Abstract
Menopause marks a significant transition in a woman’s life, typically occurring between the ages of 46 and 55 years, characterized by the cessation of menstruation and a decline in ovarian function. This article provides a comprehensive overview of menopause, examining its physiological, psychological, and social dimensions. It explores the hormonal changes, including decreased levels of estrogen and progesterone, and how these changes contribute to common symptoms such as hot flashes, sleep disturbances, and mood fluctuations. Additionally, it highlights contemporary approaches to management, including lifestyle modifications and hormonal and non-hormonal therapies. By synthesizing current research and clinical insights, this overview aims to enhance understanding and support for women navigating menopause and the healthcare professionals assisting them.
Introduction
Menopause is a universally experienced transition from a woman’s reproductive years to non-reproductive years and is gaining national attention. 1 There is a greater demand for information on menopause and for providers to implement treatment strategies to manage bothersome symptoms associated with menopause. There has also been a concurrent rise in medical misinformation about menopause. The Menopause Society (previously known as the North American Menopause Society) has emphasized the need to combat misinformation by improving clinician knowledge of current recommended guidelines for providing menopause related care. 2 Often, women present to the primary care setting for evaluation, and primary care providers need comprehensive, evidence-based resources to appropriately counsel and treat patients in the primary care setting. This article aims to provide a comprehensive review of menopause, including epidemiology, diagnosis, evaluation, and management for the primary care provider.
Epidemiology
Menopause is characterized by the permanent cessation of menstrual cycling and is clinically defined as the absence of menses for 12 months.3,4 The transition from reproductive to non-reproductive years, also known as perimenopause or the menopause transition, begins before the final menstrual period. 3 The average age of menopause in the United States is 52, with the range of typical menopause onset occurring between 46 and 55 years. 5 However, women can begin to experience symptoms of perimenopause in their 40s. 5 Those entering menopause at less than 40 years are classified as premature menopause, those entering menopause between ages 40 and 45 years are classified as early menopause, and those entering menopause after age 55 years are classified as late menopause. 6 Perimenopause and menopause may occur earlier in women with a history of oophorectomy, primary ovarian insufficiency (POI), hysterectomy, pelvic radiation therapy, or active tobacco use.6 -9 There are also variations in the transition to menopause by race and ethnicity. In a longitudinal cohort study of 3302 women in the US (Study of Women’s Health Across the Nation, SWAN study), Chinese and Japanese women had longer cycle lengths compared to Non-Hispanic White women, and Black women experienced more severe vasomotor symptoms (VMS) and a longer duration of the menopause transition. 10 In a more extensive online survey study of 68 864 US women, Black, Hispanic, Indigenous, and Middle Eastern women were found to have more bothersome menopause related symptoms compared to Non-Hispanic White women even after accounting for socioeconomic factors. 11 Given that the average life expectancy of women in the US is 80 years, 12 most women in the US will spend over a third of their lives in menopause.12 -15 Despite this prevalence, perimenopausal and menopausal symptoms are often underreported, under screened, and undertreated and can result in a significant decrease in one’s quality of life and lost productivity.13 -17 Untreated menopausal symptoms have a significant negative economic impact; it is estimated that the cost associated with the loss of work productivity associated with menopausal symptoms is $1.8 billion annually in the US. 18
Barriers to Accessing Menopause Related Care
Barriers to accessing menopause related care exist at the patient, provider, and system levels.19,20 Patient factors range from a lack of knowledge regarding menopause to the stigma surrounding the discussion of symptoms and concerns about the side effects of treatments. 21 An online survey sampling over 800 postmenopausal women (primarily from the UK) found that 94% reported never having received education on menopause, and 60% of women only began to seek information on menopause after they started to experience menopause-related symptoms.21,22 Another online survey sampling of 196 US women’s experiences of menopause and pursuit of treatment for menopause found that 25% of women did not discuss their symptoms because they felt these were a natural course of menopause. 23 Similarly, a survey analysis of women in the SWAN study experiencing urinary incontinence found that 61% did not report these symptoms because they felt symptoms were not severe and expressed that urinary incontinence is a natural part of aging. 24 Menopause-related symptoms, such as hypoactive sexual desire, dyspareunia, and genitourinary symptoms of menopause, can be perceived by patients as socially taboo. Thus, they do not feel empowered to bring these topics up in the clinical setting.25,26 Fear regarding the adverse side effects of hormone therapy may affect women’s decision to pursue menopause related care. A sub-analysis of 500 US women participating in the Vaginal Health: Insights, Views & Attitudes (VIVA) study found that a majority expressed negative associations with systemic hormone therapy with particular concerns about the risk of breast cancer or clot/stroke. 27 Improving patient knowledge of menopause and treatment options prior to the onset of symptoms may empower patients to discuss these issues. A small study utilizing a telephone, nurse educator-based intervention with an accompanying menopause booklet to inform women on menopause related symptoms and treatment choices demonstrated increased participant knowledge of menopause, decreased conflict regarding treatment options, and 94% reported feeling better prepared to talk to their provider regarding menopause symptoms. 28
