Abstract
Background:
In addition to the morbidity and mortality associated with acute infection, COVID-19 has been associated with persistent symptoms (>30 days), often referred to as Long COVID (LC). LC symptoms often cluster into phenotypes, resembling conditions such as fibromyalgia, postural orthostatic tachycardiac syndrome (POTS), and myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). LC clinics have been established to best address the needs of LC patients and continuity of care. We developed a cross-sectional survey to assess treatment response through our LC Clinic (LCC).
Methods:
A 25-question survey (1-10 Likert scale) was expert- and content-validated by LCC clinicians, patients, and patient advocates. The survey assessed LC symptoms and the helpfulness of different interventions, including medications and supplements. A total of 852 LCC patients were asked to complete the survey, with 536 (62.9%) responding.
Results:
The mean time from associated COVID-19 infection to survey completion was 23.2 ± 6.4 months. The mean age of responders was 52.3 ± 14.1 (63% females). Self-reported symptoms were all significantly improved (P < .001) from the initial visit to the LCC (baseline) to the time of the follow-up survey. However, only 4.5% (24/536) of patients rated all symptoms low (1-2) at the time of the survey, indicating low levels of full recovery in our cohort. The patients rated numerous interventions as being helpful, including low-dose naltrexone (45/77; 58%), vagal nerve stimulation (18/34; 53%), and fisetin (28/44; 64%).
Conclusions:
Patients report general improvements in symptoms following the initial LCC visit, but complete recovery rates remain low at 23.2 ± 6.4 months.
Introduction
Post-acute sequelae of SARS CoV-2 infection (PASC), also referred to as Long COVID (LC) is a condition defined by persistent symptoms and/or long-term complications lasting over 4 weeks from the onset of the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection.1,2 PASC has been subdivided into 2 categories: (1) current or subacute coronavirus disease of 2019 (COVID-19) symptoms ranging from the onset of disease to 4-12 weeks beyond the active infection, and (2) chronic or post-COVID-19 syndrome which comprises of symptoms persisting beyond 12 weeks from the onset of acute SARS-CoV-2 infection. 3 LC has been associated with as many as 84 different symptoms, including fatigue, headache, attention disorder, hair loss, cough, myalgia, joint pain, impaired mobility, cognitive impairment (which includes brain fog and memory loss), olfactory and gustatory dysfunction, sleep disorder, depression, anxiety, posttraumatic stress disorder, gastrointestinal upset, rashes, and palpitations.4 -7 While there is no consensus on the etiology of LC, several theories have emerged, including persistent reservoirs of virus or viral particles triggering a persistent immune response, immune dysfunction leading to chronic inflammation, endothelial cell dysfunction, altered perfusion due to thromboses, dysautonomia, and mitochondrial dysfunction.8 -10 Besides the severity of the acute COVID-19 illness, various sociodemographic factors and pre-existing health conditions are linked to increased risk for LC, including female sex, smoking, obesity, anxiety, depression, socioeconomic factors, and some autoimmune diseases.11,12
LC has been estimated to be present in an estimated 65 million patients worldwide, estimated to represent 6%-20% of the population who have recovered from acute COVID-19 infection.13 -15 Although LC prevalence was initially reported as decreasing; recent data does not support this. 14 The impact of LC has been devastating, with a recent study demonstrating that only one-fourth of patients resumed their pre-LC full-time work duties, and some studies have estimated the annual economic impact of LC at $170 billion in lost wages in the United States alone.16,17 Furthermore, almost half worked reduced hours, and 23% were not working at all as a direct result of LC. 16 A study evaluating patients 6 months after hospital discharge for COVID-19 found that almost 50% reported functional limitations in activities of daily living. 18
Despite the high prevalence and significant impact on quality of life, there are no current FDA approved treatments for LC. The National Institutes of Health (NIH) is funding a large multi-center research treatment and therapeutic programs as part of the Researching COVID to Enhance Recover (RECOVER) program. 19 Until there are widespread effective treatments, however, LC clinics (LCC) and primary care clinicians (PCCs) will need to use multiple approaches, including off-label medicines and interventions, many of which have been previously used for postural orthostatic tachycardiac syndrome (POTS), myalgic encephalomyelitis chronic fatigue syndrome (ME/CFS), and fibromyalgia. 20 However, data on LC therapies remains limited, and while many LC clinics (LCCs) exist, there is a paucity of knowledge about their efficacy.
