Abstract
Introduction/Objectives:
The prevalence of heart failure (HF) in the United States is currently at an all-time high and is predicted to rise to 8.5 million by 2030. Development of HF differs between men and women. We examined the risk factors associated with HF in women compared to men.
Methods:
A retrospective medical record review of adult men and women with HF. Variables were compared between women and men with Heart failure with Preserved Ejection Fraction (HFpEF) or Heart Failure with Reduced Ejection Fraction (HFrEF).
Results:
There were 460 women with HFpEF (WHFpEF) and 258 men with HFpEF (MHFpEF). Higher prevalences were observed in WHFpEF group compared to the MHFpEF group for older age (82.2 ± 9.5 vs 80.6 ± 9.6 years; P = .029), asthma (34.3 vs 20.2%; P < .001), osteoarthritis (66.5 vs 50.0%; P < .001), depression (39.6 vs 23.6%; P < .001), hypothyroidism (38.0 vs 17.1%; P < .001), and anxiety (43.0 vs 20.9%; P < .001). Greater odds of HFpEF in women was observed with increasing age per year (OR = 1.043, 95% CI = 1.027-1.059; P < .001), increasing BMI (OR = 1.037, 95% CI = 1.018-1.056; P < .001), rheumatological disorder (OR = 1.920, 95% CI = 1.457-2.530; P < .001), mental health disorder, such as depression or anxiety (OR = 1.898, 95% CI = 1.459-2.470; P < .001), asthma (OR = 1.807, 95% CI = 1.350-2.420; P < .001), and hypothyroidism (OR = 1.973, 95% CI = 1.486-2.618; P < .001). Among HFrEF patients, 225 patients were in WHFrEF group and 250 patients were in MHFrEF group. Higher prevalences were observed in WHFrEF group compared to the MHFrEF group for asthma (26.7 vs 16.8%; P = .009), depression (40.0 vs 22.0%; P < .001), hypothyroidism (32.0 vs 20.0%; P = .003), and anxiety (32.9 vs 21.6%; P = .006). Greater odds of HFrEF in women was noted for Black race (OR = 1.455, 95% CI = 1.018-2.080; P = .039).
Conclusion:
In women with HF, old age, elevated BMI and osteoarthritis are associated with HFpEF; Black race is associated with HFrEF; while asthma, depression, anxiety, and hypothyroidism are associated with both HFpEF and HFrEF.
Introduction
As per the Heart Failure Epidemiology and Outcomes Statistics published in 2023, the prevalence of heart failure (HF) in the United States is at an all-time high and is predicted to rise to 8.5 million by 2030. 1 It is a well-known fact that women are under-represented in studies on cardiovascular disease, especially HF.2-5 The American College of Cardiology, American Heart Association, and Heart Failure Society of America also explicitly acknowledge this disparity in their HF management guideline, noting that most evidence for guideline-directed medical therapy is derived from predominantly male cohorts. 6
Studies suggest that the development of HF differs between women and men. Women have a higher lifetime risk of acquiring Heart failure with Preserved Ejection Fraction (HFpEF) than Heart Failure with Reduced Ejection Fraction (HFrEF). On the other hand, HFrEF tends to be more common in men. Studies conclude that women with HF have different anatomical and physiologic features compared to men, such as lower blood pressure, less left ventricular mass, greater contractility, and less cell turnover. 7 It has also been reported that women with HF have worse quality of life compared to men with HF. 7
There is a difference in the pathophysiology of HFpEF and HFrEF. HFpEF is preceded by chronic metabolic comorbidities, whereas HFrEF is usually preceded by cardiomyocyte damage due to ischemia, genetic mutation, myocarditis, or valvular disease. 8 It is believed that compared to women, men are predisposed to macrovascular coronary artery disease and myocardial fibrosis which leads to a higher risk of HFrEF, whereas HFpEF is related to coronary microvascular dysfunction and endothelial inflammation.7,9 The current literature suggests that HFrEF is associated with male sex, history of cardiomyopathy, and myocardial infarction; whereas HFpEF is associated with atrial fibrillation, pulmonary hypertension, obesity, and valvular heart disease. 10
In addition to traditional risk factors, women have unique or disproportionately potent risk factors, including pregnancy-related complications (eg, preeclampsia and gestational diabetes), premature menopause, and exposure to certain cancer therapies (eg, breast cancer treatment-induced cardiomyopathy). 7 Nontraditional factors, such as depression, anxiety, caregiver stress, and low socioeconomic status might also contribute more to HF risk in women than in men. 6 Despite this knowledge, the medical literature highlights persistent gaps, such as sex-specific mechanisms, optimal drug dosing, and tailored risk prediction models are not fully established.7,11,12 To address these and additional gaps, it is imperative to identify and build a comprehensive list of the variables that are associated with HF in women. Additionally, considering an increasing trend of HF in both sexes, greater emphasis must be given to preventative care, which is only possible through identification of multiple risk factors to provide opportunity to modify or optimally control them. In this study, we aimed to examine multiple risk factors and comorbid medical conditions associated with HF in women compared to men.
