Abstract
Introduction:
Chronic kidney disease (CKD) is a major health problem in Thailand and health behaviors are central to its risk and progression. Because of the shortage of healthcare personnel, village health volunteers (VHVs) have been collaborating in the primary health care system. However, the contribution of VHVs to CKD reduction has not been evaluated yet. This study aimed to evaluate the efficacy of the VHV-integrated model in preventing and slowing down CKD and its risk factors.
Methods:
The population-based cohort study was conducted in a rural community of Thailand between 2017 and 2019. Baseline clinical and behavioral characteristics including CKD, diabetes, hypertension, and other high-risk factors of the participants were collected. The integrated care model was initiated by the multidisciplinary care team that facilitated, empowered, and trained VHVs targeting risk factors of CKD, health literacy, and health promotion. Then the participants were educated and trained for lifestyle modification and were monitored continuously for 18 months by VHVs. Changes in the CKD risk factors, and kidney functions before and after the application of integrated care model were compared.
Results:
A total of 831 subjects participated in the study with an average age of 57.5 years, and 69.5% were female. Among them, 222 participants (26.7%) were diagnosed as having CKD, the vast majority (95%) of which were in the early stages (G1-G3 and A1-A2). CKD risk factors such as high salt intake, smoking, alcohol consumption, self-NSAID (non-steroidal anti-inflammatory drugs) use were significantly decreased after application of the care model. Also, hemoglobin A1c was significantly reduced in diabetic patients, and blood pressure was controlled better than before in the hypertensive patients. Most importantly, a decline of estimated glomerular filtration rate of the CKD group was improved and lower than the non-CKD group.
Conclusion:
The integrated care model through VHV significantly attenuated the risk factors associated with CKD in the general and high-risk population and effectively slowed down the progression of CKD.
Keywords
Introduction
Chronic kidney disease (CKD) is a major health problem in Thailand and needed a taskforce to reduce the burden.1 -3 The disease is associated with several risk factors such as diabetes mellitus (DM), hypertension (HT), aging, family history of CKD, recurrent urinary tract infections, glomerulonephritis, renal stones, gout, regular intake of non-steroidal anti-inflammatory drugs (NSAIDs) or other nephrotoxic substances,4,5 and, among all, health behaviors are central to the risk and progression of CKD. Health behavior is any activity taken by individuals that affects their health. As an integrated care, behavioral health can be targeted in combination with other cares.6 -8 This is inevitable in the rural area of northeast Thailand, where people have CKD risk behaviors such as high salt intake, drinking inadequate amounts of water, smoking, and regular use of painkillers.2,3,9,10 The proactive screening revealed that the majority of CKD patients in the rural areas were in the early stages (stages 1-3) in which their disease progression can be slowed down by appropriate control measures. 3 Therefore, an integrated care model targeting good health behavior at the community level is a must to slow the disease progression and curb the current CKD burden in Thailand. 9
In Thailand, as well as the other underdeveloped or developing countries, the shortage of healthcare workforce such as community nurses have been partially solved by the employment of village health volunteers (VHVs). They are selected and trained according to the curriculum of the Ministry of Public Health to act as the providers of basic health care, educators for members living in their communities, and as the health reporters to subdistrict health care officers. After training, each VHV takes care of about 10 households.11,12 Because VHVs are also the members of the same community, thus as the insiders, they can catch up health problems and other factors related to public health diseases which previous studies reported the benefit of VHVs care in some diseases but scarce information for CKD. We postulated that the empowerment of VHVs by a specialized multidisciplinary team would be effective for CKD prevention and control.
Earlier, various care models to prevent or slow down the progression of CKD were designed mainly depending on the specialists such as nurse case managers in hospitals in conjunction with the multidisciplinary team.13,14 Herein, we designed an integrated care model with key features expanding and matching the Thai context, primarily be operated by VHVs together with the subdistrict health care providers and the multidisciplinary care team at the community level. Then, we aimed to investigate the efficacy of the VHV-integrated model in preventing and slowing down CKD and its risk factors are evaluated after 18 months of practice. The results could be beneficial to improve disease burden as well as reduce the country’s overall health expenses.
Methods
Study Design and Participants
This population-based intervention study was conducted in a rural community of Khon Kaen Province, Thailand, between 2017 and 2019. The study was a part of and continuation of CKD screening in the population under the Chronic Kidney Disease in the Northeast Thailand (CKDNET) project. 9 The participants in 8 villages of the Don Chang Sub-district were enrolled based on their willingness to participate, aged ≥ 18 years, and without psychopathic disorders. Of 1012 participants in the CKD screening project, 831 subjects were enrolled in this study.