In late 2020, in response to the rising prevalence of LC, we established an LCC known as the Post-COVID Care Clinic (PCoCC), which offers a multidisciplinary approach to the treatment of LC symptoms combining use of off-label medications and nonpharmacologic rehabilitative approaches, many of which have previously been used for POTS, ME/CFS, and fibromyalgia.7,21 PCoCC is staffed by General Internal Medicine physicians with expertise in treating complex and undefined chronic diseases including POTS, ME/CFS, and fibromyalgia. 22 Specialty care in areas such as Neurology, Physical Medicine & Rehabilitation, Pulmonary, and Cardiology is coordinated as needed by the PCoCC physician. We have previously published on diagnostic testing in PCoCC patients (i.e., IL-6, and PET scans)9,23 but have not assessed the efficacy of our LCC and our patients’ recovery in the months after they returned home. While many LCC multidisciplinary treatment programs exist, there is a paucity of knowledge about their efficacy. In order to address this knowledge gap in the medical literature, we performed a cross-sectional survey assessing patient outcomes after treatment in our LCC. Here, we report the outcomes of the survey and discuss their implications in the care of LC patients.
Methods
This is a cross-sectional follow-up survey of patients who presented to the PCoCC for treatment. This survey was a practice improvement initiative so was deemed exempt from Mayo Clinic Institutional Review Board review.
Setting
Patients aged 18 years or older from across the United States who presented to the LCC of a large quaternary care health center between January 1, 2021, and October 27, 2022, were identified from the electronic medical record (EMR) and invited to participate in this survey. Patients aged under 18 years old were evaluated in a separate pediatric LCC, so were not a part of this sample.
Survey Population
A total of 852 patients were identified, and an electronic survey was sent via their EMR patient portal. Those patients who did not complete the electronic survey were then contacted by nurse outreach, independent of the project team to attempt to complete the survey via a telephone call. The patient flow is shown in Figure 1.

Patient consort diagram.
Survey
The survey was developed by PCoCC physicians. We then had a group of 4 LC patients and 2 patient advocacy leaders validate the survey content and strength of the questions. This patient expert group assessed the ease of understanding the questions in the survey, the ease of answering the questions, and the level of detail provided in the survey. The feedback was reviewed by the project team (including the PCoCC physicians) and the survey was refined to its final form. This validation process followed the methodology we had developed for validating other surveys. 24
The survey included a series of questions utilizing a scale from 1 (not significant) to 10 (extremely significant) (Supplemental Appendix 1). Patients were asked to rate their symptoms when they came to the clinic for initial evaluation (recall) and their symptoms at the time of filling out the survey (present). In addition, the survey asked about treatment options for LC that were being commonly trialed at the time, including non-pharmacologic interventions (post COVID treatment program, physical therapy, occupational therapy, biofeedback, etc.), as well as medications (low-dose naltrexone (LDN), propranolol, amitriptyline, colchicine, midodrine, fludrocortisone, etc.), or supplements (fisetin, coenzyme Q10, ginseng, ashwagandha, Reishi mushrooms, specialized pro-resolving mediators [SPMs]), and how helpful they found these treatments. Of note, multiple treatments that our PCoCC physicians have been offering more recently, such as guanfacine, NAC, and L-arginine, were not captured in this survey due to the timing of the survey collection.
PCoCC Patient Flow
Patients who have had persistent symptoms after COVID-19 infections are either self- or physician-referred to the PCoCC. Our LC definition in the PCoCC was persistent symptoms >28 days following the acute COVID-19 infection, though most of our patients presented at over 6 months since their inciting infection. All PCoCC patients completed a standardized questionnaire containing 52 questions about the initial COVID-19 infection, symptoms, and treatment along with their ongoing and persistent symptoms. These patient questionnaires are then reviewed by a PCoCC clinician. Patients with symptoms limited to a single organ system are directly referred to the subspecialty team for the management of LC symptoms. Those with symptoms involving multiple organ systems are initially evaluated by a 30-minute virtual visit for the purpose of introducing the PCoCC and pre-ordering tests and consultations as appropriate. This is followed by an in-person consultation with a PCoCC physician at a later date. After receiving an individualized treatment plan from a physician, patients proceed with a virtual treatment program designed to provide education on LC pathophysiology, pacing, and sympathetic reduction techniques, along with health coaching and gentle patient-directed rehabilitation as tolerated. This is an 8-h long program delivered in two 4-hour virtual segments, accompanied by health coaching and nursing follow-up for 3 months (Post COVID Treatment Program).
