Abstract
Introduction/Objectives:
Inflammatory vasculitis (IV) is characterized by blood vessel inflammation, leading to vessel damage and potential organ failure. This study aimed to identify the risk factors and comorbidities in adults with IV.
Methods:
A retrospective review of adult patients receiving outpatient care was conducted. Variables were compared between IV and non-IV (NoIV) groups.
Results:
Among 1500 patients, 626 (41.7%) had IV. Patients in the IV group were significantly older (IV = 66.5 ± 18.8 vs NoIV = 52.9 ± 18.4 years; P < .001) and women (IV = 67.7 vs NoIV = 48.0%; P < .001). IV group had a lower prevalence of patients of White race, and higher prevalences of use of tobacco and recreational drugs. Higher frequencies of associations of comorbid medical conditions were noted in the IV group compared to NoIV group, such as hypertension (50.7 vs 29.0%; P < .001), DM (19.4 vs 10.4%; P < .001), CKD (12.1 vs 3.7%; P < .001), CAD (12.1 vs 6.2%; P < .001), CHF (5.8 vs 2.0%; P < .001), PAD (2.4 vs 0.8%; P = .013), COPD (7.8 vs 1.8%; P < .001), pulmonary fibrosis (2.4 vs 0.1%; P < .001), osteoporosis (10.4 vs 1.9%; P < .001), osteoarthritis (12.0 vs 7.1%; P = .001), GERD (21.8 vs 13.2%; P < .001), depression (14.1 vs 7.9%; P < .001), hypothyroidism (14.2 vs 8.2%; P < .001), anemia (16.9 vs 5.0%; P < .001), seizure disorder (3.0 vs 0.9%; P < .001), cancer (13.7 vs 8.5%; P = .001), asthma (15.3 vs 6.7%; P < .001), atrial fibrillation (9.6 vs 4.7%; P < .001), CVA (7.0 vs 2.5%; P < .001), SLE (2.9 vs 0.5%; P < .001), and rheumatoid arthritis (5.0 vs 0.8%; P < .001). There were significantly greater odds of IV with increasing age per year (OR 1.030, 95% CI 1.022-1.039; P < .001), recreational drug use (OR 1.953, 95% CI 1.150-3.318; P < .013), hypertension (OR 1.590, 95% CI 1.155-2.188; P = .004), DM (OR 1.621, 95% CI 1.080-2.434; P = .020), osteoarthritis-osteoporosis (OR 1.496, 95% CI 1.020-2.193; P = .039), cardiac disorders (OR 1.543, 95% CI 1.065-2.236; P = .022), pulmonary disorders (OR 3.173, 95% CI 1.663-6.054; P < .001), and rheumatological disorders (OR 3.589, 95% CI 1.780-7.236; P < .001).
Conclusion:
IV is associated with older age, female sex, non-White race, tobacco use, recreational drug use, hypertension, DM, cardiovascular disorders (CAD, CHF, PAD, CVA), pulmonary disorders (COPD, asthma, pulmonary fibrosis), osteoporosis, osteoarthritis, SLE, and rheumatoid arthritis.
