Abstract
Introduction:
Continuous Glucose Monitors (CGMs) offer critical insight into glucose trends, aiding significantly in overall type 2 diabetes (T2DM) management. Few studies have evaluated pharmacist involvement in CGM management.
Methods:
This was a retrospective study, conducted at two primary care offices within a community health system. The aim of this study was to assess pharmacist impact on the deprescribing of high-risk medications in patients with T2DM utilizing CGM data. The primary outcome was the percentage of patients that experienced deprescribing of a high-risk medication (defined as reduction or discontinuation in total daily dosage of insulin, sulfonylureas, and thiazolidinediones). The secondary outcomes were rate of hospitalizations and changes in total daily insulin dose. Chi-square tests and t-tests were utilized to analyze primary and secondary outcomes.
Results:
Among 317 participants, 58% of patients on CGMs had pharmacists involved in their care. Of patients in the pharmacist-led group, 11.4% experienced deprescribing of a high-risk medication compared to about 8.3% in the usual care group. Overall, hospitalizations were 3.2% lower in the pharmacist-led group compared to the usual care group during the study period. In addition, patients in the pharmacist-led group experienced a reduction in total daily insulin dose, while an increase in total daily insulin dosage for the usual care group was observed.
Conclusion:
While our study did not find a statistically significant difference in pharmacist-led deprescribing, there was a trend towards reduction in high-risk medication use. This suggests potential clinical significance, emphasizing the role of pharmacist involvement in prescribing practices of medications used to treat T2DM, including deprescribing high-risk medications and initiating non-high-risk medications with additional benefits. Further studies are needed to determine a difference in prescribing practice in pharmacist-led management of T2DM.
Background
Approximately 130 million adults in the United States are currently living with either diabetes or prediabetes, with an additional 1.2 million new diabetes cases diagnosed among individuals aged 18 years and older in 2021. 1 Uncontrolled diabetes leads to detrimental economic effects contributing to increased health care expenditures, particularly spending on inpatient hospital stays and prescription medications. In 2022, the total estimated cost of diabetes and associated complications in the United States was $412.9 billion. 2 One of the more recent and valuable advancements in the field of diabetes management is continuous glucose monitoring devices (CGMs). CGMs provide valuable insights into glucose trends, aiding in more effective management for individuals with difficulty controlling diabetes. The current 2024 American Diabetes Association (ADA) guidelines recommend CGM devices for all adults prescribed insulin for the treatment of diabetes. 3 In addition, the ADA recommends initiating therapy based on comorbid disease states, emphasizing the importance of minimizing the use of high-risk medications like insulin when possible. However, the complexity and rapid evolution of the diabetes technology landscape can lead to barriers in implementation and management for people with diabetes, their care partners, and the health care team.
Multiple studies have demonstrated the positive impact of pharmacists involved with managing diabetes in A1c reduction through pharmacist-led visits.4,5 A retrospective cohort study identified adult patients with uncontrolled type 2 diabetes mellitus (T2DM), defined as an A1c greater than or equal to 8%, and showed statistically significant reductions in A1c for patients with pharmacist-led visits as opposed to visits without a pharmacist involved. 4 Another study evaluated the clinical utility of CGMs monitored by a pharmacist. This retrospective analysis determined pharmacist-led visits in T2DM patients significantly reduced A1c within 6 months of implementation of a CGM. 6 In addition, pharmacists also provide education to patients transitioning to other healthcare settings with the goal of avoiding medication errors, hospital readmissions, and improving medication adherence.
A 2022 study aimed to review and update the diabetes-specific aspects of the 2009 Evaluation and Management of Adult Hypoglycemic Disorders: Endocrine Society Clinical Practice Guideline. 7 It concluded that real-time continuous glucose monitoring should be used rather than no CGM for outpatients with T2DM who take insulin and/or sulfonylureas and are at risk for hypoglycemia. This finding underscores the importance of integrating CGMs into diabetes management strategies, potentially further supporting pharmacists in their efforts to optimize diabetes care, including the crucial aspect of de-prescribing high-risk medications.