Statistical Analysis
The survey data was uploaded into Research Electronic Data Capture (REDCap, 2004 Vanderbilt). 25 The data were then analyzed using SAS version 9.4 (SAS Institute Inc, Cary, NC). Respondent characteristics are summarized using mean ± SD and median (25th, 75th) for continuous variables and frequency counts and percentages for categorical variables. Respondents were asked to rate the negative impact of individual symptoms, and overall level of functioning, when first seen in the PCoCC for LC symptoms (baseline), and at the time of the follow-up survey, using a scale from 1 to 10, with 1 being “not significant” and 10 being “extremely significant.” These symptom severity ratings were summarized using the median (25th, 75th) and compared between time periods using the signed rank test. To summarize the percentage of patients who had recovered from LC at the time of the survey, patients who reported a score of 1 or 2 for all symptoms assessed were classified as recovered. In all cases, 2-tailed tests were used with P < .05 considered statistically significant.
Results
A total of 852 patients were identified to receive a follow-up survey and 536 (74%) patients eventually completed the survey. Of the 536, 308 patients responded to the initial digital survey sent in the EMR. The remaining 228 non-responders completed the survey over the telephone with nursing outreach.
Demographics
The mean age of responders was 52.3 ± 14.1 years, with a majority (63%; 340/536) being female, white (93%), non-Hispanic (96%), and married (72.7%, 390/536). The average time between the initial COVID-19 infection and the follow-up survey was 23.2 ± 6.4 months (Table 1). Demographics did not differ significantly between responders and non-responders, except for age, with responders being significantly older than non-responders (52.3 ± 14.1 years vs 48.2 ± 15.9 years; P < .001) where non-responders ages were calculated using the median date of survey completion for responders.
Patient Characteristics. a

Continuous variables are reported as mean ± SD and median (Q1, Q3). Categorical variables are reported as number (%).
Time since the first positive test was missing for 1 patient.
Symptom Severity
Overall, symptoms reported at first presentation to the PCoCC (baseline) included fatigue, brain fog, shortness of breath, muscle pain, headache, rapid heart rate, insomnia, dizziness, balance difficulties, chest pain, and tinnitus (Table 2). Level of functioning was measured using a numeric scale of 1 to 10 (with 1 being the best and 10 being the worst imaginable). The median (IQR) level of functioning was rated as 8 ( 6, 9) at baseline and 5 ( 3, 8) at the time of the follow-up survey (P < .001) (Table 2).
Overall Level of Functioning and Symptom Severity at Baseline and Follow-up. a

Patients were asked to rate the severity of each symptom on a scale from 1 to 10, where 1 corresponded to “not significant” and 10 corresponded to “extremely significant.” Data are summarized by presenting the median (25th, 75th) and compared between time periods using the signed rank test.
Due to missing data, the number of respondents included for a given symptom ranged from 533 to 536.
All the individual symptoms assessed were significantly improved when comparing the baseline with the follow up (P < .001) (Table 2). As an example, fatigue significantly improved from a median (IQR) score of 8 (7, 10) at the baseline visit to 6 (3, 8) at the time of this survey (P < .001). Similarly, brain fog significantly improved from 8 (6, 9) at baseline to 5 (3, 8) at follow-up (P < .001). Only 4.5% (24/536) of patients who responded to the survey were determined to have recovered completely based on symptom ratings of 1 or 2 for all domains at follow-up.
While the majority of the population did experience an improvement, it is important to note that a minority of participants did experience a worsening of their overall function (Figure 2). Similar changes were seen in the specific symptoms of fatigue (Figure 3), myalgia (Figure 4), and orthostasis (Figure 5).

Overall function in LC patients treated in our LCC at baseline and at follow-up.

Fatigue in LC patients treated in our LCC at baseline and at follow-up.

Myalgia in LC patients treated in our LCC at baseline and at follow-up.

Orthostasis in LC patients treated in our LCC at baseline and at follow-up.
