Abstract
Objectives:
To evaluate uptake of lung cancer screening in an urban Native American clinic using 2 culturally targeted promotion strategies.
Methods:
Patients eligible for lung cancer screening from July 2019 to July 2021 were randomized to receive either a single culturally-targeted mailer from the clinic regarding possible eligibility for screening, or the same mailer plus a follow-up text message and additional mailing.
Results:
Overall, there were low rates of shared decision-making visit scheduling (8.5%) with no difference between promotion strategy groups (9.4% in control group vs 7.7% in culturally-targeted outreach group). Only about 50% of the lung cancer screening CT exams ordered were completed and returned to the clinic.
Conclusions:
While there was no difference between arms in this intervention, 8.5% of the sample did complete a shared decision-making visit after these low-cost interventions. The gap between the number of screening CTs ordered and number who completed the CT represents an area where further interventions should focus.
Introduction
More people die from lung cancer than colon, breast, and cervical cancer combined. 1 Lung cancer screening (LCS) is a relatively new recommended preventive health service (since 2013) for older adults who smoke or previously smoked cigarettes. 2 Despite widespread approval by professional societies, the United States Preventive Services Task Force, and all insurance payors, recent estimates are that only around 6.5% of eligible persons have been screened, 3 possibly due to the complexity of the screening process and poor understanding of eligibility and reimbursement. While the screening exam is a simple low-dose chest computed tomography (LDCT), the screening process is more complex than just the exam. Scientific and regulatory guidelines include specific eligibility criteria based on age and intensity of smoking estimated by daily smoking habits and years of smoking. Guidelines require a shared decision-making (SDM) visit to confirm eligibility and discuss risks and benefits of the screening service, as well as smoking cessation for current smokers. 4 If a patient is deemed appropriate—meaning they are both eligible and interested—a LDCT is ordered. Because of technical specifications for the LDCT this exam should only be performed at qualified imaging centers, 5 which adds additional complexity to the process.
Northern Plains American Indians (AI) experience the most extreme lung cancer disparities of all tribal regions in the US, with men having nearly double the mortality rate compared to men of other ethnicities living in the Northern Plains. 6 A recent study of American Indians/Alaska Natives (AI/AN) cancer treatment showed a significant delay in and lower rates of treatment initiation in a Medicare population, compared to non-Hispanic Whites. 7 Cigarette smoking is more prevalent in Minnesota AI communities 8 than the United States average, likely for cultural reasons and due to the legacy of settler-colonialism. Attitudes toward tobacco use and LCS are often different in the AI community than the general population. Traditionally, “tobacco is a sacred gift used for spiritual, cultural, and ceremonial practices” in some Indigenous cultures, including the Anishinaabe (Ojibwe) and Dakota in present day Minnesota. 9 Colonialism fundamentally altered access to and relationship with traditional tobacco, which for some Indigenous people has been replaced by a harmful relationship with commercial tobacco. Health messages about smoking cessation and LCS that do not differentiate between traditional and commercial tobacco use may be culturally unacceptable to some AI despite the formidable health burden that arises from the use of commercial tobacco.
Inadequate access to high quality health care also contributes to disparities in cancer screening and mortality. 10 AI remain underrepresented in medical research and have a traumatic history of interactions with medical researchers and government organizations. Trust is an important determinant of accepting recommendations for preventive care services. Trust in the recommended clinician is a facilitator, but mistrust of healthcare systems (both generally and toward specific healthcare delivery organizations) is a barrier to completing LDCT.
LCS is an opportunity to reduce AI/AN lung cancer deaths, but known barriers need to be overcome to effectively implement LCS in this population. High quality LCS starts with shared decision-making to incorporate patient values and includes commercial tobacco cessation counseling for current users. We have conducted a randomized culturally targeted LCS promotion pilot to increase uptake of LCS in an urban AI/AN clinic.
Methods
Study Design
We collaborated with an urban Native American community clinic in the Midwestern United States to pilot test the effectiveness of 2 randomly assigned LCS promotion strategies.
Setting
The clinic is a community health center that offers a full range of healthcare services. The clinic mission is to promote health and wellness of mind, body, and spirit of Native American families. The clinic is within 1.5 miles of 3 imaging centers affiliated with different large healthcare systems. In addition to a Chief Executive Officer who is of Native American identity, several staff are AI/AN and 2 AI/AN elders in residence are employed by the clinic.