Keywords
Introduction
Inflammatory vasculitis (IV) refers to a range of conditions marked by blood vessel inflammation, resulting in vessel damage and potential organ failure. These diseases present a variety of symptoms, depending on the type and size of blood vessels affected. Small vessel involvement is characteristic of antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV), which includes granulomatosis with polyangiitis (GPA), microscopic polyangiitis (MPA), and eosinophilic granulomatosis with polyangiitis (EGPA). Although rare, the incidence of AAV has increased over the past several decades, with a combined incidence rate of 33.0 per million in the United States. 1 Medium vessel vasculitis includes polyarteritis nodosa, which affects small and medium-sized vessels, and Kawasaki disease, which affects the coronary arteries. Large vessel vasculitis affects larger arteries, such as the aorta and its major branches in Takayasu arteritis and branches of the carotid, vertebral, and temporal arteries in Giant cell arteritis (GCA). 1
The pathophysiology of inflammatory vasculitis is broad and multifaceted, involving genetic predispositions and immune system dysregulation. AAV, for example, is characterized by the presence of antibodies that target neutrophils, inducing their activation and leading to inflammation and damage. 2 GCA, by contrast, is associated with the activation of vascular dendritic cells, which produce chemokines that recruit T-cells and form granulomas. 3 Types of inflammatory vasculitis have also been shown to affect individuals from varying age and ethnic demographics. Takayasu arteritis and Kawasaki disease, for example, are both prevalent in Asian populations, while the former is associated with individuals under 40 years old, and the latter predominantly affects children under 5 years old. 1
Much remains unknown about the associations between IV and a wider array of demographic factors and comorbid medical conditions. Recent studies have highlighted the elevated risk of comorbidities in patients with chronic inflammatory diseases. For instance, patients with systemic vasculitis and other disorders such as psoriasis, inflammatory bowel disease, and inflammatory arthritis were shown to have an increased risk of developing type 2 diabetes mellitus (DM), coronary artery disease (CAD), and cerebrovascular accident (CVA). 4 Elevated C-reactive protein levels additionally correlated with higher incidence of these comorbidities. 4
Patients with AAV have been shown to have a heightened prevalence of metabolic syndrome compared to healthy controls. 5 Other studies have focused specifically on comorbidities associated with GPA, revealing increased risks of hypertension, dyslipidemia, DM, and venous thromboembolism. 2 These findings emphasize the need for comprehensive monitoring and early intervention to mitigate these risks. Additionally, renal, cardiovascular, and treatment-related organ damage has been shown to accumulate over time in patients with GPA, highlighting the importance of long-term management strategies for individuals with this condition.6,7 We aimed to investigate the associations of demographic factors, personal social factors, family history of autoimmune diseases, and comorbid medical conditions with IV in adult patients.
Materials and Methods
Study Design and Setting: This study was a retrospective cohort analysis of all adult patients who received medical care at a single internal medicine practice in a suburban location. Convenience sampling was facilitated by utilizing electronic medical records to identify patients who had office visits between January 1, 2023, and December 31, 2023.
Participants: We included patients aged 18 years or older with a diagnosis of IV, such as GCA, Takayasu arteritis, Polyarteritis nodosa, GPA, EGPA, MPA, Henoch-Schönlein purpura, Cryoglobulinemic vasculitis, Bechet’s disease, Cogan’s syndrome, IgA vasculitis, Leukocytoclastic vasculitis, central nervous system (CNS) vasculitis, and Buerger’s disease. Patients under the age of 18 were excluded from the study.
Variables: For each patient, we collected data on demographics and social factors, laboratory parameters, comorbid medical conditions, and the use of specific medications. All data points were recorded using Microsoft Excel (2016, Redmond, Washington, USA).
Data Source and Access: Our Institutional Review Board (IRB) approved the study, and all collected data were used exclusively for research purposes as permitted by the IRB. Informed consent waivers were granted, and the study adhered to the IRB’s ethical standards. Investigators had access only to the selected data in the Epic healthcare software (Epic Systems Corporation, Wisconsin, USA) electronic medical records from a de-identified patient list provided by the medical informatics team, based on the inclusion criteria.
Bias: To reduce potential confounders and ensure accurate identification of IV diagnoses, we excluded patients who had suspected or unconfirmed diagnosis of IV, patients with positive antinuclear antibody (ANA) test without a confirmed diagnosis of an autoimmune disorder, and patients with a diagnosis of vasculitis secondary to a drug (drug induced vasculitis).
Study Size: We selected a sample size of 1500 adult patients. We rationalized that in the United States, the prevalence of the commonest IV, which is GCA, is about 204 per 100,000 people aged 50 and older and the prevalence of other IVs is much lower. Putting all IVs together, we estimated approximately at least 500 patients needed in the IV group and under 1000 patients in the control group of patients who did not have IV.