Materials and Methods
Study Design and Setting
This study was a retrospective, non-matched, cohort analytic study that utilized convenience sampling of the existing electronic medical records of an entire cohort of adult patients with HF who received care in the outpatient clinics of an urban non-profit tertiary healthcare system between January 1, 2023, and December 31, 2023.
Participants
The inclusion criteria for our study were adult patients who were 18 years of age or older with a documented diagnosis of HFpEF or HFrEF. HFpEF was defined as HF with a left ventricular ejection fraction (LVEF) ≥50% and objective evidence of increased left ventricular (LV) filling pressures, such as documentation of elevated natriuretic peptide levels or abnormal noninvasive/invasive hemodynamic measurements; and HFrEF was defined as HF with a LVEF ≤40%. 6 Our exclusion criteria were patients who were younger than 18 years of age, or who did not have a diagnosis of HFpEF or HFrEF, or who had a diagnosis of HF with mildly reduced EF, or patients who had documentation of both HFpEF and HFrEF in their medical records, or patients who were asymptomatic without an echocardiogram to support the diagnosis of HF.
Variables
We collected the data on demographics, social factors and family history; biometric values, such as body mass index (BMI), systolic blood pressure (SBP), and diastolic blood pressure (DBP); certain comorbid medical conditions, defined as additional diseases or disorders that coexisted with a primary or index disease in the same individual, but were not direct complications or sequelae of the index disease itself. These included hypertension, dyslipidemia, diabetes mellitus (DM), chronic kidney disease (CKD), coronary artery disease (CAD), peripheral artery disease (PAD), carotid artery disease, chronic obstructive pulmonary disease (COPD), asthma, obstructive sleep apnea (OSA), osteoarthritis (OA), rheumatological disorders, chronic liver disease, mental health disorders, hypothyroidism, hyperthyroidism, anemia, cancer, atrial fibrillation, other cardiac dysrhythmias, acquired valvular heart disease, congenital heart disease, myocardial deposition disorder, pulmonary hypertension, cerebrovascular accident (CVA), and Coronavirus disease 2019 disease (COVID-19). We utilized Microsoft Excel (2016, Redmond, Washington, USA) spreadsheet to record all the data.
Data Source
The Institutional Review Board approved this study and granted authorization to collect data for research purposes. Being a retrospective data collection, informed consents were not required. The data was accessible to all investigators from the hospital’s electronic medical records in the Epic healthcare software (Epic Systems Corporation, Wisconsin, USA).
Sample Study Size
We included the entire population of a total of 1207 patients in our healthcare system who were diagnosed with either HFpEF or HFrEF, out of which 1193 patient records accurately documented the diagnosis of HFpEF or HFrEF and had all pertinent data variables available. Hence, we included 1193 patients.
Statistical Methods
We used the SPSS (Statistical Package for the Social Sciences, version 15.01, IBM, Armonk, New York, USA) software for statistical analysis. We divided our cohort of patients into 4 groups: women with HFpEF (WHFpEF), men with HFpEF (MHFpEF), women with HFrEF (WHFrEF), and men with HFrEF (MHFrEF). We compared WHFpEF group with MHFpEF group, and WHFrEF group with MHFrEF group. For continuous variables, we ran a test of skewness to determine whether the data was normally distributed or nonparametric. Independent t-test was performed on normally distributed data and Mann-Whitney U test was performed on the non-parametric data. For the categorical variables, Pearson chi square tests were used. Additionally, in women with HF, we used logistic regression models in which women with HFpEF (WHFpEF), or women with HFrEF (WHFrEF) were the outcome variables. The model for WHFpEF included variables, such as age, race, BMI, smoking, rheumatological disorders, mental health disorders, CAD, asthma, hypothyroidism, and CKD. Similarly, the model for WHFrEF included age, race, BMI, smoking, rheumatological disorders, mental health disorders, CAD, asthma, hypothyroidism, and CKD. We defined statistical significance as a P value of <.05.
