Abstract
Introduction/objective:
As the coronavirus disease 2019 (COVID-19) pandemic progressed, the virus was found to cause long-term health complications known as long COVID (LC). This study aimed to investigate LC symptom severity and the factors associated with the likelihood of persistence beyond 1 year among COVID-19 survivors in Saudi Arabia.
Methods:
This descriptive, cross-sectional, questionnaire-based study was conducted via convenience sampling between December 1, 2023, and March 1, 2024. In-person interviews were performed, and 845 individuals with persistent symptoms after acute COVID-19 were included.
Results:
Hair loss and memory impairment were the most reported symptoms. In predicting LC persistence beyond 12 months, women were found to have higher odds of being symptomatic than men, and individuals from moderate-to-high-income households were more likely to report persistent symptoms than those from low-income households. Each additional acute COVID-19 symptom increased the likelihood of persistent symptoms by 1.14 times. Reporting more symptoms in the first 6 months post-infection significantly reduced the odds of long-term symptoms by approximately 30%.
Conclusion:
LC symptom severity varies among patients, and sociodemographic and clinical factors influence the likelihood of experiencing symptoms beyond 1 year. Understanding these factors can provide insights and help optimize management, leading to improved patient outcomes.
Introduction
Globally, the coronavirus disease 2019 (COVID-19) pandemic has had a profound and lasting impact, leading to unprecedented public health challenges.1 -3 COVID-19 primarily targets the respiratory system but also affects multiple organs, causing a broad spectrum of clinical manifestations, ranging from asymptomatic cases to severe respiratory failure.4,5 The rapid transmission and high infectivity of the virus have overwhelmed healthcare systems, leading to considerable morbidity and mortality worldwide.6,7
The impact of COVID-19 on health in Saudi Arabia has been significant. The virus spread rapidly across the country, resulting in over 400 000 infections and more than 6000 deaths as of July 2021. The concentration of cases in major regions such as Riyadh and Mecca underscore the vulnerability of densely populated areas. The Saudi government implemented strict preventive measures, including international travel bans, the closure of educational institutions, and restrictions on access to holy sites. These efforts helped mitigate further outbreaks, although the healthcare system remained under strain due to high infection rates in certain governorates. 8
Furthermore, the effects of COVID-19 on health-related quality of life (HRQoL) in Saudi Arabia have been substantial, particularly among adult patients who experienced varying degrees of symptom severity. A study conducted on 310 Saudi adults found that COVID-19 severity had an inverse relationship with HRQoL, with patients experiencing more severe symptoms reporting lower HRQoL scores. The study also highlighted that COVID-19 significantly impacts mental health, with long-term symptoms, such as fatigue and cognitive impairment, contributing to ongoing reductions in HRQoL. These findings underscore the need for continued healthcare support, particularly in mental health services, to manage the long-term effects of COVID-19 in the Saudi population. 9
Moreover, during the infection, patients reported a mean EuroQol 5 Dimension (EQ-5D-5L) index score of 0.68 and a EuroQol Visual Analog Scale (EQ-VAS) score of 63.16, indicating considerable impairments in mobility, self-care, daily activities, pain/discomfort, and anxiety/depression. However, 2 weeks after recovery, these scores improved to 0.92 and 86.96, respectively, demonstrating significant recovery across all HRQoL dimensions. Factors such as having a normal weight, being employed, not being anemic, and previous Bacillus Calmette-Guérin (BCG) vaccination were positively associated with greater improvements in HRQoL. Conversely, individuals with chronic conditions, such as asthma or obesity, experienced delayed recovery and lower HRQoL improvements. These findings suggest that socio-demographic and health-related factors significantly influence the recovery trajectory, emphasizing the need for personalized interventions to improve patient outcomes. 10 As the pandemic progressed, it became evident that the virus caused acute illness and long-term health implications. One of the most concerning aspects has been the emergence of long COVID (LC). LC refers to a constellation of symptoms that develop, relapse, or persist for at least 3 months beyond the acute phase of the infection and cannot be explained by other diagnoses.11,12 LC can affect multiple organ systems, leading to symptoms of varying severity and duration. Commonly reported symptoms include chronic fatigue, brain fog, fever, dyspnea, hair loss, and chronic pain encompassing joints and muscles.13 -19
