Authors’ Note
Merrin Mathew and Madhan Ramesh is now affiliated to Department of Clinical Pharmacy, JSS College of Pharmacy, Mysuru.
Thippeswamy Thippeswamy and Yeliyur Sreerangachar Ravi Kumar is now affiliated to Department of General Medicine, JSS Medical College, Mysuru.
Mandyam Dhati Ravi is now affiliated to Department of Clinical Research Excellence, JSS Medical College, JSS AHER.
Ethical Considerations
This study was reviewed and approved by the Institutional Ethics Committee of study site (Ref. No: JSSMC/IEC/2401005/01NCT/2022-23).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