Along with several other clinical disease state evaluations and the treatment of those chronic diseases, ambulatory care clinical pharmacy specialists (CPS) within our health system are uniquely positioned to de-prescribe high risk medications for the treatment of T2DM. Collaborative drug therapy management (CDTM), a type of collaborative practice agreement, is a tool used to provide these patient care services. Because data has shown significant improvements compared with standard of care related to pharmacist-driven therapeutic outcomes, CDTM has been adopted as a valuable component of our workflow. 8 Herein, we describe a process and workflow of pharmacist driven CGM initiation and subsequent management (Figure 1). This study describes a pharmacist driven workflow that allows for remuneration, rather than a singular pharmacist intervention. The current workflow at the study sites surrounding CPS involvement in the CGM procurement process involves several structured steps to ensure optimal patient outcomes. Patients eligible for CGM are identified during their Medicare Annual Wellness Visit (AWV) conducted by the CPS or during a routine visit with their physician. Ideally, once identified, the primary care provider communicates the need for a CGM to the CPS if the CPS was not involved in the original identification. The days that a CPS is scheduled to work at the individual sites varies. Subsequently, not all patients prescribed CGMs have CPS involvement in their care process related to diabetes management from CGM data. In this study, patients with CPS involvement in their CGM management were compared to those that did not have CPS involvement in their care. When involved in procurement, the CPS conducts a formulary lookup to confirm the patient’s insurance eligibility to obtain a CGM device. Upon verification and depending on the specific insurance coverage of the individual patient, an order is placed through a durable medical equipment (DME) ordering platform or a prescription is sent to the pharmacy. If the CGM is obtained through the DME ordering platform, a representative from the DME company contacts the patient to discuss copay details and shipping information. If the patient consents, the CGM device and supplies are shipped directly to the patient’s home. Subsequently, the patient has the option to meet with the CPS for setup of the CGM device, syncing the device sensor with a smartphone or providing setup for the CGM reader when applicable, and reviewing goals and treatment of diabetes. Follow-up appointments are conducted either in person or over the phone, utilizing platforms such as Dexcom Clarity or Freestyle Libre portal to monitor glucose readings and adjust treatment plans as needed.

Workflow diagram: pharmacist integration of CGM into diabetes management.
This comprehensive workflow allows successful implementation and management of pharmacist-driven CGM services for patients with diabetes using CDTM. While the demand for primary care providers across the United States outpaces supply, CDTM provides a way to bridge potential gaps in diabetes management, and allows utilization of CGM visits as an opportunity for pharmacotherapy optimization. 8 Given the rising prevalence of diabetes and the innovative nature of CGM technology, we aim to investigate the influence of pharmacist involvement in CGM on the prescribing practices of medications used to treat T2DM. Specifically, as the guidelines and literature support the utilization of CGMs in patients with diabetes on high-risk medications, this study aims to evaluate how pharmacist-led utilization of CGM can impact pharmacotherapy decisions surrounding these high-risk medications. Until pharmacists can consistently bill major payers for their services, it is imperative to establish reproducible workflows to carry out beneficial clinical initiatives. Pharmacists are able to perform certain duties related to CGM set-up and patient education that can be directly linked to billable services in the United States. Subsequently, this study additionally aims to describe in detail a successful workflow in which pharmacists play an integral role in assisting patients with the procurement of CGMs and management of CGM data as it relates to pharmacotherapy decisions.
Methods
Study Design
This study was a retrospective, observational, chart review comparing pharmacist-led intervention to usual care as it relates to management of CGM data in patients with T2DM.
Study Setting
The study was conducted within two primary care offices of a not-for-profit comprehensive community health network, with a total of nine billing providers.
Study Population
Patients who met all of the following criteria at any time within the study period were eligible for inclusion: an age of at least 18 years; documented diagnosis of T2DM based on prescriber diagnosis code; prescribed an approved personal CGM device including Freestyle Libre 14 day, Freestyle Libre 2, Freestyle Libre 3, Dexcom G5, Dexcom G6, or Dexcom G7; prescribed an FDA-approved glucose-lowering pharmacotherapy regimen during the study period; documented visit by CPS or provider in St. Joseph’s/Candler Health System electronic medical record (EMR) focused on T2DM; Patients who were pregnant or diagnosed with type 1 diabetes were not eligible for inclusion in the study. Those managed solely by a physician were included in the usual care group and any patient with at least 1 documented pharmacist-interaction met inclusion for the pharmacist-led group.
Outcomes
Patients were included in the study if they met all inclusion criteria at any time within the study period from June 1, 2021 to June 30, 2023.
The primary outcome of the study was the percentage of patients in each group that experienced a de-prescribing event with regards to high-risk medications in the treatment of T2DM. To obtain the primary outcome, patient prescription information was collected and documented for those prescribed high-risk medications. For patients prescribed high-risk medications at the start of the study, the name, strength, and daily dose were recorded, including total daily units of insulin or total daily milligrams of sulfonylurea and/or TZD. This information was also recorded at the end of the study.
