Abstract
Objectives:
To evaluate the performance of noninvasive, elastic scattering spectroscopy, algorithm-powered device (DermaSensor) to detect melanoma and basal and squamous cell cancers in the primary care setting.
Patients & Methods:
DERM-SUCCESS, a blinded, prospective, multicenter pivotal study, enrolled adult patients between August 17, 2020, and December 9, 2021, with lesions that their primary care physicians (PCPs) suspected of skin cancer at clinics in the US (n = 18) and Australia (n = 4). These lesions were assessed by PCPs and scanned with the DermaSensor device. Biopsy specimens were collected, and histopathologic analysis was performed by dermatopathologists. The diagnostic performance of the device, dermatopathologist discordance, and subgroup analyses of clinical interest were calculated.
Results:
Of the 1579 skin lesions enrolled, dermatopathologic analysis identified 224 (14.2%) cancers. Device sensitivity was 95.5% (95% CI, 91.7%-97.6%) overall and 96.3% (92.9%-98.4%) for patients in the FDA-approved age group 40 years and older (90.2% for melanoma, 97.8% for basal cell carcinoma, and 97.7% for squamous cell carcinoma). Device specificity was 20.7%. The negative predictive value was 96.6%, and the positive predictive value was 16.6% (NNB 6). The device misclassified as “
Conclusions:
The DermaSensor device is an easy-to-use, point-of-care, hand-held skin cancer adjunctive diagnostic device with high sensitivity and NPV to help inform PCP decision-making about skin lesions suspicious for cancer that need further evaluation and those that may be monitored.
Keywords
Introduction
The incidence of basal cell carcinoma (BCC), squamous cell carcinoma (SCC) (collectively, keratinocyte carcinomas [KC]), and malignant melanoma is increasing worldwide. Skin cancer will be diagnosed in 1 of 5 Americans during their lifetime, making it the most common type of cancer in the US. 1 In 2022, an estimated 5.4 million cases of KC were diagnosed in 3.3 million people, and an estimated 2500 to 15 000 patients died of SCC.1 -4 An estimated 99 780 new cases of invasive melanoma and 97 920 cases of melanoma in situ were diagnosed in 2022, and 7650 people died of melanoma. 1 Most skin cancer is curable if diagnosed early, including KC, melanoma in situ, and nonulcerated malignant melanoma with a Breslow depth less than 1 mm. 1
Skin biopsy and subsequent histopathologic analysis is the standard for skin cancer diagnoses. 5 The number needed to biopsy (NNB) ratio of total biopsies to identify 1 histopathologic-confirmed skin cancer is commonly used to compare the diagnostic accuracy of providers who perform skin biopsy and is the inverse of positive predictive value (PPV). 6 Training and experience in dermatologic care correlates inversely with NNB and directly with diagnostic specificity and PPV.6 -8 One study at a large academic medical center found an NNB to find 1 melanoma of 14.33 for dermatologists, 27.80 for PCPs, and 53.56 for other primary care providers. 8 And, PCP sensitivity for correctly managing (i.e., refer or biopsy) skin cancer by naked eye examination is well below that of dermatologists, ranging from 54% to 88%.9,10
With only 1 dermatologist for every 30 000 people in the US, early skin cancer detection cannot depend on dermatologists. Improved PCP performance is necessary to improve detection and avoid unnecessary biopsies and referrals of benign lesions for biopsy. 11 Dermoscopy training greatly improves PCP sensitivity and specificity;10,12 -17 however, the requirements for additional training and ongoing practice to maintain dermoscopy competency has limited its use in the US to only 8% of PCPs.18,19 Despite availability of a range of education and algorithm interventions to improve PCP diagnostic ability, outcomes are inconsistent, difficult to replicate, and challenging to maintain. 20
The Nevisense system (Scibase) is an electrical impedance spectroscopy device approved by the US Food and Drug Administration (FDA) for melanoma detection (sensitivity, 96.6%; specificity, 34.4%). Its use is restricted to dermatologists and only lesions concerning for melanoma. 21 Because seborrheic keratoses generate high electrical impedance scores, they must be excluded via prescreening.
Artificial intelligence-assisted smartphone applications that assess clinical and dermoscopic images of skin lesions are becoming increasingly available though none are authorized for use by the FDA.22,23 Although such image-based software may eventually achieve dermatologist-level sensitivity and specificity,23 -26 current performance (early 2025) is below standard of care,27,28 increasing use of health care resources without added benefits.29,30 These applications are unable to evaluate cellular characteristics like ESS.