LC severity varies widely among patients. Some individuals experience mild symptoms, whereas others experience severe debilitating conditions that greatly impact their daily lives and productivity.20,21 The severity of LC symptoms contributes to the overall burden of the disease, affecting not only individual patients but also healthcare systems and economies. 22 Severe symptoms can lead to long-term disability and increased healthcare utilization, underscoring the need for effective management strategies. 23
Notably, the number of symptoms experienced during the acute phase of COVID-19 directly correlates with LC duration. Fernández de las Peñas et al 24 indicate that patients who initially report a higher number of symptoms are more likely to experience prolonged symptoms that extend beyond 1 year. Furthermore, individuals who experience severe acute COVID-19, particularly those requiring hospitalization or intensive care, are more susceptible to LC. Preexisting health conditions also considerably influence the likelihood of developing LC symptoms. Patients with chronic illnesses such as cardiovascular diseases, diabetes, and respiratory conditions are at increased risk of LC, and the presence of multiple comorbidities exacerbates this risk. 25
Despite extensive international research on LC, there are limited studies and considerable uncertainty regarding symptom severity. This knowledge gap presents challenges in understanding the burden of LC and its implications for public health. Additionally, there is a notable gap in studies specific to Saudi Arabia. Existing literature in this region is limited and does not adequately address the long-term sequelae of COVID-19. This study aimed to investigate the LC symptom severity, and the factors associated with the risk of persistence beyond 1 year among COVID-19 survivors in Saudi Arabia. Understanding these aspects is crucial for developing targeted interventions and improving patient outcomes.
Methods
Study Design
Between December 1, 2023, and March 1, 2024, a descriptive, cross-sectional, questionnaire-based study was performed involving 845 individuals who had persistent symptoms after acute COVID-19 and were residing in Saudi Arabia. The convenience sampling method was used, and data were collected via face-to-face interviews in public community settings. The study was explained to the participants, and written informed consent was obtained. The ethical guidelines outlined in the Declaration of Helsinki were strictly adhered to throughout the data collection, handling, and storage processes, ensuring the confidentiality of all participants. The study protocol was reviewed and approved by our Institutional Review Board.
Study Sample
The study included patients aged 18 years or older who experienced persistent symptoms after the acute COVID-19 phase (the first 4 weeks since the initial infection). Patients with COVID-19 who did not experience any persistent symptoms beyond the acute phase of the disease, those who declined to provide consent or disclose all required information, and those under 18 years were excluded. With a precision of 5% and an alpha level of .05, a sample size calculation was performed using Epi Info 7.0. A minimum sample size of 251 patients was required.
Data Collection
Trained volunteers administered the survey using a tablet device to collect participant responses. The survey was designed using questionnaires derived from previously published literature.26 -29 The survey included questions regarding sociodemographic factors, risk factors, clinical manifestations of acute COVID-19, and persistent symptoms. Family physicians reviewed and refined the wording to enhance the validity. Acute COVID-19 is identified by the presence of COVID-19 signs and symptoms that persist for up to 4 weeks following the initial infection. 30 LC is a term used to describe signs and symptoms that occur during or after acute COVID-19, lasting 12 weeks or more, and cannot be attributed to any other specific diagnosis. 11
Statistical Analysis
The mean and standard deviation were used to describe continuous variables. The Kolmogorov-Smirnov test was used to assess the statistical normality assumption for the metric variables. Metric variables with statistical normality assumption violations, such as skewness, were described using median and interquartile range (IQR) scores. Categorical variables were described with frequencies and percentages. Multiple response dichotomies analysis was used to describe the variables measured with more than 1 option, such as the COVID-19 symptoms. Multivariable logistic regression was applied to the odds of having persistent COVID-19 symptoms beyond 12 months. This was performed by regressing the symptomatic state across 5 repeated periods against sociodemographic characteristics and other relevant disease-related factors and outcomes. The association between the independent predictor variables in the multivariable logistic binary regression analysis and the analyzed outcome variables was expressed as multivariable-adjusted odds ratios with 95% confidence intervals. SPSS IBM (version 2) was used for statistical analysis. P-values < .050 indicated statistical significance.