Statistical Methods: Statistical analysis was conducted using the Statistical Package for the Social Sciences (SPSS, version 15.01, IBM, Armonk, New York, USA). Patients were categorized into 2 groups: those with IV and those without IV (NoIV). Skewness testing was performed for continuous variables to assess data distribution. An independent t-test was applied for normally distributed data with skewness values between −1 and 1, while the Mann-Whitney U test was used for non-parametric data with skewness values beyond this range. Categorical variables were analyzed using chi-square tests or Fisher’s exact tests, depending on the expected data values. Logistic regression was utilized with IV as the outcome variable, and a gradient boosting model identified the most significant predictors of IV. A P-value of <.05 was considered statistically significant.
Results
There were 1500 patients in this study. The IV group had 626 (41.7%) patients and the NoIV group had the remaining 874 patients (Table 1). In the IV group, the majority of the patients had GCA (39.3%), GPA (22.8%), or Bechet’s syndrome (13.3%), followed by lesser frequencies of polyateritis nodes (4.3%), Takayasu arteritis (4.0%), EGPA (3.8%), microscopic polyangiitis (3.4%), CNS vasculitis (3.2%), Buerger’s disease (3.2%), cryoglobulinemic vasculitis (1.1%), Henoch-Schönlein purpura (0.8%), and Cogan’s syndrome (0.8%) (Table 2). The mean age was significantly greater in the IV group (IV = 66.5 ± 18.8 vs NoIV = 52.9 ± 18.4 years; P < .001) (Table 1). The IV group had significantly greater number of females than the NoIV group (67.7 vs 48.0%; P < .001) (Table 1). In both the groups, the majority of the patients identified themselves as of White race followed by Black, other and Asian races. However, IV group had lesser frequency of patients of White race (IV = 78.2% vs NoIV = 60.0%; P < .001) (Table 1). There was significantly lesser use of alcohol in the IV group compared to the NoIV group (34.3% vs 49.4%; P < .001); however, there were significantly higher use of tobacco and recreational drugs in the IV group (tobacco: IV = 35.5% vs NoIV = 28.8%; P = .007, and recreational drug: IV = 8.0% vs NoIV = 4.7%; P = .007) (Table 1). More patients in the IV group had a family history of autoimmune disorders compared to the NoIV group (7.9% vs 4.2%; P = .003) (Table 1). The mean total cholesterol and LDL-cholesterol were significantly lower in the IV group while the median WBC count, ESR, and CRP were significantly higher in the IV group compared to NoIV group (Table 1). There was no difference in the mean HDL-cholesterol triglyceride and median rheumatoid factor levels between the 2 groups.