Results
Women With HFpEF Versus Men With HFpEF
The mean age was significantly greater in the WHFpEF group (n = 460) compared to the MHFpEF (n = 258; 82.2 ± 9.5 vs 80.6 ± 9.6 years; P = .029; Table 1). In both the groups, majority of the patients identified as of White race followed by Black and other races; however, the difference was not significant (Table 1). Tobacco, alcohol and recreational drug use were significantly lower in the WHFpEF group compared to the MHFpEF group (Table 1). There were no significant differences between the 2 groups in the family history of heart failure, mean BMI, mean systolic and diastolic blood pressures (Table 1).
Baseline Characteristics of Patients With HFpEF.

Abbreviatons: BMI, Body mass index; CAD, coronary artery disease; DBP, diastolic blood pressure; HFpEF, heart failure with preserved ejection fraction; n, number of patients; SBP, systolic blood pressure; SD, standard deviation.
Analysis of associations with comorbid medical diagnoses revealed greater prevalence of several comorbidities with WHFpEF group compared to the MHFpEF group, such as asthma (34.3 vs 20.2%; P < .001), osteoarthritis (66.5 vs 50.0%; P < .001), depression (39.6 vs 23.6%; P < .001), hypothyroidism (38.0 vs 17.1%; P < .001), and anxiety (43.0 vs 20.9%; P < .001; Table 2). Additionally, we found significantly lower prevalence of some comorbidities with WHFpEF group compared to the MHFpEF group, such as CKD (50.0 vs 63.6%; P < .001), and CAD (50.0 vs 64.3%; P < .001; Table 2). We did not find significant differences in the prevalences of other comorbidities, such as hypertension, DM, dyslipidemia, PAD, carotid artery disorder, COPD, obstructive sleep apnea, chronic liver disease, bipolar disorder, hyperthyroidism, anemia, schizophrenia, cancer, atrial fibrillation, other cardiac dysrhythmias, acquired valvular heart disease, congenital heart disease, myocardial deposition disorder, pulmonary hypertension, CVA, COVID-19 disease, or prior use of chemotherapy (Table 2).
Comparative Associations of Comorbidities in Patients With HFpEF.
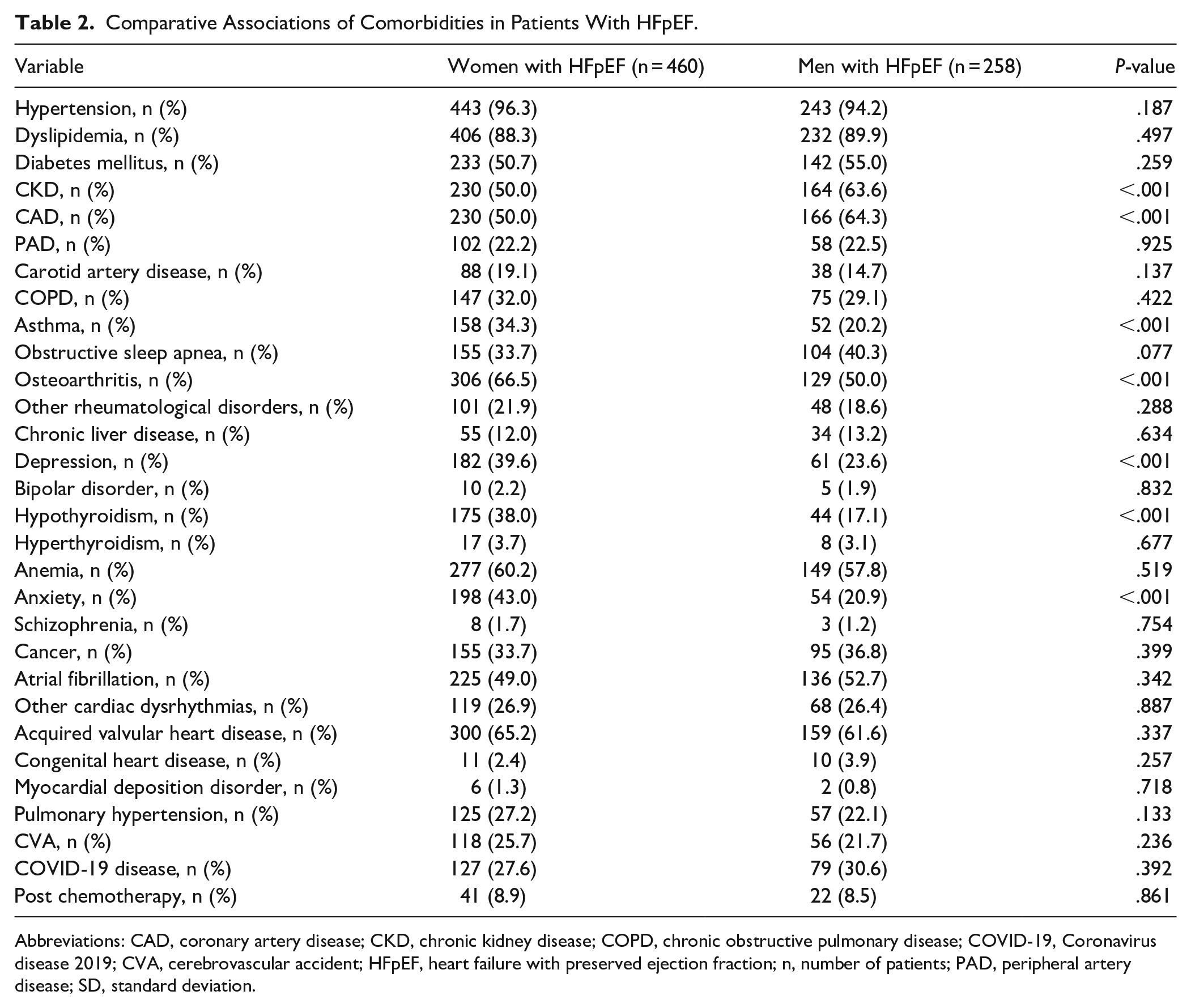
Abbreviations: CAD, coronary artery disease; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; COVID-19, Coronavirus disease 2019; CVA, cerebrovascular accident; HFpEF, heart failure with preserved ejection fraction; n, number of patients; PAD, peripheral artery disease; SD, standard deviation.