Secondary outcomes included the incidence of all-cause hospitalizations and 30-day hospital readmission. Hospitalizations were documented if reported within our health system records, noting the date and reason for each hospitalization. To capture 30-day hospital readmissions, any additional hospitalization within 30 days of the initial hospitalization was recorded. Changes in total daily doses of insulin (in units) was also recorded.
Statistical Methods
Data compilation included patients who had been de-prescribed a high-risk medication, defined as either discontinuation of or reduction in the strength of the medication by the end of the study.
The occurrence of the primary outcome, defined as any de-prescribing event or dosage decrease of a high-risk medication for the treatment of T2DM was analyzed using chi-square analysis. To quantify the change in total daily insulin dosage, a t-test was performed for analysis.
Two sets of computer-generated reports were compiled using an EMR and a durable medical equipment (DME) ordering platform. Within the EMR, patient data was extracted, and continuous glucose monitor prescriptions were filtered to include the six specified devices. The CGM devices included were the Freestyle Libre 14 day, Freestyle Libre 2, Freestyle Libre 3, Dexcom G5, Dexcom G6, and Dexcom G7. Prescription dates were further filtered to align with the study period, which spanned from June 1, 2021 to June 30, 2023. A subset data filter was then applied to identify patients with history of an appointment at either of the two study center locations. Type 1 diabetic and pregnant patients were subsequently excluded from the analysis. A total of 318 patients met the inclusion criteria at the first clinic location and 167 patients met inclusion criteria for the second clinic location. Additionally, 205 patients were excluded due to duplicate patient charts or were not treated with an FDA approved diabetes pharmacotherapy agent during the study period.
Similarly, the DME ordering platform was utilized to compile a data report by filtering the timeline of the study period, which spanned from June 1, 2021 to June 30, 2023. Using a subset filter for diabetic supplies, the 6 specified CGM devices were then included. Type 1 diabetic and pregnant patients were also removed from this dataset. A total of 57 additional patients met inclusion criteria. Additionally, 20 patients were excluded either due to duplicate patient information, not on a Food and Drug Administration (FDA) approved diabetes pharmacotherapy agent, or no longer on a CGM device. Patient demographics such as gender, age, weight, height, and medications during the study period were collected. Comorbidity data including chronic kidney disease, heart failure, and atherosclerotic disease were collected.
Results
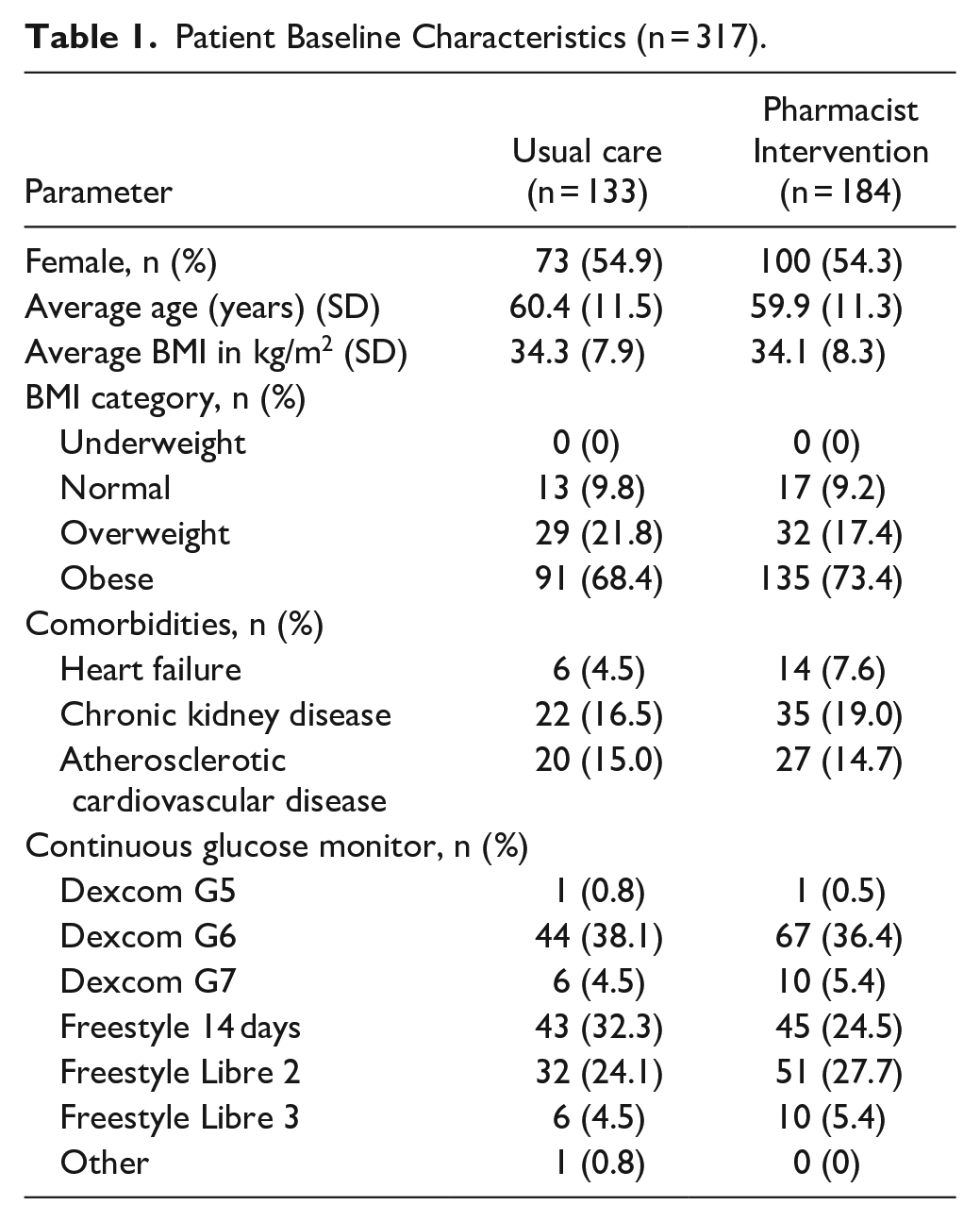
The study included a total of 317 participants. Baseline characteristics of the 317 patients included in the study are further described in Table 1. There was a total of 184 participants (58%) in the pharmacist-led group compared to 133 participants (42%) receiving usual care. Among those receiving usual care, 54.9% were female, with an average age of 60.4 years and an average BMI of 34.3. Prevalence of comorbidities was noted as follows: 4.5% had heart failure (HF), 16.5% had chronic kidney disease (CKD), and 15.0% had atherosclerotic cardiovascular disease (ASCVD). The most