Study Population
The study population were patients of the clinic who were possibly eligible for LCS. Possibly eligible here is here defined as age 50 to 80 years old with current or past commercial tobacco smoking documented in the electronic health record (EHR). We obtained a list from the EHR of such patients seen at the clinic from July 2019 to July 2021. Since the electronic health record does not reliably document quantitative pack-years smoked or time since quitting (the primary factors to determine which participants are truly eligible for LCS) it is often difficult or impossible to identify eligible individuals from EHR review alone. Patients were excluded from the study sample if they had received a CT (low dose or otherwise) of the chest in the last 12 months, as lung cancer screening CTs are obtained at 12-month intervals.
Human subjects’ protection approval was obtained by a commercial IRB (Advarra) and informed consent was waived. The clinic holds a Federal-Wide Assurance which ensures that research done at this site will be conducted in compliance with the federal regulations for the protection of human subjects in research.
Randomization and Treatment
Randomization to control versus intensive outreach group was done 1:1 in a block method, stratified by sex and reported smoking status. Randomization schedules were generated by the study statistician and tracked within the secure web-based Research Electronic Data Capture (REDCap).
All participants were mailed a letter from the clinic indicating they may be eligible for a new health service along with instructions for scheduling an appointment, a clinic-logo branded facemask, and a LCS brochure (Figure 1). The brochure was developed in collaboration with the clinic and the American Indian Cancer Foundation based on a previously published qualitative study. 11 Additionally, the clinic utilized existing social media accounts to inform about screening eligibility and benefits and ultimately, promote lung cancer screening during the study. Patients randomized to the control group received only this simple intervention.

Lung Cancer Screening brochure developed in collaboration with NACC and the American Indian Cancer Foundation.
Patients randomized to the intensive outreach group also received an additional promotion strategy which included a follow-up text message and a second mailing. This mailing included a bundle of traditional medicine (sage, cedar, or sweetgrass) and a story book about traditional tobacco developed by the Great Lakes Inter-Tribal Council. 12
Eligible patients who called the clinic to inquire about LCS after the mailing date were scheduled for a shared decision-making (SDM) visit, either via telehealth or in-person. Clinic providers were asked to use a standard template to guide SDM, including confirmation of eligibility and discussion of risks and benefits of screening. Clinic staff received education and resources from the study team, including guidance from a pulmonologist expert in LCS regarding elements of the SDM visit mandated by the Centers for Medicare and Medicaid Services, eligibility criteria, personalized risk assessment tools, LCS referrals, insurance coverage, use of note template during the SDM visit, and management of results.
Eligible and appropriate patients received a referral for the LDCT exam. Results are returned to the clinic, but any abnormal findings would also be managed by the usual process of evaluating lung nodules at each participating healthcare organization. Importantly, patients were offered their choice of referral location for LDCT as many of this clinic’s patients have strong preferences for where they receive specialty care.
Outcomes
The primary outcome of this study is completion rates of the LCS exam (standard of care health service) within 6 months of initial promotion. Secondary outcomes include the difference between the promotion strategy groups for the following outcomes within 6 months: participation in shared decision-making (SDM) visit (including scheduled or attended), order placed by clinical staff for LCS exam (LDCT), completion of LDCT, and recommended follow-up for abnormal LCS exam (including with a provider and/or traditional healer).
Data Collection and Statistical Analysis
Manual chart review was used to collect information on primary and secondary outcomes as well as patient characteristics such as smoking status, insurance payor, age, details about SDM visit, and details about LDCT exam. Data was collected and stored in REDCap.
Based on the average rate of screening completion for LCS-eligible persons in Minnesota (8%), we expected the LCS completion rate in the control group to be as low as 10% and considered an increase to 30% to be clinically meaningful (additional 20% of eligible persons screened). The clinic staff initially estimated 400 patients met study eligibility criteria, and so we assumed a total sample size of at least 350 with equal enrollment in each group (175 per group). For a 2-sided α of .05, this would provide a power of 99%.
Patients whose mailings were returned to the clinic were excluded from the study. Participant characteristics were summarized using mean (SD) for continuous and count (%) for categorical variables. We compared proportions of participants in each group using Chi-square tests. Exploratory analysis was conducted to determine the association between personal characteristics and the primary outcome, using Chi-Square tests (Fisher’s exact tests in cases of small cell counts) for categorical variables and t-tests for continuous variables. All analysis was completed in R, version 3.6.1.
