Abstract
Previous reviews of strategies to increase cervical cancer screening are more than 10 years old, the U.S. continues to fall short of the Healthy People 2030 cervical cancer screening goal, and guidelines were revised in 2018, therefore an updated review of the existing literature is needed. We conducted a scoping review using electronic databases PubMed, Scopus, and Ovid Medline that included publication dates between 2012 and 2021 to answer the question, “Which strategies implemented in U.S. primary care settings have been most successful in increasing rates of cervical cancer screening since the 2012 US Preventative Services Task Force cervical cancer screening guidelines were published?” We mapped findings to pre-specified implementation strategy categories. After initially identifying 399 articles, we excluded 350 due to duplicates or not meeting review criteria, leaving 49 articles for full review. We excluded 37 of these during full-text review and identified 2 additional articles from the manual search of reference lists for a total of 14 studies for abstraction. Eleven articles reported on strategies resulting in increased cervical cancer screening, and 3 did not. Clinic workflow re-design strategies showed the greatest promise in improving cervical cancer screening rates, education strategies for patients had mixed results, and quality management strategies were not effective. These findings suggest clinical workflow re-structures and patient education strategies can increase cervical cancer screening in primary care settings. Results are particularly important in settings that care for underserved populations, as these settings may need additional implementation strategies to decrease cervical cancer screening disparities.
Keywords
Background
In 2023, 13 960 new cases of invasive cancer will be diagnosed in the United States (U.S.), and 4310 women will die from this disease. 1 Since the development of the Papanicolaou test (Pap test) in the 1950s, cervical cancer deaths have significantly decreased. Indeed, cervical cancer screening, which now includes both the Pap and the human papilloma virus (HPV) screening tests, is effective in detecting cancer early and identifying high risk groups and is an evidence-based intervention. This allows for targeted surveillance, early treatment and reductions in mortality.2,3 Approximately 80% of all eligible adults are screened for cervical cancer, 4 yet rates differ according to socioeconomic attributes. For example, screening rates are lower for racial and ethnic minorities, those with lower income, and individuals who experience challenges accessing healthcare compared to those who are non-Hispanic white, of high-income, and have regular healthcare access. Because of these inequities, disadvantaged groups are more likely to be diagnosed with late-stage invasive cervical cancer, leading to increased mortality.5,6 The overall rate of attributable deaths has remained the same over the last 20 years4,7 indicating barriers to screening persist for disadvantaged groups.
Several barriers to receipt of guideline-concordant care have been identified, 8 including patient-level barriers due to lack of knowledge, fear of screening, problems with transportation or time-off,9,10 and lack of physician recommendations. 11 System-level barriers also exist, including lack of funding for follow up care, healthcare personnel shortages, limited patient outreach, lack of equipment, and inconsistent compensation for screening. 12
Implementation strategies, which are operations that support adoption of evidence based interventions, 13 have been found to improve recommended cancer screenings,14,15 including invitation letters, reminder calls, and educational materials.16,17 Additionally, several evidence-based cancer screening programs aimed at facilitating access to underserved populations currently exist. 18 The United States Preventive Services Task Force (USPSTF) Community Guide previously found supportive evidence for strategies such as one-on-one education and client reminders, and sufficient evidence for provider assessment and feedback.14,19 However, these reviews are now more than 10 years old. The U.S. continues to fall short of the Healthy People 2030 cervical cancer screening goal, 20 and new cervical cancer screening guidelines were published in 2018. 21 In addition, primary care clinics serving disadvantaged populations have not improved cervical cancer screening rates over the last 10 years.22,23 Thus, updating evidence on new or existing implementation strategies that could improve screening rates is needed and could reduce disparities.
We conducted a scoping review to identify new evidence for implementation strategies evaluated in primary care settings to improve cervical cancer screening rates. Results from an updated review will inform implementation strategies that can be used in the primary care setting to increase cervical cancer screening rates for both HPV and Pap testing and potentially reduce disparities.
Methods
We followed Arksey and O’Malley’s 24 5 step scoping review methodology to guide this work.