Baseline Characteristics.
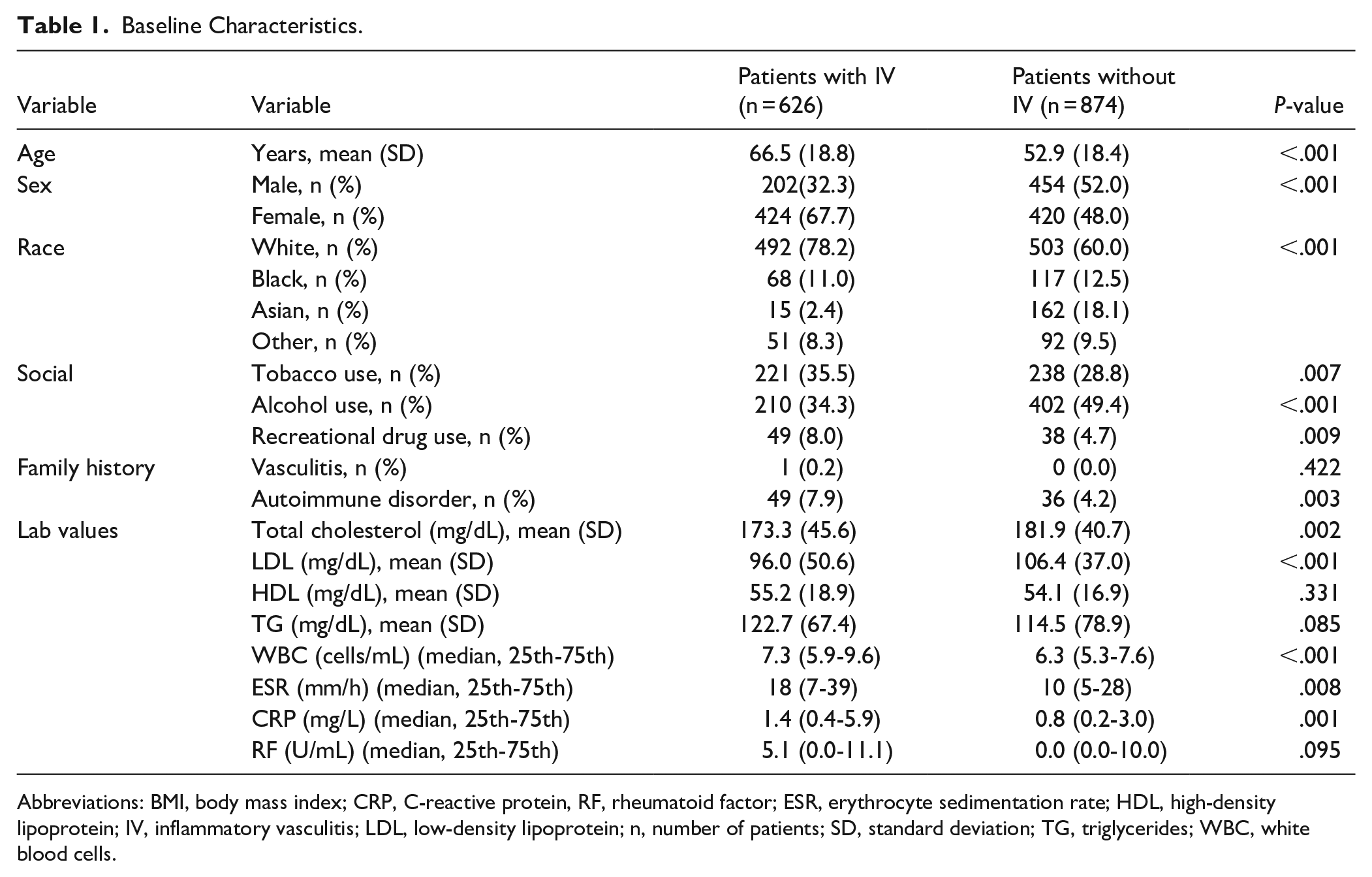
Abbreviations: BMI, body mass index; CRP, C-reactive protein, RF, rheumatoid factor; ESR, erythrocyte sedimentation rate; HDL, high-density lipoprotein; IV, inflammatory vasculitis; LDL, low-density lipoprotein; n, number of patients; SD, standard deviation; TG, triglycerides; WBC, white blood cells.
Types of Inflammatory Vasculitis.

Abbreviation: n, number of patients.
Analysis of associations with comorbid medical diagnoses revealed significantly higher frequencies of associations of several comorbidities in the IV group in comparison to NoIV group, such as hypertension (50.7 vs 29.0%; P < .001), DM (19.4 vs 10.4%; P < .001), chronic kidney disease (CKD) (12.1 vs 3.7%; P < .001), CAD (12.1 vs 6.2%; P < .001), congestive heart failure (CHF) (5.8 vs 2.0%; P < .001), peripheral arterial disease (PAD) (2.4 vs 0.8%; P = .013), chronic obstructive pulmonary disease (COPD) (7.8 vs 1.8%; P < .001), pulmonary fibrosis (2.4 vs 0.1%; P < .001), osteoporosis (10.4 vs 1.9%; P < .001), osteoarthritis (OA) (12.0 vs 7.1%; P = .001), gastroesophageal reflux disorder (GERD) (21.8 vs 13.2%; P < .001), depression (14.1 vs 7.9%; P < .001), hypothyroidism (14.2 vs 8.2%; P < .001), anemia (16.9 vs 5.0%; P < .001), seizure disorder (3.0 vs 0.9%; P < .001), cancer (13.7 vs 8.5%; P = .001), asthma (15.3 vs 6.7%; P < .001), atrial fibrillation (9.6 vs 4.7%; P < .001), CVA (7.0 vs 2.5%; P < .001), systemic lupus erythematous (SLE) (2.9 vs 0.5%; P < .001), rheumatoid arthritis (5.0 vs 0.8%; P < .001), and inflammatory bowel disease (IBD) (1.8 vs 0.5%; P = .014) (Table 3). The frequencies of association of dyslipidemia, chronic liver disease, obesity, and COVID-19 disease were significantly lower in the IV group compared to the NoIV group (Table 3). We did not find significant differences in other comorbidities, such as dementia, anxiety, psoriasis, hepatitis C virus infection, and cancer chemotherapy between the 2 groups (Table 3). The usage of certain medications in the group with IV was higher than the NoIV group, such as hydralazine (2.2% vs 0.8%; P < .022) and TNF-α inhibitor (4.0 vs 0.2%; P < .001) (Table 3).