Procedures
After CKD screening process, all participants received their individual health reports and counselling from the multidisciplinary team. Then the procedures were conducted in a 2-year study period, that is, 6-month preintervention and 18-month intervention phases. The preintervention phase included a situation analysis to identify causes and risk factors of CKD in this population and an establishment of the appropriate and feasible model that was well-accepted by the community population. The intervention phase was applying of VHV-integrated model which continued for 18 months. A flow diagram of the study design is illustrated in Figure 1.
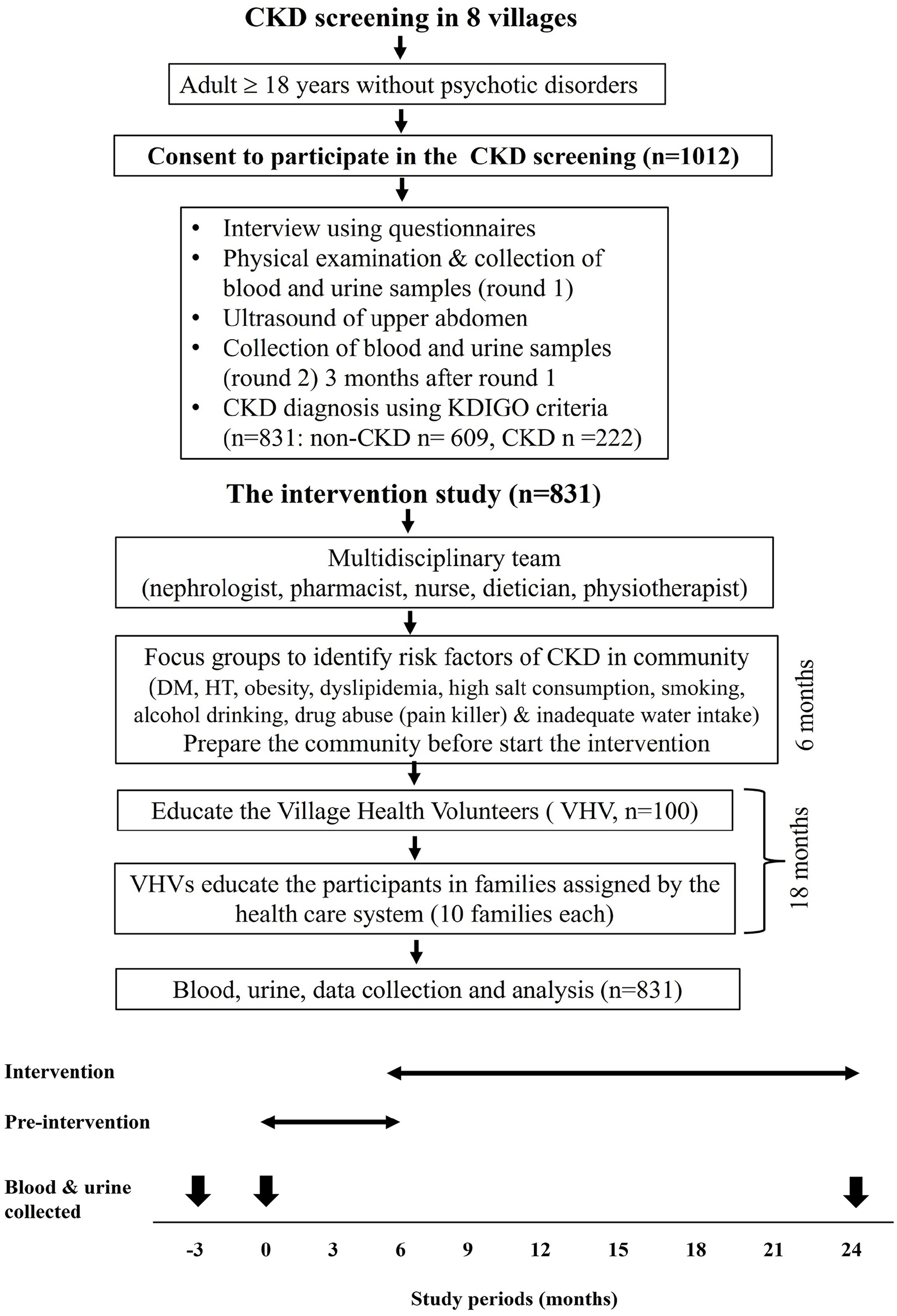
A flow diagram of study design.
