Abstract
Introduction/Objectives:
In the context of enrolling patients from primary care practices into a weight loss research study, this study describes the percentage of patients primary care clinicians (PCCs) considered appropriate for referral and study participation rates among those referred.
Methods:
An electronic health record review identified 5322 patients aged 18 to 75 years with BMI ≥ 30 kg/m2 as eligible for review by community, attending, and resident PCCs for referral to a weight loss study. Referrals and patient interest in participating in the study are reported overall and by PCC type.
Results:
The mean (SD) age was 53.1 (13.9) years, mean BMI 36.5 (6.2) kg/m2, 41.5% were male, and 30.9% were African American. PCCs reviewed 3882 patients and referred 2762 (71.1%) to the study. Of 1120 not referred, 260 (23.2%) were not known well enough by PCCs to assess for referral. The most common reasons for not referring included too much comorbidity (171, 15.3%), psychological reasons (170, 15.2%), and non-English speaking (125, 11.2%). Among 1897 patients invited to participate, 676 (35.6%) expressed interest and 360 (19.0%) were enrolled.
Conclusions:
PCCs referred 71.1% of their eligible patients. Among those referred, interest in participation was modest and enrollment was low.
TRIAL REGISTRATION:
ClinicalTrials.gov Identifier: NCT04302727
Keywords
Introduction
Among adults with obesity, modest weight loss can prevent or delay the onset of diabetes and improve blood lipids and blood pressure. 1 Accordingly, the US Preventive Services Task Force (USPSTF) “recommends that clinicians offer or refer adults with a body mass index of 30 kg/m2 or higher to intensive, multicomponent behavioral interventions.” 2 In formulating this recommendation, the USPSTF found adequate evidence that intensive behavioral weight loss interventions yield significant weight loss. However, most weight loss studies cited in the USPSTF Evidence Report 1 did not enroll participants based on referral from primary care clinicians (PCCs). In addition, the provision of weight loss counseling in routine primary care practice is quite low, typically less than 20%.3,4 Thus, the USPSTF Recommendation Statement on behavioral weight loss interventions 2 recognized the need for further research on weight loss interventions in primary care settings.
While the USPSTF recommendations apply to all patients, it is not known (1) what percentage of patients are considered appropriate for referral to intensive behavioral weight loss interventions by their PCC and (2) among those referred, what the uptake of weight loss counseling would be. In studies included in systematic reviews by Booth et al 5 and Yoong et al 6 assessing behavioral weight loss programs delivered in primary care settings, none initiated enrollment based on a complete listing of practice patients with BMI ≥ 30 kg/m2. Similarly, more recent weight loss studies enrolling participants in primary care settings7 -9 have also not followed a systematic approach to participant enrollment. Understanding PCC referrals for intensive weight loss interventions and patient uptake of such referrals is needed to inform individual clinician and population health (practice and/or health care system wide)10 -12 efforts to address obesity in the primary care setting. In this paper, we report findings on a systematic recruitment/enrollment strategy for a weight loss trial that (1) identified all patients at 7 primary care practices whose BMI was ≥30 kg/m2 and (2) invited PCCs to refer their identified patients, as appropriate, to a study comparing 2 intensive weight loss counseling programs.
Methods
Study Design Overview
The referral findings described in this paper took place in the context of conducting an efficacy randomized controlled trial (RCT) in which 360 participants were randomized to a behavioral weight loss intervention promoting a Mediterranean-style dietary pattern or to the commercially-available weight loss program, WW™ (previously Weight Watchers™), with the primary outcome weight loss at 24 months. Enrollment took place from 06/12/2020 to 05/11/2022. This study was approved and monitored by the University of North Carolina (UNC)—Chapel Hill Institutional Review Board (IRB).
Recruitment Framework and Participant Selection
Study sites included 1 large academic internal medicine practice and 6 community practices in central North Carolina. All invited study sites agreed to participate. We were granted a limited HIPAA waiver by our IRB to use electronic health record (EHR) data to enumerate all patients at these practices meeting initial entry criteria and to contact those so identified by phone and email to assess eligibility. To initiate recruitment at a particular site, we used the EHR to identify all patients at that site with BMI ≥ 30 kg/m2, age 18 to 75 years (inclusive), estimated glomerular filtration rate >30 mL/min/1.73 m2, at least 1 patient care visit at the study site within the prior 2 years, and weight <500 pounds (added 10/09/2020 due to weight range limitation of our scales).