Results
The baseline sociodemographic characteristics of the study participants (n = 845) are summarized in Table 1. The sample comprised 51.7% females and 48.3% males, with a mean age of 34.10 ± 12.8 years. Of the participants, 56.4% were married, and 57.3% had a university degree or higher. Regarding socioeconomic status, 0.8% had a very low household income, 6.2% had a low income, and 93.1% had a medium or higher income. The mean body mass index (BMI) was 26.61 ± 5.17. Regarding smoking habits, 73.4% of the participants were never smokers. Finally, 44.6% of the participants had no chronic diseases, 29.1% had 1 chronic disease, and 24.1% had 2 or more chronic diseases.
Baseline Sociodemographic Characteristics of Study Participants (n = 845).

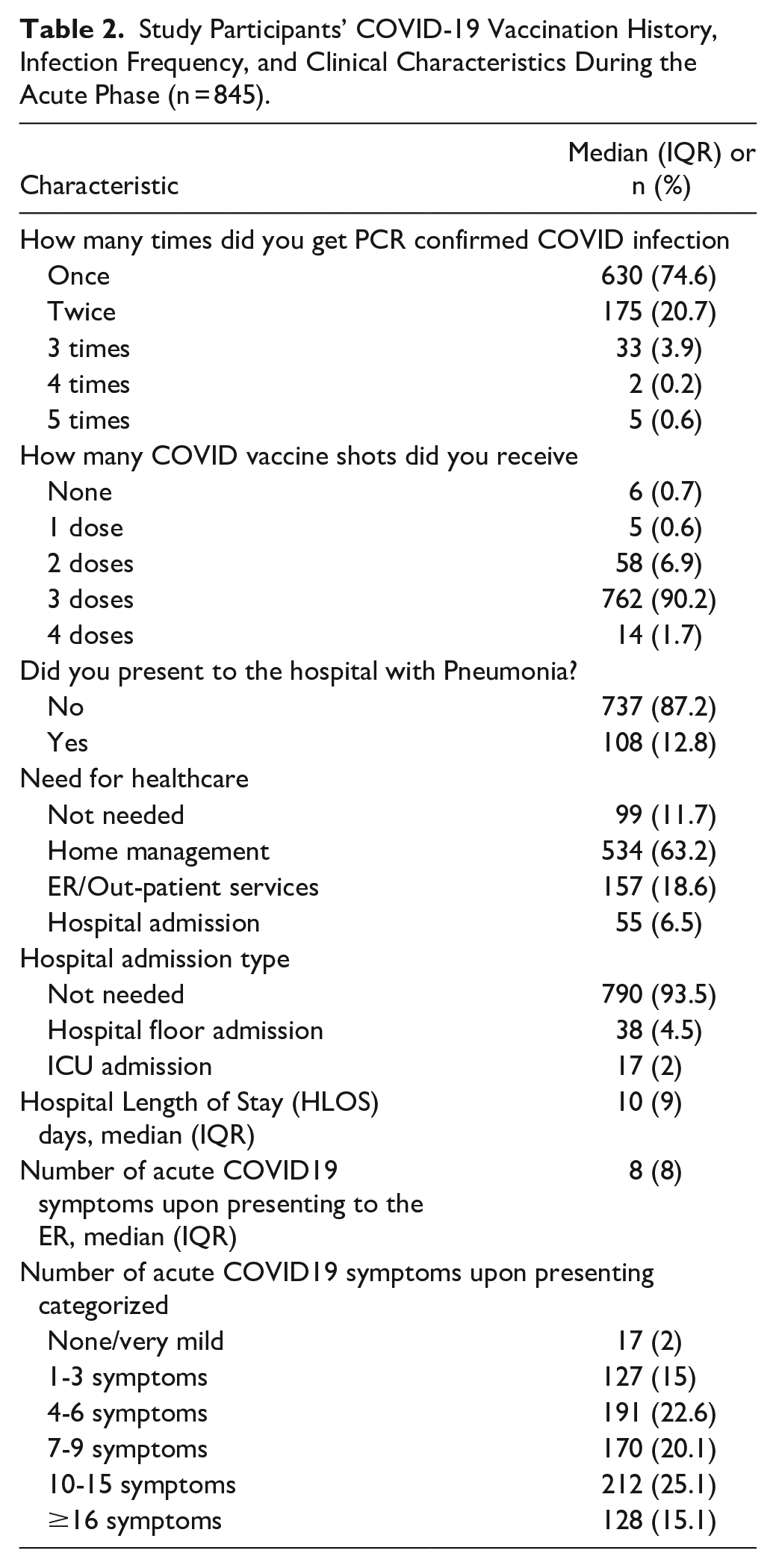
Table 2 presents a descriptive analysis of the participants’ experiences with COVID-19 and its main outcomes during the acute phase (≤1 month). Most participants (74.6%) were diagnosed with COVID-19 only once. Regarding COVID-19 vaccination, 91.9% received at least 3 doses. During the acute phase, only 12.8% of patients presented to the hospital with pneumonia. Furthermore, 63.2% managed their condition through home quarantine, whereas 6.5% required hospital admission. Among those hospitalized, 4.5% were admitted to general floors, and 2% required intensive care unit admission, with a median hospital length of stay of 10 days (IQR = 9 days). Eight was the median (IQR = 8) number of COVID symptoms upon presenting to the emergency room.
Study Participants’ COVID-19 Vaccination History, Infection Frequency, and Clinical Characteristics During the Acute Phase (n = 845).
The most frequent acute COVID-19 symptoms were fever (79.2%), cough (77.5%), fatigue (61.5%), headache (50.0%), shortness of breath (47.7%), anosmia (45%), and sore throat (42.9%). A comprehensive overview of the prevalence of acute COVID-19 symptoms is shown in Figure 1.

Percentage of participants reporting acute signs and symptoms of COVID-19 (n = 845).