Treatment
Treatments utilized varied greatly among the 576 participants who responded to the survey. These treatments could have been recommended by the PCoCC physicians, local physicians, or trialed independently by the patient at home. The most common treatment modality was the PCoCC Post COVID Treatment Program (404/576, 75%), with 285/404 (71%) finding it to be helpful. Physical therapy and occupational therapy were reported as being helpful by 72% (169/236) and 71% (88/124), respectively, of the patients who had tried them. Other non-pharmacologic treatments that were reported as helpful included biofeedback (85% [82/96]), vagal nerve stimulators (53% [18/34]), and plasmapheresis (100% (9/9)) (Table 3).
Interventions, Medications, and Supplements. a
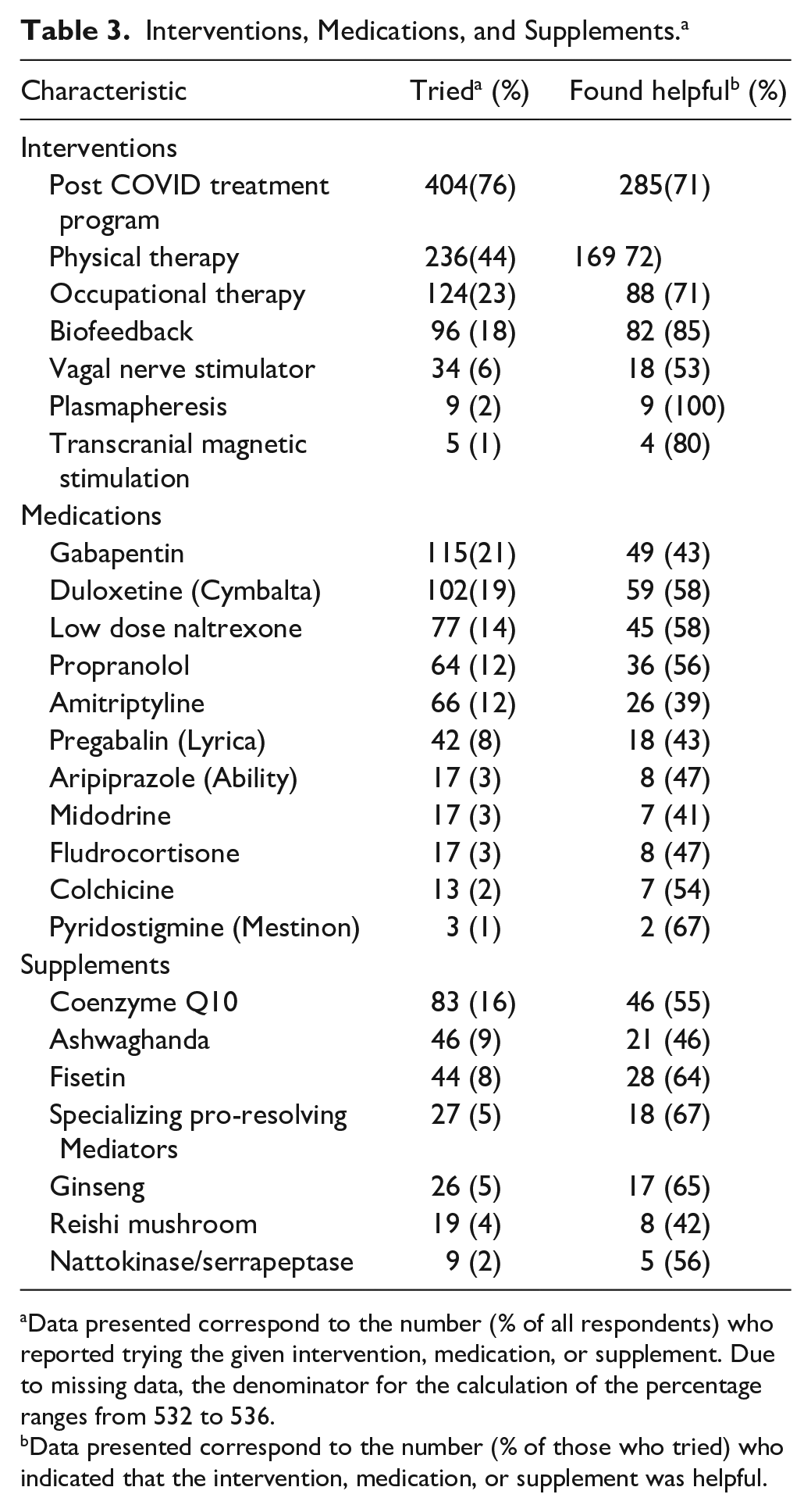
Data presented correspond to the number (% of all respondents) who reported trying the given intervention, medication, or supplement. Due to missing data, the denominator for the calculation of the percentage ranges from 532 to 536.