Step 1: Research Question
We chose the research question, “Which strategies implemented in U.S. primary care settings have been most successful in increasing rates of cervical cancer screening since publication of the 2012 US Preventative Services Task Force cervical cancer screening guidelines?”
Step 2: Literature Search Approach, Including Inclusion, and Exclusion Criteria
We searched electronic databases PubMed, Scopus, and Ovid Medline in December 2021 for the time period 1/1/2012 to 12/30/21 and conducted manual searches of reference collections and systematic reviews. We consulted a health science librarian and used a combination of the following keywords that included MeSH terms: “Early Detection of Cancer” AND “United States” AND “Pap test” AND “Interventions.” Additional search terms such as or “human papillomavirus” (“HPV”), “HPV testing,” “adherence,” “health knowledge,” “attitudes,” and “practice patterns” did not yield additional studies. Studies that included either Pap or HPV testing or both were included. The searches were limited to studies published in English and those conducted in the U.S. because healthcare delivery differs outside the U.S. We removed duplicates and those that did not meet the inclusion criteria of publication date, setting, or U.S. based on the title and abstract. Survivorship studies were also removed. Additionally, we manually searched studies included in reviews and the reference lists of relevant papers and PubMed related articles search feature.
We included articles if they were original research, had publication dates in 2012 to 2021, were conducted in a primary care clinic setting, involved subjects aged 18 to 65 years, included an implementation strategy, and had cervical cancer screening/detection as its primary outcome. We excluded meta-analysis/systematic reviews, gray literature (information produced outside of traditional publishing/distribution channels, such as reports, policy literature, newsletters, government documents, speeches, white papers, urban plans).
Step 3: Abstract and Full Paper Review
Our initial search strategy using the publication dates we selected (2012-2021) identified 399 articles. We discarded 142 duplicate articles and 7 articles for studies not conducted in the U.S. Four members of the research team independently reviewed article titles and abstracts based on inclusion criteria and relevance to interventions increasing cervical cancer screening. Of 250 articles, 201 were excluded after abstract review, leaving 49 for full-text review (Figure 1). When it was unclear whether an article should be included, the research team discussed the article(s) as a group and came to consensus on inclusion versus exclusion.

Consort diagram of scoping review article identification, inclusions, and exclusions.
After title and abstract review, five members of the research team independently reviewed the full-text of the remaining 49 articles to finalize decisions regarding abstraction. Each paper was discussed by the entire research team, which resulted in identifying 12 articles that met inclusion criteria for abstraction. The remaining 37 articles were excluded because they were: (1) systematic, meta-analysis, or prior scoping reviews (n = 7); (2) not conducted in a primary care setting (n = 28); (3) not implementation studies (n = 1); and (4) not studies where the primary outcome was cervical cancer screening (n = 1). The manual search of reference lists identified 2 additional articles that were also included for full abstraction for a total of 14 articles.
Step 4: Data Abstraction
We created a data abstraction form that included: authors, publication year, study design, approach (eg, randomization type, study groups, setting and location, study period, inclusion of disadvantaged population (if applicable), intervention, duration of intervention, number of subjects, and primary outcome (did cancer screening rates increase?), intervention effect size (how much did rates increase?), and limitations. We followed the National Heart, Lung, and Blood Institute’s (NHLBI) 25 Study Quality Assessment Tools to measure the study’s internal validity, assigning each study a rating of good, fair, or poor quality to assess studies for flaws and limitations, such as insufficient power, selection bias, or inadequate adjustment for confounding.
Step 5: Collating, Summarizing, and Reporting the Results
The research team discussed the data extracted from the included articles. During this discussion, we identified strategies implemented, what worked and what did not work, and study limitations. We also mapped the strategies to the Expert Recommendations for Implementing Change (ERIC) project 6 implementation strategy categories that represent 68 implementation strategies, which include plan, educate, restructure, finance, quality management, and attend to policy contexts. 26 Studies focusing on underserved populations were indicated in the tables to highlight strategies implemented in settings that serve hard to reach or underserved populations.