1 to 3 months: The most reported persistent COVID-19 symptoms within 1 to 3 months after infection were cough (11.2%), anosmia (6.6%), bodily fatigue (6.6%), headaches (5.3%), shortness of breath (3.9%), and exertional dyspnea (3.0%).
3 to 12 months: The most frequently experienced symptoms were fatigue (17%), cough (14.4%), hair loss (7.8%), anosmia (7.1%), and memory issues (4.1%).
>12 months: The most common persistent COVID-19 symptoms were hair loss (13.6%), memory impairment (9.1%), concentration problems (7.9%), fatigue (7.9%), and exertional dyspnea (5.6%) (Table 3).
Proportion of People with Persistent COVID-19 Symptoms Across Time Points Post-Infection (n = 845).

Symptom Patterns Across Time Points
In examining the symptom patterns over different time points, some symptoms, such as cough and memory issues, showed noteworthy trends and patterns. Cough was reported in 11.2% of patients at 1 to 3 months, increased to 14.4% at 3 to 12 months, and decreased to 2.5% after 12 months indicating an initial high prevalence that further increased before significantly dropping after 12 months. In contrast, memory impairment showed a consistent increase over time, starting from 0.4% at 1 to 3 months, increasing to 4.1% at 3 to 12 months, and increasing to 9.1% after 12 months. Anosmia and headaches showed a general decline, with headaches slightly increasing after 12 months. These patterns underscore the dynamic nature of symptom prevalence over different periods, with some symptoms peaking early and then diminishing, whereas others steadily increased over time.
Figure 2 illustrates the perceived severity of the most commonly reported persistent COVID-19 symptoms. The symptoms with the highest perceived severity were hair loss (38.4%) followed by memory impairment (19.1%). Fatigue, cough, and exertional dyspnea were rated moderately severe by 56.9%, 51.9%, and 40.2% of the participants, respectively. Mild symptoms were also frequently reported, including anosmia (54.6%), headaches (45.3%), and cough (40.1%). These results indicate a varied perception of symptom severity among participants.