Results
There were 523 patients at the clinic eligible for the study and 469 patients were randomized (Figure 2). Baseline characteristics are shown in Table 1. Most of the sample were female (57.4%), reported ongoing commercial tobacco smoking (62.5%) and nearly all lived within the metropolitan area (95.1%). The average age was 70 years old, with 46.8% of the sample reporting Medicaid as their insurance. Demographic characteristics were similar between the control and intervention groups.
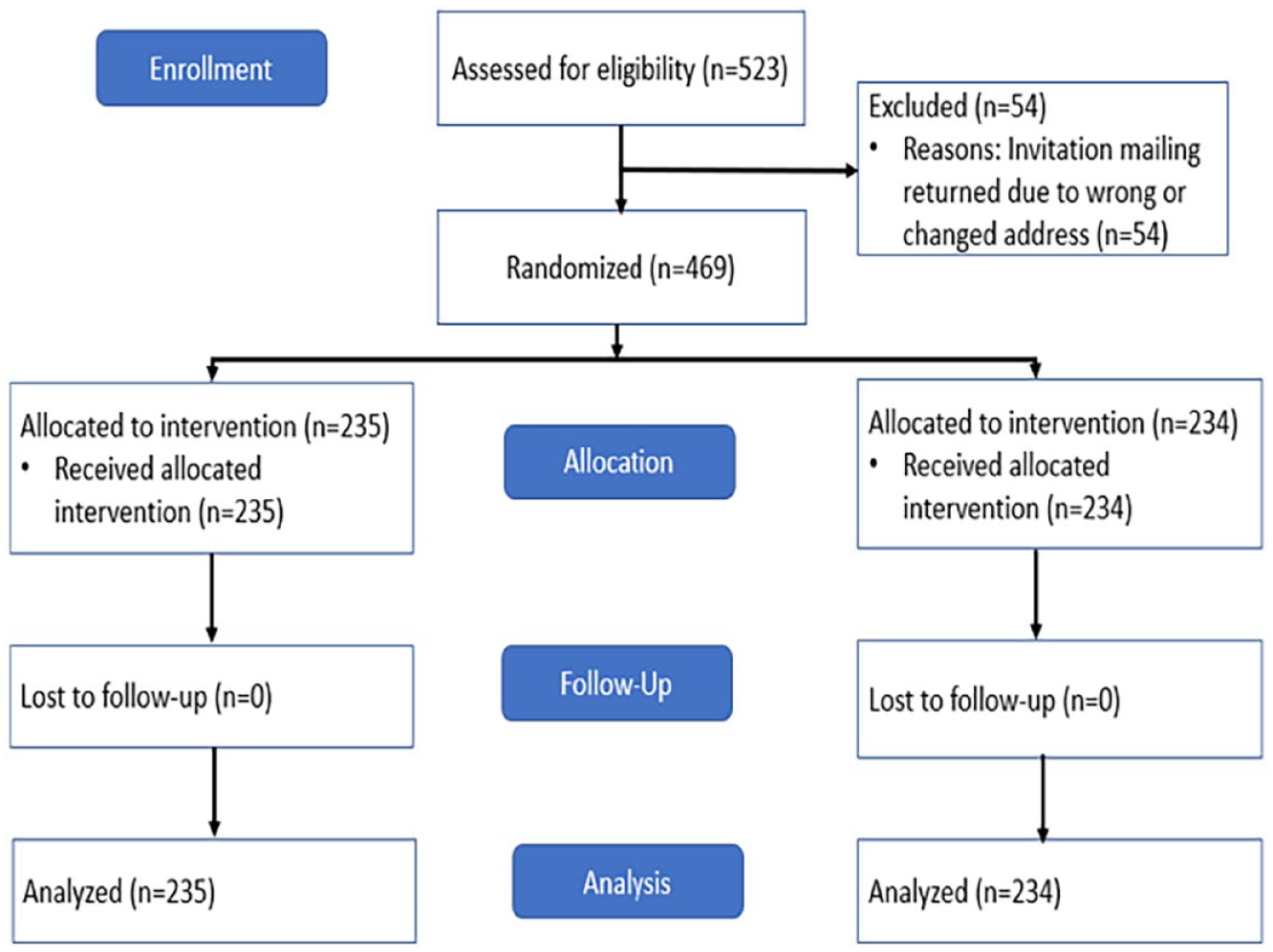
CONSORT diagram.