From a provider standpoint, lack of knowledge/confidence in prescribing MHT, communication barriers, time constraints, and personal attitudes or beliefs regarding menopause can become significant barriers to prescribing hormonal or non-hormonal therapy to manage menopause. 29 A survey of US trainees in family medicine, internal medicine, and obstetrics and gynecology found that residents self-reported minimal curriculum on menopause hormone therapy and had knowledge gaps on when to prescribe menopause hormone therapy. 30 Similarly, a survey of US primary care providers and gynecologists revealed that primary care providers had less knowledge and confidence in managing vulvovaginal atrophy compared to gynecologists, and clinician-reported barriers to management included lack of time, tools for diagnosis and management, and educational materials for patients. 31 Inadequate communication is also a barrier, Shepherd and colleagues found that approximately 42% of women who discussed their menopause related symptoms with a physician received no treatment advice and 23% reported feeling that their symptoms were dismissed or were not adequately addressed. 23 Strategies to combat such issues can include implementing menopause education into provider curriculum, which has been shown to increase trainee knowledge and confidence in providing menopause related care. 32
At the systems level, a recent cross-sectional analysis of National Health and Nutrition Examination Survey (NHANES) data on menopause hormone therapy (MHT) prescribing practices in the US among 13 048 postmenopausal women from 1999 to 2020 found a significant decline in MHT prescribing with women belonging to racial and ethnic minority groups having lower rates of MHT use compared to Non-Hispanic White women. 33 Non-Hispanic Black women had the lowest rates of MHT use among all groups evaluated. Women who were insured, more educated, and had higher incomes had greater odds of MHT use. 33 These disparities in MHT prescription suggest that women belonging to populations experiencing health disparities have greater struggles in receiving menopause related care and social determinants of health must also be taken into account when delivering menopause related care.22,29
Pathophysiology of Menopause
The pathophysiology of menopause can be complex. The depletion of ovarian follicles starts prior to birth and continues throughout a female’s entire lifespan. 6 Prior to menopause, granulosa cells of preantral and antral ovarian follicles produce high amounts of inhibin B and anti-mullerian hormone (AMH). 6 These limit the number of follicles available for ovulation each month to preserve the follicular pool over a woman’s “reproductive” years. 6 Eventually, the number and size of follicles reach a point where the restraints on follicular growth, inhibin B and AMH, begin to reduce, leading to exponential depletion of ovarian follicles and the beginning of the menopause transition. 6 At first, this results in longer luteal phases of menstrual cycles and irregular menstrual bleeding patterns. 6 Specifically, the menopause transition is often recognized by the development of increased menstrual cycle irregularity of greater than or equal to 7 days. 6 The menopause transition tends to consist of significant hormonal fluctuation, including estradiol level variation, at which time the symptoms of menopause will often begin. 6 Maturation of the ovarian follicle, known as folliculogenesis and estradiol production, progressively declines, resulting in increased menstrual irregularity and symptoms of estrogen deficiency over the menopause transition. 6 Eventually, folliculogenesis can no longer occur, and the production of estradiol and progesterone ceases, resulting in sustained amenorrhea. 6
Diagnosis, Clinical Features, and Presentation
The prevalence and severity of menopausal symptoms and complications, which include physical and psychological symptoms, vary widely among women.34 -36 Perimenopause and menopause symptoms can range from VMS, menstrual irregularity, weight changes, and skin changes to genitourinary symptoms, mood changes, and decreased libido (Table 1).14,34,35,37 The decline in estrogen levels and the accumulation of central obesity during menopause also elevate the risk of chronic conditions, including cardiovascular disease, dyslipidemia, type 2 diabetes, osteoporosis, and cognitive disorders.35,38 -40 Approximately 80% of postmenopausal women experience VMS, such as hot flashes and night sweats, while up to 50% report genitourinary symptoms, including vulvovaginal atrophy, vaginal dryness, dyspareunia, incontinence, and urinary urgency. 35 Additionally, around 39% of women suffer from depression during or after menopause. 41
Clinical Symptoms of Menopause.