Distribution of severity of top-reported persistent COVID-19 symptoms (n = 845).
Multivariable binary logistic regression analysis revealed several significant predictors of COVID-19 symptoms 12 months post-infection. Sex was a significant factor, with women being 1.825 times more likely to be symptomatic than men (P < .01). Socioeconomic status also played a role, as moderate-to-high-income households were 1.449 times more likely to report symptoms after 12 months than those from low-to very low-income households (P < .001). The number of acute COVID-19 symptoms was positively associated with the odds of persistent symptoms, with each additional acute symptom increasing the likelihood by a factor of 1.143 times (or 14.3% times higher) on average (P < .001). Conversely, the number of symptoms reported in the first 1 to 3 months post-infection was negatively associated with long-term symptomatology, with a 36.5% reduction in the odds of each additional symptom (P < .001). The symptoms reported at 3 to 6 months also showed a significant negative association with the odds of being symptomatic after 12 months (P < .001). However, age, BMI, and chronic disease were not significantly associated with persistent symptoms (P > .05). Symptoms during the 6- to 12-month period and other measured predictor variables did not significantly impact long-term symptomatology when tested in different iterative analysis models (Table 4).
Multivariable Logistic Regression Analysis of People’s Odds of Having Persistent COVID-19 Symptom After 12 Months (n = 845).

Outcome: symptomatic state after 12 months versus previous period
Discussion
In this community-based study of LC among adults in Saudi Arabia, we examined persistent symptoms following acute infection by assessing their severity and identifying significant risk factors linked to symptom persistence beyond 1 year.
The most frequent LC symptoms in our sample were fatigue, hair loss, cough, memory impairment, anosmia, concentration problems, exertional dyspnea, and headaches, which are comparable to those reported in the literature. 27
In line with previous studies, we identified female sex as a significant risk factor for LC.27,31 -34 Autoimmunity has been established as a characteristic feature of LC, 35 which could explain its higher occurrence in women. 36 Additionally, women are more likely to disclose symptoms to their healthcare providers. 37 Numerous studies have demonstrated an association between advancing age, high BMI, and preexisting chronic diseases with the development of LC.3,13,38 Obesity induces a hyperinflammatory state, leading to dysfunctional immune responses that may prolong recovery time. 39 Furthermore, obesity is often associated with comorbidities such as hypertension and cardiovascular disease, 39 which further exacerbate this condition. 40 Similar mechanisms of immune dysregulation and chronic inflammation are observed with aging, potentially explaining the higher risk of LC in older individuals. 41 In our study, age was found not to be a significant predictor of symptom persistence. Moreover, the BMI and number of chronic diseases showed a positive, although non-significant, association with persistent symptoms. The lack of statistical significance may be attributable to several factors, including the limited variability in the above-mentioned characteristics of our study population, especially the low proportions of participants with older age, higher BMI (in the obese categories), and a greater number of comorbidities, as well as the multifactorial nature of LC. Notably, nearly half of the participants (50.1%) were from a younger age group (20-30 years), while 78.5% had a BMI categorized as overweight or below. Additionally, only 24.1% of participants reported having comorbidities, defined as 2 or more chronic diseases. This limited variability may have restricted our ability to adequately assess associations between higher values of these factors and symptom persistence. Despite the non-significant results, the positive trends observed for BMI and the number of chronic diseases suggest these factors may still contribute to the persistence of LC symptoms. Many studies have shown a correlation between higher levels of socioeconomic deprivation and an increased risk of LC.31,34,38 However, our data revealed that persistent symptoms were most prevalent among individuals in the moderate-to-high socioeconomic groups. This trend is consistent with the findings of Rabiee Rad et al, 42 who observed that non-hospitalized patients from lower socioeconomic backgrounds reported a lower incidence of LC-related cardiovascular symptoms. Potential explanations include the increased cost of healthcare services, which is likely to discourage disadvantaged groups from seeking medical care, resulting in the underreporting of symptoms. 43 Conversely, individuals from higher socioeconomic backgrounds have better access to healthcare, 44 allowing them to rule out other potential causes of their symptoms, with LC remaining the most plausible explanation. Lastly, it is worth mentioning that patients with moderate-to-high socioeconomic status represented the majority of the sample (93.1%) and could have received more emphasis.