Identification of CKD-Risk Factors and Establishment of the VHV-Integrated Model
A survey of the focused groups was conducted to identify the causes of CKD and related conditions in the community. This supervision model with in-depth interview and observation was applied for 3 months when data were collected from a total of 62 primary information providers consisting of CKD patients and their relatives or care givers, VHVs, community leaders, and subdistrict health personnel. Moreover, information was also acquired by interviewing the owners of village pharmacies, grocery stores, and food shops in the community to assess the risk factors involving non-prescribed drugs and diet. The homes of the CKD patients were also visited to assess their living environment. The results of the survey revealed behaviors at risks for CKD were NSAID intake (self-prescribed), high salt and seasonings intake, the use of unstandardized herbal products, herbicide and pesticide uses, smoking, and alcohol intake as the common risk factors related to CKD in the village. Based on these findings, after 3 months of preparation, the integrated care model was developed by the collaboration and social agreement of the rural community and the multidisciplinary research team of the tertiary hospital, Srinagalind Hospital, Khon Kaen University. The key elements of this model were the empowerment of the target groups through community participation, public health literacy, and lifestyle modification (Figure 2).

The components of the integrated care model. The model comprised a multidisciplinary team designed to empower the target population through village health volunteers, focusing on participation, public health literacy, and lifestyle modification.
Action
After the process of 6-month pre-intervention, the VHV-integrated model had continued for 18 months when a total of 100 VHVs attempted to bring the changes, cognition, and self-care attitude effectively in the participants. In-depth details, the multidisciplinary team including nephrologists, primary physicians, nurses, nutritionists, pharmacists, and physical therapists acted as the facilitators and mentors to empower, train, coach, and educate the VHVs for 4 times covering the basic knowledges of multifaceted subjects especially risk factors and early signs and symptoms of CKD, and health promotion. The VHVs conducted a series of activities in the community by incorporation with routine works under supervision of the subdistrict health officials every 2 to 4 weeks guided by the multidisciplinary team to promote and adjust the health behaviors of the participants for prevention and slowing down CKD, DM, and HT. Posters, brochures, flip sheets, spot announcement, and manuals containing detailed information on the diseases, appropriate diet including low salt intake, harmful effects of painkillers, illegal herbs, herbicide, pesticide, smoking, and alcohol drinking, were distributed. Also, basic exercise steps to get rid of muscle pains were demonstrated to the villagers to reduce/avoid NSAID intake. The VHVs were provided with salinity meters, sphygmomanometers, and weight scales to monitor continuously the salt intake, blood pressure, and weight gain of the inhabitants of the households under their responsibility. When abnormal parameters were detected, the VHVs consulted the multidisciplinary team and/or advised the participants to seek primary care personnel for further management. With this model, the participants were in continuous touch with the multidisciplinary team and subdistrict health care officers through the VHVs for any health concerns or problems encountered while under interventions (Supplemental Figure 1).
Data and Sample Collection
Data from the participants were collected before and after implementation of the integrated care model. Socio-epidemiological data were collected from the participants by interviewing based on the pre-designed questionnaire. Body weight (BW) and height were recorded, and the systolic (SBP) and diastolic (DBP) blood pressures were measured using automated digital sphygmomanometers (Terumo Co., Tokyo, Japan). Overnight fasting blood and urine samples of the participants were collected twice at the 3 months interval to screen for CKD at the beginning of the study (−3 and 0 months of the study period), and the third time sampling was done at the end of the 18-month-intervention period as shown in Figure 1. The samples were analyzed for serum creatinine, blood glucose, hemoglobin A1c (HbA1C), urinalysis and urine albumin-creatinine ratio (UACR) using standard laboratory methods and machines as previously described. 3
Definitions
CKD was defined according to the guideline of the Kidney Disease Improving Global Outcomes (KDIGO), indicating the presence of either kidney damage (UACR ≥ 30 mg/g, urine red blood cells (RBC) ≥3 to 5 cells/high power field (hpf), structural abnormalities discovered by renal ultrasonography examination) and/or reduction of estimated glomerular filtration rate (eGFR) <60 ml/min/1.73 m2 for over 3 months.15,3 CKD staging was defined as GFR categories (G1-G5) and albuminuria categories (A1-A3). 16 HT was considered when the participant has persistent SBP ≥ 140 mm Hg or DBP ≥ 90 mm Hg, or were diagnosed by physicians followed by medication. DM was considered either the participant was diagnosed by physicians and being on medication, or fasting plasma glucose (FPG) level of ≥126 mg/dl or HbA1C ≥ 6.5% twice.
Outcomes
Post-intervention changes of clinical parameters, that is, serum creatinine, eGFR, UACR, FPG, HbA1C, SBP, DBP, BW, and the behavior risks for CKD in the study population and in the subgroups of CKD and high-risk groups for CKD were determined.