Participant Characteristics.

Overall, there were low rates of SDM visit scheduling (8.5%) with no difference between promotion strategy groups (9.4% in control group vs 7.7% in culturally targeted outreach group). Though not statistically significant, those who did schedule an SDM visit were more likely to be female (67.5%vs 56.5%) and have Medicaid insurance (50%vs 44.3%) (Table 2). Compared to those who did not schedule an SDM visit, those who did schedule were more likely to be current users of commercial tobacco (80%vs 60.8%, P-value = .026).
Proportion of Current Smokers.

There was no difference between the control and intervention groups for completion of SDM, eligibility of LCS, ordering LDCT, completing LCT, and LDCT results (Figure 3). Of the participants who scheduled an SDM visit, nearly all completed the visit, were eligible for lung cancer screening, and had a CT ordered. However, only about 50% of the LDCT exams ordered were completed and returned to the clinic.
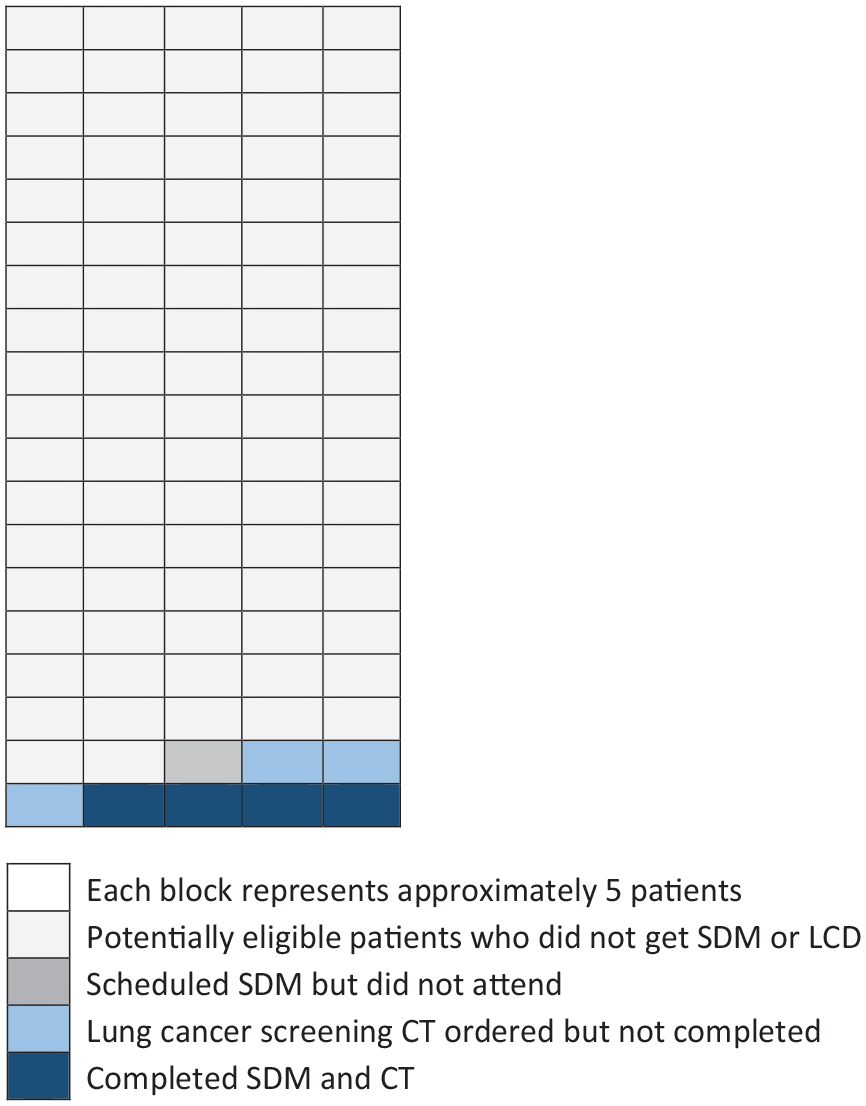
Number of patients who completed each step of the lung cancer screening process (out of 469 who were sent a mailed LCS brochure) showing a sharp decline between number of CTs ordered and number of CTs completed.