Comparative Association of Comorbidities and Medications.

Abbreviations: CAD, coronary artery disease; CHF, congestive heart failure; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; COVID-19, coronavirus disease 2019; CVA, cerebrovascular accident; GERD, gastroesophageal reflux disorder; n, number of patients; PAD, peripheral artery disease; SLE, systemic lupus erythematosus; TNF-α inhibitor, tumor necrosis factor-α inhibitor.
A logistic regression model demonstrated greater odds of IV with increasing age per year (OR 1.030, 95% CI 1.022-1.039; P < .001), recreational drug use (OR 1.953, 95% CI 1.150-3.318; P < .013), hypertension (OR 1.590, 95% CI 1.155-2.188; P = .004), DM (OR 1.621, 95% CI 1.080-2.434; P = .020), osteoarthritis-osteoporosis (OR 1.496, 95% CI 1.020-2.193; P = .039), cardiac disorders (OR 1.543, 95% CI 1.065-2.236; P = .022), pulmonary disorders (OR 3.173, 95% CI 1.663-6.054; P < .001), and rheumatological disorders (OR 3.589, 95% CI 1.780-7.236; P < .001) (Table 4).
Influence of Variables on Inflammatory Vasculitis.

Discussion
Our findings of older age and female sex are consistent with the available literature suggesting that certain vasculitis, such as GCA, primarily affect older individuals, while IV associated with autoimmune diseases generally have a higher prevalence in females. 8
Racial distribution analysis showed that although patients identified as of White race represented the majority in both the groups, their proportion was lower in the IV group compared to the NoIV group. This suggests that IV may disproportionately affect certain racial and ethnic groups, warranting further exploration of genetic and environmental risk factors. These findings are supported by a study which highlights distinct racial/ethnic patterns for various vasculitides, including AAV and large-vessel vasculitis (LVV). 9 The study notes that non-White populations, such as those of Asian and African descent, exhibit different susceptibilities and disease presentations compared to the White populations. 9
Lifestyle factors, such as tobacco and recreational drug use were higher in our patients with IV. A study examined the relationship between cigarette smoking and AAV and found that patients with AAV were more likely to be current or former smokers, with a dose-response relationship particularly strong in those with myeloperoxidase (MPO)-ANCA-positive disease. 10 This suggests a potential link between smoking and IV pathogenesis, consistent with previous reports implicating smoking in immune dysregulation and chronic inflammation.
We found that having a family history of autoimmune diseases was associated with IV which suggests that genetic predisposition may play a role in the development of IV. Authors in a study emphasized the significance of genetic factors in vasculitis, noting that familial aggregation and genetic associations with disease susceptibility have been consistently observed. 11
In our study, hypertension was associated with IV. The association of vascular inflammation and development of hypertension has been vastly reported. Particularly, vascular inflammation of the renal arteries has been reported to be associated with hypertension. 12 It has also been suggested that degenerative predisposition to generalized vascular inflammation associated with IV may be involved in the hypertensive injury. 13 The role of inflammation-driven endothelial dysfunction and platelet activation on accelerated atherosclerosis and hypertension has been established in patients with (AAV). 14 It is believed that inflammatory vasculitis alters the vascular endothelial function in multiple ways, such as inflammasome activation, oxidative stress, protein misfolding and immune mediated vascular responses, which in turn result into microvascular damage which results into hypertension and other cardiovascular disorders. 15 A meta-analysis of 122 studies that investigated the association of hypertension and cardiovascular complications in autoimmune disorders and IV concluded that the prevalence of hypertension was higher in these groups of patients compared to the control population. 16 Several studies indicate that hypertension involves low-grade inflammation and immune cell infiltration in the vasculature, kidneys, and heart, contributing to vascular dysfunction and remodeling.17 -20 This inflammatory response in hypertension is mediated by various immune cells, including T cells, B cells, macrophages, and cytokines such as IL-17 and TNF-α.17 -19 Our finding of high association of hypertension in patients with IV aligns with the existing literature.