Statistical Analysis
Data were presented as frequency, mean ± standard deviation (SD), median (interquartile range; IQR), or percentage. Comparisons of outcomes before and after the intervention within the same group were analyzed by the McNemar test for categorical variables and the Student’s paired t-test or Wilcoxon Signed Ranks test for continuous variables, as applicable. Independence of 2 or more groups were compared by Chi-square test or Fisher’s Exact test for categorical variables and Student’s t-test or Mann-Whitney U-test or Analysis of variance (ANOVA) for continuous variables, as appropriate. Statistical analyses were performed using STATA version 17.0 and the predictive value of ≤.05 was considered a statistical significance.
Results
Baseline Characteristics of the Participants
Overall, a total of 1012 subjects from 8 villages participated in the CKD screening of the CKDNET project, of which 831 cases were included in this care model, accounting for an 82.1% response rate. Their characteristics are given in Table 1. The average age of the participants was 57.5 years and 69.5% were females. Among the participants, farming was the major occupation, and the majority (75%) of them had a primary-level education. In term of an economic situation, 86.9% of the participants had a monthly income of <10 000 Baht (<$282 a month or <$3386 a year). HT (32.7%) and DM (17.8%) were the major underlying CKD-related conditions.
Baseline Characteristics of the Participants.

Abbreviations: CVD, cardiovascular disease; DM, diabetes mellitus; HT, hypertension; SD, standard deviation; SLE, systemic lupus erythematosus.
Prevalence of Chronic Kidney Disease Before the Intervention
Among 831 participants, 222 (26.7%) were defined as CKD according to the KDIGO criteria with 1 criterion (163 cases, 19.6%), 2 criteria (39 cases, 4.69%), 3 criteria (18 cases, 2.17%), and 4 criteria (2 cases, 0.24%). Proportions of CKD defined as eGFR <60 ml/min/1.73 m2 were 35.1%, albuminuria 45.9%, hematuria 9.0%, and abnormal renal ultrasound 49.1%. Comparison of the baseline characteristics between CKD and non-CKD participants is demonstrated in the Supplemental Table 1, in which CKD participants were older, more proportions of male and unemployed status, lesser education level, lower renal function, and higher percentages of anemia and hyperuricemia. The top 3 causes of CKD were DM (69 cases, 31.1%), HT (a total of 125 cases, 57 cases (56.3%) of which were diagnosed with DM), and renal stones (35 cases, 15.8%).
Staging distribution of CKD categorized by the levels of eGFR and albuminuria is demonstrated in Table 2. The proportion of the participants with CKD at the early stage (G1-G3) was 94.7% and that of A1 and A2 was 91.4%.
Staging of CKD Categorized by Levels of eGFR and Albuminuria.
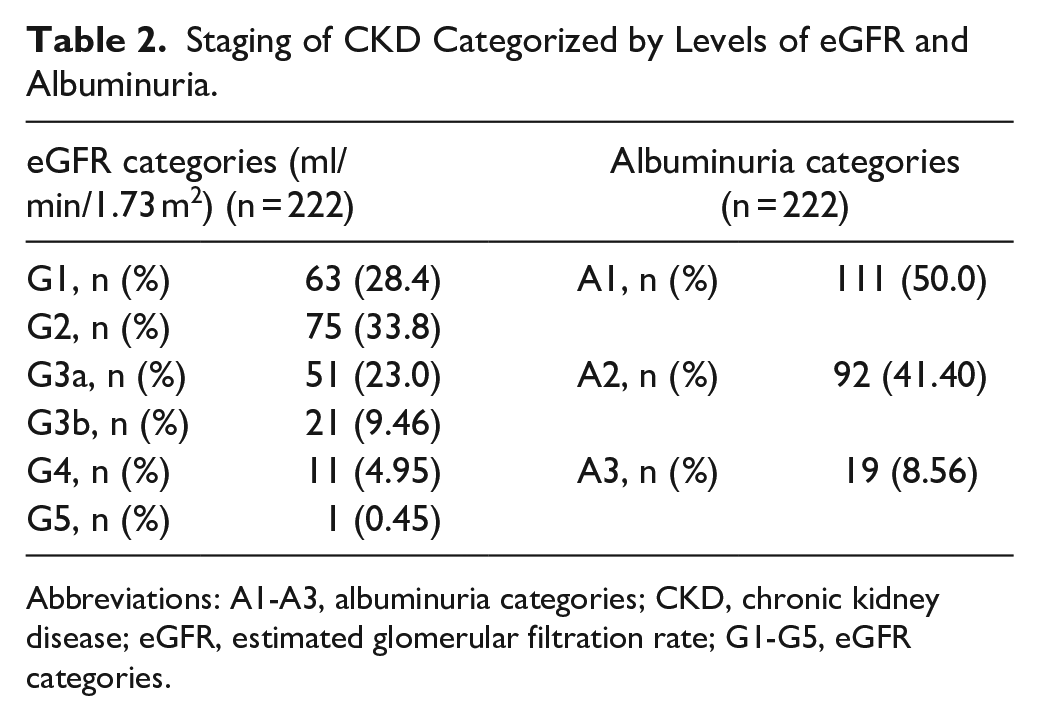
Abbreviations: A1-A3, albuminuria categories; CKD, chronic kidney disease; eGFR, estimated glomerular filtration rate; G1-G5, eGFR categories.