Data presented correspond to the number (% of those who tried) who indicated that the intervention, medication, or supplement was helpful.
Among the participants who reported the use of pharmacologic therapies to treat their LC symptoms, beneficial effects were reported for gabapentin (43% [49/115]), duloxetine (58% [59/102]), low-dose naltrexone (LDN) (58% [45/77]), propranolol (56% [36/64]), amitriptyline (39% [26/66]), pregabalin (43% [18/42]), aripiprazole (47% [8/17]), and colchicine (54% [7/13]) were all found to be helpful (Table 3). Among supplements used to help with LC symptoms, coenzyme Q10 (56% [46/83]), ashwagandha (46% [21/46]), fisetin (64% [28/44]), specialized pro-resolving mediators (SPM’s) (67% [18/27]), ginseng (65% [17/26]), Reishi mushrooms (42% [8/19]), and nattokinase/serrapeptase (56% [5/9]) were all reported to be helpful (Table 3).
Discussion
This report presents important and novel findings from a cross-sectional survey of LC patients treated in an LCC. There is a paucity of data that evaluates LCC treatment effectiveness. Most patients (71%; 285/404) reported improvement from the PCoCC integrated pacing rehabilitation approach delivered by our nursing personnel, physical therapists, and wellness coaches. Although there are no FDA-approved treatments for LC, patients in this survey also reported symptom benefits from numerous supplements, prescription medications, and devices such as vagus nerve stimulators. These included LDN (58%; 44/77), duloxetine (58%; 59/102), propranolol (56%; 36/64), SPMs (67%; 18/27), ginseng (65%; 17/26), and fisetin 64% (28/44). The overall level of functioning and other key symptoms such as fatigue, brain fog, and dysautonomia improved from the time they were initially evaluated to the time of the follow-up survey (mean time from positive test to survey 23 ± 6.4 months). These findings suggested that the integrated pacing rehabilitation approach combined with pharmacotherapies and supplements targeted at neuroinflammation may be at least partially beneficial to LC patients. Unfortunately, only 4.5% (24/536) of patients rated symptoms low (1-2) in all domains suggesting very few of our patients have recovered from LC completely.
A similar-sized (n = 341) 2-year prospective study was conducted with the goal of characterizing the clinical symptoms and factors associated with LC, as well as assessing factors associated with recovery. 26 This study was conducted in one of the largest LCCs in Spain that had provided multidisciplinary care to >1200 patients. 26 The 344 participants with LC (WHO definition) enrolled in the study were followed for a median of 23 months (16.5, 23.5). 26 Of these, only 7.6% (n = 26/341) of the LC patients were deemed to be recovered, which is similar to that observed in our study (4.5%). A patient with LC was considered to be recovered when all persistent symptoms were remitted for ≥3 consecutive months. In addition, participants with LC symptoms before diagnosis was made were considered recovered when they returned to baseline levels (pre-COVID). 26 The authors found the recovered participants were more likely to be male (RR 3.24 95% CI 1.56-6.75; P = .002), have been admitted to the ICU (RR 8.30 95% CI 2.84-24.27; P < .001), and have vascular comorbidities (RR 1.99 95% CI 1.01-3.92; P = .048). 26 Although we did not run a detailed analysis of participants who recovered, this should be a focus of future studies.