An affordable, automated point-of-care device is needed to improve PCP skin cancer detection and reduce unnecessary referrals for biopsy. Elastic scattering spectroscopy (ESS) is a noninvasive method for obtaining a spectral recording of photons scattered by chromophores in tissues, facilitating detection of cellular elements indicative of malignant neoplasms in skin 31 and other tissues.32 -36
Methods
Trial Design
The DermaSensor Study of Primary Care Physician Use of Elastic Scattering Spectroscopy (DERM-SUCCESS) was a prospective, blinded validation trial designed in collaboration with the FDA for clearance of a novel ESS, algorithm-powered device (DermaSensor device, manufactured by DermaSensor, Inc.) for evaluating lesions suggestive of skin cancer. The primary aim of the DERM-SUCCESS trial was to measure the performance of the device for lesions suggestive of melanoma, BCC, and/or SCC. The secondary aim was to compare device sensitivity to a performance goal of 90%, which is based on the published dermatologist sensitivity range of 81% to 96%.37 -40
This study was reviewed and approved by the 3 Institutional Review Boards, each overseeing separate sites. Written informed consent was obtained for all participants before enrollment. Study participants received a small stipend.
Setting
Participants were enrolled in a convenience sample of suburban and rural family medicine and internal medicine practices, 18 in the US and 4 in Australia, of experienced PCPs who conduct a high volume of skin exams and perform their own skin biopsies from August 17, 2020, through December 9, 2021. Nine of the 22 study sites were in towns with populations under 15 000 people.
Participants
All patients seeing study PCPs during study enrollment with a suspicious lesion who met eligibility criteria, and were willing to participate were enrolled (Figure 1). Eligibility criteria included patients aged 22 years or older with a skin lesion(s) suggestive of melanoma, BCC, and/or SCC (based on visual inspection by PCP) requiring biopsy. Excluded lesions included those less than 2.5 mm or greater than 15 mm in diameter (smaller lesions were excluded due to risk of the 2.5 mm probe tip placement on perilesional skin; larger lesions were excluded since the 5 recordings sample a limited volume of the lesional tissue); located on mucosal or acral skin, within 1 cm of the eye, at a previous biopsy or surgical site, or in acute sunburned skin; completely covered with thick crust, erosion, or ulceration (without a nonulcerated area ≥2.5 mm) or containing foreign matter. Dermoscopy was not used for lesion assessment. After target numbers of KCs were reached (see protocol below), participating PCPs were instructed to continue to enroll patients with pigmented lesions until the number of melanomas required by the FDA for this trial were enrolled. Participant sex, age, ethnicity, and self-reported race were recorded, and Fitzpatrick skin type was assessed by the patient and recorded by PCPs.

Flow diagram of included and excluded study data. The ITT population comprised all patients and lesions that were included in the safety analysis, and the mITT population comprised all patients and lesions that were included in the effectiveness analysis.
Device
The battery-operated, handheld DermaSensor device (Figure 2) emits pulses of light (200 microseconds/pulse) that span wavelengths from near-ultraviolet, through visible, to near-infrared. Two optical fibers are enclosed in a biocompatible 2.5-mm sterilizable fiberoptic tip that is placed in contact with the lesion surface. The volume of tissue evaluated with the device for each spectral recording was estimated computationally with Monte Carlo simulations to be approximately 0.7 × 0.4 × 0.5 mm (length × width × depth). Five spectral scans at various points on the lesion can be performed in about 10 s total time per lesion to record light reflectance of tissue structure and architecture (e.g., nuclear and chromatin characteristics) with the algorithm, then comparing those spectral readings to lesions in the training data set.

Elastic scattering spectroscopy device for analysis of suspected skin cancer lesions. An example patient number (#234-1234), with a negative monitor or positive investigate further result, is shown. To investigate further results, a representative spectral score and corresponding definition are shown. (© 2023 DermaSensor, Inc; used with permission.).
The device AI developed algorithm was trained on more than 10 000 spectral scans and 2000 lesions. In accordance with requirements by the FDA for a pivotal study, the algorithm was developed before study enrollment. The algorithm was upgraded and then used for analysis of the blinded dataset based on data from 3 other clinical trials before study completion. The device output to which investigators and dermatopathologists were blinded is either
Study Sequence
The PCPs electronically recorded a lesion(s) description. The lesion(s) was then scanned with the ESS device. Both PCPs and patients were blinded to the results. A biopsy specimen was collected according to the PCP standard of care and provided to an independent dermatopathologic core laboratory for histopathologic analysis. As with the PCPs, the dermatopathologists were blinded to device output.