Plan strategies help develop the capacity to implement a strategy. Educate strategies can be at the organizational, staff or patient level and help relay information about the intervention. Restructure strategies focus on changes to staffing, workflows, change physical structures, and utilization of electronic health systems. Finance strategies can offer incentives to patients or clinicians and can include funding mechanisms. Quality management strategies evaluate and change systems to improve care and workflows. 26
Results
Of the 14 fully abstracted articles, several research designs were used, including 10 randomized controlled trials and 3 observational pre-post studies (Table 1). One study was a quality improvement initiative that used four rapid plan-do-study-act cycles. Of the 14 studies, 9 were of good quality, and 5 were fair quality. 25 Five studies were conducted in Federally Qualified Health Centers (FQHC), and three were done in self-described safety-net clinics. Each study was conducted in a single-state, all of which were within the Continental U.S. Eleven implemented strategies that resulted in increased cervical cancer screening,27 -37 whereas three found no effect.38 -40 Three studies focused exclusively on Latinx patients,27,28,38 one on Latinx and Haitian women, 36 and a fifth focused on an Appalachian Ohio population. 35 The remaining studies included general primary care patient populations.
Description of Included Studies, Interventions, and Outcomes Mapped to ERIC Implementation Strategy Categories.
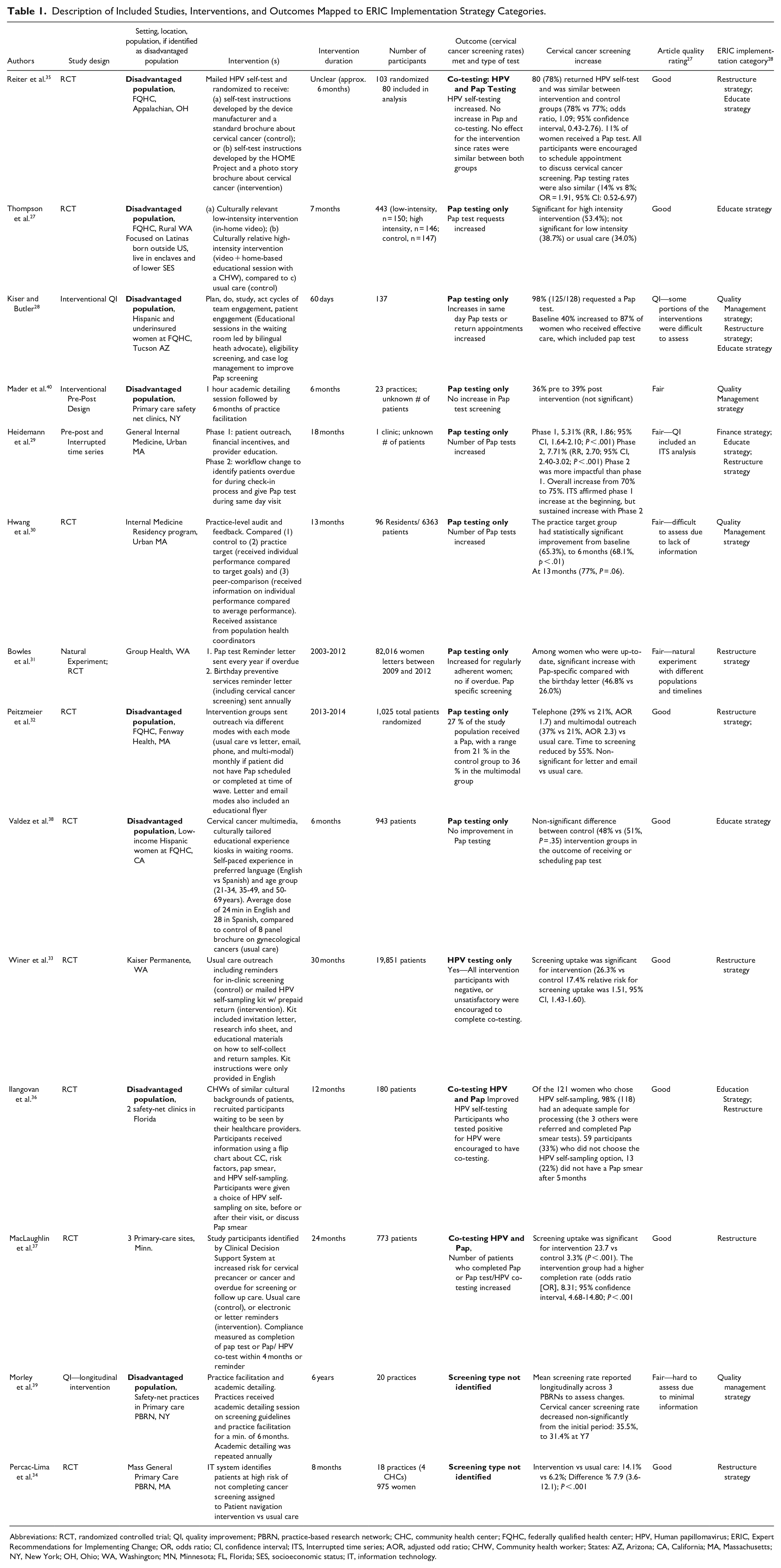
Abbreviations: RCT, randomized controlled trial; QI, quality improvement; PBRN, practice-based research network; CHC, community health center; FQHC, federally qualified health center; HPV, Human papillomavirus; ERIC, Expert Recommendations for Implementing Change; OR, odds ratio; CI, confidence interval; ITS, Interrupted time series; AOR, adjusted odd ratio; CHW, Community health worker; States: AZ, Arizona; CA, California; MA, Massachusetts; NY, New York; OH, Ohio; WA, Washington; MN, Minnesota; FL, Florida; SES, socioeconomic status; IT, information technology.
Using the 6 ERIC categories, 26 we classified 7 of the strategies as education strategies (predominantly educating patients), 4 as quality management strategies, and 9 as re-structure strategies. One study included a finance strategy, and none included plan or policy strategies. The strategies were not mutually exclusive; half of the studies (n = 7) employed multiple strategies, and half (n = 7) employed a single intervention (Table 1).
Education Strategies