Additional studies have supported the low overall recovery rate in LC in the current setting, with few effective therapies available. The largest was a cross-sectional study conducted in Scotland using a cohort of 33 281 (31 486 symptomatic) laboratory-confirmed (PCR) acute SARS-CoV-2 infections compared to 62 957 never-infected individuals in a national database. 27 Greater than 33% (n = 10 489) of symptomatic participants reported symptom duration >4 weeks.20,27 Participants were asked to complete additional surveys at 6, 12, and 18 months evaluating the same 26 symptoms and limitations in daily living. 27 Recovery was assessed by asking participants to self-report their status of recovery (full, partial, or none). A total of 3744 participants completed both the 6 and 12-month surveys, and the number recovered at those time points was 1683 (45%) and 1736 (46%), respectively. There was little change in those reporting partial recovery (6 months = 1766, 47%; 12 months = 1705, 46%) or not recovered (6 months = 295, 8%; 12 months = 303, 8%). 27 Of those participants who filled out both surveys, only 53 additional patients reported being fully recovered at 12 months (53/3444 = 1.4%). 27 This large cross-sectional study, therefore, shows similar trends of low recovery rates in community LC patients. One additional important aspect of our low recovery findings is patients seen in our LCC had a mean of 23.2 ± 6.4 months between the initial visit and COVID-19 positive test. During that time, most of the patients in our clinic had seen numerous local specialists (e.g., neurology, cardiology, pulmonology, rheumatology, and infectious diseases) along with primary care physicians (PCP) who lacked the experience in treating LC.
Our approach to the treatment of LC patients is based on the cumulative experience of treating thousands of patients with fibromyalgia, ME/CFS, and POTS and other complex medical conditions in the Consultative Medicine (CM) Clinic of General Internal Medicine (GIM).10,22,28,29 Our CM physicians specialize in seeing patients with complex, multisystem diseases, many of which have evaded diagnosis locally. Additionally, the majority of our PCoCC physicians were participants and leaders in our institution’s acute COVID-19 efforts. These included the COVID Frontline Care Team (CFCT), which followed over 50 000 acute COVID-19 cases, pairing high risk patients with at-home monitoring, and the Monoclonal Antibody Treatment Team which delivered >20 000 doses to patients across our health system.30,31 The experience in caring for patients with ME/CFS, fibromyalgia, and POTS coupled with the breadth of expertise associated with acute COVID-19 care made this group of physicians the ideal group to take care of LC patients.
Our institution’s early experience with ambulatory LC care involved sending patients to multiple specialists (most commonly, neurology, pulmonology, cardiology, rheumatology, and infectious diseases). Testing, however, would largely result in non-specific findings that were mildly abnormal but not intervenable (i.e., sinus tachycardia, orthostatic intolerance, air trapping, D-dimer elevation, mild inflammatory marker elevation). Our LCC was developed to incorporate GIM CM’s expertise in the management of chronic post-infectious illnesses to help diagnose, treat, and coordinate subspecialty care when needed. Our LCC physicians began utilizing advanced inflammatory markers (i.e., clinical cytokine panels) and specialized testing, including PET scans and transcranial Doppler exams, autonomic tilt table testing, and nuclear joint scans in LC patients to help with diagnosis and develop treatment plans.9,23,32
As described, our model centers on developing LC expertise based on our CM model and experience gained from acute COVID-19 care and treatment teams. Other models include PCP-centric versus wide direct access to specialty care.33 -35 A recent analysis of LC care delivered at a large tertiary Midwest institution reported the results and observations from a direct access to specialty care LCC approach. 33 The cohort of 1802 LC patients was seen a median of 8 months after the initial onset of COVID-19 with 12 specialty clinics being available for consultation. 33 LC patients obtained appointments through physician referral, hospital follow up, or self-referral. 33 Neurology, cardiology, and pulmonology were the most frequently referred specialists (>85% of all visits). The authors concluded that their approach provided optimal access to specialists and testing compared to centralized internal medicine approaches.33,34 This direct entry to specialty care approach is often applied by medical centers, including our institution for complex medical care and indeed was the approach we trialed for LC early in 2020. Although this approach provided access to specialists and diagnostic testing, identified abnormalities were often only mild and not intervenable, and detailed coordinated treatment plans were lacking. In addition, even when patients were transitioned to local care with detailed treatment recommendations, there were significant barriers to the implementation of these recommendations by the PCPs, with factors such as lack of knowledge of LC, lack of FDA approved treatments, and unwillingness to prescribe medications “off-label” being most commonly cited.