commonly prescribed glucose monitoring devices were Dexcom G6, followed by Freestyle 14 day, and Freestyle Libre 2. For patients with pharmacist intervention, 54.3% were female, with an average age of 59.9 years and an average BMI of 34.1. Rates of comorbidities were slightly higher compared to the usual care group: heart failure (7.6%), CKD (19.0%), and ASCVD (14.7%). Dexcom G6 was the most utilized device, followed by Freestyle Libre 2, and Freestyle Libre 14 day. There were no statistically significant differences in baseline demographics.
Patient Baseline Characteristics (n = 317).
Primary and secondary outcome results are noted in Table 2. In the pharmacist-led group, 11.4% of participants were de-prescribed a high-risk medication compared to 8.3% in the usual care group (P = .359). Of the 75 total hospitalized patients, six patients were hospitalized for diabetes related events. One patient in the pharmacist-led group was hospitalized for hypoglycemia. The remaining 5 patients were in the usual care group and consisted of one patient hospitalized for hypoglycemia, two patients hospitalized for diabetic ketoacidosis, and two patients hospitalized for hyperglycemia. Of the total patients hospitalized during the study, a total of 6.7% were readmitted within 30 days of initial hospitalization, two patients in the usual care group and 3 patients in the intervention group. Insulin was the most commonly prescribed high-risk medication, and the mean change in the total daily dose was decreased in the pharmacist-led group by −2.8 units compared to an increase in the usual care group (+4.8 units). The number of prescribed non-high-risk medications was similar between groups, with metformin being most common in usual care (44.7%) and GLP-1 agonists in the pharmacist-led group (41.6%).
Primary and Secondary Outcomes Results.

Discussion
Medications collected during the study period included high-risk and non-high-risk pharmacotherapy for diabetes management. High-risk pharmacotherapy to manage diabetes was defined as medication leading to hypoglycemia and/or weight gain. This included insulin, sulfonylureas, and thiazolidinediones (TZDs). Insulin promotes glucose uptake into cells, decreasing glucose which may lead to hypoglycemia and weight gain due to increased glycogen and fat storage. 8 Sulfonylureas stimulate insulin release, increasing the risk of hypoglycemia and promoting weight gain through insulin’s anabolic effects. TZDs enhance insulin sensitivity, which may cause hypoglycemia if not balanced with reduced insulin secretion or increased glucose production, and may lead to weight gain by promoting fat storage.8,9 Non-high-risk pharmacotherapy were defined as medications with low risk of hypoglycemia or weight gain. These included sodium-glucose cotransporter-2 inhibitors (SGLT2i), glucagon-like peptide-1 agonist (GLP-1 agonist), dipeptidyl peptidase-4 inhibitor (DPP4i), and metformin. 10
The study found no statistically significant difference in primary or secondary outcomes between the usual care and pharmacist-led groups. However, the pharmacist intervention group showed a higher percentage of patients who were de-prescribed a high-risk medication and a lower percentage of patients hospitalized during the study period. Specifically, for insulin there was a reduction in total daily units among participants who received pharmacist interventions compared to the usual care group.