We invited all PCCs at a participating study site to attend a virtual 30-min meeting providing a brief overview of the study, specific information on how to make referrals to the study, and information on compensation to study participants ($60 for each of the 4 measurement visits attended). We then sent each PCC a spreadsheet listing their patients meeting initial study entry criteria along with detailed instructions on how to use the spreadsheet to refer patients to the study. Referrals were made from a review of this list and not during office visits. In thinking about referrals, we advised PCCs to “consider each of your patient’s clinical and social context. As the intervention lasts for 2 years, please only refer patients who you think could take part in an intervention of this duration.” Our protocol included 2 follow-up reminders by the study PI to those who had not returned their enrollment listing. Community practices and PCCs received modest compensation for participation in the study as described in eMethods 3 in the Supplemental Materials.
We also invited PCCs to provide the reason(s) for not referring a patient to the study. This could be (1) because the PCC did not know the patient well enough to make a determination about referral, (2) because the patient does not speak English (materials for the Mediterranean-style dietary intervention only available in English), (3) for study-specific exclusion criteria (selected because they are common reasons a PCC may not refer a patient for intensive weight loss counseling), or (4) for other reasons. Study-specific exclusion criteria were: (1) multi-comorbidity (defined as a positive response to not referring for “too much medical comorbidity” such that a weight loss intervention is not a clinical priority), (2) not referred for psychological or psychiatric reasons, (3) type 1 diabetes (typically these patients are already receiving intensive dietary counseling to achieve optimal glycemic control), (4) prior bariatric surgery (typically these patients are followed in post-bariatric surgery clinics that offer behavioral weight loss counseling), and (5) diagnosis of malignancy or (6) alcohol or substance abuse disorder. More detail on inclusion/exclusion criteria and PCC referrals is provided in the eMethods 1 to 3 in Supplemental Materials.
As males, African Americans, and those with type 2 diabetes mellitus (T2DM) are often underrepresented in weight loss trials,5 -9 an objective of this study was to enrich our sample with participants from these sub-groups to better understand intervention acceptability and effectiveness among these sub-groups. Accordingly, we attempted to contemporaneously enrich our sample so that at study enrollment, at least 40% of study participants would be male, 40% would be African Americans, and 40% would be persons with T2DM. We categorized race as it was recorded in the EHR based on patient self-report: African American, other (Asian, American Indian or Alaska Native, native Hawaiian or Pacific Islander, patient refused, or unknown), or White. Our protocol for achieving sub-group enrollment goals is provided in the eMethods 4 in Supplemental Materials. Early during enrollment, we observed several patients were referred who did not meet inclusion criteria (eg, patient had recent diagnosis of cancer). Thereafter, as outlined in eMethods 2 in Supplemental Materials, a study investigator conducted a chart review of participants who were referred, excluding those who did not meet eligibility criteria. Also, while the study was enrolling at a particular site, we allowed PCCs to refer patients to the study who met initial inclusion criteria but were not included in our EHR-based listing (eg, patient may have initiated care at the practice after our EHR listing was created).
Enrollment for this study began immediately after UNC-Chapel Hill ended the COVID-19 embargo on non-urgent clinical research on 06/01/2020. We initiated participant enrollment by sending all patients referred to the study a recruitment brochure by US mail and also sending this brochure by email to those with an email on file in the EHR (90.5%). Recruitment materials clearly indicated that referral to this study was made by the individual patient’s PCC. We invited potential participants to contact study staff by email or phone to let us know if they were interested in learning more about the study or if they did not want to participate. If we did not hear from potential participants within 7 to 10 days of initial outreach, we attempted to contact them by phone.
Data Collection
Study staff conducted eligibility calls with potential participants to further assess eligibility criteria as outlined in eMethods 2 in Supplemental Materials. Those interested and eligible were scheduled for a consent call, during which study staff reviewed key components of the study. Those wishing to provide e-consent were invited to do so with the others signing the consent form at their enrollment visit.
Outcomes and Statistical Analysis
In these analyses, the primary study outcomes were the percentage of patients with BMI ≥ 30 kg/m2 who were considered appropriate candidates for intensive behavioral weight loss interventions by their PCC and, among those referred, the percentage interested in taking part in the study, both overall and across our sub-groups of interest. Characteristics of potential participants and those referred are described by counts and percentages for categorial variables and means and standard deviations (SD) for continuous variables. For comparisons of characteristic and referral behaviors across the 3 groups of clinicians, 1-way ANOVA was used for continuous variables and the Chi-square test for categorial variables. Pair-wise comparisons were also performed if the overall test was statistically significant. All analyses were conducted using SAS (SAS v9.4, SAS Institute, Cary, NC). A P-value <.05 was considered statistically significant.