All specimens were analyzed by 2 dermatopathologists who had to independently reach diagnostic consensus using MPATH-Dx criteria for melanocytic lesions 41 Discordance (benign or malignant) between the first 2 dermatopathologists was adjudicated by a third dermatopathologist and by 2 additional dermatopathologists for lesions diagnosed by at least 2 of the initial dermatopathologists as highly atypical, with differing diagnoses (malignant, benign, or atypical) by each of the initial 3 dermatopathologists, and/or diagnosed as malignant by 1 dermatopathologist but not as malignant by the adjudicator. If the 2 additional dermatopathologists disagreed, the lesion was excluded from analysis. Patients’ participation in the study was complete after their enrolled lesion(s) was biopsied. All biopsy results were communicated to patients by their PCP and standard of care followed.
Data Analysis
Device sensitivity was tested with an α level of .025 and compared by using a method-of-moments approach for clustered matched-pair data
41
to account for possible within-patient correlation for patients with multiple lesions. The device’s ability to detect malignant lesions better than random chance (sensitivity + specificity > 1) was tested with generalized estimating equations logistic regression with a noninferiority margin of 10% by using a 1-sided exact binomial test with an α level of .025, with each patient treated as a random effect and assuming an exchangeable within-patient correlation structure to account for multiple within-patient lesions. The secondary hypothesis was tested for performance in comparison with a 90% threshold. Level of agreement between dermatopathologists was assessed with the Cohen κ statistic. For all other statistical inferences, 2-sided Wilson or exact confidence intervals were determined as appropriate, and unless specified,
Results
A total of 1028 patients (1598 lesions) consented to participate in the study (Figure 1), of whom 7 did not meet the inclusion criteria and 16 (19 lesions) were excluded because their lesions did not meet eligibility criteria, device deficiencies, or a dermatopathologic consensus was not reached. The resulting 1005 patients (1579 lesions, 1451 in the US and 128 in Australia) were included in our analyses.
Slightly more than half of participants were women (51.5%), and the majority (97.1%) were White (Table 1). Mean (SD) participant age was 58.5 (15.1) years, and 138 patients (13.7%) were younger than 40 years. Most participants (72.5%) had Fitzpatrick skin types I through III, with most (35.0%) having type III skin. Most patients had 1 (65.4%) or 2 (20.6%) eligible lesions analyzed in the study. Of 1579 lesions, 998 (63.2%) had darker pigmentation, and 581 (36.8%) had lighter pigmentation. Nearly two-thirds (60.6%) of lesions were elevated or papular, and 67.8% were discovered by the patient. Mean (SD) lesion size was 6.7 (2.9) mm in length and 5.2 (2.2) mm in width. All biopsy specimens collected were clinically suspicious for a malignant neoplasm. Histopathologic analysis by the dermatopathologists indicated that 1355 lesions were benign. Of 224 malignant lesions, 176 were KCs (90 BCC and 86 SCC) and 48 were melanocytic high-risk lesions (19 severely atypical nevi/atypical junctional melanocytic proliferations and 29 invasive and in situ melanomas) (Table 2). Of the 48 melanocytic lesions, 7 were on patients younger than 40 years. No KCs were identified for patients younger than 40 years. Severely atypical nevi, atypical junctional melanocytic, and melanoma in situ lesions were grouped in the analysis as high-risk melanocytic lesions with melanomas by FDA requirement to ensure referral to dermatology due to the close histopathological relationship and lack of reproducibility of diagnostic differentiation between them on histopathology.41,42
Patient (n = 1005) and Lesion (n = 1579) Characteristics.

Patient age, lesion length, and lesion width summarized as mean (SD), and all other variables summarized as No. (%) of patients or lesions.
Dermatopathologic Risk Classifications and Diagnoses (n = 1579).