Awareness of CKD and Related Diseases
Although CKD and the related diseases were highly prevalent in this rural study area, awareness of these diseases was quite low. Only 8 of 222 CKD cases (3.6%) had perceived their renal dysfunction and structural abnormality. In other words, 96.4% of them were unaware of their CKD. High percentages of the participants unrecognized their HT (26.8%), DM (21.6%), and renal stones (59.1%).
Risk Factor Changes After the Adoption of VHV-Integrated Care Model
There were significant changes between pre-and post-intervention values in the risk factors related to CKD such as smoking, alcohol consumption, self NSAIDs intake, and herb use (Table 3). The amount of daily intake of salt and seasonings was significantly decreased whereas daily water intake was increased after the intervention. Both over-BW and BMI were significantly improved after the intervention. HbA1C level of DM patients was significantly reduced and, in the same way, SBP of HT patients was controlled well at the end of the study in both CKD and non-CKD groups. DBP was also significantly decreased after the intervention in overall and HT patients, especially in the non-CKD group. Although statistically not significant, a tendency of reduction of DBP was observed in the HT patients in the CKD group.
Changes in Risk Factors and Behaviors Related to CKD After the Intervention.
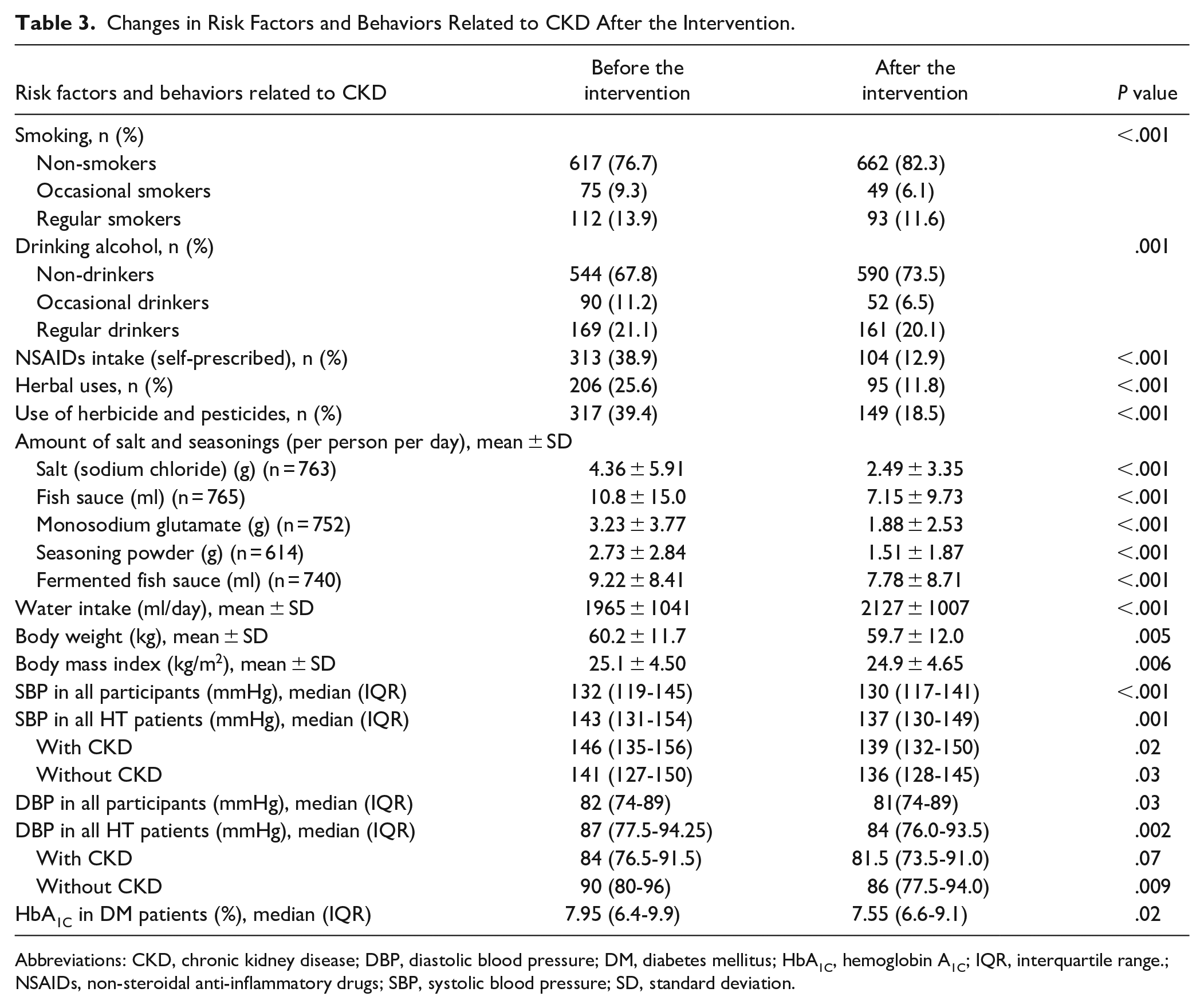
Abbreviations: CKD, chronic kidney disease; DBP, diastolic blood pressure; DM, diabetes mellitus; HbA1C, hemoglobin A1C; IQR, interquartile range.; NSAIDs, non-steroidal anti-inflammatory drugs; SBP, systolic blood pressure; SD, standard deviation.
During the 3-month period of CKD screening before intervention, the CKD group showed more mean eGFR decline compared with the non-CKD group (−0.70 ml/min/1.73 m2 vs −0.38 ml/min/1.73 m2). If the eGFR reduction rate were postulated to be stable, the next 24-month mean eGFR changes would be −5.61 ml/min/1.73 m2 in the CKD groups and −3.02 ml/min/1.73 m2 in the non-CKD group (Supplemental Figure 2).