Results
Sample for Enrollment
A total of 5233 patients were identified for study recruitment (5214 from EHR; 19 referred by PCCs during enrollment) with detail on enrollment by practice site and for sub-groups of interest provided in eMethods 5 in Supplemental Materials. Characteristics of this sample are shown in Table 1 stratified by the type of referring PCC: those from community practices, academic faculty providers (attendings), and residents. The average age was 53.1 years, average BMI 36.5 kg/m2, 41.5% were male, 30.9% African American, and 28.6% had T2DM (P < .0001). Patients seen in community clinics were younger, more likely to be male, and less likely to be African American or have T2DM when compared to attendings and residents (P < .05 for these pair-wise comparison). Within the academic practice, patients served by resident PCCs were younger, had a higher BMI, and were more likely to be male and African American (P < .05 for these pair-wise comparisons).
Characteristics of Patients Identified for Study Recruitment by Clinician Type.

Abbreviations: ANOVA, analysis of variance; att, attending clinician; com, community clinician; res, resident clinician.
Attending indicates faculty clinician at academic medical center.
Resident indicates categorial internal medicine trainee.
Other includes Asian, American Indian or Alaska Native, native Hawaiian or Pacific Islander, or patient refused, or unknown.
Referral for Weight Loss Intervention by PCCs
As shown in Table 2, of 112 PCCs invited to refer patients to this study, 88.4% made referrals. As depicted in Figure 1, the study CONSORT diagram, of 5233 patients meeting initial study entry criteria, 4824 were included in referral spreadsheets distributed to PCCs who were instructed to review their patient listing and make referrals to the study, as appropriate. As depicted in Table 3, a total of 3882 patients were reviewed by PCCs for eligibility, of which 2762 (71.1%) were referred to this study evaluating intensive weight loss counseling and 1120 (28.9%) were not referred. Of note, community clinicians were much more likely to refer (81.7% of patients referred) than attendings (66.3%) or residents (47.4 %)—P < .0001 for Chi-square test and all pair-wise comparisons. Table 3 also shows the reasons for non-referral by PCC provider type. Among those not referred, PCCs either (1) did not provide a reason for non-referral (88; 7.9%); (2) felt they did not know the patient sufficiently well enough to refer (260; 23.2%), or (3) provided at least 1 reason for non-referral (772; 68.9%). Attendings were much more likely to know their patients well enough to assess for referral compared to community and resident clinicians (89.9%, 71.3%, and 58.0%, respectively, P < .0001). Also, attendings and community physicians were more likely to provide a reason for not referring, compared to residents (76.4%, 70.1%, and 52.7%, respectively, P < .0001). Among choices from which PCCs could indicate a reason for non-referral, multi-comorbidity (171; 15.3%), psychological reasons (170; 15.2%), and does not speak English (125; 11.2%) were most common. Notably, community physicians saw far fewer Sanish speaking patients (P < .0001).
Participation Rates for Clinicians Invited to Refer Patients to Study.
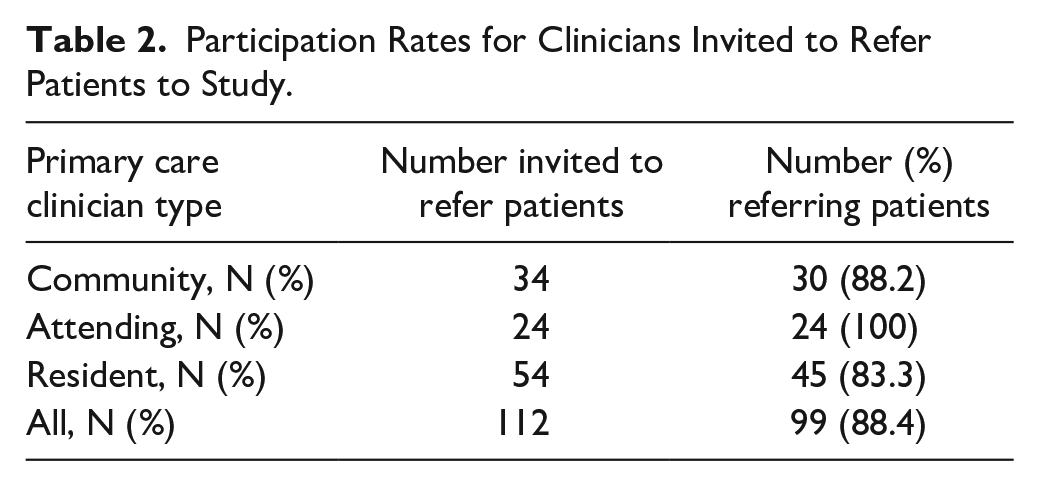

Flow of participants through screening.