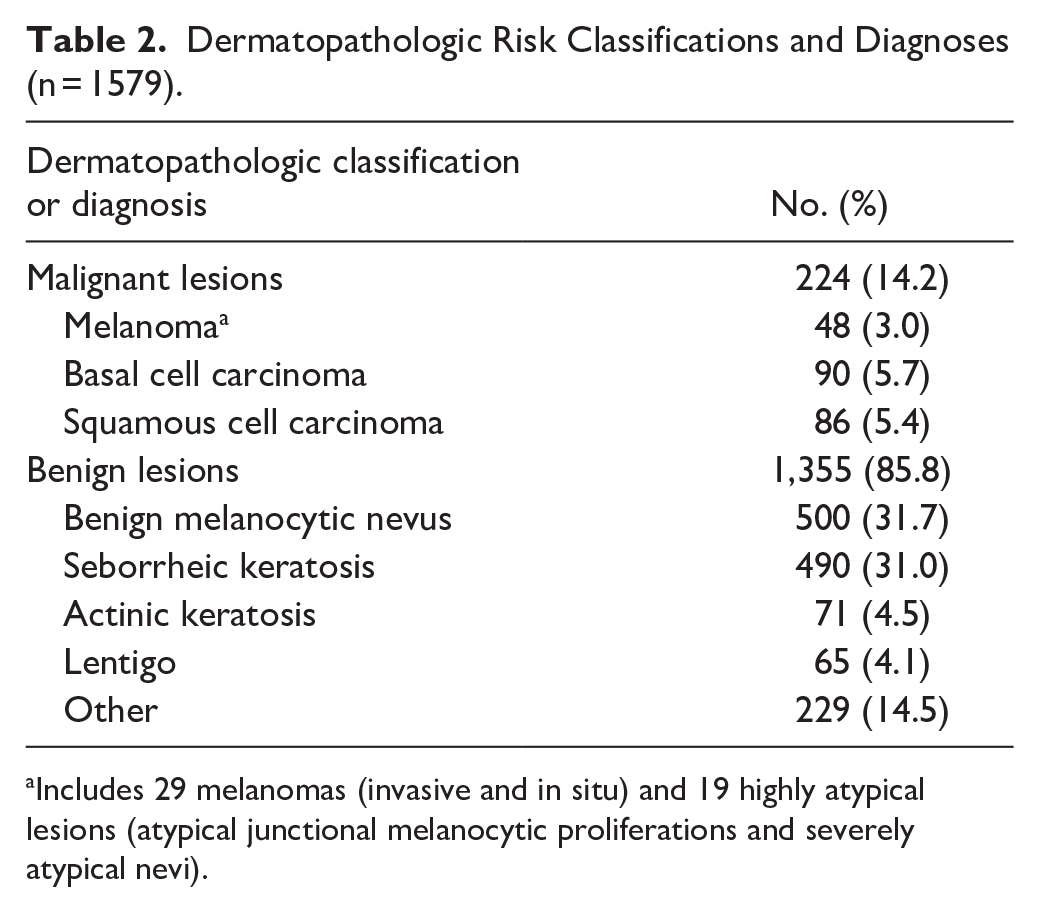
Includes 29 melanomas (invasive and in situ) and 19 highly atypical lesions (atypical junctional melanocytic proliferations and severely atypical nevi).
The level of agreement between the 2 core laboratory dermatopathologists was 81.3% (Cohen κ, –0.1; 95% CI, –0.2 to 0.0); they agreed that 39 of the 48 melanomas were melanoma (or its near-equivalent, e.g., severe atypia). One diagnosed 44 of 48 (91.7%) melanomas; the other diagnosed 43 of 48 (89.6%) melanomas. For patients 40 years and older, the sensitivity of the 2 dermatopathologists was 90.2% (37 of 41 melanomas). Thus, the device sensitivity of 90.2% for melanoma was identical to that of both dermatopathologists (i.e., all 3 correctly diagnosed 37 of 41 melanomas). Five lesions excluded from primary analysis because of a lack of consensus had various diagnoses on the spectrum from compound melanocytic nevi to severely atypical nevi. Exclusion of these lesions was documented before study database lock and unblinding of the study results.