Evaluation
Formal evaluation via laboratory testing is not indicated or recommended to note a patient’s menopausal status for most patients. 42 It is important to note that other causes of amenorrhea, such as pregnancy, lactation, and secondary causes of amenorrhea (medications for ovarian suppression and contraceptive use), are reasonably excluded prior to declaring a patient as menopausal. Patients presenting in premature menopause warrant further history and evaluation to rule out secondary causes of amenorrhea. 6
On occasion, assessment of menopausal status may help guide discussion on the need for ongoing contraception in patients where it is unclear if they have met clinical criteria for menopause. In women in their 40s and 50s, using hormonal contraceptives with irregular or absent bleeding cannot reliably indicate menopausal status, as it may result from the hormonal effects of the contraceptives. 43 Measurement of follicle stimulating hormone (FSH) and estradiol in perimenopausal women on oral contraceptives may be unreliable, and an accurate assessment of these labs would be ideally performed after a break from hormonal contraceptives.44,45 An elevated FSH level of >30 IU/L and low estradiol levels may indicate menopause when off of OCPs. 46
OCPs suppress the hypothalamic-pituitary-ovarian axis with suppression of FSH and LH levels. 44 Data from pre-menopausal women undergoing in vitro fertilization treatment who pre-treated with OCP the month prior to oocyte harvest experienced rebound surges of FSH after cessation of OCP, which returned to the control group (no OCP pre-treatment) after approximately 5 days. 45 However, these findings are challenging to extrapolate into the perimenopausal and menopausal population but suggest that measurement of FSH and LH immediately after cessation of OCP may be unreliable. Older studies indicate that FSH remains unaffected, and estradiol levels may be higher in the mid-luteal phase in women using hormonal intrauterine contraception (IUC), but these need further study. 47
In summary, the menstrual history and age of the patient remain the most predictive markers for determining menopausal status. 46 Laboratory evaluation may be helpful in clinical decision making regarding the ongoing need for contraception, but data is limited on the timing of this measurement after cessation of OCP or in the context of a hormonal IUC.
Management/Treatment
Pharmacologic
Hormonal therapy
The most effective way to treat VMS is systemic MHT. However, since the release of data from the Women’s Health Initiative (WHI) in 2002, the prescribing of MHT drastically declined.33,48,49 Despite newer evidence supporting the safety of MHT, many providers are still hesitant to initiate. The FDA currently only approves MHT for the treatment of moderate-to-severe VMS, prevention of postmenopausal osteoporosis, and in premature menopause.50,51
Estrogen therapy is effective in alleviating symptoms common to perimenopause. Vaginal estrogen is indicated for the management of moderate-to-severe symptoms of genitourinary syndrome of menopause (GSM) and does not require endometrial protection with a progestogen. Transdermal or oral estrogen is indicated for VMS. In women with a uterus, a progestogen must be added to mitigate the risk of endometrial hyperplasia.52,53 A few FDA-approved oral progestogens can be given continuously or cyclically. A progestin containing IUC is often utilized for uterine protection but is an off-label use.52,54