One of the myriad challenges we face in LC care delivery is the lack of FDA-approved treatments. Our early phenotyping approach focused on identifying overlapping features of commonly post-infectious diagnoses, such as ME/CFS, POTS, and fibromyalgia, in order to guide treatment. 20 For example, if LC patients meet the criteria for these complex chronic illnesses, we would diagnose them as LC induced ME/CFS if we determine COVID-19 to be the inducing factor. Treatments of LC would then be based on modalities that we have found to be helpful in these phenotypes (e.g., LDN for ME/CFS and propranolol for hyperadrenergic POTS). This approach differs from the predominantly symptom clustering phenotyping recently done by NIH RECOVER. 36 In addition to guiding treatment choices with some modicum of success, this approach has helped when LC patients file for medical disability.
In the current environment, with no FDA approved therapeutics for LC and multiple therapeutic interventions being proposed for the treatment of LC, listening to the lived experience of LC patients is key to identifying what treatments and supplements may be potentially efficacious. One of the challenges with this approach is the off-label drug use (OLDU) of these non-approved therapeutics. Because the FDA does not regulate the practice of medicine, OLDU occurs in almost every specialty, particularly in areas such as LC, with patient populations that suffer from significant symptom burden but lack clear pathophysiologic understanding and a lack of pivotal clinical trials. 37 Patients seen in our LCC trialed multiple therapies ranging from nonpharmacological interventions such as physical therapy, occupational therapy, biofeedback, vagal nerve stimulator (VNS), plasmapheresis, and transcranial magnetic stimulation (TCMS); to medications like LDN, propranolol, amitriptyline, colchicine, midodrine, and fludrocortisone; and supplements such as fisetin, coenzyme Q10, ginseng, ashwagandha, Reishi mushrooms, and specialized pro-resolving mediators, among others. Interestingly, upon evaluation in our questionnaire, patients did perceive benefit from these therapies, ranging from as high as 100% reporting benefit from plasmapheresis (n = 9) to only 39% reporting benefit from a trial of amitriptyline (n = 66). Benefits from therapeutic strategies ranging from 50% to 80% were noted by patient’s utilizing physical therapy, occupational therapy, VNS, TCMS and using medications LDN, duloxetine, propranolol, colchicine, coenzyme Q10, fisetin, ginseng, and nattokinase/serrapeptase.
Having this type of patient-centric OLDU data has informed us about which therapeutics to trial in small clinical trial pilots, such as VNS. In addition, it has led to some clinicians being more open to accepting the risk of OLDU prescribing, both in our own clinic as well as local PCCs. Although current efforts are rightfully focused on finding novel therapeutics, there is a dire need to have some of the current commonly used therapies recognized as standard LC treatments. This approach will potentially allow commonly prescribed OLDU therapies to be more widely accepted by clinicians and thus be disseminated to more LC patients. When an FDA label does exist, such as with VNS for headaches, we favor trialing these therapies with potential crossover effects as first line strategies in LC patients with these conditions.
Our survey has a number of limitations. First, data on the severity of COVID-19 infection, including the need for oxygen and admission/ICU status was not routinely collected in this patient cohort, which limited the ability to assess whether LC is more prevalent in patients with severe disease, as demonstrated in previous studies. Second, we were unable to precisely delineate the relationship of severity of LC symptoms with time, so ability to advise on a generalized timeline of symptom burden is limited. Third, there is the existence of recall bias as these patients might not correctly recall symptoms at the time of LCC evaluation and indeed the reported symptoms may even have predated their COVID-19 infection. Further, recall of symptoms at a particular time point may be difficult, especially with the cognitive dysfunction associated with LC. It should also be noted that the survey was created specifically for LC patients and designed to be adaptable to future studies as treatment options change. While it would have been ideal to use previously validated studies, there were no previously validated studies that would have obtained the information we were attempting to obtain. We attempted to mitigate this limitation by having the questionnaire validated by patient and content matter experts. Future studies should administer the questionnaire real time to minimize assessments. Fourth, with a response rate of 62.9%, there is a possibility of non-response bias and therefore the findings from our survey sample may not be representative for all patients who presented to the PcoCC for treatment, especially as non-respondents tended to be younger with less comorbidities. A 63% response rate however is relatively high for a survey, and previous work suggests that the distribution of data does not differ significantly between studies with a response rates between 40% and 80%.38,39 Fifth, we did not collect data on repeat infections, which may have had implications for those patients who reported worsening of symptoms. And finally, the generalizability of these results coming from a retrospective survey at a single institute cannot be extrapolated to the general population.