After the 24-month study period, eGFR levels were significantly decreased whereas serum creatinine and UACR were significantly increased in both CKD and non-CKD groups. The magnitude of eGFR deterioration, however, was significantly lower in the CKD group (mean difference and median difference of GFR −2.42 and −2.04 ml/min/1.73 m2 in the CKD group vs −3.44 and −3.00 ml/min/1.73 m2 in the non-CKD group, P = .04) as shown in Table 4. In additional analysis, 87 (14.3%) participants of the 609 initial non-CKD group newly developed CKD during the intervention period. Supplemental Table 2 shows the comparison of the characteristics among 3 groups, that is, non-CKD (n = 522), newly developed CKD (n = 87) and established CKD (n = 222) groups. Apparently, baseline proportions of main CKD risk factors (such as aging, HT, DM, renal stone), anemia, hyperuricemia, and levels of renal function in the newly developed CKD group were lesser than the established CKD group but were greater than the non-CKD group. Furthermore, the highest GFR decline during the study period was observed in this newly developed CKD group (−4.92 ml/min/1.73 m2), while the established CKD and non-CKD groups had a similar lower rate of GFR decline (Supplemental Table 3). However, if compared with the rate of GFR decline before the intervention, both newly developed CKD and established CKD groups had lesser slopes of GFR changes as shown in Supplemental Figures 2 and 3.
Comparison of GFR and Albuminuria Levels Between Pre-and Post-Intervention (24 Months Apart) in CKD and Non-CKD groups.

Abbreviations: ACR, albumin creatinine ratio; CKD, chronic kidney disease; IQR, interquartile range; SD, standard deviation.
The subgroup analysis of the CKD group regarding etiologies of CKD revealed a significantly lesser reduction of GFR in the HT subgroup [−0.9 (−4.92 to 3.35) ml/min/1.73 m2] versus the non-HT subgroup [−3.20 (−7.7 to 1.80) ml/min/1.73 m2, P = .04].
Likewise, a higher increase of UACR was observed in both established and newly developed CKD groups (Supplemental Table 3). A doubling of UACR, one of the indicators of CKD progression,17,18 was observed in 18.8%, 43.7%, and 34.7%, respectively, of the non-CKD, newly developed CKD, and established CKD groups. Significant albuminuria progression was associated with older, more NSAIDs use, lesser levels eGFR and hemoglobin level, and higher blood pressure and serum levels of creatinine, uric acid, and HbA1C.