A logistic regression model demonstrated greater odds of HFpEF in women with increasing age per year (OR = 1.043, 95% CI = 1.027-1.059; P < .001), increasing BMI (OR = 1.037, 95% CI = 1.018-1.056; P < .001), having a rheumatological disorder (OR = 1.920, 95% CI = 1.457-2.530; P < .001), having a mental health disorder, such as depression or anxiety (OR = 1.898, 95% CI = 1.459-2.470; P < .001), having asthma (OR = 1.807, 95% CI = 1.350-2.420; P < .001), and having hypothyroidism (OR = 1.973, 95% CI = 1.486-2.618; P < .001; Table 3).
Influence of Variables in Women With HFpEF.

Women With HFrEF Versus Men With HFrEF
The mean age and race distribution were comparable between the WHFrEF group (n = 225) and MHFrEF group (n = 250; Table 4). In both the groups, majority of the patients identified as of White race followed by Black and other races; however, the difference was not significant (Table 4). Use of tobacco and alcohol were significantly lower in the WHFrEF group compared to the MHFrEF group, while we found no difference in the use of recreational drugs (Table 4). There were no differences in the frequencies of family history of heart failure, mean BMI, and mean SBP and mean DBP between the 2 groups (Table 4).
Baseline Characteristics of Patients With HFrEF.

Abbreviations: BMI, Body mass index; CAD, coronary artery disease; DBP, diastolic blood pressure; HFpEF, heart failure with preserved ejection fraction; n, number of patients; SBP, systolic blood pressure; SD, standard deviation.
Analysis of associations with comorbid medical diagnoses in the WHFrEF group compared to the MHFrEF group revealed higher prevalence of asthma (26.7 vs 16.8%; P = .009), depression (40.0 vs 22.0%; P < .001), hypothyroidism (32.0 vs 20.0%; P = .003), and anxiety (32.9 vs 21.6%; P = .006; Table 5). Additionally, we found significantly lower prevalence of some comorbidities with WHFrEF group compared to the MHFrEF group, such as CKD (47.6 vs 58.8%; P = .014), CAD (60.0 vs 77.2%; P < .001), OSA (21.3 vs 29.2%; P = .049), atrial fibrillation (47.1 vs 56.8%; P = .035), and other cardiac dysrhythmias (32.4 vs 41.2%; P = .049; Table 5). We did not find significant differences in the prevalences of hypertension, DM, dyslipidemia, PAD, carotid artery disorder, COPD, osteoarthritis, other rheumatological disorders, chronic liver disease, bipolar disorder, hyperthyroidism, anemia, schizophrenia, cancer, acquired valvular heart disease, congenital heart disease, myocardial deposition disorder, pulmonary hypertension, CVA, COVID-19 disease, or prior use of chemotherapy (Table 5).