Association of IV with DM has been suggested in some studies with the notion that chronic vascular inflammation might lead to atherosclerotic cardiovascular disease and insulin resistance, which in turn may lead to metabolic syndrome and type-2 DM.21,22 DM has been observed in certain IV, such as GCA and GPA which is believed to be due to corticosteroid treatment. 23 Other studies suggest that vascular injury, such as endothelial dysfunction, altered vascular tone, and inflammation are common pathophysiological mechanisms that link both DM and IV. 24 Consensus is that DM is associated with an increased risk of IV, primarily through mechanisms involving chronic inflammation and oxidative stress. Hyperglycemia in DM leads to the formation of advanced glycation end products (AGEs), which interact with their receptors (RAGE) on vascular cells, promoting inflammation and endothelial dysfunction.25,26 This interaction triggers the release of pro-inflammatory cytokines and reactive oxygen species (ROS), contributing to vascular inflammation. Additionally, DM enhances the activation of Toll-like receptors (TLRs), particularly TLR2 and TLR4, which are involved in innate immune response. This activation leads to increased production of inflammatory mediators such as tumor necrosis factor-alpha (TNF-α) and interleukins (IL-1β, IL-6), further exacerbating vascular inflammation. 27 The chronic inflammatory state in DM also involves the recruitment and activation of immune cells, such as macrophages and lymphocytes, which infiltrate the vascular tissue and contribute to the pro-inflammatory environment. 25 This persistent inflammation can lead to vascular complications, including IV. Our study establishes a strong association of DM with IV as well.
A strong association of cardiovascular diseases (CVD), such as CAD, CHF, peripheral arterial disease, and CVA with IV has been widely reported.28,29 Patients with IV, especially AAV, Takayasu arteritis, and GCA, demonstrate a significantly increased risk of CVD, such as CAD, CVA, and venous thromboembolism. Risk estimates range from 1.6- to over 3-fold higher than the general population, with the risk being particularly elevated around the time of diagnosis and during periods of active disease.30 -33 Cardiac involvement in IV contributes to increased morbidity and mortality, hence early recognition and intervention are critical, as subclinical cardiovascular involvement is frequent and may precede overt events.34 -36 CVD happens to be the consequence of vasculitis and vascular damage from chronic vascular inflammation which results into a sustained procoagulant state and accelerated atherosclerosis. 28 Other studies assessed coronary microvascular dysfunction (CMD) on myocardial tissue perfusion by assessing the coronary flow reserve (CFR) in patients with IV, which provided a measure of the integrated hemodynamic effects of epicardial coronary artery disease, diffuse atherosclerosis, and vessel remodeling.29,37 They reported significantly impaired CFR and CMD in patients with IV even though these patients had lower coronary artery calcium scores, indicating that even though the patients with IV did not have evidence of a higher atherosclerotic burden, nevertheless they had more coronary vasomotor dysfunction. 29
We found a strong association of pulmonary disorders, such as COPD, asthma, and pulmonary fibrosis with IV. Several IVs have strong association with pulmonary disorders, particularly the AAV. Pulmonary involvement in these conditions can manifest as diffuse alveolar hemorrhage, interstitial lung disease (ILD), pulmonary nodules, and pleural effusions.38 -40 In GPA, pulmonary manifestations are common, with up to 90% of patients experiencing respiratory tract involvement, including nodules, cavitations, and diffuse alveolar hemorrhage. 40 MPA frequently presents with ILD, particularly with a usual interstitial pneumonia (UIP) pattern, and is often associated with myeloperoxidase (MPO)-ANCA positivity.39,41 EGPA is characterized by asthma and eosinophilic lung infiltrates, which can precede systemic vasculitis by several years. 40 Pulmonary involvement in large and medium vessel IV such as Takayasu arteritis and GCA, is less common but can include pulmonary artery aneurysms and pulmonary hypertension. 42 Similarly, Behçet’s disease is associated with pulmonary artery aneurysms and in situ thrombosis. 40 The pathogenesis of pulmonary involvement in IV involves immune dysregulation, leading to vascular inflammation and tissue damage. ANCA and anti-GBM autoantibodies, neutrophil activation, and the formation of neutrophil extracellular traps (NETs) play significant roles in this process. 43