Supplemental Table 4 is demonstrated the significant improvement of participants’ perspective regarding points of view in self-care, and confidence and satisfaction in VHVs works and in shopkeepers who sold the over-counter drugs.
Discussion
In Thailand, the National Health Examination surveys (NHES) were periodically performed and the survey in Thai population aged ≥20 years showed the rising prevalences of DM 7.7 % to 9.9% and hypertension 22.0% to 28.5% from 2004 to 2014.10,19 The higher prevalences of DM and CKD were noted in Bangkok and northeastern regions which predominated in rural areas.2,10,20 Furthermore, the proportion of uncontrolled glycemia was also highlighted in this region, 21 therefore, strategies to improve primary care in rural areas are needed.
In this study, an integrated care model was developed to prevent and slow down progression of CKD and related conditions or diseases in the rural Thai community. This model achieved synergistic empowerment of VHVs and community people with better health knowledge, understanding, and practice, as reflected by optimal outcomes. The potential risk factors related to CKD such as smoking, alcohol consumption, self NSAIDs use, utilizations of herb, pesticide and herbicide, daily salt, and other seasonings intake among the participants were lowered after implementation of the integrated care model. The HbA1C levels (in DM patients), BMI, and blood pressure were also reduced at the end of the study for 18 months. Daily water intake was increased after intervention. Therefore, our integrated care model was effective in reducing the risk factors related to CKD and related non-communicable diseases in the rural community.
Any community-based kidney disease intervention programs serve as a resource for people living with the disease. Our model was distinct from other intervention programs because of the central role of VHVs who are guided consistently by the multidisciplinary team of general physicians, nephrologists, nurses, pharmacists, dieticians, and physiotherapists. It should be highlighted that VHVs, and sub-district health center personnel have a better understanding of local community dynamics and can effectively contribute to the prevention and control of diseases by linkage between villagers and the Thai healthcare system at the sub-district or district level. VHVs and local health personnel have contributed to the primary healthcare screening and control for several diseases, for example, tuberculosis, dengue infection, parasitic diseases, cancer screening and prevention, elderly care, and obesity management.22 -28 Even recently, their contribution was clearly shown in the fight against COVID-19. 29 Nevertheless, reports of improved control of CKD, DM, and HT by VHVs-central care were limited. The present results clearly showed the importance of collaboration of trained VHVs and local health personnel with the experts as a multidisciplinary team for controlling CKD at the community level. Furthermore, this VHV-integrated model may be a solution to correct some reported barriers to CKD care, such as deficient CKD awareness, shortages of skilled healthcare professionals, and requirement for potential facilitators for community coordination. 30
Although some interventional studies on CKD patients in different settings have reported an improvement of some risk factors, others have found no effects.31 -33 This might be due to differences in the characteristics of interventions such as the mode of delivery, intensity, content, and educator. In this study, our integrated care model decreased the CKD risk factors such as uncontrolled HT, DM and obesity, salt intake, smoking, and use of potential nephrotoxins. This might enhance health literacy and empower the participants via the direct home-based continuous guidance, education, and surveillance as well as monitoring of the risk factors by VHVs who live within the community. Supporting the model setting of this study, patient education at the primary care level by a model of care delivery has been recommended in an earlier report. 34
Previous studies revealed an inconsistent effect of lifestyle interventions on kidney function, including no, negative, or positive effects.35,36 In our study, eGFR was significantly decreased after intervention in both CKD and non-CKD groups. Partly, this could be a physiological eGFR decrease due to aging as about 50% of the participants in this study were >60 years. During the 3-month period of pre-intervention, the CKD group had a nearly 2-fold decrease of eGFR compared with the non-CKD group (−0.70 ml/min/1.73 m2 in the CKD group vs −0.38 ml/min/1.73 m2 in the non-CKD group). However, after the 24-month study period, the degree of eGFR reduction was lower in the CKD group (−2.04 ml/min/1.73 m2 in the CKD group vs −3.00 ml/min/1.73 m2 in the non-CKD group, P = .04). Similarly, in primary hospital setups under multidisciplinary care including lifestyle modification, CKD stage III and IV patients were stated to have a lower annual decline in eGFR.37 -39 A systemic review also supported the usefulness of appropriate educational interventions for CKD patients which may improve knowledge, self-management, and patient outcomes. 40 In this study, 14.3% of the initial non-CKD cases who had baseline renal function almost reached the CKD criteria developed definite-CKD later during the 18-month intervention and had worst GFR deterioration compared with the initial CKD participants. This finding suggests that aggressive intervention should also be performed in those who have high risks and in the pre-CKD stage. Furthermore, behavioral change and life-style modification were more effective if the participants had been already aware of their illness. In addition to slow CKD progression, an application of the model in long term on a wider population or even in different geographical locations will be interesting because this integrated care model was also effective to control DM and HT which are the major risk factors of CKD.