The present study revealed a positive association between the number of acute-phase symptoms and the probability of LC symptoms after 12 months. This was documented in a previous study in which LC was associated with having more than 5 symptoms during the first week of illness. 13 This independent risk factor is predictive in all age groups and sexes,13,45 with an increased risk of 1.3 times for every additional acute symptom. 46 However, LC can manifest in patients with mild illness. 45 In terms of the association between the timing at which symptoms develop and their chances of persisting, a recent study found that symptoms are likely to last longer if they develop between weeks 5 and 12 following COVID-19 diagnosis. 47
Moreover, another study found a positive association between symptoms experienced 3 months after infection and their persistence for over a year; however, this was prevalent in only 15.1% of patients. 48 Contrary to previous studies, we found a negative association between the number of persistent symptoms experienced in the first 6 months following acute infection and the odds of staying symptomatic beyond 1 year. To our knowledge, this is the first study to examine the relationship between the number of symptoms during the first 6 months and the chances of persistence beyond 12 months.
This study quantified the severity of LC symptoms, an area that remains underexplored in the current literature, particularly in Saudi Arabia. The most prevalent respiratory symptoms of LC are dyspnea and cough, with prevalence rates ranging from 24.1 to 52.46% and 18.01 to 43.9%, respectively.49 -51 In our study, cough and exertional dyspnea were the most common symptoms observed in 16.9% and 8.6% of participants, respectively. The prevalence of respiratory symptoms is notably lower, which may be attributed to the high vaccination rate in our sample. Receiving 2 doses of the vaccine before infection has substantially reduced the incidence of LC, 52 with risk reduction ranging from 41% to 50%.53,54 However, 1 study suggested that these doses only offer partial protection, 55 whereas another study revealed no significant disparities in LC risk between vaccinated and unvaccinated individuals. 56 Also, 1 study reported worsening of LC symptoms following vaccination in some patients. 57
COVID-19 vaccines are effective in addressing LC in several ways. First, the primary infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) can be prevented. 58 Second, the severity of breakthrough infections can be mitigated. 55 Lastly, the vaccine can provide therapeutic benefits to individuals already experiencing persistent symptoms. 52 Respiratory symptoms were among the least reported severe LC symptoms, with exertional dyspnea being severe in 9.3% and cough in 8% of cases, which may also be attributed to vaccination.
Hair loss, memory impairment, fatigue, anosmia, and headache were the most severe among the persistent symptoms. Approximately 38.4% of the participants reported severe hair loss, similar to reports from another study where 44% of women and 9% of men reported experiencing moderate or severe hair loss. 59 SARS-CoV-2 utilizes angiotensin-converting enzyme 2 receptors and transmembrane protease serine 2 co-receptors to invade cells, 60 possibly destroying hair follicles directly, or damage could occur as a result of a cytokine storm. 61 Furthermore, psychological stress caused by the COVID-19 pandemic might have also played a role.62,63
The prevalence of LC memory impairment in our sample was 13.2%, which was within the previously reported range of 11% to 72.8%.49,64,65 Memory impairment is equally prevalent in all age groups. 49 Our findings indicate that the likelihood of developing memory issues progressively increased following acute COVID-19, aligning with the trends observed in previous studies. 49 Our study found that 19.1% of participants experienced severe memory impairment, which had major effects on daily activities. 49 Damage to the hippocampus, which plays an essential role in memory, 66 directly or through inflammatory mediators or hypoxia,67,68 may cause memory impairment in patients with LC.