Conclusions
Long COVID is a heterogenous condition characterized by a wide range of symptoms, often in recognizable phenotypes resembling post-infectious complex chronic diseases such as POTS, ME/CFS, and fibromyalgia, that may persist for years following the onset of acute COVID-19. Complete recovery from LC in our patient population is low and attributable to the complex cases we see as well as the lack of novel therapeutics. The LCC approach that has worked best for our institution, and the thousands of LC patients we have seen at the time of this paper, is centering the care with specialized GIM physicians partnered with a multidisciplinary team and, when needed, multispecialty care (e.g., neurology, cardiology, and pulmonology). Pragmatic trials of commonly prescribed medications (i.e., LDN, guanfacine, and propranolol) and devices (VNS) to set a standard of care for LC need to be supported in addition to developing novel therapeutics.
Supplemental Material
sj-pdf-1-jpc-10.1177_21501319241258671 – Supplemental material for Longitudinal Progression of Patients with Long COVID Treated in a Post-COVID Clinic: A Cross-Sectional Survey
Supplemental material, sj-pdf-1-jpc-10.1177_21501319241258671 for Longitudinal Progression of Patients with Long COVID Treated in a Post-COVID Clinic: A Cross-Sectional Survey by Ryan T. Hurt, Siddhant Yadav, Darrell R. Schroeder, Ivana T. Croghan, Michael R. Mueller, Stephanie L. Grach, Christopher A. Aakre, Elizabeth A. Gilman, Christopher R. Stephenson, Joshua Overgaard, Nerissa M. Collins, Donna K. Lawson, Ann M. Thompson, Lasonya T. Natividad, Osman Mohamed Elfadil and Ravindra Ganesh in Journal of Primary Care & Community Health
Supplemental Material
sj-pdf-2-jpc-10.1177_21501319241258671 – Supplemental material for Longitudinal Progression of Patients with Long COVID Treated in a Post-COVID Clinic: A Cross-Sectional Survey
Supplemental material, sj-pdf-2-jpc-10.1177_21501319241258671 for Longitudinal Progression of Patients with Long COVID Treated in a Post-COVID Clinic: A Cross-Sectional Survey by Ryan T. Hurt, Siddhant Yadav, Darrell R. Schroeder, Ivana T. Croghan, Michael R. Mueller, Stephanie L. Grach, Christopher A. Aakre, Elizabeth A. Gilman, Christopher R. Stephenson, Joshua Overgaard, Nerissa M. Collins, Donna K. Lawson, Ann M. Thompson, Lasonya T. Natividad, Osman Mohamed Elfadil and Ravindra Ganesh in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
The PCOCC study team would like to thank the Division of General Internal Medicine for their support of this project, our registered nurses for their assistance with survey conduct and data abstraction, and our excellent team of allied health professionals in the Consultative Medicine and Integrative Medicine and Health Clinics for their tireless support of our LC patients. In addition, we would like to thank the following study team who conducted phone interviews for those individuals not responding to the emailed surveys. This includes Barbara Bachman Lentz RN and Rachel Huber RN. Finally, a special thanks to all study participants, without their participation, this study would not have been possible.
Authors’ Contributions
All the authors participated in the study concept and design, collection, analysis and interpretation of data, drafting and revising the paper for important intellectual content, and have seen and approved the final version of the manuscript. Each author will take public responsibility for the entire work.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RTH is a consultant for Nestle Nutrition. All authors declare no support from any organization for the submitted work; no financial relationships with any organizations that might have an interest in the submitted work in the previous 3 years; and no other relationships or activities that could appear to have influenced the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication was supported in large part by the GHR Foundation (Minneapolis, MN). In addition, the publication was supported by grant number UL1 TR002377 from the National Center for Advancing Translational Sciences (NCATS). Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIH.
Ethics and Consent to Participate
This project was a practice improvement initiative. Institutional Review Board review was not needed.
Availability of Data and Materials
Deidentified study data that supports the results can be shared beginning 9 to 36 months following publication provided the investigator who proposes to use the data has approval from an Institutional Review Board (IRB), Independent Ethics Committee (IEC), or Research Ethics Board (REB), as applicable, and executes a data use/sharing agreement with Mayo Clinic and the first author/corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