Comparative Associations of Comorbidities in Patients With HFrEF.

Abbreviations: CAD, coronary artery disease; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; COVID-19, coronavirus disease 2019; CVA, cerebrovascular accident; HFpEF, heart failure with preserved ejection fraction; n, number of patients; PAD, peripheral artery disease; SD, standard deviation.
Greater odds of HFrEF in women was noted for Black race (OR = 1.455, 95% CI = 1.018-2.080; P = .039; Table 6).
Influence of Variables in Women With HFrEF.

Discussion
We found that compared to men, women with HFpEF had higher prevalence of old age, elevated BMI, asthma, osteoarthritis, depression, anxiety, and hypothyroidism; while women with HFrEF had higher prevalence of Black race, asthma, depression, anxiety, and hypothyroidism.
In our population of patients, women with HFpEF were older in age compared to the men with HFpEF. Although, there happens to be a similar lifetime risk of HF in both men and women; nevertheless, reported studies showed increased prevalence of HFpEF in women, when compared to men and the prevalence increased with age.7,13,14 A study that analyzed 3, large HFpEF trials revealed that patients who were younger were mostly men who were obese, while patients who were older comprised of women with comorbid medical conditions. 15 Although there is no clear mechanism which explains this phenomenon, there are several proposed hypotheses: (1) greater inflammation of the myocardial muscles in women associated with microvascular dysfunction; (2) presence of risk factors that were sex-specific, such as adverse pregnancy outcomes, reproductive factors and early menopause; and (3) the fact that traditional HF risk factors, such as CAD, DM, obesity, and hypertension tend to contribute to the development of HFpEF preferentially in women. 13 Moreover, estradiol regulates inflammatory factors, hence a declining level of estradiol during menopause leads to changes in blood pressure, lipid content, and body fat, which are all known risk factors in the development of HFpEF. 16 Although, older age has been reported as a risk factor for HFpEF in both men and women; however, there are no reports that suggest a predilection toward women more than men. Our study underscores a greater association of older age with women who had HFpEF compared to men with HFpEF.
It has been reported that up to 80% of all individuals with HFpEF, irrespective of sex, tend to be either overweight or obese. Obesity-related pathophysiological changes include increased plasma volume, right ventricular dysfunction, and concentric left ventricular remodeling in such population.17-19 In our study, we found that when compared to men, women with HFpEF had a higher association with elevated BMI which has not been explicitly reported. Women with HFpEF and elevated BMI tend to exhibit greater visceral adipose tissue (VAT) accumulation, which has been implicated with severe hemodynamic abnormalities during exercise, including higher pulmonary capillary wedge pressure (PCWP). 20 This suggests that VAT might play a distinct role in the pathophysiology of HFpEF in women, contributing to decreased exercise capacity and higher filling pressures.20,21 We also found that higher BMI was more strongly linked to HFpEF in women than HFrEF. This association underscores the importance of managing obesity and its related conditions to mitigate the risk and severity of HFpEF in women.
We found that osteoarthritis was strongly associated with HFpEF in women which has not been reported so far. One study reported that osteoarthritis was linked to increased cardiovascular risk factors such as obesity, hypertension, and systemic inflammation, which happen to be the risk factors for HFpEF. 22 This association was believed to be due to the higher prevalence of systemic inflammation and metabolic syndrome in women with osteoarthritis, which could exacerbate the pathophysiological mechanisms underlying HFpEF. 23
It has been reported that HFrEF happens to be more prevalent in individuals who identify as of the Black race when compared to other racial groups. 24 In the Multi-Ethnic Study of Atherosclerosis (MESA) regarding individuals without baseline cardiovascular disease, individuals identified as of Black race had the highest risk of developing HF, followed by individuals of Hispanic, White, and Chinese race categories. 25 Similar findings were observed in the Atherosclerosis Risk in Communities (ARIC) study, in which Black American men and women had a higher burden of new-onset HF compared to White men and women. 26 Multiple studies have shown that HFrEF is more prevalent in Black men than in Black women, with Black women having higher rates than White women but still lower than Black men.6,7,27-32 However, in our study, we found that Black race had a stronger association with women with HFrEF compared to HFrEF in men and women of any other race. Several factors might contribute to this association in Black race and women with HFrEF, such as a higher burden of modifiable risk factors, including DM, hypertension, obesity, and other non-ischemia related causes. 33
Asthma has been reported to be associated with an increased risk of developing cardiovascular disease, especially in women.34,35 A prospective study of over 200 000 patients found that asthma was associated with a 2.14-fold increased hazard of HF (HFpEF and HFrEF), with stronger associations observed with women compared to men. 36 A meta-analysis of 18 cohort studies reported a pooled risk ratio of 2.1 for HF in individuals with asthma compared to non-asthmatics, and noted that women with asthma had a higher risk, suggesting a possible sex-specific effect for cardiovascular risk. 37 There are other epidemiological reviews that have supported the link between asthma and increased cardiovascular risk, including HF, and have highlighted the role of systemic inflammation.38,39 Our observation of association of asthma with HF in women aligns with the existing literature.