When considering MHT in individuals with ovaries, there are concerns about how it will affect breast and cardiovascular health. Oral estrogen goes through the first pass metabolism and increases coagulation factors. 55 Transdermal preparations, such as patches or creams, have decreased the risk of venous thromboembolism compared to oral estrogen and have not been shown to increase blood pressure. 55 When the WHI was released, the use of MHT was abruptly discontinued in many women due to concerns about the risk of breast cancer and thromboembolism. However, age stratified analysis of long-term outcomes showed that women aged 50 to 59 years did not have an increase in all-cause, cardiovascular, or cancer mortality at the 18-year follow-up. 56 Since the WHI, multiple studies have shown similar findings. A Cochrane review including 19 high-quality trials of MHT showed that the risk of a cardiovascular adverse event differed by age. 57 Many of these trials also used oral estrogens and progestins, which likely impose a greater risk of venous thromboembolism and cerebrovascular disease than a transdermal estrogen preparation or micronized progesterone. These findings lead to the current “timing hypothesis” (sometimes referred to as the “critical window theory”), which proposes that women in early menopause have healthier blood vessels, which undergo vasodilation and experience overall anti-inflammatory effects in response to estrogen. 37 With age, the vasculature becomes more stenotic and stiff. This damaged endothelium has impaired vasodilation; estrogen has pro-inflammatory effects on this substrate and can destabilize plaques. 37 The timing hypothesis has led to the current guidelines that MHT should be initiated in people with ovaries less than 60 years of age and within 10 years of menopause.37,58
Although the WHI initially showed an increased incidence of breast cancer, continued follow-up revealed that with the use of conjugated equine estrogens (CEE) alone, there were 1.4 fewer cases per 100 women. 59 In the group of CEE plus medroxyprogesterone acetate (MPA), there was a slight increase after 5+ years of use. 59 This showed an increased risk of 1 to 2 additional cases per 1000 women. 59 The Collaborative Group, a meta-analysis of 58 prospective and retrospective observation studies, noted similar results. 60 However, contrary to the WHI, there was an increase in breast cancer in estrogen-alone hormone therapy, although not as high of an increase in the combined therapy group (estrogen plus progestogen). 60 This has led to the thought that a progestogen may be the culprit.
There is some thought that different types of progestogens may have a lower risk for breast cancer, but the data is mixed. A study completed in the UK evaluated hormone replacement and risk of breast cancer. It evaluated 4 types of progestogens combined with estrogen and noted an increase in breast cancer with all types, although some had a lower risk than others. 61 A French cohort study reviewed estrogen with multiple types of progestogens and noted progesterone and dydrogesterone had the lowest risk for breast cancer. 62 The use of an IUC has mixed data on its risk for breast cancer.63,64 Lastly, a study by Abenhaim et al 65 noted that synthetic progestins seemed to have the highest risk for breast cancer. Due to the mixed results from multiple studies, progestogen therapy alone is often not recommended to treat menopausal symptoms.62,63
There are very few contraindications to MHT, but a list of known contraindications is included in Table 2: advance stage endometrial cancer, unexplained vaginal bleeding, personal history of breast cancer, uncontrolled hypertension, personal history of unprovoked venous thromboembolism, some clotting disorders, cirrhosis, active hepatitis, and menopause onset >10 years ago. 37 Other health conditions are not contraindicated but should be considered and discussed prior to starting MHT, as shown in Table 2: untreated hyperlipidemia, diabetes, smoking, personal history of provoked VTE, chronic inflammatory states, human immunodeficiency virus, known genetic mutation increasing the risk of breast or ovarian cancer, and age greater than or equal to 60 years.37,66
Considerations When Initiating Menopause Hormone Therapy.