Although there is a strong association of asthma and pulmonary fibrosis with IV, the association with COPD is not well established. While both COPD and IV involve significant inflammatory processes, there is no direct evidence in the literature to suggest a specific association between COPD and IV. The immune responses and inflammatory pathways in COPD, such as the presence of autoantibodies and T-cell responses to lung antigens, suggest potential for autoimmune mechanisms. 44 However, these mechanisms do not directly link COPD to IV. Our finding of a strong association of COPD and IV is unique, which has not been reported so far.
Osteoporosis is associated with IV due to the direct effects of chronic vascular inflammation which can lead to increased bone resorption and decreased bone formation, and the adverse impact of glucocorticoid therapy for IV on bone health.45,46
The association of OA with IV has not been well reported; however, we found a strong association. One study suggests an association between vascular pathology and the risk of hand and knee OA, with a potential causal relationship for knee OA. 47 Several intersecting pathophysiological aspects may explain such association, such as presence of inflammatory mediators and endothelial dysfunction are common in both OA and IV.48,49 Rheumatological disorders, such as RA, SLE, systemic sclerosis, Sjögren’s syndrome, and idiopathic inflammatory myositis are frequently associated with IV, hence classified as “vasculitis associated with systemic disease.”50 -52 Our findings align with the existing literature.
Additionally, we found a higher prevalence of certain medications in the IV group, particularly hydralazine and TNF-α inhibitors. Hydralazine has been linked to drug-induced vasculitis, particularly AAV, and TNF-α inhibitors have been associated with paradoxical inflammatory reactions. Hydralazine is among the drugs most frequently linked to drug-induced AAV, with a notably high reporting rate in pharmacovigilance databases. 53
The limitations of our study include the fact that being a retrospective study, we had to solely rely on the documentation of the clinicians regarding the presence or history of IV and the comorbidities. Additionally, being a single center study limits the generalizability of our findings as our findings might not represent the variability of comorbid medical conditions in different settings as well as other demographic profiles, and our findings could not include the differences in the specific social determinants of health in other settings, such as access to healthcare. The major strength of our study was a large database of patients who followed over decades in a single healthcare institution in which the patient records were available to all clinicians who could accurately document the onset of the diagnosis or history of IV and each variable over a long period of time.
Conclusion
We conclude that IV is associated with older age, female sex, non-White race, tobacco use, recreational drug use, hypertension, DM, cardiovascular disorders (CAD, CHF, PAD, CVA), pulmonary disorders (COPD, asthma, pulmonary fibrosis), osteoporosis, osteoarthritis, SLE, and rheumatoid arthritis. Further research is needed to explore the mechanisms of the comorbidities, optimization of the modifiable risk factors, and identification of therapeutic targets which may offer better patient care and outcomes.
Footnotes
Informed Consent
Not applicable. Being a retrospective chart review study, the Institutional Review Board waived the need for informed consent.
Author Contributions
IM and SR made substantial contributions to the study design, drafting, data acquisition, data analysis, and manuscript writing. All authors contributed to data collection and manuscript writing. KH analyzed the data. SR contributed by revising the manuscript critically for improved intellectual content, and final approval for the version to be published.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The authors declare that data supporting the findings of this study are available within the article.