Reasons Not Referred to Weight Loss Study, by Primary Care Clinician (PCC) Type.

Abbreviations: att, attending clinician; com, community clinician; res, resident clinician.
Attending indicates faculty clinician at academic medical center.
Resident indicates categorial internal medicine trainee.
Clinician may indicate 1 or more reasons for not referring.
Enrollment Results for Those Referred to Study
As outlined in Figure 1, 2621 patients referred to the study were considered eligible. To achieve an enrollment sample of 360 for the randomized trial, we invited 1897 (72.4%) to participate, enriching the sample of those invited with patients from our sub-groups of interest. Of these 1897, 887 (46.8%) were reached by phone and completed the assessment for eligibility. Of these 887, 211 (23.8%) were not interested in taking part in this weight loss study and 676 (76.2%) expressed interest in taking part or learning more about the study. Of these, 42 (6.2%) did not complete screening, 157 (23.2%) were not eligible (for reasons outlined in Figure 1) and 479 (70.6%) were interested and eligible. Of these 479, 360 (75.2%) completed the enrollment protocol and were randomized. Overall, among 1897 patients invited to participate, 676 (35.6%) expressed interest and 360 (19.0%) were enrolled. Among the sub-group of 887 who were invited and successfully contacted about study participation, 676 (76.2%) expressed interest in participating or learning more about the study and 360 (40.6 %) were enrolled.
Enrollment of Sub-groups of Interest
Figure 2 depicts the composition of the patients over the course of enrollment. Overall, 52.8% of those invited to participate were male, yielding a final percentage of 44.2% males enrolled. Of note, throughout the enrollment sequence the percentage of males was much higher for community PCCs compared to residents. Overall, 36.7% of those invited to participate and 42.2% of those expressing interest were African American yielding a final enrollment percentage of 33.6%. Throughout the enrollment process, the percentage of African American was substantially higher for residents. Overall, 34.2% of those invited to take part had T2DM yielding 30.8% enrolled with this condition. Counts of participants by sub-group of interest at each enrollment stage are detailed in eTable 1 in Supplemental Materials.

Composition of patients by enrollment stage. This Figure displays the percentage of the study sample composed of subgroups of interest at each enrollment time point. Panel A displays the composition of male patients over the course of enrollment. Panel B displays the composition of African American patients over the course of enrollment. Panel C displays the composition of Persons with diabetes over the course of enrollment. Note that Stage 3, “Invited to participate,” represents the sample enriched with subgroups of interest (males, African Americans, and those with diabetes). The frequency counts for these subgroups are available in eTable 1.”
Discussion
In this study we used the EHR to prepare a comprehensive list of patients with BMI ≥ 30 kg/m2 for each PCC at 7 primary care practices. We invited these PCCs to review their listing and refer patients, as appropriate, to a weight loss study offering intensive behavioral weight loss counseling. PCCs referred the large majority of their listed patients (71.1%) to this study. For those sufficiently well known to their PCC to consider referral, the most common reasons for not referring were multi-comorbidity and psychological conditions. Among patients invited to take part in this study, 35.6% expressed interest and 19.0% were enrolled.
Though many weight loss trials have been conducted in primary care settings,5 -9,13 none to our knowledge have undertaken a systematic enrollment protocol where PCCs were invited to review and refer from a comprehensive listing of their patients with BMI ≥ 30 kg/m2. Clark et al 13 report on a different type of systematic approach to referrals for weight loss counseling in a retrospective analysis of a quality improvement weight management program conducted at 5 community health centers from 2006 through 2008. An EHR based approach was used to identify consecutive patients eligible for referral to an in-clinic lifestyle behavior change counselor. For all patients ≥18 years of age and with BMI ≥ 25 kg/m, at the time during a clinic visit when they were entering orders into the EHR, PCCs received an electronic prompt to refer to the weight loss program. Of 12,487 so identified as overweight and obese, 40.3% received a provider referral and 15.6% had weight loss program contact. In this program, the PCCs were not asked to indicate why they did not make a referral.