Discussion
We found no difference in the rate of shared decision-making visit or low dose CT completion between groups. Because this was an implementation study to increase uptake of an approved screening process, the benefit to this clinic is further reaching than the number of CT scans obtained during the study period. Patients who attended an SDM visit and use commercial tobacco had an opportunity to receive commercial tobacco cessation assistance. Providers at this clinic were trained in SDM, indications for lung cancer screening, and the process for referral to a lung cancer screening center.
While there was no difference between arms in this intervention, 8.5% of the sample did complete a SDM visit by the end of the study period. If each of these eligible patients had undergone CT, this would be on par with Minnesota state LCS uptake estimates, 13 using the low-cost intervention of a culturally targeted mailing. However, only 3.8% had a confirmed completion of LDCT by the end of the study period, which is below the national average. Thus, the gap between the number of screening CTs ordered and number of patients who completed the CT represents an area where further interventions should focus. While virtual SDM visits were offered, twice as many patients chose to have an in-person visit, which perhaps demonstrates the value patients see with an in-person visit and the potential barriers to care that virtual visits entail, such as not having a stable internet connection. Importantly, the screening LDCTs could not be conducted at NACC, but rather were scheduled and conducted at another location. This required the patients to travel to and receive care within a different local healthcare system. This may have contributed to added patient burden resulting in incomplete screening.
In this sample, those who completed the SDM visit (80%) reported ongoing commercial tobacco use, compared to the sample at large (62%). In the National Lung Screening Trial, about half of the participants continued to smoke cigarettes. We interpret this finding (higher rates of ongoing commercial tobacco use in individuals willing to engage in SDM) to represent little to no stigma surrounding cigarette smoking. Although people who smoke in the US population at large experience perceived and internalized stigma around cigarette smoking, we believe older Indigenous Minnesotans do not perceive such stigma. The cultural practices and beliefs stemming from colonization and commercialization of a sacred medicine (tobacco), may have insulated 11 against wider stigmatizing messages associated with public messaging to promote tobacco control. Looking at tobacco use within the context of colonization and commercialization of a sacred medicine, these findings represent a unique opportunity to end the misperception that AI/AN who choose to use commercial tobacco do not care about their health. SDM visits provide an opportunity to explain the benefits of reclaiming traditional tobacco and honoring bodily autonomy through appropriate cancer screening.
The study has several limitations including that it took place at a single site. Because the control group included a culturally targeted brochure with description of eligibility for lung cancer screening, we cannot make any conclusions about how many of the participants in the clinic were eligible for screening overall. While most other screening promotion mail interventions have low to modest effect, they are low-cost. Additionally, combining with a financial incentive has shown to be effective. 14
The National Lung Screening Trial 15 showed a 20% relative reduction in lung cancer mortality within eligible individuals using a LDCT compared to the control group who received radiography for lung cancer screening. Nearly a quarter of AI/AN nationally over 50 are expected to be eligible for LCS, a greater proportion than any other racial group. 16 Of note, only 190 of the 53 456 participants in the NLST identified as AI/AN. Due to structural and individual racism and discrimination, AI/AN individuals have justifiable mistrust of medical researchers and government organizations, and are often underrepresented in medical research.
The clinic recognizes its important role in conducting meaningful, ethical research within its population. Previously, patients of this clinic reported barriers to LCS including a mistrust of health systems in general (but a strong trust in their clinic), diagnosis knowledge avoidance, low-perceived personal value for a diagnosis yet a high-perceived community value for wellbeing, and practical resource barriers such as transportation. 11 This information guided the process of making and testing a culturally targeted brochure, used within this study. Culturally targeted approaches to LCS, such as the intervention described within this study, are needed to ensure that contextually appropriate care is provided to historically oppressed communities.
Efforts should continue to identify and resolve barriers to lung cancer screening in AI/AN and to ensure this group is equitably engaged in shared decision-making regarding lung cancer screening.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Institutes of Health’s National Heart, Lung, and Blood Institute grant T32 HL007741-23 (CR), the National Center for Advancing Translational Sciences grant UL1TR002494 (AB), and the University of Minnesota Program in Health Disparities Research. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Health or any of the authors’ affiliated academic institutions.