Additionally, participants who received CPS interventions showed a 1.7% increase in the prescribing of non-high-risk medications compared to the usual care group. The increase in non-high-risk pharmacotherapy may contribute to improved glycemic control, reducing the risk of complications associated with uncontrolled diabetes such as hospitalizations and hypoglycemia. Additionally, many non-high-risk medications offer cardiovascular benefits, particularly SGLT2 inhibitors and GLP-1 agonists, which have shown reduction of major adverse cardiovascular events in high-risk patients. Furthermore, the weight management effects of GLP-1 agonists and the cardiovascular and renal protective properties of SGLT2 inhibitors given their FDA approval for HF and CKD, add to the overall benefits of this approach in patients with or without T2DM. 11 Therefore, this study highlights how pharmacists may simultaneously decrease high-risk medication use while promoting alternative therapies with multifactorial benefits for individual patients. The results of our study are aligned with previous studies that have shown the benefit of pharmacist involvement in high-risk T2DM patients. Halalau et.al. previously found the average decrease in A1c for patients with pharmacist care compared to the control group at 6 months was −2.85% versus −1.32%, (P = .0051). 12 An additional meta-analysis analyzed pharmacist impact on various lab values, including A1c. This study found that overall, pharmacist care was thought to reduce A1c by an average of −1.07% (95% CI [−1.32, −0.83]; P < .001). 13 While these previous studies established that pharmacists can successfully reduce A1c in high risk T2DM patients, our study further specifies how pharmacists can additionally reduce high-risk medication and increase utilization of non-high-risk medications while contributing to reimbursable services through CDTM utilization.
Study limitations include a small sample size which may have contributed to the lack of statistical significance in the study findings. Additionally, most patients within the study had multiple visits during the study period with their provider and the pharmacist (if applicable). It is difficult to discern whether or not each individual deprescribing event was solely attributed to pharmacist-driven intervention. Nevertheless, the absolute reduction in high-risk medication use in the pharmacist group compared to the usual care group suggests that adding pharmacists to the care team for patients with T2DM utilizing CGMs can improve pharmacotherapy optimization and potentially decrease hospitalization rates. Pharmacists have demonstrated a key role in the procurement process of CGMs for patients in the workflow described at this institution. The workflow highlighted within presents an opportunity for other institutions to initiate reproducible workflows for pharmacists to adjust pharmacotherapy under CDTM agreements in response to CGM data for patients with T2DM. While some states allow pharmacists to bill for clinical services, very few opportunities exist for direct billing due to lack of provider status under Medicare Part B. Because of this, it is essential to identify opportunities where pharmacists can contribute to billable services under the provider while utilizing their clinical training and expertise to promote safe and effective medication use.
Conclusion
These findings suggest the potential benefits of pharmacist-driven interventions in medication management through CDTM utilizing CGM data, particularly in the management of T2DM. The reduction of high-risk medication use in the treatment of T2DM aligns with recent guideline updates and presents a reproducible visit type for pharmacists in the outpatient setting to contribute to pharmacotherapy management. Further long-term studies are needed to determine the long-term impact of pharmacist-led interventions on prescribing practices utilizing CGM data. Future studies completed on a larger scale may provide more robust data and further show decrease in high risk medication use with pharmacist-led interventions using CGM data. Additional outcomes evaluating the effect of pharmacist-led interventions including the occurrence and severity of hypoglycemia may further show benefit of this service. Overall, our study highlights the potential beneficial implications of collaborative care, and the importance of pharmacist involvement in utilizing patient specific data from CGMs to optimize medication therapy, particularly with regards to deprescribing high-risk medications and initiating alternative non-high-risk medications.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: I declare that there is no conflict of interest regarding the publication of this paper. I, corresponding author on behalf of all contributing authors, hereby declare that the information given in this disclosure is true and complete to the best of my knowledge and belief.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