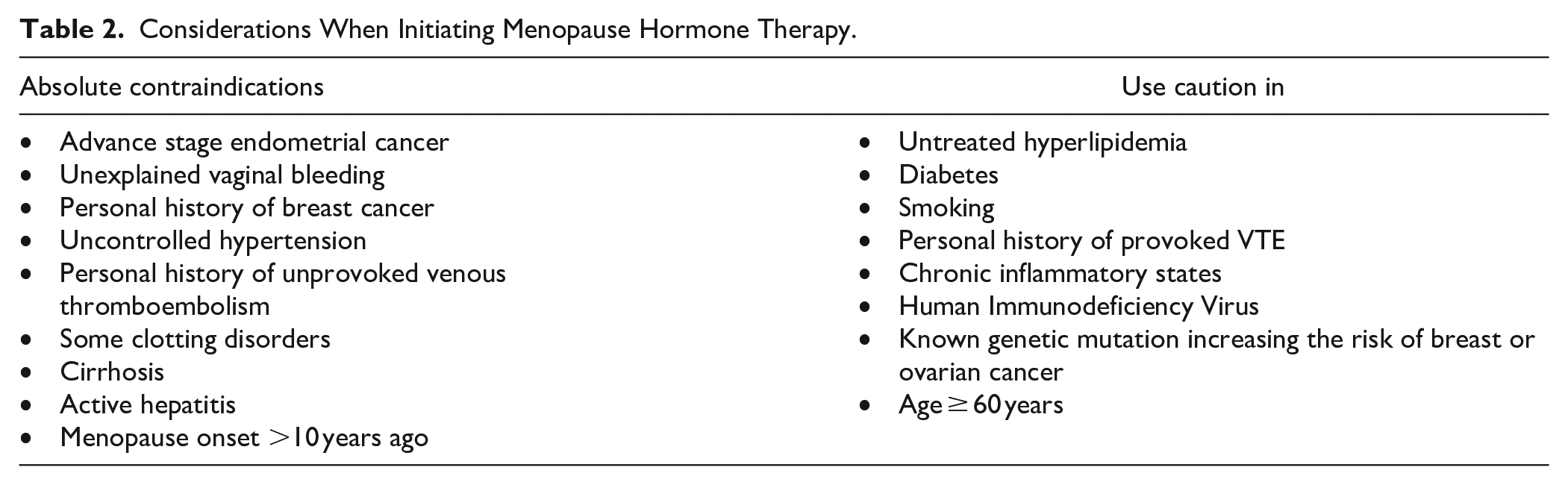
Women who experience premature or early menopause due to primary ovarian insufficiency, hypothalamic hypogonadism, radiation, chemotherapy, bilateral salpingo-oophorectomy, or any other causes of a low estrogen state, should be treated with MHT, even in the absence of bothersome menopausal symptoms.67 -69 In this population, MHT supports cardiovascular, bone, cognitive health, sexual dysfunction, and overall mortality until the average age of menopause.67,69 Even for patients who have known genetic mutations such as BRCA1 and BRCA2 and are at increased risk of breast cancer, MHT should be utilized if the patient has early or premature menopause. 70
The Menopause Society recommends utilizing the lowest dose possible to alleviate symptoms. Please refer to Table 3 for options for starting MHT. When deciding between cyclic progesterone (administered nightly days 1-12 of each month) and nightly progesterone, the menopausal status of the patient is helpful. Patients who are perimenopausal and still have ovarian function are likely to have irregular bleeding if using a nightly regimen. 71 Using cyclic progesterone makes the bleeding profile more predictable. As patients enter further into menopause, they are less likely to experience a bleed with the cyclic regimen, and it is at this point that they can transition to nightly progesterone. Uterine bleeding can occur in 40% of those on estrogen therapy with a continuous progestogen in the first 3 to 6 months. 6 If there is persistent bleeding with MHT use while doing estrogen with nightly progesterone, further evaluation is warranted. 67 Postmenopausal bleeding can be investigated with a pelvic ultrasound, hysteroscopy, and/or endometrial biopsy. 42 Oral progesterone should be taken at night as it will likely cause drowsiness. There are transdermal products that contain progestogens, but these are synthetic. Due to the concern that progestins may higher increase for breast cancer, natural progesterone is favored.
Options for Starting Menopause Hormone Therapy (MHT).

Offers information on methods of delivery of menopause hormone therapy and suggested dosing regimens for patients experiencing bothersome menopause related symptoms. We recognize that this is not an exhaustive list of treatment options for dosing or delivery methods and that each patient’s treatment regimen is individualized based on their presentation. This table aims to provide the primary care clinician with a broad conception of dose and delivery methods.