We recognize our findings on PCC referrals are for a research study offering intensive weight loss intervention and contextually may differ from referrals made in routine practice or as part of a practice or health system-based quality improvement program. We think PCC referrals in this study are likely to be similar to referrals made in routine practice from patient listings of those with BMI ≥ 30 kg/m2 (if intensive weight behavioral weight loss programs were available without cost to patients) as our study specific exclusion criteria align with common reasons a PCC may not refer for intensive behavioral weight loss counseling. Also, a substantial percentage of patients (23.2%) were not referred because the PCC did not know the patient sufficiently well to make a determination about participation; if referrals were made in the context of clinic visits as might be the case in some systematic enrollment programs, this percentage would be much lower.
Uptake of referral to this weight loss study was low but consistent with previously reported engagement with both weight loss counseling offered in routine clinical practice3,4 and studies conducted in primary care settings. As noted, enrollment was 15.6% in a large weight loss quality improvement project 13 and within a large integrated health system that examined weight loss referrals and engagement, among 160 163 adults with a median BMI of 35 kg/m2, 17% were referred to a weight loss program, of which 29% attended 1 visit. 14 While enrollment percentages of those screened varies among research projects, for many larger studies9,15 -20 enrollment yield is less than one-third, as we observed in our study.
This study enrolled participants from 2 distinct practice settings, community versus academic, and within the academic setting from attendings and residents. Difference in patient populations between settings and clinicians were expected. Overall, patients served by community clinicians were younger, had less severe obesity, and were less likely to be African American or have diabetes. Within the academic setting, resident patients had greater BMI and were more likely to be African American and have diabetes. The referral rate for community clinicians (81.7%) and attendings (66.3%) was much higher than for residents (47.4%) due in part to the anticipated finding that resident did not know their patients as well as community clinicians and attendings.
The COVID-19 pandemic may have also influenced the recruitment and enrollment process for our study. Patients may have been reluctant to participate due to concerns about potential or perceived SARS-CoV-2 virus infection risks. On the contrary, uptake may have been enhanced by those seeking to address weight gain that may have resulted from changes in physical activity and/or eating patterns during the pandemic21,22 or to simply escape the pandemic-induced social isolation. 23
Our efforts to enrich our sample with sub-groups typically under-represented in weight-loss interventions were only partially successful. We enrolled greater than 40% male participants primarily because the sample of men available for enrollment was very large. For African Americans and those with T2DM, enrollment approached, but did not achieve the desired 40% threshold. Further research is warranted to better understand barriers to enrolling patients from these subgroups.
Since publication of the USPSTF recommendations for weight loss management in 2018, the landscape of effective weight loss options has changed substantially. Given the increasing effectiveness of medical and surgical interventions to achieve weight loss, patients may be less likely to accept referrals/recommendations for intensive behavioral weight loss interventions. Most importantly has been the approval of glucagon-like peptide (GLP-1)24,25 and dual acting GLP-1 and gastric inhibitory polypeptide (GIP) 26 agonist medication for weight loss. These medications typically yield meaningful weight loss, improve cardiometabolic profiles,25,26 and reduce risk for CVD events. 27 There has also been increasing use of bariatric surgery, 28 which similarly improves cardiometabolic profiles and clinical outcomes.29,30 In addition, there is an increasing appreciation that a healthful dietary pattern improves cardiometabolic profiles and clinical outcomes without weight loss.31 -33 Given the multiple options of effective interventions to reduce cardiometabolic risk and improve clinical outcomes, new paradigms are needed to assist the PCC and patients in making informed decisions about interventions targeting weight management and/or chronic disease risk reduction. While some patients might prefer lifestyle interventions, others may prefer medications or surgery if costs are not prohibitive. Decision aids could be helpful in this context.
Although our findings on patient uptake have relevance for individual clinicians who refer for weight loss counseling, our findings on referral rates and uptake may be most relevant for practices and health systems that may be considering a population health approach addressing weight management for patients identified from the EHR with BMI ≥ 30 kg/m2.10,11 Based on our findings and others13,14 such programs should anticipate that a substantial percentage of patients identified from the EHR based on a BMI criterion would not be referred by their PCC for intensive weight loss counseling and that program uptake will be modest among those referred.