Fatigue is the most common LC symptom.15,69 In the present study, the overall prevalence of fatigue was 24.9% and was described as moderate or severe in 70.4% of patients. Of these, 31.8% reported fatigue lasting longer than 12 months after the acute COVID-19 infection. This was less than the previously reported prevalence of 41% after more than 12 months. 29 Fatigue can result from autonomic nervous system dysfunction, 70 and psychological factors are likely to be involved. 71
Approximately 11% of the participants in our study reported anosmia as an LC symptom, which is significantly lower than the previously reported 30.3%. 72 In the current study, more than half (54.6%) of the patients experienced only mild disruption, whereas 12.5% reported severe symptoms. Severe anosmia was slightly higher than the 8.5% observed in previous studies. 72 Anosmia can occur before the appearance of respiratory symptoms, 73 and may be the sole manifestation of COVID-19.74,75 Anosmia is postulated to result from the viral invasion of neuroepithelial cells and subsequent inflammation.76 -78
LC headaches were reported by 8.5% of the participants, compared with previously documented 8.4% to 10.6%, 79 with 54.7% being moderate or severe. The risk of developing headaches after recovery is not associated with the severity of acute COVID-19. 80 Headaches cause significant disability in patients with LC, and those who experience headaches before COVID-19 may have exacerbated symptoms. 81 Evidence suggests that SARS-CoV-2 can enter the brain via the nasal cavity or bloodstream, leading to neuroinflammation.69,82 Headaches following COVID-19 can be attributed to infection-induced irritation of the trigeminal nerve83,84 or neurotoxin release associated with cytokine storms. 85
LC symptoms markedly affect quality of life47,86,87 and activities of daily living. Seropositive individuals experience a significant impact on their social and home lives, 88 and even mild cases of COVID-19 can lead to persistent activity limitations months after acute infection. 89 Approximately 20% of hospitalized patients with COVID-19 require readmission,90,91 and over one-third require an average of 2 additional medical visits, 92 contributing to the burden of LC on healthcare systems. Furthermore, a notable proportion of formerly healthy workers experience difficulties returning to their jobs93 -96 or need to reduce their work schedules after recovery. 49 These issues highlight the need for specialized services for LC and pose medical challenges, financial consequences, and substantial increases in healthcare costs.87,92
Since the effects of COVID-19 can persist long after the initial infection, impacting most bodily systems and significantly hindering the quality of life, long-term studies are needed to determine the duration of these effects and the necessary medical interventions. Standardizing definitions for LC will advance symptom detection and improve patient outcomes.
This study has several strengths. First, trained volunteers conducted face-to-face interviews, enhancing the response accuracy. Second, a wide range of LC symptoms was investigated, encompassing all body systems. Third, information regarding the onset, duration, and severity of symptoms was collected. Finally, the study was performed more than 3 years after the World Health Organization classified COVID-19 as a pandemic. This duration provided ample time for the development of LC symptoms. Regarding limitations, recall bias could have resulted owing to the retrospective nature of the study. Unmeasured risk factors, such as the participants’ occupations, were also present, which might have contributed to LC.
Conclusion
The effects of COVID-19 can last beyond the initial period of infection, including changes in quality of life and persistence of symptoms. Prolonged symptoms can result from direct tissue damage caused by the virus, additional coagulation and inflammatory processes associated with acute COVID-19, or psychological factors. The effects of LC pose economic and health concerns. Extended longitudinal investigations are necessary to determine the duration of the lingering effects of COVID-19 and the specific medical attention required for these patients.
Future studies should aim for conducting nationwide research involving more regions of Saudi Arabia, including more diverse populations, particularly with greater representation of older individuals, those with higher BMI, and patients with more comorbidities, to better assess their potential associations with LC symptom persistence. This could provide a better understanding and representation of the population and, thus, guide future interventions. Additionally, the multifactorial nature of LC underscores the need for more comprehensive approaches that account for a wider range of contributing factors. Developing consistent definitions and diagnostic criteria for LC will enable standardized symptom identification and comparability of results across different studies. Health policies should include these findings in clinical care pathway designs to meet the needs of individuals with LC.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study protocol was reviewed and approved by the Institutional Review Board of Imam Abdulrahman Bin Faisal University, located in the eastern province of Saudi Arabia (approval number: IRB-2024-01-209).
Consent to Participate
All patients provided written informed consent.
Consent to Publish
Not applicable.
Data Availability Statement
This study’s data, including participant data and a data dictionary, will be made available to researchers with a methodologically sound proposal and approved data access agreement.