The multidisciplinary interventions in our care model were unable to stop UACR increase in both established-CKD and new-CKD groups which suggests that more extensive reduction of risk factors and stringent control to reach targeted blood pressure (<130/80 mmHg) 41 and glucose levels (HbA1C ≤ 7% in early stages of CKD) 42 are mandatory for further attenuation of albuminuria. In this regard, a decrease in UACR after nurse-based lifestyle intervention has been reported in patients with a high risk of CKD in a 12-month follow-up study. 43
The authors are aware of the limitations of this study. First, the nature of the study led us to compare the outcomes before and after multidisciplinary interventions without the use of a control group. Second, the majority of the participants were in an early stage of CKD, which otherwise might have caused differences in the findings. In general, the individuals at the earlier stage of CKD may give a lower priority to education than those with the advanced stages, therefore, gaining more attention may be the reason supporting the positive results.
The important issue is the sustainability of this intervention model. Since the key success of the model is a linkage among multidisciplinary team, VHVs, and participants, the telehealth platform for continuous education and the application for VHVs to follow participants were developed to sustain the model, especially during the contagious occurrence such as the pandemic infection of the COVID-19 in the past few years. Additionally, with the participation and empowerment concepts made for the local health personnel and VHVs, they realize their important roles and what they should continue for the better health of community, therefore, enhancing the sustainability of this model.
Conclusions
The integrated CKD care model was developed where village health volunteers play the central role for slowing down and prevention of CKD at the community level. The proposed model could attenuate the risk factors associated with CKD in the general and high-risk population and effectively slow down CKD progression. Further studies with the control group will strengthen this model and increase the level of impact.
Supplemental Material
sj-pdf-1-jpc-10.1177_21501319241240355 – Supplemental material for Integrated Care Model by the Village Health Volunteers to Prevent and Slow down Progression of Chronic Kidney Disease in a Rural Community, Thailand
Supplemental material, sj-pdf-1-jpc-10.1177_21501319241240355 for Integrated Care Model by the Village Health Volunteers to Prevent and Slow down Progression of Chronic Kidney Disease in a Rural Community, Thailand by Ampornpan Theeranut, Nonglak Methakanjanasak, Sunee Lertsinudom, Pattama Surit, Nichanun Panyaek, Saisamon Leeladapattarakul, Peangtikumporn Nilpetch, Pattapong Kessomboon, Chalongchai Chalermwat, Watcharapong Rintara, Wudipong Khongtong, Pawich Paktipat, Pannee Banchonhattakit, Darunee Chunlertrith, Amod Sharma, Ubon Cha’on and Sirirat Anutrakulchai in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
The authors would like to thank Professor Yukifumi Nawa for editing the manuscript via Publication Clinic KKU, Thailand. We would like to thank the provincial governor, provincial health officers, Don Chang local authorities, and participants for their active participation in the project.
List of Abbreviations
CKD, Chronic Kidney Disease; GFR, Glomerular Filtration Rate; UACR, Urine Albumin Creatinine Ratio; NSAIDs, Non-steroidal Anti-inflammatory Drugs; DM, Diabetes Mellitus; HT, Hypertension; CVD, Cardiovascular Disease; SLE, Systemic Lupus Erythematosus; KDIGO, Kidney Disease Improving Global Outcomes; CKDNET, Chronic Kidney Disease Prevention in the Northeast of Thailand; DCC model, Don Chang Care model
Author Contributions
AT, NM, PS, SA, SuL, and UC; conceptualization, project administration, and supervision. AT, NM, PS, SA, NP, SaL, PN, PaK, CC, WR, PhK, PP, PB, DC, and SuL; investigation, methodology, and data curation. SA and UC; funding acquisition, resource, formal analysis, and validation. NM, SA, AS, and UC writing the original draft. All authors were involved in refining study protocols, interpretation of data, critically revising the paper, and approving the final version.
Availability of Data and Materials
The data used and analyzed in the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Khon Kaen University through the Chronic Kidney Disease Northeast Thailand research project (CKDNET 2560, 2561, 2562). The funder has no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Ethical Approval and Consent to Participate
Ethical approval and consent to participate Ethical approval to conduct the study was obtained from the Ethics Committee for Human Research, Khon Kaen University (HE601035, HE622068). All methods were carried out in accordance with the relevant guidelines and regulations laid down in the Declaration of Helsinki. All participants provided informed consent at the start of the baseline survey.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