Five of the studies focused on patient education strategies with mixed results. These studies ranged from educational kiosks in waiting rooms to a video plus a home-based educational session.27,38 Strategies that had a positive impact on cervical cancer screening rates included a video in Spanish that provided education about cervical cancer screening, including a home-based educational session, 27 utilization of community health workers (CHW) of similar cultural backgrounds in healthcare waiting rooms, 28 and another study that provided education with a CHW in the waiting room, plus HPV self-testing kits. 36 Although these strategies were found to be effective, they were time and staff intensive to implement. Conversely, the multimedia education kiosks experience in the recipient’s preferred language did not increase cervical cancer screening, 38 nor did tailored instructions for mailed HPV self-screening kit (standard vs design by HOME Project). 35 Only one study included clinician education as part of a multiphase and multimodal intervention. 29
Re-structure Strategies
Restructuring strategies varied from workflow changes that transferred responsibilities from clinicians to medical assistants, 29 or incorporating the use HPV self-tests in the office or by mail,33,36 use of information technology to identify and track patients, 37 and mailing reminders to targeted patients overdue or at high risk.31,32 Workflow changes had medical assistants identifying patients in need of screening during appointment check-in, discussing whether or not the patient was willing to be screened, and providing the patient with a gown for pap testing. 29 These workflow changes exhibited sustained improvement of cervical cancer screening rates throughout the 9-month implementation period.
Ilangovan’s et al. 36 study included a multimodal intervention strategy that improved cervical cancer screening rates with Latinx and Haitian women who were offered to complete a HPV self-sampling kits while waiting for their appointment, combined with culturally tailored education from a CHW of similar cultural background inside 2 safety-net clinic waiting rooms. Winer et al. 33 and Reiter et al. 35 both found that providing HPV self-sampling kits with pre-paid returned envelopes led to improved cervical cancer screening rates.
A high intensity, multimodal intervention utilized an information technology system to identify and track patients overdue for screening and provided patient navigation services in the patient’s preferred language assisted them in successfully completing screening. 34 In comparison, studies that provided reminders to patients to complete Pap testing versus usual care were also successful. These included a yearly Pap test-specific reminder letter for patients who are up to date with cervical cancer screening, 31 a letter or an electronic reminder for those identified by a Clinical Decision Support System as overdue or at high-risk for pre-cancer, 37 and a telephone and multi-modal reminders that was performed monthly. 32 While successful, the multi-modal reminders intervention resulted in 27% of participants completing a Pap test during the intervention, 32 23.7% for the patients identified by the Clinical Decision Support System, 37 and 46% for the Pap-specific letter for women who were up to date on other preventive services, 31 indicating more research is needed with hard-to-reach patients.