We found a strong association of depression and anxiety disorder in women with HF (both HFpEF and HFrEF). Several studies have reported that depression and anxiety were common in women with HF and were associated with poor outcomes, including increased morbidity, mortality, and reduced quality of life.40,41 One study reported that compared to men with HF, women with HF were more likely to develop depression, and depressive symptoms in women were strongly linked to higher risk of cardiovascular death. 42 Changes in depression severity over time also impacted the prognosis. 42 It has also been observed that in women with HFrEF, association of depression at baseline had a stronger association with cardiovascular death. 34 Another prospective cohort study reported that among individuals with anxiety disorder only, depression only, and individuals with both conditions, the risk of HF was greater than in the individuals without these conditions. 43 A large cohort Veteran’s Administration study concluded that anxiety disorder, depression, and co-occurring anxiety with depression were associated with HF in patients who were free of cardiovascular disease at baseline. 44 The investigators also found that the risk of HF was greater after accounting for the protective effects of psychotropic medications. 44 Despite their impact, depression and anxiety are often underdiagnosed and undertreated in HF, highlighting the need for routine screening and targeted psychosocial interventions, particularly in women.
Hypothyroidism has been reported to be associated with an increased risk of HF, with women being at a higher risk than men for developing HF after hypothyroidism.45-51 Both subclinical and overt hypothyroidism have been reported to be linked to adverse cardiac changes, such as diastolic dysfunction, increased systemic vascular resistance, and progression to HFpEF and HFrEF. 46 We found a similar association. Women with hypothyroidism, particularly after iodine exposure, were found to have a significantly higher risk of developing HF compared to men. 47 While thyroid hormone replacement improved cardiac function in some cases, evidence has been mixed regarding whether treating subclinical hypothyroidism might reduce HF risk, or not.49,50 Further research is needed to clarify sex-specific risks and treatment benefits.
Being a retrospective study, our study had a few limitations. We had to solely rely on the documentation of the clinical care team regarding the onset of HF and the onset of the comorbidities, while inadequate or duplication of HF diagnosis in the medical records forced us to exclude several patients who had no concurring supportive echocardiogram evidence. Since the study patients belonged to the medical offices in urban settings, our results cannot be generalized. Moreover, diet, physical activity, and socioeconomic status could not be included in the study which might have influenced the sex-specific risk of HF. However, in this study, we could review a large database of existing patients who received their medical care in the offices of a single healthcare institution which allowed the care teams to document events and comorbidities in a chronological order over a long duration of follow up.
Conclusion
We provide a comprehensive list of risk factors associated with HF in women and conclude that in women with HF, old age, elevated BMI, and osteoarthritis are associated with HFpEF; Black race is associated with HFrEF; while asthma, depression, anxiety, and hypothyroidism are associated with both HFpEF and HFrEF when compared to men with HF. Further research in different settings may allow clinicians to identify, modify, and optimize the sex specific risk factors associated with HF in women.
Footnotes
Consent to Participate
Not applicable. Being a retrospective chart review study, the Institutional Review Board waived the need for informed consent.
Author Contributions
KY and SR made substantial contributions to the study design, drafting, data acquisition, data analysis, and manuscript writing. All authors contributed to data collection and manuscript writing. KH analyzed the data. SR contributed by revising the manuscript critically for improved intellectual content, and final approval for the version to be published.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The authors declare that data supporting the findings of this study are available within the article.