Overall sensitivity of the ESS device for detecting skin cancer was 95.5% (95% CI, 91.7%-97.6%) (
Table 3
). For the FDA-approved target group of patients aged 40 years and older, the device sensitivity for skin cancer was 96.3% (95% CI, 92.9%-98.4%), including 90.2% (95% CI, 76.9%-97.3%) for melanoma, 97.8% (92.2-99.7%) for BCC, and 97.7% (91.9%-99.7%) for SCC. For all ages, device sensitivity was 87.5% (95% CI, 76.4%-93.8%) for melanoma. Patient-level sensitivity, defined as the proportion of participants with skin cancer for whom the device correctly detected at least 1 malignant lesion was 97.4% (95% CI, 94.2%-99.2%) for all skin cancers and 95.1% (95% CI, 83.5%-99.4%) for melanoma for patients aged 40 years and older (see Table 3). For patients aged 40 years and older, patient-level sensitivity was 97.4% (95% CI, 94.2%-99.2%) for all skin cancers and 95.1% (95% CI, 83.5%-99.4%) for melanoma for patients aged 40 years and older. When compared with the performance goal of 90%, device sensitivity was both noninferior (
Sensitivity of Device Using Dermatopathologic Diagnoses as Standard
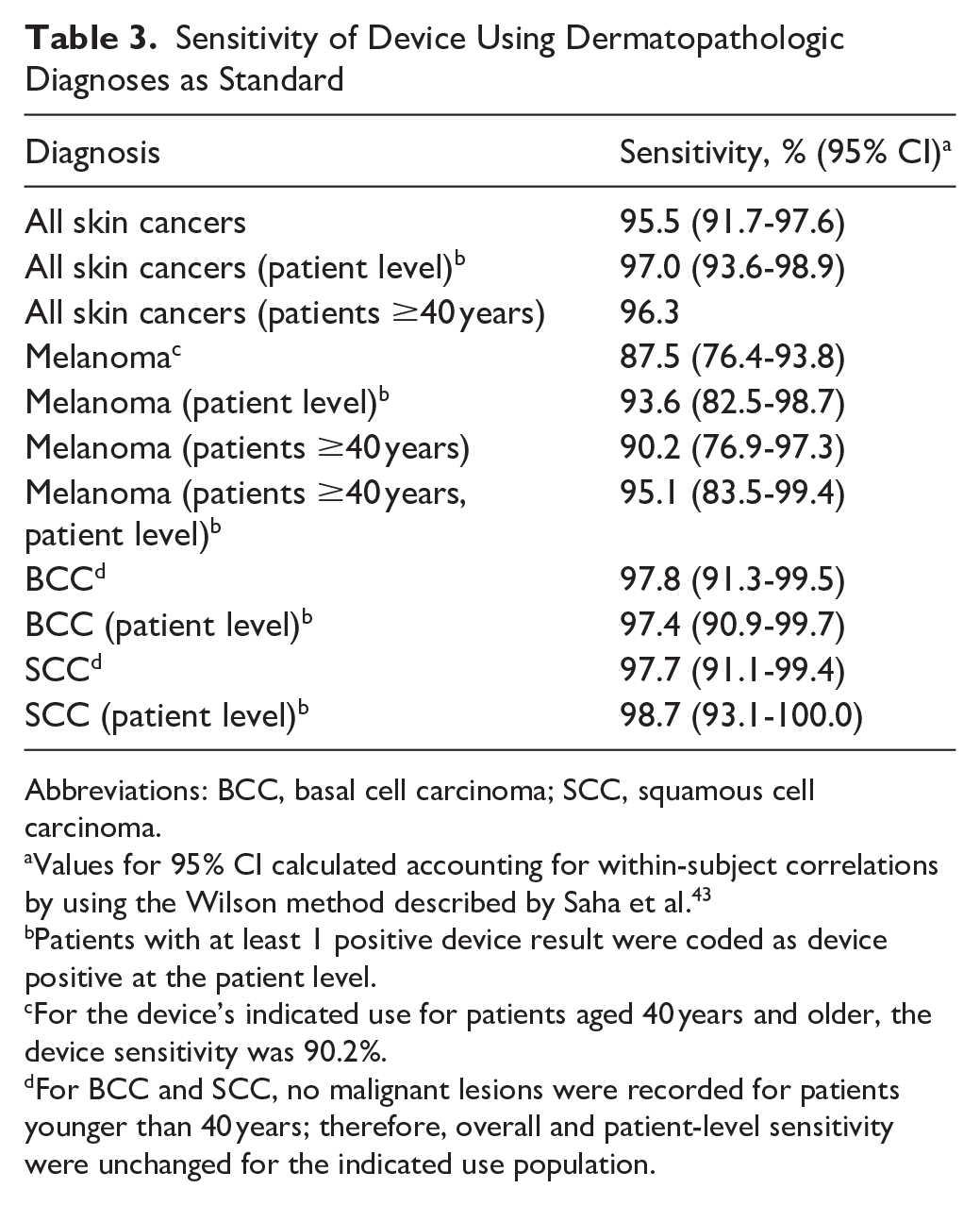
Abbreviations: BCC, basal cell carcinoma; SCC, squamous cell carcinoma.