Quality Management Strategy
Two studies included quality management strategies aimed at clinicians (Morley et al. 39 and Mader et al. 40 ), which comprised of academic detailing (medical staff trained by educator to review guidelines and strategies to improve clinical performance) and practice facilitation (practices assisted by a trained quality improvement professional) to: (1) review/adapt workflows, (2) assess clinical performance, and (3) assess organization policies, all of which occurred over time. These strategies did not yield an increase in cervical cancer screening.
Hwang et al. 30 reported an increase in cervical cancer screening in their 3-arm randomized clinical trial testing an intervention where all internal medicine residents in the study received information on how to contact a population health coordinator for assistance in identifying patients overdue for a service and scheduling follow up visits. Arm 2 residents received additional personalized information on their screening rates compared to target goals at the practice level (Practice Target group), while arm 3 residents received their screening rates in comparison to their peer’s screening rates (Peer Comparison group). The difference between the intervention and control groups was not statistically significant, but the Practice Target group screening rates increased from their baseline of 65.3% to 77.0% at 13 months, while the peer comparison group did not show an increase in screening rates compared to their baseline. Unfortunately, the impact of the population health coordinators was not directly measured.
Discussion
Evidence indicating that cervical cancer screening can reduce disease mortality is indisputable. So why has it been so difficult to increase receipt of these screenings, especially in disadvantaged populations? Findings from this scoping literature review of publications following the 2012 USPSTF updated Cervical Cancer Screening Guidelines through 2018 identified minimal support for education strategies directed at clinicians, and educational strategies aimed at patients had mixed results. Studies that included re-design strategies showed the most promise in improving cervical cancer screening rates showing increases by 40%. 28 Successful strategies included: (1) mailing HPV self-sampling kits to patients; (2) providing self-sampling HPV tests combined along with patient education in waiting rooms; (3) providing strategies for resident physicians to identify and follow up-with patients for screening; (4) having medical assistants, instead of clinicians, discuss Pap testing with patients, and prepare them for the procedure at the beginning of a visit; and (5) patient navigation.
Eight of the 14 published studies were done in federally qualified health centers or safety-net clinics which typically serve under-resourced diverse communities and patients who are uninsured or publicly insured. FQHCs face many challenges such as time limitations for visits, clinician shortages and a lack of continuity of patient care. Five of the studies showed increases in cervical cancer screening with successful interventions that used self-sampling HPV testing or CHWs who provided culturally tailored information and alongside same-day Pap tests, which can provide insights into which types of intervention research may work with clinics serving these under-resourced communities and often complex patients. Likely because reducing travel, time off from work and other existing barriers, and getting everything done at a single visit was an effective strategy to increase screening rates. Successful interventions with disadvantaged populations addressed patient level barriers, such as cost, time, financial constraints, comprehension, and privacy concerns. For example, free same-day Pap tests offered during the check-in process eliminating the need to schedule another appointment, travel time off from work, and expenses. Mailed self-sampling HPV tests with standard instructions and brochure from the HPV testing company compared with modified instructions and a photo story brochure did not increase cervical cancer screening, yet overall 78% of women in the study returned the HPV test with follow up patient surveys indicating the self-sampling HPV test was convenient to use. 35 Ilangovan’s et al. 36 intervention of self-sampled HPV self-tests within the office setting, along with CHW who explained the procedure in the waiting room, also increased cervical cancer screening with patients citing ease of use, privacy, and speed.
While the use of HPV self-testing is a successful intervention, it is not FDA-approved nor guideline approved to date, and may not meet Health Resources and Services Administration (HRSA) requirements. 41 Currently there are 3 testing options endorsed by USPSTF, American Cancer Society and the American College of Obstetricians and Gynecologists; Pap, Pap/HPV co-testing, and Primary HPV. HPV testing assesses risks related to viral exposure and developing the disease, while Pap tests identify cells that have a range of diagnostic possibilities. Co-testing has been identified as beneficial in women 30 to 65 to fully understand patient risk and ongoing monitoring since cervical cells can change. 42 Our review found only 2 studies that examined both screening tests.