This study has limitations. As noted, the research context may have impacted PCC referrals for an intensive weight loss intervention, though we think this impact was likely minimal to modest. A more important limitation pertains to patient interest in and uptake of an intensive behavioral weight loss intervention in the research context. Many may not have been interested due to this context, though our yield from referrals is similar to that reported in a non-research context. Also, outcomes of referrals for weight loss counseling may be different if based on shared decision making during a face-to-face discussion versus referrals from a listing. That said, in our current health care context, many population health programs enumerate those who qualify for preventive health interventions and request approval for enrollment into such programs via EHR derived listings provided to the PCC. The major strength of this study is assessment of weight loss referrals from PCCs reviewing a comprehensive listing of their patients with a BMI ≥ 30 kg/m2.
Conclusions
In summary, PCCs did not refer almost 30% of their patients with a BMI ≥ 30 kg/m2 to our study providing intensive weight loss counseling. Among those referred, interest in participation was modest and enrollment was low, but consistent with rates observed in other behavioral weight loss research and quality improvement projects. New paradigms are needed to inform PCC and patient decision-making about weight loss and other lifestyle interventions designed to improve cardiometabolic profiles and clinical outcomes.
Supplemental Material
sj-docx-1-jpc-10.1177_21501319241308054 – Supplemental material for An Evaluation of Primary Care Clinician Referrals for Behavioral Weight Loss Counseling
Supplemental material, sj-docx-1-jpc-10.1177_21501319241308054 for An Evaluation of Primary Care Clinician Referrals for Behavioral Weight Loss Counseling by Veeral M. Saraiya, Seth A. Berkowitz, Larry F. Johnston, David J. Couper, Daniela Sotres-Alvarez, Kiira M. Lyons, Cheyenne K. Thaxton, Harlyn G. Skinner, Carmen D. Samuel-Hodge and Thomas C. Keyserling in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
We thank the study participants and study staff without whom this work would not have been possible. We thank Dr. Qiang Wu for his contributions to the statistical analyses reported in this paper.
Author Contributions
VMS and TCK had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis; Concept and design: VMS, SAB, DC, DS-A, CDS-H, and TCK; Acquisition, analysis, or interpretation of data: VMS, SAB, LFJ, QW, DC, DS-A, KML, CKT, HGS, CDS-H, and TCK; Drafting of the manuscript: VMS and TKC; Critical review of the manuscript for important intellectual content: VMS, SAB, LFJ, DC, DS-A, KML, CKT, HGS, CDS-H, and TCK; Statistical analysis: VMS, and TCK; Obtained funding: CDS-H and TCK; Administrative, technical, or material support: LFJ, KML, and CKT; Supervision: VMS, KML, and TCK.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Berkowitz reports research grants from NIH/NHLBI, Blue Cross Blue Shield of North Carolina, North Carolina Department of Health and Human Services, American Diabetes Association, Rockefeller Foundation, and the American Heart Association outside of the submitted work. He reports consulting fees from Kaiser Permanente outside of the submitted work. Dr. Couper reports research grants from NIH/NHLBI, NIH/NIDDK, and the COPD Foundation outside of the submitted work. Dr. Sotres-Alvarez reports research grants from NIH/NHLBI, NIH/NIDDK, and NIH/NIMHD outside of the submitted work. Dr. Samuel Hodge reports research grants from NIH/NIMHD and CDC outside of the submitted work. Dr. Keyserling reports research grants from NIH/NIDDK, NIH/OD/NICHD, CDC, and the Duke Endowment outside of the submitted work. Dr. Keyserling’s wife was founder and now volunteer advisor to a company that makes healthful frozen foods. She does not hold equity in the company.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Grants R61 HL142680 and R33 HL142680 from the National Heart, Lung, and Blood Institute (Dr. Samuel-Hodge, Dr. Keyserling); The funder had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Ethical Considerations
This study was approved by the University of North Carolina at Chapel Institution Review Board (IRB; Ethics Code: 19-1712) on Octtober 2, 2019.
Consent to Participate
All participants provided written or digital informed consent prior to enrollment in the study.
Consent to Publish
This research was conducted ethically in accordance with the World Medical Association Declaration of Helsinki.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are not publicly available due to study participant privacy but are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