Values for 95% CI calculated accounting for within-subject correlations by using the Wilson method described by Saha et al. 43
Patients with at least 1 positive device result were coded as device positive at the patient level.
For the device’s indicated use for patients aged 40 years and older, the device sensitivity was 90.2%.
For BCC and SCC, no malignant lesions were recorded for patients younger than 40 years; therefore, overall and patient-level sensitivity were unchanged for the indicated use population.
Clinical Characteristics and Histopathologic Findings for Missed Melanomas.

The device algorithm internally generates a numeric value based on a spectral score between 0 and 1, which is then converted to the device output of either a negative result of
Although the dataset included fewer patients with Fitzpatrick skin type IV through VI (27.5%), no meaningful differences in sensitivity and specificity were evident between patients with Fitzpatrick skin type I through III (sensitivity, 96.5%; 95% CI, 92.6%-98.7%; specificity, 18.7%; 95% CI, 16.2%-21.5%) and IV through VI (sensitivity, 92.2%; 95% CI, 81.1%-97.8%; specificity, 25.1%; 95% CI, 20.9%-29.7%).
The AUROC curve for the device was calculated as 0.7796 which would be classified as
The overall NPV of the device was 96.6% (95% CI, 93.5%-98.2%), which indicates that a lesion with a device result of

Positive Predictive Value (PPV) According to Spectral Score for Investigate Further Results. Device PPV ranged from 6.4% (95% CI, 3.7%-10.9%) for the lowest spectral score of 1 to 61.3% (95% CI, 48.3%-72.9%) for the highest score of 10. The device PPV was 7.4% (95% CI, 5.5%-9.9%; NNB, 13.5) for low (1-5) spectral scores and 33.2% (95% CI, 28.8%-37.9%; NNB, 3.0) for high (6-10) scores (
Discussion
The DermaSensor device’s 96.3% sensitivity for detecting skin cancer and 96.6% NPV classifies it a very good diagnostic device for risk stratifying suspicious skin lesions. The device was approved by the FDA in January 2024 based on this data for skin cancer risk assessment for patients 40 years and older, making it the first such device available for use by PCPs. 23
Since spectral scores are directly related to increasing malignancy risk, the device may find good utility for prioritizing timing of further care (e.g., urgency of referral to dermatology). The NNB for all skin cancers in this data set skewed towards a high percentage of melanocytic lesions was 7.1 for PCPs and 6.0 for the device. If PCPs had been unblinded and device output used to inform biopsy decisions, they would have decreased their biopsy frequency in the approved 40 years and older population by 18.4% (1288 vs 1579 biopsies) and would have monitored rather than investigated further 3.7% of cancers (4 KCs and 4 melanomas or 0.5% of lesions). For the 37% of
The device correctly classified 90.2% of melanomas as
Patient-level device sensitivity was 95.1% for the device to read
By analyzing ESS data from suspicious lesions, the device provides immediate risk information to the PCP to inform lesion management. This may improve referrals by PCPs and enhance referral adherence by patients. Pigmented lesions of concern for melanoma with a
Limitations
Our study was limited by the exclusion criteria (e.g., fully crusted lesions which may have excluded some squamous cell cancers and very small and large lesions) and included only lesions identified as concerning and warranting biopsy by PCPs who frequently perform biopsies for skin cancer and who were blinded to device results. Enrollment of only lesions suspicious for melanoma after achieving the target sample size for BCC and SCC resulted in a high proportion of melanocytic lesions (about 2/3) making overall comparisons of NNB to an average PCP practice inaccurate. Other limitations of our study included a patient population that was only 12.7% Fitzpatrick V-VI.
Conclusions
The DermaSensor device is an easy-to-use, point-of-care, hand-held, AI-developed, algorithm-powered skin cancer adjunctive diagnostic device with high sensitivity and NPV for use in the primary care setting. PCPs can use the device to prioritize skin lesions that merit further evaluation and those that may be monitored.