Education strategies for patients showed promise in many settings, including at home, community-based, and clinical settings, though some have not been tested across multiple settings. While not all unimodal or population-specific forms of education were successful at improving cervical cancer screening, overall results suggest that a multi-modal approach to educate patients and providing screening options, such as to self-sample a HPV test on site increased screening rates as seen in Ilangovan et al. 36 Two community-based studies outside of primary care settings, which focused on Korean American or Vietnamese patients, illustrated important improvements to screening when the educational interventions where culturally tailored to the population being served, and supplemented by additional support mechanisms.43,44 Percac-Lima’s et al. 34 intervention provided patient navigators who educated patients in their preferred language over the phone, as well as assisted with transportation and other support, which was successful across multiple cancer prevention services in high risk patients. Similarly, a study of Hispanic women that looked at the use of culturally tailored narrative film as an effective means to communicate information about cancer prevention showed significant increases in new knowledge and favorable attitudes toward Pap test expense compared with those who viewed a non-narrative film, though increased receipt of cancer screening was not statistically significant. 45 Some of these strategies could, however, be adapted for use in the primary care setting and retested with more robust study designs.
Finance strategies for patients showed success when combined with other strategies, 29 while policy strategies or plan strategies were not noted in any of the reviewed studies.
Quality management intervention strategies for clinicians (Mader et al. 40 and Morley et al. 39 ), such as academic detailing and practice facilitation were not successful in increasing cervical cancer screening. While Mader’s et al. 40 intervention was difficult to assess due to its short study duration of 6 months, Morley’s et al. 39 intervention was measured for 6 years and actually saw a decrease in cervical cancer screening rates.
Results from the intervention trials included in this review may be influenced by factors that affected the uptake of the intervention. Thus, a next step is determining what will work for specific organizations attempting to increase cancer screening rates, including the 3 guideline-endorsed options (Pap, Pap/HPV co-test, and Primary HPV) or self-sampling HPV testing. Use of an implementation science framework would help with devising an appropriate strategy tailored to the setting and the patients they serve. For example, Le’s et al. 41 used the Consolidated Framework for Implementation Framework (CFIR) 46 to provide better insights on both the internal and external contextual features of the implementation of self-collection of HPV testing in FQHCs.
This scoping review of published literature between 2012 and 2021 excluded gray literature, which may have led to missing information. Additionally, although we consulted with an academic librarian and used terms to ensure comprehensive capture, we may have inadvertently missed some literature. The sample sizes of 5 of the studies were small and limits generalizability of the findings. Finally, the ERIC strategies compilation is not exhaustive, and many additional strategies may be relevant dependent on the populations and healthcare settings.
In conclusion, more research is needed to further decipher the best ways to facilitate cervical cancer screening for patients, especially for those in under-resourced settings and not up to date on screening, for sustainable improvements in cervical cancer screening rates that may include Pap, Pap/HPV co-test, Primary HPV, or self-sampling HPV testing.
Footnotes
Author Contributions
KB drafted paper and participated in conducted the scoping review and selection process. She also contributed to several drafts of the manuscript and approved the version being submitted. HA oversaw study design, scoping review, selection process, and drafted the paper. She contributed to several drafts of the manuscript and approved the version being submitted. SM participated in conducted the scoping review, selection process, and data charting. She also contributed to several drafts of the manuscript and approved the version being submitted. HH participated in the selection process, data charting, and contributed to several drafts of the manuscript. She has approved the version being submitted. JE participated in the selection process, data charting, and contributed to several drafts of the manuscript. He has approved the version we are submitting. NH and JD critically revised several drafts of the manuscript and in the acquisition of the funding, and approved the version being submitted. PC critically revised several drafts of the manuscript and approved the version being submitted.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this [publication/press release] was supported by the National Cancer Institute of the National Institutes of Health under award number P50CA244289. This program is supported by funding provided through the Cancer MoonshotSM. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