Non-hormonal prescriptions
Several non-hormonal prescriptions are recommended by the Menopause Society (TMS) to treat VMS. Paroxetine 7.5 mg daily and fezolinetant 45 mg daily are the only 2 FDA-approved treatments for this indication. Fezolinetant, a neurokinin 3 receptor antagonist, is the only FDA-approved non-hormonal prescription developed specifically to treat hot flashes by binding to the kisspeptin/neurokinin B/dynorphin (KNDy) neuron to modulate its activity in the thermoregulatory center. Hypersecretion of neurokinin B from KDNy neurons was discovered to affect the hypothalamus’s thermoregulatory functions, resulting in temperature dysregulation and VMS. 72 Adverse effects include headaches and, in rare cases, elevated liver enzymes. It is advised that liver enzymes be assessed before prescribing this medication and monitored every 3 months for the first year. Fezolinetant should not be given to those with ALT or AST levels greater than or equal to 2 times the upper limit of normal or if the total bilirubin is elevated. This medication is contraindicated in those with severe renal impairment, end-stage renal disease, cirrhosis, or concurrent use of CYP1A2 inhibitors.
Though paroxetine is the only approved selective serotonin-reuptake inhibitor (SSRI) for treating hot flashes, other medications in this class can also be helpful, such as escitalopram. Serotonin-norepinephrine reuptake inhibitors (SNRIs), including venlafaxine and desvenlafaxine, can also help reduce vasomotor symptoms. 73 While some antidepressants can cause weight gain or lower libido, low doses of paroxetine (7.5 mg/d) can lessen VMS and improve sleep disruption without causing these side effects. 73 SSRIs, such as paroxetine, should not be given to those taking tamoxifen due to inhibition of CYP2D6. Contraindications to SSRI and SNRI use include concurrent use of monoamine oxidase inhibitors, history of neuroleptic syndrome, and serotonin syndrome. 73
Furthermore, oxybutynin and gabapentin are recommended due to their ability to reduce VMS and limit adverse effects.50,74 Oxybutynin, an anticholinergic used to treat overactive bladder syndrome and urinary urge incontinence, has been shown to improve VMS significantly.50,74 Dry mouth is a common side effect, and urinary retention can occur. Though most apparent in older persons, studies show an association between long-term use of anticholinergics and cognitive decline.50,75 Gabapentin has also been shown to reduce VMS frequency and severity.50,76 It can cause side effects of drowsiness and dizziness, which usually attenuate over time and resolve in 4 weeks. Given this, starting at low doses and gradually titrating up is often favored based on tolerance and efficacy. 50 When given at bedtime, this medication can be beneficial for those with primary concerns of VMS and disturbed sleep and those with concurrent neuropathy or neuralgias. 50
Over-the-counter (OTC)
OTC supplements are difficult to recommend as limited research and data are available. With the lack of FDA approval, there is concern about the purity of each product. There are multiple supplements often used for managing VMS. These include Soy, Black Cohosh, Rhubarb, and Pollen extract. Given the lack of quality evidence-based scientific research supporting the use of OTC supplements and herbal products, their use to manage menopausal symptoms is not recommended. 50
Lifestyle modifications
Leading a healthy lifestyle may decrease the severity of menopausal symptoms for some women. For women experiencing mild symptoms, it is reasonable to offer lifestyle modifications such as exercise, mind/body activities, cognitive behavioral therapy, alcohol consumption reduction, and dietary changes. 50 Some dietary products that are more likely to contribute to vasomotor symptoms include caffeine, hot beverages, processed sugar, and alcohol. 50 Unfortunately, there is limited evidence from clinical trials to support lifestyle modifications as management for bothersome menopausal symptoms. 50
Referrals
The majority of symptomatic menopausal women can be successfully managed in the primary care setting. However, a few unique populations may warrant referral to a menopausal specialist. Populations to consider referring to a menopause specialist include patients with premature ovarian insufficiency, iatrogenic menopause, secondary amenorrhea evaluation, strong family history or increased genetic risk of estrogen-sensitive cancers, history of or increased risk of cardiovascular disease, and history of endometriosis or leiomyomas.
Conclusions
The symptoms of perimenopause and menopause can average between 4 and 10 years in duration, and it can significantly impact one’s quality of life and overall health. Educating providers and patients on the symptoms and treatment options is important to reduce barriers, improve access to menopause related care, and improve a woman’s overall well-being in midlife. Discussing the risks and benefits of MHT and demystifying old data can help enhance one’s overall health and quality of life.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