Footnotes
Acknowledgements
We commend Joseph Massaro, PhD (deceased), for writing the initial study statistical plan and for his contributions to the study design. We acknowledge Irving Bigio, PhD, Eladio Rodriguez-Diaz, PhD, and Preston Hoang, MS, for their contributions developing the device technology. We thank Na Wang, PhD, Margaret Shea, Kanisha Mittal, Lindsey Furton, Kaitlin Hartlage, Cason Hucks, and Matthew Bullard for their contributions to the statistical analyses. We also thank Phillip Leboit, MD, Maxwell Fung, MD, and Jennifer McNiff, MD, for serving as expert dermatopathology panelists for this study. All persons acknowledged above or their institutions were compensated for their contributions to the study. We especially thank all study participants and investigators; without their participation, this study would not have been possible.
Cody Simmons, MS (cofounder and chief executive officer of DermaSensor, Inc), and Kiran Chatha, MD, MPH (DermaSensor, Inc employee), reviewed the manuscript and provided data verification and validation support. Nisha Badders, PhD, ELS, Mayo Clinic, provided editorial suggestions on an earlier draft of the manuscript
Authors’ Note
Subsets of the data were presented as posters at (1) the 2nd Annual Innovations in Dermatology Fall Conference; November 3-5, 2022; Las Vegas, Nevada; (2) Maui Derm Hawaii 2023; January 23-27, 2023; Maui, Hawaii; presented as (3) a podium presentation at the 11th Annual American Dermoscopy Meeting; July 13-15, 2023; Stowe, Vermont; (4) as a podium presentation at the American Academy of Dermatology Innovation Academy Meeting; August 10-13, 2023; Tampa, Florida; and (5) the Skin of Color Update conference; October 6-8, 2023; New York, New York. Another subset of the data was presented as a podium presentation at the Society of Teachers of Family Medicine (STFM) Annual Spring Conference; April 29-May 3, 2023; Tampa, Florida.
Portions of this manuscript have been published in abstract form: Cutis. 2022;110(6 Suppl):31; J Clin Aesthet Dermatol. 2023;16(4 Suppl 1):S16; and J Am Acad Dermatol. 2023;89(3 Suppl):AB233.
ORCID iDs
Ethics and Considerations
This study was reviewed and approved by the Western Institutional Review Board-Copernicus Group Review Board (#20182730), the Australian Bellberry Human Research Ethics Committee (#2018-08-620), and the Mayo Clinic Institutional Review Board (#20-011463).
Consent to Participate
Before enrollment, written informed consent was obtained for all participants. Study participants received a small stipend.
Author Contributions
Concept and design: Leffell
Acquisition of data: Merry, Croghan, McCormick, Considine, Thompson
Analysis and interpretation of data: Dukes, Merry, Croghan, Leffell
Drafting of the manuscript: Merry, Croghan
Critical revision of manuscript: Duvall, McCormick, Considine, Thompson, Leffell, Dukes
Statistical analysis: Dukes
Obtained funding: The institutions of Dukes, Merry, Croghan, Duvall, McCormick, Considine, and Thompson received funding for conducting the clinical trial
Administrative, technical, and material support: Thompson
Study supervision: Leffell
No use of artificial intelligence-assisted technologies were used in the production of this manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by DermaSensor, Inc. The sponsor designed the study and hired a contract research organization to oversee the study conduct and data collection and management. Data analysis was completed by an external biostatistics group at Boston University. The sponsor did not prepare the manuscript but did review it.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs Merry and Croghan conducted the study at Mayo Clinic in Rochester, Minnesota, with the support of the sponsor DermaSensor, Inc. Dr Dukes performed statistical analysis under contract from the sponsor at Boston University School of Public Health. Drs McCormick and Considine were site principal investigators at their respective family medicine private practices in Virginia and Australia. Dr Thompson performed dermatopathologic analyses under contract. None have received personal remuneration from the company. Dr Leffell is a member of the DermaSensor Scientific Advisory Board and is compensated by the company.
Abbreviations
AI = artificial intelligence
AUROC = area under the receiver operating characteristic
APP = advanced practice provider
BCC = basal cell carcinoma
DERM-SUCCESS =
ESS = elastic scattering spectroscopy
FDA = US Food and Drug Administration
KC = keratinocyte carcinoma
NNB = number needed to biopsy
NPV = negative predictive value
PCP = primary care physician
PPV = positive predictive value
SCC = squamous cell carcinoma
Trial Registry Information
Trial registration: NCT06690086
Data Availability Statement
All data supporting the study findings are contained in this manuscript. Kimberly A. Dukes, PhD, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Stephen P. Merry, MD, MPH, takes responsibility for the integrity of the work as a whole, from inception to published article.
